Saudi Arabia: These 1 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Saudi Arabia
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: Saudi Arabia: These 1 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| VELPHORO | sucroferric oxyhydroxide | 2027-11-16 |
| INTRAROSA | prasterone | 2027-08-10 |
| BRILINTA | ticagrelor | 2026-08-21 |
| PREPOPIK | citric acid; magnesium oxide; sodium picosulfate | 2027-10-12 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: Saudi Arabia: These 1 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Saudi Arabia Patent 112,340,044
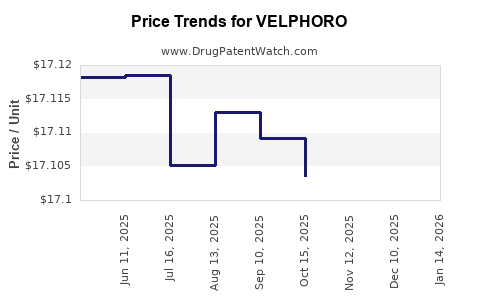
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Saudi Arabia Patent 111,320,726
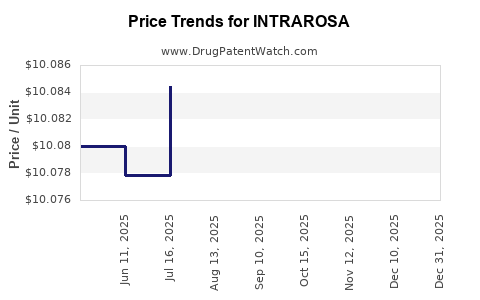
This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: Saudi Arabia Patent 7,280,442
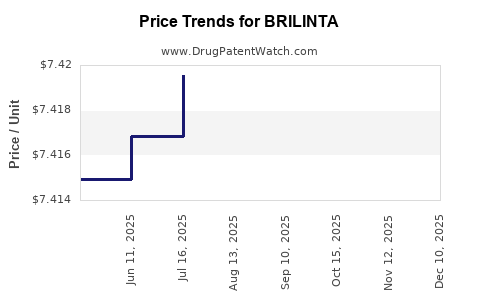
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: Saudi Arabia Patent 8,290,670
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
