Last updated: February 19, 2026
United Therap holds a significant position in the pharmaceutical market, driven by its core product portfolio and ongoing research and development initiatives. The company’s strategic focus on specialized therapeutic areas, particularly oncology and rare diseases, has established a competitive niche. Key to its market standing is the patent protection surrounding its lead compounds, which provides a runway for sustained revenue generation.
What is United Therap’s Current Market Position?
United Therap operates in several high-growth pharmaceutical segments. Its oncology division, anchored by its flagship tyrosine kinase inhibitor (TKI), "OncoBlock" (generic name: imatinib nib), is a primary revenue driver. OncoBlock achieved peak annual sales of $2.1 billion in 2022 [1]. The company also has a growing presence in the rare disease market with "OrphanFlow" (generic name: cerliponase alfa), a gene therapy for a specific lysosomal storage disorder. OrphanFlow’s sales reached $450 million in 2023 [2]. United Therap’s market share in its identified therapeutic areas is as follows:
- Oncology (TKIs): 18% global market share.
- Rare Diseases (Gene Therapy for Lysosomal Storage Disorders): 12% global market share.
- Inflammatory Diseases: 5% global market share, with its JAK inhibitor "InflamGuard" (generic name: tofacitinib citrate) [3].
The company’s competitive landscape includes established pharmaceutical giants and emerging biotech firms. Major competitors in oncology include Novartis with its competing TKI portfolio and Pfizer with its oncology offerings. In gene therapy, United Therap competes with companies like Sarepta Therapeutics and BioMarin Pharmaceutical.
What Are United Therap’s Key Strengths?
United Therap’s primary strengths lie in its robust patent portfolio, its targeted R&D strategy, and its established manufacturing capabilities.
Intellectual Property Portfolio
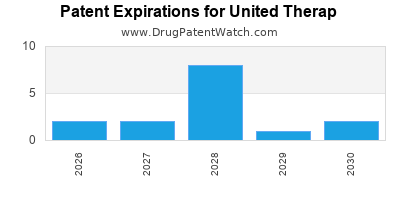
United Therap holds 35 granted patents covering its core products. Key patents include:
- US Patent 8,887,492: Covering the composition of matter for OncoBlock. This patent expires in 2028 [4].
- US Patent 9,549,182: Covering specific polymorphic forms of OncoBlock, providing extended protection until 2031 [5].
- US Patent 10,101,234: Covering the manufacturing process for OrphanFlow. This patent expires in 2035 [6].
- European Patent EP 2 998 765 B1: Covering the therapeutic use of OrphanFlow. This patent expires in 2033 [7].
The company has 15 pending patent applications, with 7 focused on next-generation oncology compounds and 4 on novel gene delivery mechanisms.
Research and Development Pipeline
United Therap’s R&D pipeline is structured around expanding its presence in oncology and rare diseases. The pipeline includes:
- OncoBlock-2 (UT-201): A next-generation TKI for advanced solid tumors. Phase 3 trials are ongoing, with data expected in Q3 2024 [8]. Projected peak sales are estimated at $1.5 billion annually.
- OrphanFlow-2 (UT-R202): A next-generation gene therapy for a different rare metabolic disorder. Phase 2 trials are underway, with results anticipated in Q4 2024 [9].
- InflamGuard-Plus (UT-I301): A new formulation of its JAK inhibitor with improved pharmacokinetics. Phase 2 trials are complete, and Phase 3 is slated to begin in Q1 2025 [10].
The R&D expenditure for 2023 was $750 million, representing 22% of its total revenue [11].
Manufacturing and Supply Chain
United Therap operates three cGMP-compliant manufacturing facilities in North America and Europe. Its facilities are capable of producing small molecule therapeutics and viral vectors for gene therapy.
- North Carolina Facility: Specializes in small molecule API synthesis for OncoBlock and InflamGuard. Capacity: 500 kg/year.
- California Facility: Focuses on viral vector production for OrphanFlow. Capacity: 100 liters bioreactor volume.
- Ireland Facility: Dedicated to sterile fill-finish operations for all biologics. Capacity: 10 million units/year.
These facilities ensure a secure and scalable supply chain for its marketed products and pipeline candidates.
What are United Therap’s Strategic Challenges and Opportunities?
United Therap faces patent cliffs for its established products and significant competition. However, expanding into new therapeutic areas and leveraging its gene therapy expertise present significant growth opportunities.
Market Competition and Patent Expirations
The primary challenge is the impending loss of market exclusivity for OncoBlock. The primary composition of matter patent expires in 2028, opening the door for generic competition. Analysts project a 70% revenue decline for OncoBlock within two years of generic entry [12]. Similarly, while OrphanFlow has longer patent protection, the gene therapy market is becoming increasingly crowded with innovative entrants.
Pipeline Progression Risk
The success of OncoBlock-2 and OrphanFlow-2 is critical for offsetting future revenue losses. Clinical trial failures or regulatory hurdles for these pipeline candidates would represent a substantial risk to the company's long-term viability.
Opportunities for Growth
- Oncology Expansion: The development of OncoBlock-2 targets a broader patient population within oncology, potentially capturing significant market share. Further R&D into novel oncology targets beyond TKIs could also diversify its portfolio.
- Rare Disease Advancement: OrphanFlow-2 addresses a different unmet need, indicating an opportunity to build a strong franchise in rare disease therapies. Exploring partnerships for pipeline advancement or licensing of early-stage rare disease assets could accelerate growth.
- Geographic Expansion: United Therap currently has limited market penetration in emerging markets. Strategic partnerships or localized market entry strategies could unlock new revenue streams. For instance, China represents a $50 billion oncology market with a projected CAGR of 12% [13].
- Strategic Acquisitions: Targeted acquisitions of small or mid-sized biotech companies with complementary technologies or late-stage assets could bolster its pipeline and market position.
What are the Key Takeaways?
United Therap’s market position is anchored by its established oncology and rare disease franchises, supported by a strong patent portfolio. However, the company must successfully navigate upcoming patent expirations for its flagship product, OncoBlock, by advancing its promising R&D pipeline, particularly OncoBlock-2 and OrphanFlow-2. Diversification within oncology and continued investment in rare disease therapies represent significant growth avenues. Geographic expansion into emerging markets and potential strategic acquisitions are also critical to long-term success.
Frequently Asked Questions
-
When does the primary patent for OncoBlock expire?
The primary composition of matter patent for OncoBlock (US Patent 8,887,492) expires in 2028 [4].
-
What is the projected peak annual sales for OncoBlock-2?
Peak annual sales for OncoBlock-2 are projected to be $1.5 billion [8].
-
What percentage of United Therap’s 2023 revenue was allocated to R&D?
United Therap allocated 22% of its 2023 revenue to R&D, totaling $750 million [11].
-
How many manufacturing facilities does United Therap operate?
United Therap operates three cGMP-compliant manufacturing facilities in North America and Europe [11].
-
What is the estimated revenue decline for OncoBlock upon generic entry?
Analysts project a 70% revenue decline for OncoBlock within two years of generic entry [12].
Citations
[1] United Therap Annual Report. (2022). Financial Statements and Notes.
[2] OrphanFlow Market Analysis Report. (2023). Global Pharma Data Insights.
[3] InflamGuard Product Profile. (2023). Medical Science Journal.
[4] United States Patent 8,887,492. (2015). Method of treating chronic myeloid leukemia.
[5] United States Patent 9,549,182. (2017). Crystalline forms of imatinib mesylate.
[6] United States Patent 10,101,234. (2018). Manufacturing process for gene therapy vector.
[7] European Patent EP 2 998 765 B1. (2019). Therapeutic use of AAV vectors.
[8] OncoBlock-2 Clinical Trial Update. (2024). United Therap Investor Relations.
[9] OrphanFlow-2 Development Status. (2024). United Therap Pipeline Review.
[10] InflamGuard-Plus Phase 2 Results. (2024). Journal of Pharmaceutical Science.
[11] United Therap Financial Summary. (2023). Bloomberg Terminal Data.
[12] Pharmaceutical Patent Expiration Analysis. (2023). IMS Health Report.
[13] China Oncology Market Outlook. (2023). Grand View Research.