South Korea: These 57 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in South Korea
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: South Korea: These 57 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| CREON | pancrelipase (amylase;lipase;protease) | 2026-08-15 |
| PANCREAZE | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| ZENPEP | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| EMBEDA | morphine sulfate; naltrexone hydrochloride | 2027-06-19 |
| RYTARY | carbidopa; levodopa | 2027-12-28 |
| ESBRIET | pirfenidone | 2026-09-22 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: South Korea: These 57 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: South Korea Patent 101,302,841
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: South Korea Patent 101,344,546
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 101,670,016
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 101,837,010
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 101,670,016
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 101,837,010
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 101,486,228
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: South Korea Patent 101,451,564
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: South Korea Patent 101,569,604
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: South Korea Patent 101,675,651
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: South Korea Patent 101,493,102

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: South Korea Patent 101,451,993

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: South Korea Patent 101,593,579

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: South Korea Patent 101,638,130
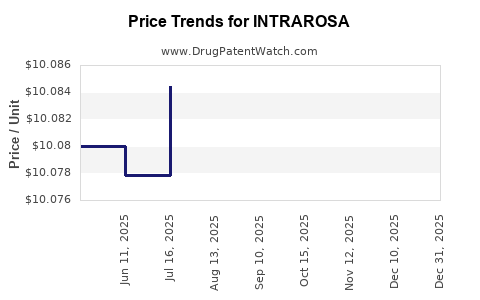
This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: South Korea Patent 101,680,287

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: South Korea Patent 101,850,125

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: South Korea Patent 101,420,450
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 28, 2026
Generic Entry Controlled by: South Korea Patent 101,608,755
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 101,598,402
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 101,916,024
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 21, 2026
Generic Entry Controlled by: South Korea Patent 101,260,554
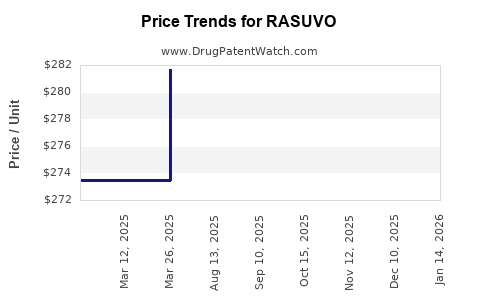
This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can DYANAVEL XR (amphetamine) generic drug versions launch?
Generic name: amphetamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 15, 2027
Generic Entry Controlled by: South Korea Patent 101,495,146
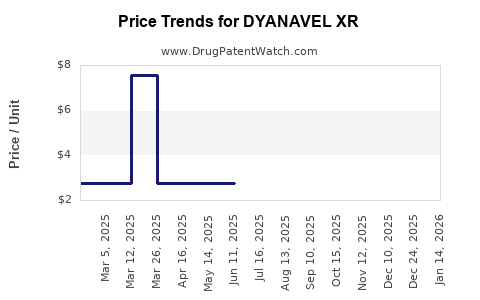
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering DYANAVEL XR
See drug price trends for DYANAVEL XR.
The generic ingredient in DYANAVEL XR is amphetamine. There are fifty-five drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the amphetamine profile page.
When can TUZISTRA XR (chlorpheniramine polistirex; codeine polistirex) generic drug versions launch?
Generic name: chlorpheniramine polistirex; codeine polistirex
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 15, 2027
Generic Entry Controlled by: South Korea Patent 101,495,146
TUZISTRA XR is a drug marketed by Tris Pharma Inc. There are two patents protecting this drug.
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering TUZISTRA XR
See drug price trends for TUZISTRA XR.
The generic ingredient in TUZISTRA XR is chlorpheniramine polistirex; codeine polistirex. There are twenty-nine drug master file entries for this API. Additional details are available on the chlorpheniramine polistirex; codeine polistirex profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: South Korea Patent 101,438,071
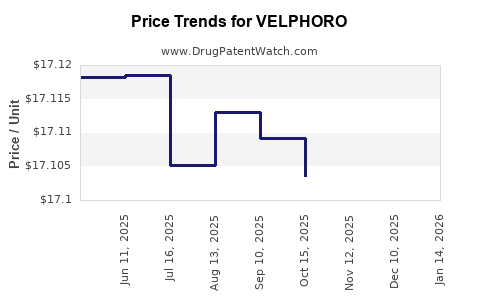
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: South Korea Patent 101,590,115

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: South Korea Patent 101,539,467
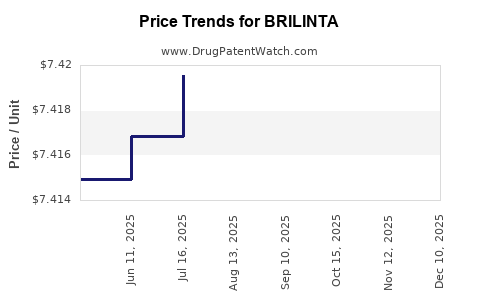
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: South Korea Patent 101,493,102
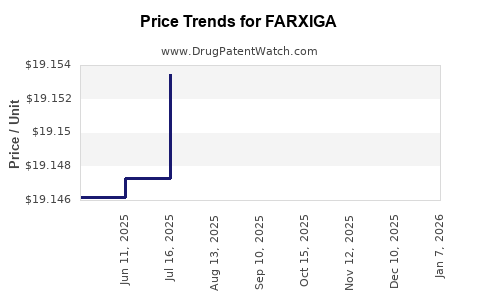
This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 05, 2027
Generic Entry Controlled by: South Korea Patent 101,237,588
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can JAKAFI (ruxolitinib phosphate) generic drug versions launch?
Generic name: ruxolitinib phosphate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 13, 2027
Generic Entry Controlled by: South Korea Patent 101,549,876
JAKAFI is a drug marketed by Incyte Corp. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and thirty-six patent family members in forty-six countries. There has been litigation on patents covering JAKAFI
See drug price trends for JAKAFI.
The generic ingredient in JAKAFI is ruxolitinib phosphate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ruxolitinib phosphate profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: South Korea Patent 101,491,554

This drug has four hundred and twenty-nine patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can KAZANO (alogliptin benzoate; metformin hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; metformin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 19, 2027
Generic Entry Controlled by: South Korea Patent 101,536,786

This drug has ninety patent family members in forty-one countries. There has been litigation on patents covering KAZANO
See drug price trends for KAZANO.
The generic ingredient in KAZANO is alogliptin benzoate; metformin hydrochloride. There are ten drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; metformin hydrochloride profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 29, 2026
Generic Entry Controlled by: South Korea Patent 101,473,022
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the macitentan profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 01, 2027
Generic Entry Controlled by: South Korea Patent 101,486,091

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: South Korea Patent 101,475,971
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: South Korea Patent 101,537,200
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: South Korea Patent 101,632,851
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: South Korea Patent 101,493,102
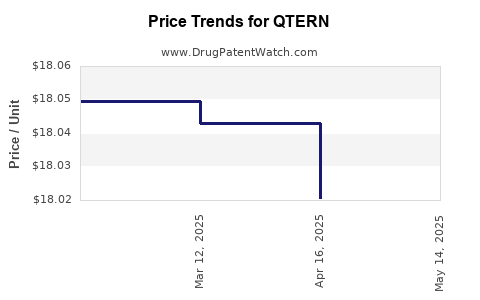
This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 05, 2026
Generic Entry Controlled by: South Korea Patent 101,514,700
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 23, 2026
Generic Entry Controlled by: South Korea Patent 101,442,272

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: South Korea Patent 101,445,514
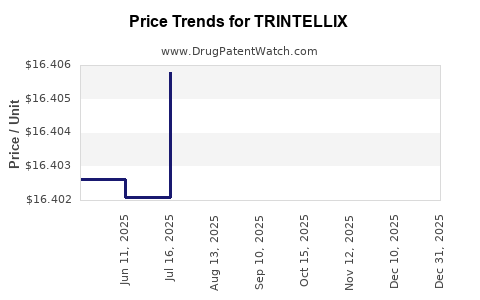
This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: South Korea Patent 101,459,168

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: South Korea Patent 101,627,901

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: South Korea Patent 101,410,291
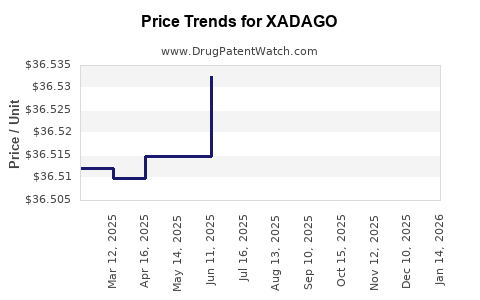
This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: South Korea Patent 101,491,541

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can ZEMDRI (plazomicin sulfate) generic drug versions launch?
Generic name: plazomicin sulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 21, 2027
Generic Entry Controlled by: South Korea Patent 101,296,099
ZEMDRI is a drug marketed by Cipla Usa. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries. There has been litigation on patents covering ZEMDRI
See drug price trends for ZEMDRI.
The generic ingredient in ZEMDRI is plazomicin sulfate. Three suppliers are listed for this generic product. Additional details are available on the plazomicin sulfate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 20, 2027
Generic Entry Controlled by: South Korea Patent 101,478,593
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: South Korea Patent 101,495,192

This drug has ninety-four patent family members in thirty countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: South Korea Patent 101,544,624
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: South Korea Patent 101,553,418
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: South Korea Patent 101,513,736
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: South Korea Patent 101,513,737
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: South Korea Patent 101,513,842
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: South Korea Patent 20,090,023,643

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: South Korea Patent 20,090,023,643

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: South Korea Patent 20,130,100,381
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: South Korea Patent 20,090,015,968
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: South Korea Patent 20,140,142,335
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: South Korea Patent 20,150,082,690
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: South Korea Patent 20,160,032,264
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: South Korea Patent 20,090,028,764
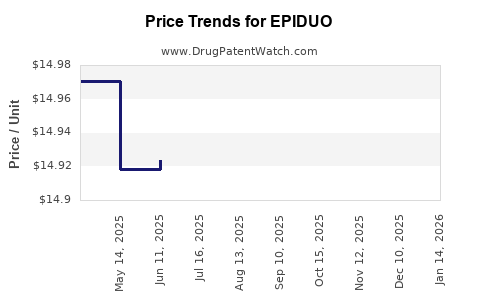
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: South Korea Patent 20,150,003,917

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: South Korea Patent 20,160,120,352

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: South Korea Patent 20,090,021,392

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: South Korea Patent 20,140,027,563

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 20, 2027
Generic Entry Controlled by: South Korea Patent 20,090,123,882
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 11, 2027
Generic Entry Controlled by: South Korea Patent 20,090,127,376
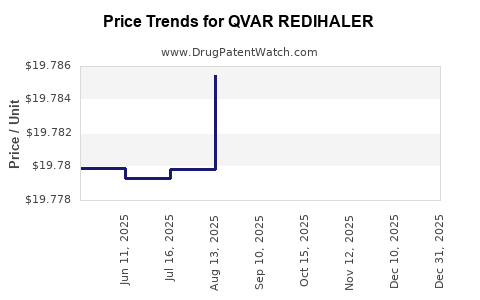
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can LEXISCAN (regadenoson) generic drug versions launch?
Generic name: regadenoson
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 02, 2027
Generic Entry Controlled by: South Korea Patent 20,080,090,491
LEXISCAN is a drug marketed by Astellas. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has twenty-eight patent family members in eighteen countries. There has been litigation on patents covering LEXISCAN
See drug price trends for LEXISCAN.
The generic ingredient in LEXISCAN is regadenoson. There are nine drug master file entries for this API. Seventeen suppliers are listed for this generic product. Additional details are available on the regadenoson profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 28, 2026
Generic Entry Controlled by: South Korea Patent 20,080,042,084
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 20,090,122,254
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 20,150,038,322
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 20,090,122,254
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: South Korea Patent 20,150,038,322
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 20,100,031,512
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 20,150,013,305
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 20,160,025,044
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 20,160,150,646
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 20,180,061,401
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: South Korea Patent 20,180,122,483
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: South Korea Patent 20,140,088,617

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 29, 2026
Generic Entry Controlled by: South Korea Patent 20,090,057,009
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the macitentan profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: South Korea Patent 20,100,099,731
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: South Korea Patent 20,130,113,533
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: South Korea Patent 20,150,133,854
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.