VAFSEO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Vafseo, and when can generic versions of Vafseo launch?
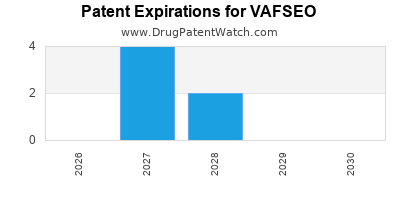
Vafseo is a drug marketed by Akebia and is included in one NDA. There are fourteen patents protecting this drug.
This drug has two hundred and sixty-four patent family members in forty-eight countries.
The generic ingredient in VAFSEO is vadadustat. One supplier is listed for this compound. Additional details are available on the vadadustat profile page.
DrugPatentWatch® Generic Entry Outlook for Vafseo
Vafseo will be eligible for patent challenges on March 27, 2028. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be March 31, 2036. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VAFSEO?
- What are the global sales for VAFSEO?
- What is Average Wholesale Price for VAFSEO?
Summary for VAFSEO
| International Patents: | 264 |
| US Patents: | 14 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 1 |
| Patent Applications: | 247 |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VAFSEO |
| What excipients (inactive ingredients) are in VAFSEO? | VAFSEO excipients list |
| DailyMed Link: | VAFSEO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VAFSEO
Generic Entry Date for VAFSEO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VAFSEO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| USRC Kidney Research | PHASE3 |
| Akebia Therapeutics | PHASE3 |
Pharmacology for VAFSEO
US Patents and Regulatory Information for VAFSEO
VAFSEO is protected by fourteen US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VAFSEO is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-003 | Mar 27, 2024 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-003 | Mar 27, 2024 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-003 | Mar 27, 2024 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-002 | Mar 27, 2024 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-003 | Mar 27, 2024 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-002 | Mar 27, 2024 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Akebia | VAFSEO | vadadustat | TABLET;ORAL | 215192-001 | Mar 27, 2024 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VAFSEO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Akebia Europe Limited | Vafseo | vadadustat | EMEA/H/C/005131Vafseo is indicated for the treatment of symptomatic anaemia associated with chronic kidney disease (CKD) in adults on chronic maintenance dialysis. | Authorised | no | no | no | 2023-04-24 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VAFSEO
When does loss-of-exclusivity occur for VAFSEO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 4157
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 16243700
Estimated Expiration: ⤷ Start Trial
Patent: 20273282
Estimated Expiration: ⤷ Start Trial
Patent: 23200252
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2017021097
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 81176
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 17002456
Estimated Expiration: ⤷ Start Trial
Patent: 19001927
Estimated Expiration: ⤷ Start Trial
China
Patent: 7645953
Estimated Expiration: ⤷ Start Trial
Patent: 5607521
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 17011200
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 170450
Estimated Expiration: ⤷ Start Trial
Patent: 210248
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0211862
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 170126
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 24869
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 77270
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 17073209
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 6920
Estimated Expiration: ⤷ Start Trial
Patent: 1792182
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 77270
Estimated Expiration: ⤷ Start Trial
Patent: 95138
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 48590
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 56958
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 4710
Estimated Expiration: ⤷ Start Trial
Patent: 2262
Estimated Expiration: ⤷ Start Trial
Patent: 5910
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 29785
Estimated Expiration: ⤷ Start Trial
Patent: 70688
Estimated Expiration: ⤷ Start Trial
Patent: 29049
Estimated Expiration: ⤷ Start Trial
Patent: 18510178
Estimated Expiration: ⤷ Start Trial
Patent: 21181478
Estimated Expiration: ⤷ Start Trial
Patent: 23093684
Estimated Expiration: ⤷ Start Trial
Patent: 25069280
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 77270
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 2705
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 4909
Estimated Expiration: ⤷ Start Trial
Patent: 6796
Estimated Expiration: ⤷ Start Trial
Patent: 17012460
Estimated Expiration: ⤷ Start Trial
Patent: 20009415
Estimated Expiration: ⤷ Start Trial
Patent: 21012155
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 863
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 5973
Estimated Expiration: ⤷ Start Trial
Patent: 3901
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 180189
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 017501789
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 77270
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 77270
Estimated Expiration: ⤷ Start Trial
Saudi Arabia
Patent: 7390054
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201707994U
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 77270
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1706681
Estimated Expiration: ⤷ Start Trial
Patent: 2006107
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2647879
Estimated Expiration: ⤷ Start Trial
Patent: 2866021
Estimated Expiration: ⤷ Start Trial
Patent: 170132865
Estimated Expiration: ⤷ Start Trial
Patent: 240038132
Estimated Expiration: ⤷ Start Trial
Patent: 250150155
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 00481
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1642839
Estimated Expiration: ⤷ Start Trial
Patent: 2140001
Estimated Expiration: ⤷ Start Trial
Patent: 2320770
Estimated Expiration: ⤷ Start Trial
Patent: 2421140
Estimated Expiration: ⤷ Start Trial
Patent: 18138
Estimated Expiration: ⤷ Start Trial
Patent: 94725
Estimated Expiration: ⤷ Start Trial
Patent: 36883
Estimated Expiration: ⤷ Start Trial
Patent: 81714
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3308
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VAFSEO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 2327696 | Inhibiteurs de la prolyl hydroxylase et procédés d'utilisation (Prolyl hydroxylase inhibitors and methods of use) | ⤷ Start Trial |
| Costa Rica | 20150660 | ⤷ Start Trial | |
| South Korea | 20220151008 | ⤷ Start Trial | |
| Taiwan | 201605801 | Solid forms of {[5-(3-chlorophenyl)-3-hydroxypyridine-2-carbonyl]amino}acetic acid, compositions, and uses thereof | ⤷ Start Trial |
| Colombia | 6170355 | ⤷ Start Trial | |
| European Patent Office | 3995138 | ⤷ Start Trial | |
| Canada | 2981176 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VAFSEO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3007695 | 122024000039 | Germany | ⤷ Start Trial | PRODUCT NAME: VADADUSTAT (VAFSEO); REGISTRATION NO/DATE: EU/1/23/1725 20230424 |
| 3007695 | C202430024 | Spain | ⤷ Start Trial | PRODUCT NAME: VADADUSTAT; NATIONAL AUTHORISATION NUMBER: EU/1/23/1725; DATE OF AUTHORISATION: 20230424; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/23/1725; DATE OF FIRST AUTHORISATION IN EEA: 20230424 |
| 3007695 | 202440023 | Slovenia | ⤷ Start Trial | PRODUCT NAME: VADADUSTAT; NATIONAL AUTHORISATION NUMBER: EU/1/23/1725; DATE OF NATIONAL AUTHORISATION: 20230424; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| 3007695 | 24C1025 | France | ⤷ Start Trial | PRODUCT NAME: VADADUSTAT; NAT. REGISTRATION NO/DATE: : EU/1/23/1725 20230425; FIRST REGISTRATION: EU/1/23/1725 20230425 |
| 3007695 | 301278 | Netherlands | ⤷ Start Trial | PRODUCT NAME: VADADUSTAT; REGISTRATION NO/DATE: EU/1/23/1725 20230425 |
| 3007695 | C20240023 | Finland | ⤷ Start Trial | PRODUCT NAME: LUTEETSIUM(177LU)VIPIVOTIIDTETRAKSETAAN;REG NO/DATE: EU/1/22/1703 12.12.2022 |
| 3007695 | PA2024521 | Lithuania | ⤷ Start Trial | PRODUCT NAME: VADADUSTATAS; REGISTRATION NO/DATE: EU/1/23/1725 20230424 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
VAFSEO Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.