DUZALLO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Duzallo, and what generic alternatives are available?
Duzallo is a drug marketed by Ironwood Pharms Inc and is included in one NDA. There are eight patents protecting this drug.
This drug has one hundred and forty-four patent family members in thirty-nine countries.
The generic ingredient in DUZALLO is allopurinol; lesinurad. There are twenty-two drug master file entries for this compound. Additional details are available on the allopurinol; lesinurad profile page.
DrugPatentWatch® Generic Entry Outlook for Duzallo
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 26, 2028. This may change due to patent challenges or generic licensing.
There have been five patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
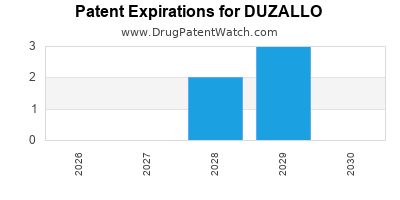
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for DUZALLO?
- What are the global sales for DUZALLO?
- What is Average Wholesale Price for DUZALLO?
Summary for DUZALLO
| International Patents: | 144 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Drug Prices: | Drug price information for DUZALLO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DUZALLO |
| DailyMed Link: | DUZALLO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for DUZALLO
Generic Entry Date for DUZALLO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
US Patents and Regulatory Information for DUZALLO
DUZALLO is protected by eight US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of DUZALLO is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ironwood Pharms Inc | DUZALLO | allopurinol; lesinurad | TABLET;ORAL | 209203-001 | Aug 18, 2017 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Ironwood Pharms Inc | DUZALLO | allopurinol; lesinurad | TABLET;ORAL | 209203-001 | Aug 18, 2017 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Ironwood Pharms Inc | DUZALLO | allopurinol; lesinurad | TABLET;ORAL | 209203-001 | Aug 18, 2017 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for DUZALLO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ironwood Pharms Inc | DUZALLO | allopurinol; lesinurad | TABLET;ORAL | 209203-002 | Aug 18, 2017 | ⤷ Start Trial | ⤷ Start Trial |
| Ironwood Pharms Inc | DUZALLO | allopurinol; lesinurad | TABLET;ORAL | 209203-001 | Aug 18, 2017 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for DUZALLO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Grunenthal GmbH | Duzallo | allopurinol, lesinurad | EMEA/H/C/004412Duzallo is indicated in adults for the treatment of hyperuricaemia in gout patients who have not achieved target serum uric acid levels with an adequate dose of allopurinol alone. | Withdrawn | no | no | no | 2018-08-23 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for DUZALLO
When does loss-of-exclusivity occur for DUZALLO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 9753
Estimated Expiration: ⤷ Start Trial
Patent: 3548
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 08329673
Estimated Expiration: ⤷ Start Trial
Patent: 09289646
Estimated Expiration: ⤷ Start Trial
Patent: 09289647
Estimated Expiration: ⤷ Start Trial
Patent: 12203172
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0819847
Estimated Expiration: ⤷ Start Trial
Patent: 0918584
Estimated Expiration: ⤷ Start Trial
Patent: 0918586
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 06858
Estimated Expiration: ⤷ Start Trial
Patent: 35828
Estimated Expiration: ⤷ Start Trial
Patent: 36117
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 08003529
Estimated Expiration: ⤷ Start Trial
Patent: 12000594
Estimated Expiration: ⤷ Start Trial
China
Patent: 1918377
Estimated Expiration: ⤷ Start Trial
Patent: 2186827
Estimated Expiration: ⤷ Start Trial
Patent: 2186832
Estimated Expiration: ⤷ Start Trial
Patent: 2643241
Estimated Expiration: ⤷ Start Trial
Patent: 3058944
Estimated Expiration: ⤷ Start Trial
Patent: 3819419
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 31333
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0140950
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16286
Estimated Expiration: ⤷ Start Trial
Patent: 19010
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 17577
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 10010310
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8193
Estimated Expiration: ⤷ Start Trial
Patent: 3500
Estimated Expiration: ⤷ Start Trial
Patent: 1000897
Estimated Expiration: ⤷ Start Trial
Patent: 1100440
Estimated Expiration: ⤷ Start Trial
Patent: 1100441
Estimated Expiration: ⤷ Start Trial
Patent: 1270666
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 17577
Estimated Expiration: ⤷ Start Trial
Patent: 28874
Estimated Expiration: ⤷ Start Trial
Patent: 28879
Estimated Expiration: ⤷ Start Trial
Patent: 42948
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 60843
Estimated Expiration: ⤷ Start Trial
Patent: 72326
Estimated Expiration: ⤷ Start Trial
Patent: 80337
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 900009
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 5974
Estimated Expiration: ⤷ Start Trial
Patent: 1429
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 99794
Estimated Expiration: ⤷ Start Trial
Patent: 05934
Estimated Expiration: ⤷ Start Trial
Patent: 18327
Estimated Expiration: ⤷ Start Trial
Patent: 11504935
Estimated Expiration: ⤷ Start Trial
Patent: 12184234
Estimated Expiration: ⤷ Start Trial
Patent: 12502049
Estimated Expiration: ⤷ Start Trial
Patent: 12502050
Estimated Expiration: ⤷ Start Trial
Patent: 14196373
Estimated Expiration: ⤷ Start Trial
Patent: 15042647
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 2019003
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0103
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 7370
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 10005776
Estimated Expiration: ⤷ Start Trial
Patent: 11002449
Estimated Expiration: ⤷ Start Trial
Patent: 11002450
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 294
Estimated Expiration: ⤷ Start Trial
Patent: 996
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 936
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 5583
Patent: NOVEL COMPOUNDS AND COMPOSITIONS AND METHODS OF USE
Estimated Expiration: ⤷ Start Trial
Patent: 5035
Patent: Substituted diazole and triazole compounds and compositions and methods of use
Estimated Expiration: ⤷ Start Trial
Patent: 1774
Patent: Substituted diazole and triazole compounds and compositions and methods of use
Estimated Expiration: ⤷ Start Trial
Patent: 1786
Patent: Novel compounds and compositions and methods of use
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 16016
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 17577
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 17577
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 569
Patent: NOVA JEDINJENJA I KOMPOZICIJE, TE POSTUPCI ZA UPOTREBU (NOVEL COMPOUNDS AND COMPOSITIONS AND METHODS OF USE)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 3721
Patent: NOVEL COMPOUNDS AND COMPOSITIONS AND METHODS OF USE
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 17577
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1003769
Patent: NOVEL COMPOUNDS AND COMPOSITIONS AND METHODS OF USE
Estimated Expiration: ⤷ Start Trial
Patent: 1101626
Patent: COMPOUNDS,COMPOSITIONS AND METHODS OF USING SAME FOR MODULATING URIC ACID LEVELS
Estimated Expiration: ⤷ Start Trial
Patent: 1203779
Patent: COMPOUNDS, COMPOSITIONS AND METHODS OF USING SAME FOR MODULATING URIC ACID LEVELS
Estimated Expiration: ⤷ Start Trial
Patent: 1204031
Patent: COMPOUNDS, COMPOSITIONS AND METHODS OF USING SAME FOR MODULATING URIC ACID LEVELS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1291643
Estimated Expiration: ⤷ Start Trial
Patent: 1294872
Estimated Expiration: ⤷ Start Trial
Patent: 1411806
Estimated Expiration: ⤷ Start Trial
Patent: 100085195
Estimated Expiration: ⤷ Start Trial
Patent: 110050708
Estimated Expiration: ⤷ Start Trial
Patent: 110050709
Estimated Expiration: ⤷ Start Trial
Patent: 120084787
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 13390
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0927733
Patent: Novel compounds and compositions and methods of use
Estimated Expiration: ⤷ Start Trial
Patent: 1018463
Patent: Compounds, compositions and methods of using same for modulating uric acid levels
Estimated Expiration: ⤷ Start Trial
Patent: 1022216
Patent: Compounds, compositions and methods of using same for modulating uric acid levels
Estimated Expiration: ⤷ Start Trial
Patent: 77200
Estimated Expiration: ⤷ Start Trial
Patent: 15840
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 10000237
Patent: NOVEL COMPOUNDS AND COMPOSITIONS AND METHODS OF USE
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 2826
Patent: ПРОИЗВОДНЫЕ ТРИАЗОЛА И ИХ ПРИМЕНЕНИЕ КАК ЛЕКАРСТВЕННОГО СРЕДСТВА (TRIAZOLE DERIVATIVES AND USE THEREOF AS A MEDICAMENT)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering DUZALLO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 2658846 | ⤷ Start Trial | |
| Taiwan | 201302718 | Polymorphic forms of 2-(5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-ylthio)acetic acid and uses thereof | ⤷ Start Trial |
| China | 102186827 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for DUZALLO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2135608 | C20160027 00203 | Estonia | ⤷ Start Trial | PRODUCT NAME: LESINURAAD;REG NO/DATE: EU/1/15/1080 22.02.2016 |
| 2135608 | CR 2016 00034 | Denmark | ⤷ Start Trial | PRODUCT NAME: LESINURAD ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU1/15/1080 20160222 |
| 2135608 | PA2016024,C2135608 | Lithuania | ⤷ Start Trial | PRODUCT NAME: LESINURADAS ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/15/1080 20160218 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Duzallo
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.