Japan: These 80 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Japan
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: Japan: These 80 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| QVAR REDIHALER | beclomethasone dipropionate | 2027-04-11 |
| CREON | pancrelipase (amylase;lipase;protease) | 2026-08-15 |
| PANCREAZE | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| ZENPEP | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| EMBEDA | morphine sulfate; naltrexone hydrochloride | 2027-06-19 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: Japan: These 80 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 11, 2027
Generic Entry Controlled by: Japan Patent 2,010,523,184
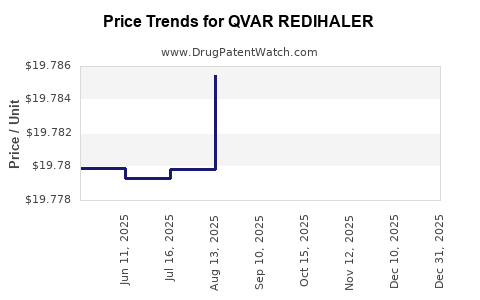
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 11, 2027
Generic Entry Controlled by: Japan Patent 5,246,968

This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: Japan Patent 2,009,504,709
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: Japan Patent 5,284,092
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: Japan Patent 5,452,918
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Japan Patent 2,010,519,217
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Japan Patent 2,014,074,053
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Japan Patent 5,966,202
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Japan Patent 2,010,519,217
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Japan Patent 2,014,074,053
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Japan Patent 5,966,202
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Japan Patent 2,009,541,320
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Japan Patent 5,566,102
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Japan Patent 2,011,507,956
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Japan Patent 2,015,187,178
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Japan Patent 2,017,014,295
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Japan Patent 5,783,725
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Japan Patent 6,033,373
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Japan Patent 6,328,205
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: Japan Patent 2,009,509,962
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: Japan Patent 5,715,101
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: Japan Patent 5,837,732
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Japan Patent 2,009,540,006
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Japan Patent 2,013,139,476
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Japan Patent 2,015,164,968
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can BUNAVAIL (buprenorphine hydrochloride; naloxone hydrochloride) generic drug versions launch?
Generic name: buprenorphine hydrochloride; naloxone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 13, 2026
Generic Entry Controlled by: Japan Patent 5,586,151

This drug has eighty-three patent family members in twenty-six countries. There has been litigation on patents covering BUNAVAIL
See drug price trends for BUNAVAIL.
The generic ingredient in BUNAVAIL is buprenorphine hydrochloride; naloxone hydrochloride. There are twenty-nine drug master file entries for this API. Twenty-seven suppliers are listed for this generic product. Additional details are available on the buprenorphine hydrochloride; naloxone hydrochloride profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: Japan Patent 5,654,873
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 2,009,545,525

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 2,013,209,394

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 2,015,071,636

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 2,016,172,758

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 2,017,222,681

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 5,313,889

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 5,666,651

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Japan Patent 5,937,187

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Japan Patent 2,009,542,779
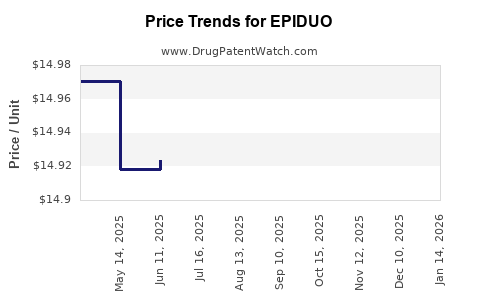
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: Japan Patent 2,016,029,094

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Japan Patent 2,009,542,779
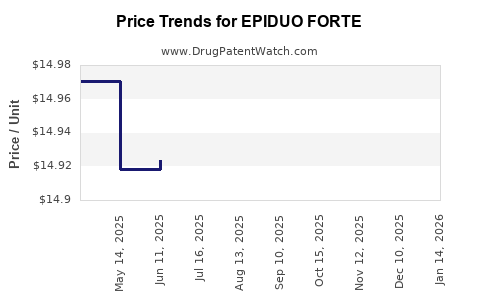
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: Japan Patent 2,016,029,094

See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: Japan Patent 2,009,519,314

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: Japan Patent 2,013,049,715

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: Japan Patent 5,484,733

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: Japan Patent 5,709,826

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can VIZAMYL (flutemetamol f-18) generic drug versions launch?
Generic name: flutemetamol f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 30, 2027
Generic Entry Controlled by: Japan Patent 2,010,536,931
VIZAMYL is a drug marketed by Ge Healthcare. There is one patent protecting this drug.
This drug has twenty-three patent family members in twenty countries.
See drug price trends for VIZAMYL.
The generic ingredient in VIZAMYL is flutemetamol f-18. One supplier is listed for this generic product. Additional details are available on the flutemetamol f-18 profile page.
When can VIZAMYL (flutemetamol f-18) generic drug versions launch?
Generic name: flutemetamol f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 30, 2027
Generic Entry Controlled by: Japan Patent 5,367,708
VIZAMYL is a drug marketed by Ge Healthcare. There is one patent protecting this drug.
This drug has twenty-three patent family members in twenty countries.
See drug price trends for VIZAMYL.
The generic ingredient in VIZAMYL is flutemetamol f-18. One supplier is listed for this generic product. Additional details are available on the flutemetamol f-18 profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Japan Patent 2,010,535,717
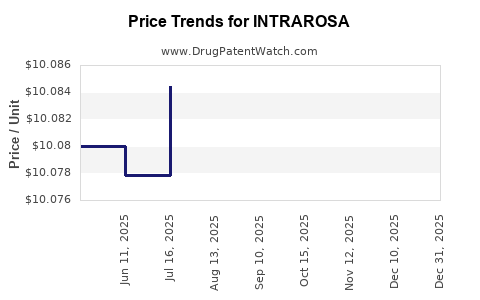
This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Japan Patent 2,013,060,475

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Japan Patent 5,725,283

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Japan Patent 6,197,292

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: Japan Patent 5,230,423
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: Japan Patent WO2008016077
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can SIGNIFOR LAR (pasireotide pamoate) generic drug versions launch?
Generic name: pasireotide pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 24, 2027
Generic Entry Controlled by: Japan Patent 5,507,447
SIGNIFOR LAR is a drug marketed by Recordati Rare. There are three patents protecting this drug.
See drug price trends for SIGNIFOR LAR.
The generic ingredient in SIGNIFOR LAR is pasireotide pamoate. One supplier is listed for this generic product. Additional details are available on the pasireotide pamoate profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,011,516,607

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,011,518,155

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,011,521,896

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,014,132,032

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,014,132,033

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,014,133,751

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,014,221,808

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,016,128,515

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,016,153,432

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,017,141,310

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,018,127,485

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 2,020,023,546

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Japan Patent 5,808,037

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can LEXISCAN (regadenoson) generic drug versions launch?
Generic name: regadenoson
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 02, 2027
Generic Entry Controlled by: Japan Patent 5,326,156
LEXISCAN is a drug marketed by Astellas. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has twenty-eight patent family members in eighteen countries. There has been litigation on patents covering LEXISCAN
See drug price trends for LEXISCAN.
The generic ingredient in LEXISCAN is regadenoson. There are nine drug master file entries for this API. Seventeen suppliers are listed for this generic product. Additional details are available on the regadenoson profile page.
When can TRIFERIC (ferric pyrophosphate citrate) generic drug versions launch?
Generic name: ferric pyrophosphate citrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 20, 2027
Generic Entry Controlled by: Japan Patent 2,009,108,027
TRIFERIC is a drug marketed by Rockwell Medical Inc. There are two patents protecting this drug.
This drug has thirteen patent family members in eleven countries.
See drug price trends for TRIFERIC.
The generic ingredient in TRIFERIC is ferric pyrophosphate citrate. There are twenty drug master file entries for this API. Additional details are available on the ferric pyrophosphate citrate profile page.
When can TRIFERIC (ferric pyrophosphate citrate) generic drug versions launch?
Generic name: ferric pyrophosphate citrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 20, 2027
Generic Entry Controlled by: Japan Patent 5,489,196
TRIFERIC is a drug marketed by Rockwell Medical Inc. There are two patents protecting this drug.
This drug has thirteen patent family members in eleven countries.
See drug price trends for TRIFERIC.
The generic ingredient in TRIFERIC is ferric pyrophosphate citrate. There are twenty drug master file entries for this API. Additional details are available on the ferric pyrophosphate citrate profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Japan Patent 2,010,530,876
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Japan Patent 2,013,075,918
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Japan Patent 5,589,102
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 21, 2026
Generic Entry Controlled by: Japan Patent 2,009,544,636
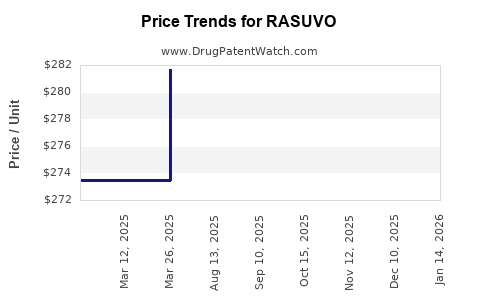
This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 21, 2026
Generic Entry Controlled by: Japan Patent 5,683,810

This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can DYANAVEL XR (amphetamine) generic drug versions launch?
Generic name: amphetamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 15, 2027
Generic Entry Controlled by: Japan Patent 2,009,530,298
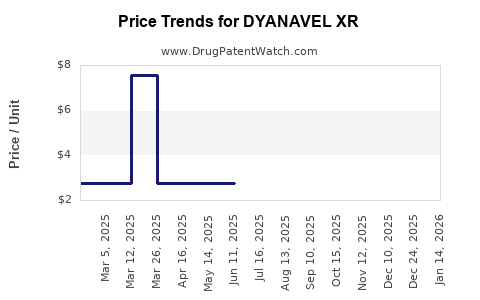
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering DYANAVEL XR
See drug price trends for DYANAVEL XR.
The generic ingredient in DYANAVEL XR is amphetamine. There are fifty-five drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the amphetamine profile page.
When can DYANAVEL XR (amphetamine) generic drug versions launch?
Generic name: amphetamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 15, 2027
Generic Entry Controlled by: Japan Patent 5,479,086

This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering DYANAVEL XR
See drug price trends for DYANAVEL XR.
The generic ingredient in DYANAVEL XR is amphetamine. There are fifty-five drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the amphetamine profile page.
When can TUZISTRA XR (chlorpheniramine polistirex; codeine polistirex) generic drug versions launch?
Generic name: chlorpheniramine polistirex; codeine polistirex
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 15, 2027
Generic Entry Controlled by: Japan Patent 2,009,530,298
TUZISTRA XR is a drug marketed by Tris Pharma Inc. There are two patents protecting this drug.
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering TUZISTRA XR
See drug price trends for TUZISTRA XR.
The generic ingredient in TUZISTRA XR is chlorpheniramine polistirex; codeine polistirex. There are twenty-nine drug master file entries for this API. Additional details are available on the chlorpheniramine polistirex; codeine polistirex profile page.
When can TUZISTRA XR (chlorpheniramine polistirex; codeine polistirex) generic drug versions launch?
Generic name: chlorpheniramine polistirex; codeine polistirex
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 15, 2027
Generic Entry Controlled by: Japan Patent 5,479,086
TUZISTRA XR is a drug marketed by Tris Pharma Inc. There are two patents protecting this drug.
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering TUZISTRA XR
See drug price trends for TUZISTRA XR.
The generic ingredient in TUZISTRA XR is chlorpheniramine polistirex; codeine polistirex. There are twenty-nine drug master file entries for this API. Additional details are available on the chlorpheniramine polistirex; codeine polistirex profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Japan Patent 2,011,503,148
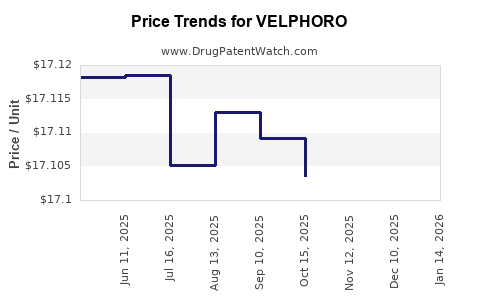
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Japan Patent 2,014,062,120

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Japan Patent 2,017,114,873

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Japan Patent 2,019,089,805

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Japan Patent 2,021,001,192

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Japan Patent 5,961,601