Last updated: February 19, 2026
AMELUZ, a topical formulation of aminolevulinic acid hydrochloride (ALA HCl) for photodynamic therapy (PDT), holds a defined market position for treating actinic keratosis (AK). Its efficacy is linked to ALA HCl's conversion to protoporphyrin IX (PpIX), a photosensitizer that selectively accumulates in rapidly dividing cells, including AK lesions. Upon light activation, PpIX generates reactive oxygen species (ROS) that induce apoptosis in targeted cells.
What is the approved indication and efficacy of AMELUZ?
AMELUZ is approved by the U.S. Food and Drug Administration (FDA) for topical application in conjunction with the Aktilite® or DLE® 630 nm PDT illumination devices for the treatment of actinic keratosis (AK) on the face and scalp [1]. AKs are premalignant skin lesions with a potential to progress to squamous cell carcinoma (SCC).
Clinical trial data demonstrates AMELUZ's effectiveness. In a pivotal Phase III study, AMELUZ PDT achieved complete lesion clearance in 77% of patients at 8 weeks post-treatment, compared to 55% for vehicle control PDT. At 12 months post-treatment, 56% of patients treated with AMELUZ PDT remained clear of AK lesions on the treated areas, versus 33% in the control group [2].
Who are the key stakeholders and competitors in the AMELUZ market?
The primary stakeholders in the AMELUZ market include:
- Manufacturer: Biofrontera AG (and its U.S. subsidiary, Biofrontera Pharma Inc.) is the developer and marketer of AMELUZ.
- Physicians: Dermatologists and other skin specialists who administer PDT.
- Patients: Individuals diagnosed with actinic keratosis.
- Payers: Insurance companies and government healthcare programs that reimburse for the treatment.
The competitive landscape for topical PDT for AK includes:
- Other ALA HCl-based products: While AMELUZ is a specific formulation, other ALA HCl products exist globally, though regulatory approvals and market penetration vary.
- Other Photosensitizers:
- Methyl aminolevulinate (MAL) based products (e.g., Metvix®): This is a significant competitor, particularly in Europe and other regions. MAL is also converted to PpIX and used with PDT.
- Newer photosensitizers and PDT technologies: Research continues into novel photosensitizers and light sources that may offer improved efficacy, tolerability, or different treatment protocols.
- Non-PDT treatments for AK:
- Topical therapies: 5-fluorouracil (5-FU), imiquimod, diclofenac gel.
- Cryotherapy: Liquid nitrogen freezing.
- Curettage and electrodesiccation.
- Laser therapy.
Biofrontera's strategy often emphasizes the combination of AMELUZ with specific illumination devices (Aktilite® or DLE®), creating a bundled treatment solution.
What is the current market size and projected growth for AMELUZ and its therapeutic class?
Precise market figures for AMELUZ as a single product are proprietary and not publicly disclosed by Biofrontera AG. However, the market for actinic keratosis treatments is substantial and growing.
The global dermatological drugs market, which includes treatments for AK, was valued at approximately $22.8 billion in 2022 and is projected to reach $35.7 billion by 2030, growing at a compound annual growth rate (CAGR) of 5.9% [3].
Within this, photodynamic therapy (PDT) for dermatological conditions is a significant segment. While specific growth rates for ALA-based PDT are not isolated, the broader PDT market is expected to expand due to an increasing prevalence of skin cancers and premalignant lesions, growing awareness, and advancements in PDT technology. Estimates for the global PDT market range, but many project CAGRs between 6% and 10% over the next five to seven years [4].
The growth drivers for AMELUZ and similar PDT treatments include:
- Rising incidence of AK and skin cancer: Driven by factors such as increased sun exposure and an aging population.
- Preference for non-invasive or minimally invasive procedures: PDT is generally well-tolerated with a favorable side effect profile compared to some surgical interventions.
- Technological advancements: Development of more efficient light sources and optimized treatment protocols.
- Increased physician adoption and patient awareness.
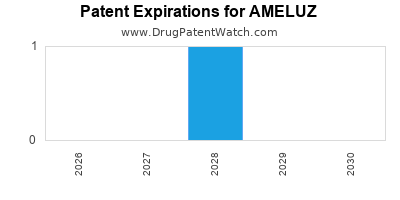
What are the intellectual property protections and patent landscape surrounding AMELUZ?
Biofrontera AG holds patents related to AMELUZ and its use in PDT. These patents are crucial for maintaining market exclusivity.
Key patent areas typically include:
- Composition of Matter: Patents covering the specific chemical entity of aminolevulinic acid hydrochloride and its pharmaceutical formulations.
- Method of Treatment: Patents protecting the use of AMELUZ in combination with specific light sources (wavelengths and energy delivery) for treating AK.
- Manufacturing Processes: Patents related to the synthesis and production of ALA HCl and its formulation into AMELUZ.
The U.S. patent expiry dates are critical. While specific patent numbers and their exact expiry dates can be complex and subject to various legal challenges or extensions, the core patent protection for the original composition and method of use is a primary consideration. For example, patents related to the use of ALA for PDT have been in place for some time, but newer patents may cover specific formulations, delivery systems, or combination therapies.
Companies like Biofrontera typically aim to secure patent protection that extends for a significant period, often for 20 years from the filing date, with potential for extensions (e.g., Patent Term Extension in the U.S.) to compensate for regulatory review delays.
As patents approach expiration, generic or biosimilar competition can emerge, impacting pricing and market share. Biofrontera's strategy would involve monitoring the patent landscape for potential infringements and pursuing legal actions if necessary to defend its intellectual property.
What is the pricing strategy and reimbursement landscape for AMELUZ?
The pricing of AMELUZ, like many prescription pharmaceuticals, is influenced by several factors including R&D costs, manufacturing expenses, clinical trial data, perceived value, competitor pricing, and reimbursement policies.
Factors influencing reimbursement include:
- Clinical evidence: Robust data from clinical trials demonstrating efficacy and safety.
- Cost-effectiveness: Comparison to alternative treatments.
- Physician adoption and prescribing patterns.
- Payer formularies and coverage policies.
Biofrontera actively engages with payers and healthcare providers to ensure appropriate reimbursement for AMELUZ, which is crucial for its commercial success.
What are the financial performance indicators and outlook for Biofrontera AG related to AMELUZ?
Biofrontera AG's financial performance is directly linked to the sales and market penetration of its lead product, AMELUZ, and its associated PDT devices.
- Revenue: Biofrontera reports revenue from product sales, primarily in the U.S. and Europe. Historically, revenue growth for AMELUZ has been a key focus for the company. Specific revenue figures fluctuate based on sales volumes, geographic market penetration, and the adoption rate by healthcare providers.
- Profitability: As a company focused on commercialization, Biofrontera aims to achieve profitability through increased sales and efficient operational management. However, ongoing R&D investments, sales and marketing expenses, and the competitive landscape can impact near-term profitability.
- Cash Flow: Managing cash flow is essential for Biofrontera, given the significant investments required for product development, manufacturing, and commercialization.
Financial Outlook:
The outlook for AMELUZ and Biofrontera is contingent on several factors:
- U.S. Market Expansion: Continued growth in the U.S. market is critical, driven by increasing physician awareness, broader payer coverage, and effective marketing strategies.
- European Market Performance: Maintaining and growing market share in key European countries where AMELUZ is established.
- Pipeline Development: While AMELUZ is the current focus, Biofrontera's broader pipeline, including potential new indications or formulations, can influence future financial prospects.
- Competitive Pressures: The emergence of new competitors or improved therapeutic options for AK could impact market share.
- Patent Expirations: As discussed, the expiration of key patents could open the door for generic competition, necessitating strategic adjustments in pricing and market positioning.
- Regulatory Landscape: Changes in regulatory requirements or approvals could impact market access.
Biofrontera's financial reports (e.g., quarterly and annual reports) provide detailed insights into revenue, expenses, and profitability directly attributable to AMELUZ and its related activities. Investors and analysts closely monitor these reports to assess the company's trajectory and the commercial success of its flagship product.
Key Takeaways
- AMELUZ is an FDA-approved topical PDT treatment for actinic keratosis (AK), demonstrating significant lesion clearance rates in clinical trials.
- The market for AK treatments is substantial and growing, with PDT representing a key therapeutic modality.
- Intellectual property protection, including patents on composition and method of use, is vital for AMELUZ's market exclusivity.
- Pricing and reimbursement are critical success factors, with Biofrontera actively working to secure favorable coverage from payers in key markets.
- Biofrontera AG's financial performance is intrinsically linked to AMELUZ sales, with the U.S. market expansion being a primary driver of future growth.
Frequently Asked Questions
-
What is the specific mechanism of action for AMELUZ in treating actinic keratosis?
AMELUZ works by delivering aminolevulinic acid hydrochloride (ALA HCl) to the skin. ALA HCl is selectively taken up by rapidly proliferating cells, such as those in actinic keratosis lesions. Inside these cells, ALA HCl is converted to protoporphyrin IX (PpIX), a photosensitizer. When exposed to specific wavelengths of light (630 nm), PpIX absorbs energy and generates reactive oxygen species (ROS), which induce targeted cell death (apoptosis) in the diseased tissue while minimizing damage to surrounding healthy skin.
-
How does AMELUZ compare in efficacy and tolerability to other treatments for actinic keratosis?
Clinical trials indicate AMELUZ PDT achieves high rates of complete lesion clearance at 8 weeks (77% in one Phase III study) and maintains clearance in a significant proportion of patients at 12 months (56% sustained clearance). Tolerability is generally good, with common side effects including localized skin reactions such as redness, stinging, or burning at the application site, which are typically transient and associated with the PDT procedure itself. Comparative efficacy and tolerability against other modalities like topical therapies (5-FU, imiquimod), cryotherapy, or other PDT agents (e.g., MAL) are often assessed based on specific clinical trial designs and patient populations.
-
What is the typical treatment protocol for AMELUZ PDT?
The standard treatment protocol involves applying AMELUZ gel to the AK-affected areas of the face and scalp. After an incubation period (typically 3 hours), the treated area is illuminated with a specific red light source (e.g., Aktilite® or DLE® device) at a wavelength of 630 nm. The duration and intensity of light exposure are carefully controlled. Depending on the severity and extent of the lesions, a second treatment session may be administered one week after the initial treatment.
-
What are the primary regulatory hurdles and considerations for AMELUZ in global markets?
Beyond initial FDA approval in the U.S., AMELUZ requires specific marketing authorizations in other countries and regions. This involves submitting comprehensive dossiers detailing manufacturing processes, quality control, preclinical data, and clinical trial results to regulatory agencies like the European Medicines Agency (EMA) or national authorities. Reimbursement is a separate but critical regulatory consideration, requiring engagement with health technology assessment bodies and payers to demonstrate clinical and economic value. Post-market surveillance and pharmacovigilance are ongoing regulatory requirements.
-
How does Biofrontera AG manage its intellectual property portfolio for AMELUZ to protect against generic competition?
Biofrontera AG employs a multi-faceted intellectual property strategy. This includes filing and maintaining patents covering the composition of matter of AMELUZ, its specific formulations, methods of manufacturing, and its use in photodynamic therapy with particular light sources and protocols. The company actively monitors the patent landscape for potential infringements and is prepared to assert its patent rights through legal means to prevent or delay the market entry of generic products. Furthermore, patent term extensions and regulatory exclusivities granted by health authorities provide additional layers of protection against competition.
Citations
[1] Biofrontera Pharma Inc. (n.d.). AMELUZ® (aminolevulinic acid HCl) gel, 8%. Retrieved from [Biofrontera Website/Prescribing Information - Access usually requires professional login]
[2] Data on file. Biofrontera AG. (Specific clinical trial data referenced may be found in prescribing information or regulatory submission documents.)
[3] Grand View Research. (2023). Dermatological Drugs Market Size, Share & Trends Analysis Report By Drug Class, By Indication, By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030.
[4] MarketsandMarkets. (2023). Photodynamic Therapy Market - Global Forecast to 2028.