CRESEMBA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cresemba, and when can generic versions of Cresemba launch?
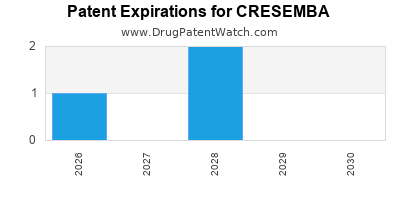
Cresemba is a drug marketed by Astellas and is included in two NDAs. There are three patents protecting this drug and two Paragraph IV challenges.
This drug has thirty-two patent family members in nineteen countries.
The generic ingredient in CRESEMBA is isavuconazonium sulfate. One supplier is listed for this compound. Additional details are available on the isavuconazonium sulfate profile page.
DrugPatentWatch® Generic Entry Outlook for Cresemba
Cresemba was eligible for patent challenges on March 6, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 8, 2030. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for CRESEMBA?
- What are the global sales for CRESEMBA?
- What is Average Wholesale Price for CRESEMBA?
Summary for CRESEMBA
| International Patents: | 32 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 51 |
| Clinical Trials: | 5 |
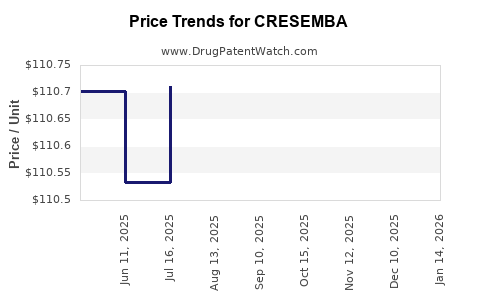
| Drug Prices: | Drug price information for CRESEMBA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CRESEMBA |
| What excipients (inactive ingredients) are in CRESEMBA? | CRESEMBA excipients list |
| DailyMed Link: | CRESEMBA at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for CRESEMBA
Generic Entry Dates for CRESEMBA*:
Constraining patent/regulatory exclusivity:
TREATMENT OF INVASIVE MUCORMYCOSIS IN PEDIATRIC PATIENTS 6 YEARS OF AGE AND OLDER WHO WEIGH 16 KG AND GREATER NDA:
Dosage:
CAPSULE;ORAL |
Generic Entry Dates for CRESEMBA*:
Constraining patent/regulatory exclusivity:
TREATMENT OF INVASIVE MUCORMYCOSIS IN PEDIATRIC PATIENTS 1 YEAR OF AGE AND OLDER NDA:
Dosage:
POWDER;INTRAVENOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for CRESEMBA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Pfizer | Phase 4 |
| Jeffrey Jenks, MD, MPH | Phase 3 |
| Astellas Pharma Global Development, Inc. | Phase 3 |
Pharmacology for CRESEMBA
| Drug Class | Azole Antifungal |
| Mechanism of Action | Cytochrome P450 3A4 Inhibitors Organic Cation Transporter 2 Inhibitors P-Glycoprotein Inhibitors |
Paragraph IV (Patent) Challenges for CRESEMBA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CRESEMBA | Capsules | isavuconazonium sulfate | 186 mg | 207500 | 4 | 2024-09-06 |
| CRESEMBA | For Injection | isavuconazonium sulfate | 372 mg/vial | 207501 | 2 | 2024-09-06 |
US Patents and Regulatory Information for CRESEMBA
CRESEMBA is protected by three US patents and twelve FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of CRESEMBA is ⤷ Start Trial.
This potential generic entry date is based on TREATMENT OF INVASIVE MUCORMYCOSIS IN PEDIATRIC PATIENTS 6 YEARS OF AGE AND OLDER WHO WEIGH 16 KG AND GREATER.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astellas | CRESEMBA | isavuconazonium sulfate | POWDER;INTRAVENOUS | 207501-001 | Mar 6, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Astellas | CRESEMBA | isavuconazonium sulfate | POWDER;INTRAVENOUS | 207501-001 | Mar 6, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astellas | CRESEMBA | isavuconazonium sulfate | CAPSULE;ORAL | 207500-002 | Nov 22, 2022 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Astellas | CRESEMBA | isavuconazonium sulfate | CAPSULE;ORAL | 207500-001 | Mar 6, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astellas | CRESEMBA | isavuconazonium sulfate | CAPSULE;ORAL | 207500-001 | Mar 6, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for CRESEMBA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astellas | CRESEMBA | isavuconazonium sulfate | CAPSULE;ORAL | 207500-002 | Nov 22, 2022 | ⤷ Start Trial | ⤷ Start Trial |
| Astellas | CRESEMBA | isavuconazonium sulfate | CAPSULE;ORAL | 207500-001 | Mar 6, 2015 | ⤷ Start Trial | ⤷ Start Trial |
| Astellas | CRESEMBA | isavuconazonium sulfate | POWDER;INTRAVENOUS | 207501-001 | Mar 6, 2015 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for CRESEMBA
When does loss-of-exclusivity occur for CRESEMBA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 07302320
Patent: Active ingredient containing stabilised solid medicinal forms and method for the production thereof
Estimated Expiration: ⤷ Start Trial
Austria
Patent: 59342
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 63941
Patent: FORMES GALENIQUES SOLIDES STABILISEES, ET LEUR PROCEDE DE PRODUCTION (STABILIZED SOLID FORMS AND METHOD FOR THE PRODUCTION THEREOF)
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 68842
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 02708
Patent: Compositions pharmaceutiques stabilisées et solides contenant au moins un médicament et procédé d'élaboration (Drug comprising stabilized pharmaceutical solid compositions and processes for their preparation)
Estimated Expiration: ⤷ Start Trial
Patent: 68842
Patent: FORMES GALÉNIQUES SOLIDES STABILISÉES CONTENANT UN PRINCIPE ACTIF ET DICITRATE DU TRIMAGNÉSIUM COMME DESSICCATEUR ET LEUR PROCÉDÉ DE PRODUCTION (ACTIVE INGREDIENT AND TRIMAGNESIUMDICITRATE AS DESICCANT CONTAINING STABILISED SOLID MEDICINAL FORMS AND METHOD FOR THE PRODUCTION THEREOF)
Estimated Expiration: ⤷ Start Trial
Germany
Patent: 2007003037
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering CRESEMBA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 6980759 | ⤷ Start Trial | |
| Australia | 2835201 | ⤷ Start Trial | |
| Japan | 2019516310 | ⤷ Start Trial | |
| World Intellectual Property Organization (WIPO) | 2017218785 | ⤷ Start Trial | |
| Canada | 2663941 | FORMES GALENIQUES SOLIDES STABILISEES, ET LEUR PROCEDE DE PRODUCTION (STABILIZED SOLID FORMS AND METHOD FOR THE PRODUCTION THEREOF) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CRESEMBA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1280795 | 15C0096 | France | ⤷ Start Trial | PRODUCT NAME: ISAVUCONAZOLE; REGISTRATION NO/DATE: EU/1/15/1036 20151015 |
| 1280795 | 122014000122 | Germany | ⤷ Start Trial | PRODUCT NAME: SITAGLIPTIN IN ALLEN DEM SCHUTZ DES GRUNDPATENTS UNTERLIEGENDEN FORMEN; REGISTRATION NO/DATE: EU/1/07/383/001-018 20070321 |
| 1280795 | 92939 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: ISAVUCONBAZOLE EN TANT QUE SULFATE D'ISAVUCONAZONIUM OU EN TANT QUE SEL D'ISAVUCONAZONIUM AVEC TOUT AUTRE ANION PHARMACEUTIQUEMENT ACCEPTABLE, OPTIONNELLEMENT SOUS FORME D'UN SEL, HYDRATE OU SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE; FIRST REGISTRATION: 20151019 |
| 1280795 | 2015/073 | Ireland | ⤷ Start Trial | PRODUCT NAME: ISAVUCONAZOLE AS ISAVUCONAZONIUM SULFATE OR AS ISAVUCONAZONIUM SALT WITH ANY OTHER PHARMACEUTICALLY ACCEPTABLE ANION, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, HYDRATE, OR SOLVATE; REGISTRATION NO/DATE: EU/1/15/1036 20151015 |
| 1280795 | CR 2016 00002 | Denmark | ⤷ Start Trial | PRODUCT NAME: ISAVUCONAZOLE AS ISAVUCONAZONIUM SULFATE OR AS AN ISAVUCONAZONIUM SALT WITH ANY OTHER PHARMACEUTICALLY ACCEPTABLE ANION, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, HYDRATE, OR SOLVATE; REG. NO/DATE: EU/1/15/1036 20151019 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Cresemba (Isavuconazonium Sulfate): Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.