Hungary: These 31 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Hungary
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: Hungary: These 31 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| PANCREAZE | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| ZENPEP | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| EMBEDA | morphine sulfate; naltrexone hydrochloride | 2027-06-19 |
| EMBEDA | morphine sulfate; naltrexone hydrochloride | 2026-06-19 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: Hungary: These 31 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Hungary Patent E026168
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Hungary Patent E026168
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Hungary Patent E031590
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Hungary Patent E032156
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Hungary Patent E033058
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 28, 2026
Generic Entry Controlled by: Hungary Patent E027156
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: Hungary Patent E038174
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: Hungary Patent E043502
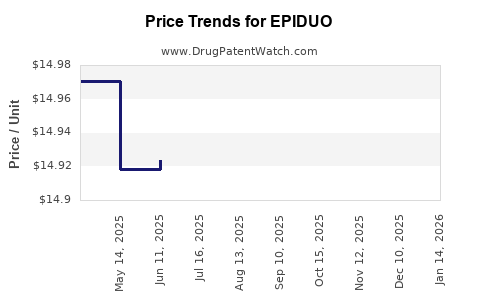
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: Hungary Patent E043502
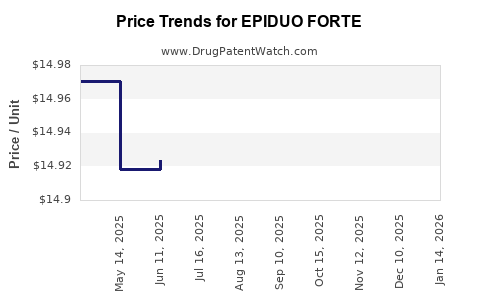
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: Hungary Patent E020998

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: Hungary Patent E047985
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Hungary Patent E025909
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Hungary Patent E041429
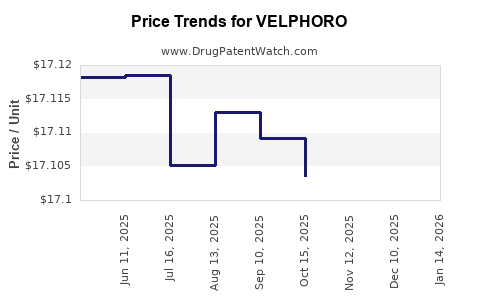
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 15, 2026
Generic Entry Controlled by: Hungary Patent E027128
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 15, 2026
Generic Entry Controlled by: Hungary Patent E032012
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: Hungary Patent E031939
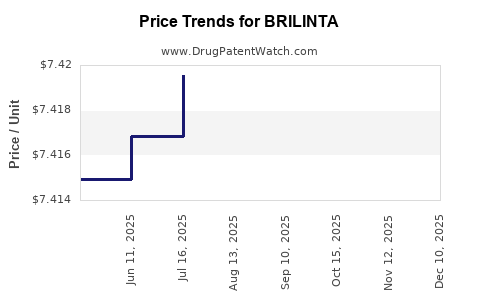
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: Hungary Patent E030158

This drug has four hundred and twenty-eight patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: Hungary Patent S1700020

This drug has four hundred and twenty-eight patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can LETAIRIS (ambrisentan) generic drug versions launch?
Generic name: ambrisentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Hungary Patent E025355
LETAIRIS is a drug marketed by Gilead. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-one patent family members in twenty-six countries. There has been litigation on patents covering LETAIRIS
See drug price trends for LETAIRIS.
The generic ingredient in LETAIRIS is ambrisentan. There are nine drug master file entries for this API. Thirteen suppliers are listed for this generic product. Additional details are available on the ambrisentan profile page.
When can LETAIRIS (ambrisentan) generic drug versions launch?
Generic name: ambrisentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Hungary Patent S1600027
LETAIRIS is a drug marketed by Gilead. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-one patent family members in twenty-six countries. There has been litigation on patents covering LETAIRIS
See drug price trends for LETAIRIS.
The generic ingredient in LETAIRIS is ambrisentan. There are nine drug master file entries for this API. Thirteen suppliers are listed for this generic product. Additional details are available on the ambrisentan profile page.
When can PRESTALIA (amlodipine besylate; perindopril arginine) generic drug versions launch?
Generic name: amlodipine besylate; perindopril arginine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 26, 2027
Generic Entry Controlled by: Hungary Patent E027898
PRESTALIA is a drug marketed by Adhera. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has thirty patent family members in twenty-seven countries. There has been litigation on patents covering PRESTALIA
The generic ingredient in PRESTALIA is amlodipine besylate; perindopril arginine. There are fifty drug master file entries for this API. Additional details are available on the amlodipine besylate; perindopril arginine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 01, 2027
Generic Entry Controlled by: Hungary Patent E027209
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Hungary Patent E030233
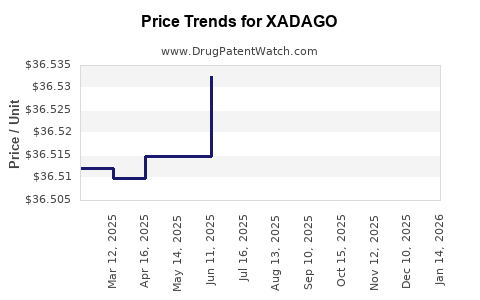
This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can ZEMDRI (plazomicin sulfate) generic drug versions launch?
Generic name: plazomicin sulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 21, 2027
Generic Entry Controlled by: Hungary Patent E030523
ZEMDRI is a drug marketed by Cipla Usa. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries. There has been litigation on patents covering ZEMDRI
See drug price trends for ZEMDRI.
The generic ingredient in ZEMDRI is plazomicin sulfate. Three suppliers are listed for this generic product. Additional details are available on the plazomicin sulfate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 20, 2027
Generic Entry Controlled by: Hungary Patent S2100001
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: Hungary Patent E031550
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: Hungary Patent E040391
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Hungary Patent E026206

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Hungary Patent E026507

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Hungary Patent E044237

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Hungary Patent E030519
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Hungary Patent S2200024
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Hungary Patent E027896
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Hungary Patent E053098
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Hungary Patent S1800010
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can LOCAMETZ (gallium ga-68 gozetotide) generic drug versions launch?
Generic name: gallium ga-68 gozetotide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 17, 2027
Generic Entry Controlled by: Hungary Patent E047200
LOCAMETZ is a drug marketed by Novartis. There are two patents protecting this drug.
This drug has fifty-five patent family members in sixteen countries.
See drug price trends for LOCAMETZ.
The generic ingredient in LOCAMETZ is gallium ga-68 gozetotide. There are sixteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the gallium ga-68 gozetotide profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: Hungary Patent S2200045
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: Hungary Patent E031550
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: Hungary Patent E040391
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 29, 2026
Generic Entry Controlled by: Hungary Patent S2400046
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: Hungary Patent E067736
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Hungary Patent S2500052

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: Hungary Patent E072202
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can AMYVID (florbetapir f-18) generic drug versions launch?
Generic name: florbetapir f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 26, 2027
Generic Entry Controlled by: Hungary Patent E032660
AMYVID is a drug marketed by Avid Radiopharms Inc. There are two patents protecting this drug.
This drug has fifty-one patent family members in thirty-three countries. There has been litigation on patents covering AMYVID
The generic ingredient in AMYVID is florbetapir f-18. One supplier is listed for this generic product. Additional details are available on the florbetapir f-18 profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 11, 2026
Generic Entry Controlled by: Hungary Patent E029145

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 11, 2026
Generic Entry Controlled by: Hungary Patent E037802

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 11, 2026
Generic Entry Controlled by: Hungary Patent S1500008

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can ORKAMBI (ivacaftor; lumacaftor) generic drug versions launch?
Generic name: ivacaftor; lumacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 07, 2027
Generic Entry Controlled by: Hungary Patent E031913
ORKAMBI is a drug marketed by Vertex Pharms Inc. There are twenty-three patents protecting this drug.
This drug has four hundred and sixty-one patent family members in thirty-six countries. There has been litigation on patents covering ORKAMBI
See drug price trends for ORKAMBI.
The generic ingredient in ORKAMBI is ivacaftor; lumacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; lumacaftor profile page.
When can ORKAMBI (ivacaftor; lumacaftor) generic drug versions launch?
Generic name: ivacaftor; lumacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 07, 2027
Generic Entry Controlled by: Hungary Patent E049642
ORKAMBI is a drug marketed by Vertex Pharms Inc. There are twenty-three patents protecting this drug.
This drug has four hundred and sixty-one patent family members in thirty-six countries. There has been litigation on patents covering ORKAMBI
See drug price trends for ORKAMBI.
The generic ingredient in ORKAMBI is ivacaftor; lumacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; lumacaftor profile page.
When can ORKAMBI (ivacaftor; lumacaftor) generic drug versions launch?
Generic name: ivacaftor; lumacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 07, 2027
Generic Entry Controlled by: Hungary Patent S2000030
ORKAMBI is a drug marketed by Vertex Pharms Inc. There are twenty-three patents protecting this drug.
This drug has four hundred and sixty-one patent family members in thirty-six countries. There has been litigation on patents covering ORKAMBI
See drug price trends for ORKAMBI.
The generic ingredient in ORKAMBI is ivacaftor; lumacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; lumacaftor profile page.
When can DUZALLO (allopurinol; lesinurad) generic drug versions launch?
Generic name: allopurinol; lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: Hungary Patent S1900009

This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering DUZALLO
See drug price trends for DUZALLO.
The generic ingredient in DUZALLO is allopurinol; lesinurad. There are twenty-two drug master file entries for this API. Additional details are available on the allopurinol; lesinurad profile page.
When can ZURAMPIC (lesinurad) generic drug versions launch?
Generic name: lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: Hungary Patent S1900009
ZURAMPIC is a drug marketed by Ironwood Pharms Inc. There are eight patents protecting this drug.
This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering ZURAMPIC
See drug price trends for ZURAMPIC.
The generic ingredient in ZURAMPIC is lesinurad. Additional details are available on the lesinurad profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 08, 2026
Generic Entry Controlled by: Hungary Patent S1500049
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can INVEGA SUSTENNA (paliperidone palmitate) generic drug versions launch?
Generic name: paliperidone palmitate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 19, 2027
Generic Entry Controlled by: Hungary Patent E053904
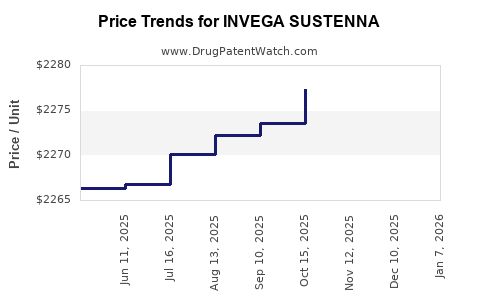
This drug has thirty-eight patent family members in twenty-nine countries. There has been litigation on patents covering INVEGA SUSTENNA
See drug price trends for INVEGA SUSTENNA.
The generic ingredient in INVEGA SUSTENNA is paliperidone palmitate. There are thirty-eight drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the paliperidone palmitate profile page.
When can ONPATTRO (patisiran sodium) generic drug versions launch?
Generic name: patisiran sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 27, 2027
Generic Entry Controlled by: Hungary Patent E034483
ONPATTRO is a drug marketed by Alnylam Pharms Inc. There are thirteen patents protecting this drug.
This drug has two hundred and fifty patent family members in thirty-one countries. There has been litigation on patents covering ONPATTRO
See drug price trends for ONPATTRO.
The generic ingredient in ONPATTRO is patisiran sodium. One supplier is listed for this generic product. Additional details are available on the patisiran sodium profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent E025565
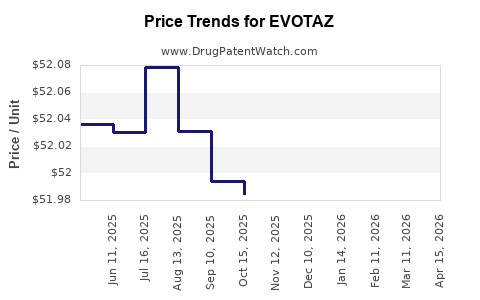
This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent E029842

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent E029843

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent E029866

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent E031707

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent S1600052

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent S1600053

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent S1600054

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: Hungary Patent S2000013

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 09, 2027
Generic Entry Controlled by: Hungary Patent E026145
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 09, 2027
Generic Entry Controlled by: Hungary Patent E027630
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 09, 2027
Generic Entry Controlled by: Hungary Patent E055205
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can ALIQOPA (copanlisib dihydrochloride) generic drug versions launch?
Generic name: copanlisib dihydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 05, 2027
Generic Entry Controlled by: Hungary Patent E028773
ALIQOPA is a drug marketed by Bayer Healthcare. There are three patents protecting this drug.
This drug has one hundred and five patent family members in forty-eight countries.
See drug price trends for ALIQOPA.
The generic ingredient in ALIQOPA is copanlisib dihydrochloride. Additional details are available on the copanlisib dihydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 26, 2026
Generic Entry Controlled by: Hungary Patent E028987
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can PYLARIFY (piflufolastat f-18) generic drug versions launch?
Generic name: piflufolastat f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 08, 2026
Generic Entry Controlled by: Hungary Patent E026216
PYLARIFY is a drug marketed by Progenics Pharms Inc and AphelionThere are six patents protecting this drug.
This drug has one hundred and sixteen patent family members in twenty-seven countries.
See drug price trends for PYLARIFY.
The generic ingredient in PYLARIFY is piflufolastat f-18. One supplier is listed for this generic product. Additional details are available on the piflufolastat f-18 profile page.
When can PYLARIFY (piflufolastat f-18) generic drug versions launch?
Generic name: piflufolastat f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 07, 2027
Generic Entry Controlled by: Hungary Patent E039199
PYLARIFY is a drug marketed by Progenics Pharms Inc and AphelionThere are six patents protecting this drug.
This drug has one hundred and sixteen patent family members in twenty-seven countries.
See drug price trends for PYLARIFY.
The generic ingredient in PYLARIFY is piflufolastat f-18. One supplier is listed for this generic product. Additional details are available on the piflufolastat f-18 profile page.
When can PYLARIFY (piflufolastat f-18) generic drug versions launch?
Generic name: piflufolastat f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 08, 2026
Generic Entry Controlled by: Hungary Patent E059645
PYLARIFY is a drug marketed by Progenics Pharms Inc and AphelionThere are six patents protecting this drug.
This drug has one hundred and sixteen patent family members in twenty-seven countries.
See drug price trends for PYLARIFY.
The generic ingredient in PYLARIFY is piflufolastat f-18. One supplier is listed for this generic product. Additional details are available on the piflufolastat f-18 profile page.
When can PYLARIFY TRUVU (piflufolastat f-18) generic drug versions launch?
Generic name: piflufolastat f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 08, 2026
Generic Entry Controlled by: Hungary Patent E026216
PYLARIFY TRUVU is a drug marketed by Aphelion. There are six patents protecting this drug.
This drug has one hundred and sixteen patent family members in twenty-seven countries.
The generic ingredient in PYLARIFY TRUVU is piflufolastat f-18. One supplier is listed for this generic product. Additional details are available on the piflufolastat f-18 profile page.
When can PYLARIFY TRUVU (piflufolastat f-18) generic drug versions launch?
Generic name: piflufolastat f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 07, 2027
Generic Entry Controlled by: Hungary Patent E039199
PYLARIFY TRUVU is a drug marketed by Aphelion. There are six patents protecting this drug.
This drug has one hundred and sixteen patent family members in twenty-seven countries.
The generic ingredient in PYLARIFY TRUVU is piflufolastat f-18. One supplier is listed for this generic product. Additional details are available on the piflufolastat f-18 profile page.
When can PYLARIFY TRUVU (piflufolastat f-18) generic drug versions launch?
Generic name: piflufolastat f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 08, 2026
Generic Entry Controlled by: Hungary Patent E059645
PYLARIFY TRUVU is a drug marketed by Aphelion. There are six patents protecting this drug.
This drug has one hundred and sixteen patent family members in twenty-seven countries.
The generic ingredient in PYLARIFY TRUVU is piflufolastat f-18. One supplier is listed for this generic product. Additional details are available on the piflufolastat f-18 profile page.
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
