Taiwan: These 44 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Taiwan
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: Taiwan: These 44 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| PANCREAZE | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| ZENPEP | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| FARYDAK | panobinostat lactate | 2026-06-12 |
| PREPOPIK | citric acid; magnesium oxide; sodium picosulfate | 2027-10-12 |
| BYDUREON | exenatide synthetic | 2026-06-28 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: Taiwan: These 44 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Taiwan Patent I421090
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Taiwan Patent I421090
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Taiwan Patent I453196
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Taiwan Patent I515179
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: Taiwan Patent I418352
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I421245

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I466876

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I519528

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Taiwan Patent I388328
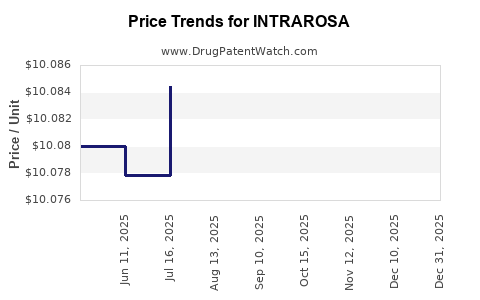
This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Taiwan Patent I609688

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: Taiwan Patent I414299
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can SIGNIFOR LAR (pasireotide pamoate) generic drug versions launch?
Generic name: pasireotide pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 24, 2027
Generic Entry Controlled by: Taiwan Patent I412373
SIGNIFOR LAR is a drug marketed by Recordati Rare. There are three patents protecting this drug.
See drug price trends for SIGNIFOR LAR.
The generic ingredient in SIGNIFOR LAR is pasireotide pamoate. One supplier is listed for this generic product. Additional details are available on the pasireotide pamoate profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 28, 2026
Generic Entry Controlled by: Taiwan Patent I371439
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Taiwan Patent I503327
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Taiwan Patent I503396
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Taiwan Patent I468167
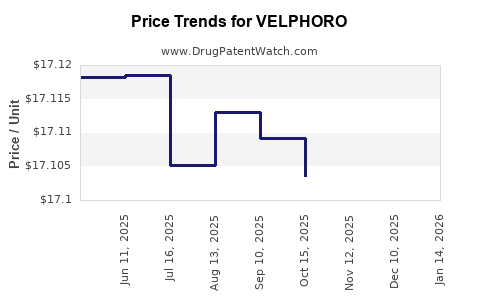
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Taiwan Patent I592159

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: Taiwan Patent I482772
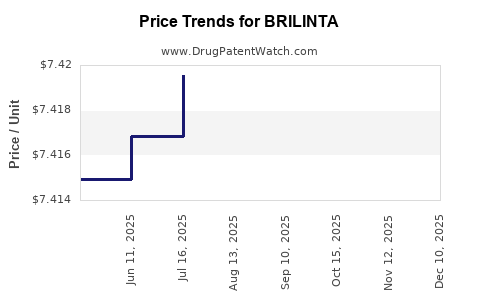
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2027
Generic Entry Controlled by: Taiwan Patent I421245
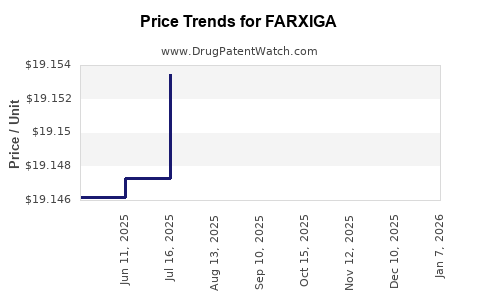
This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I466876

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I519528

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 05, 2027
Generic Entry Controlled by: Taiwan Patent I481602
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: Taiwan Patent I472325

This drug has four hundred and twenty-eight patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 29, 2026
Generic Entry Controlled by: Taiwan Patent I388556
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 01, 2027
Generic Entry Controlled by: Taiwan Patent I453041

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 01, 2027
Generic Entry Controlled by: Taiwan Patent I439267
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: Taiwan Patent I538674
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2027
Generic Entry Controlled by: Taiwan Patent I421245
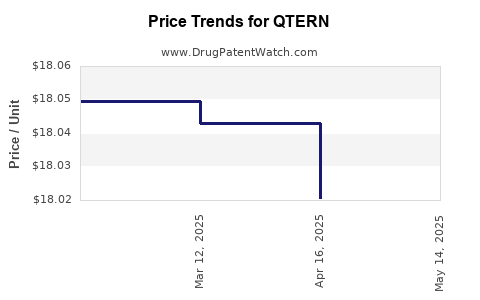
This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I466876

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent I519528

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 05, 2026
Generic Entry Controlled by: Taiwan Patent I417098
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 22, 2027
Generic Entry Controlled by: Taiwan Patent I457137

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: Taiwan Patent I443091
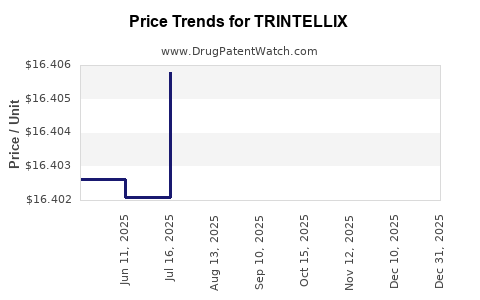
This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 22, 2027
Generic Entry Controlled by: Taiwan Patent I350185
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 15, 2027
Generic Entry Controlled by: Taiwan Patent I393700
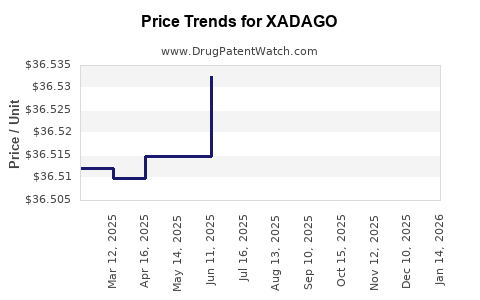
This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Taiwan Patent I412512

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can ZEMDRI (plazomicin sulfate) generic drug versions launch?
Generic name: plazomicin sulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 21, 2027
Generic Entry Controlled by: Taiwan Patent I425947
ZEMDRI is a drug marketed by Cipla Usa. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries. There has been litigation on patents covering ZEMDRI
See drug price trends for ZEMDRI.
The generic ingredient in ZEMDRI is plazomicin sulfate. Three suppliers are listed for this generic product. Additional details are available on the plazomicin sulfate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 20, 2027
Generic Entry Controlled by: Taiwan Patent I406657
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Taiwan Patent I501768
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 200,811,127

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,406,743

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,509,927

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,546,054

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 200,811,127

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,406,743

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,509,927

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,546,054

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Taiwan Patent 200,815,344
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Taiwan Patent 201,441,190
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Taiwan Patent 200,815,322

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Taiwan Patent 201,313,662

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 20, 2027
Generic Entry Controlled by: Taiwan Patent 200,904,402
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Taiwan Patent 200,843,795
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Taiwan Patent 201,422,233
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Taiwan Patent 200,843,795
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Taiwan Patent 201,422,233
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Taiwan Patent 200,918,550
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Taiwan Patent 201,534,684
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 29, 2026
Generic Entry Controlled by: Taiwan Patent 200,823,198
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 22, 2027
Generic Entry Controlled by: Taiwan Patent 200,902,087
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: Taiwan Patent 200,817,412

This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: Taiwan Patent 200,924,759
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 27, 2026
Generic Entry Controlled by: Taiwan Patent 200,808,379

See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 200,811,127

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,406,743

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,509,927

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Taiwan Patent 201,546,054

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: Taiwan Patent 200,914,030

This drug has four hundred and twenty-eight patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: Taiwan Patent 201,436,798

This drug has four hundred and twenty-eight patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 05, 2026
Generic Entry Controlled by: Taiwan Patent 200,838,527
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Taiwan Patent 200,906,409
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Taiwan Patent 200,927,136

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: Taiwan Patent 201,212,922

This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 22, 2027
Generic Entry Controlled by: Taiwan Patent 200,808,358

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: Taiwan Patent 200,817,340

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 05, 2027
Generic Entry Controlled by: Taiwan Patent 200,911,781
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Taiwan Patent 200,817,013
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 01, 2027
Generic Entry Controlled by: Taiwan Patent 200,843,742
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 01, 2027
Generic Entry Controlled by: Taiwan Patent 201,410,240
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can LYRICA CR (pregabalin) generic drug versions launch?
Generic name: pregabalin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 01, 2026
Generic Entry Controlled by: Taiwan Patent 200,803,831

This drug has thirty-seven patent family members in thirty-three countries.
See drug price trends for LYRICA CR.
The generic ingredient in LYRICA CR is pregabalin. There are forty-one drug master file entries for this API. Fifty-five suppliers are listed for this generic product. Additional details are available on the pregabalin profile page.
When can SIGNIFOR LAR (pasireotide pamoate) generic drug versions launch?
Generic name: pasireotide pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 24, 2027
Generic Entry Controlled by: Taiwan Patent 200,906,437
SIGNIFOR LAR is a drug marketed by Recordati Rare. There are three patents protecting this drug.
See drug price trends for SIGNIFOR LAR.
The generic ingredient in SIGNIFOR LAR is pasireotide pamoate. One supplier is listed for this generic product. Additional details are available on the pasireotide pamoate profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Taiwan Patent 200,938,210

This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Taiwan Patent 201,509,423