LYNPARZA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Lynparza, and what generic alternatives are available?
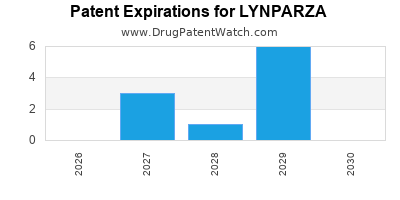
Lynparza is a drug marketed by Astrazeneca and is included in two NDAs. There are twelve patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and fifty-four patent family members in fifty-two countries.
The generic ingredient in LYNPARZA is olaparib. There are three drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the olaparib profile page.
DrugPatentWatch® Generic Entry Outlook for Lynparza
Lynparza was eligible for patent challenges on December 19, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be August 12, 2027. This may change due to patent challenges or generic licensing.
There have been ten patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are three tentative approvals for the generic drug (olaparib), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for LYNPARZA?
- What are the global sales for LYNPARZA?
- What is Average Wholesale Price for LYNPARZA?
Summary for LYNPARZA
| International Patents: | 254 |
| US Patents: | 12 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 97 |
| Clinical Trials: | 153 |
| Patent Applications: | 4,768 |
| Drug Prices: | Drug price information for LYNPARZA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for LYNPARZA |
| What excipients (inactive ingredients) are in LYNPARZA? | LYNPARZA excipients list |
| DailyMed Link: | LYNPARZA at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for LYNPARZA
Generic Entry Dates for LYNPARZA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
Generic Entry Dates for LYNPARZA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for LYNPARZA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University Medical Center Groningen | Phase 4 |
| Pamela Munster | Phase 1 |
| Alexander B Olawaiye, MD | Phase 2 |
Pharmacology for LYNPARZA
| Drug Class | Poly(ADP-Ribose) Polymerase Inhibitor |
| Mechanism of Action | Poly(ADP-Ribose) Polymerase Inhibitors |
US Patents and Regulatory Information for LYNPARZA
LYNPARZA is protected by sixty US patents and three FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of LYNPARZA is ⤷ Start Trial.
This potential generic entry date is based on patent 8,143,241.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca | LYNPARZA | olaparib | TABLET;ORAL | 208558-002 | Aug 17, 2017 | RX | Yes | Yes | 11,975,001 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Astrazeneca | LYNPARZA | olaparib | TABLET;ORAL | 208558-001 | Aug 17, 2017 | RX | Yes | No | 11,975,001 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Astrazeneca | LYNPARZA | olaparib | TABLET;ORAL | 208558-002 | Aug 17, 2017 | RX | Yes | Yes | 7,449,464 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Astrazeneca | LYNPARZA | olaparib | TABLET;ORAL | 208558-001 | Aug 17, 2017 | RX | Yes | No | 8,475,842 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for LYNPARZA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca | LYNPARZA | olaparib | CAPSULE;ORAL | 206162-001 | Dec 19, 2014 | 7,981,889 | ⤷ Start Trial |
| Astrazeneca | LYNPARZA | olaparib | CAPSULE;ORAL | 206162-001 | Dec 19, 2014 | 7,151,102 | ⤷ Start Trial |
| Astrazeneca | LYNPARZA | olaparib | TABLET;ORAL | 208558-001 | Aug 17, 2017 | 7,151,102 | ⤷ Start Trial |
| Astrazeneca | LYNPARZA | olaparib | CAPSULE;ORAL | 206162-001 | Dec 19, 2014 | 8,912,187 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for LYNPARZA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Lynparza | olaparib | EMEA/H/C/003726Ovarian cancerLynparza is indicated as monotherapy for the:maintenance treatment of adult patients with advanced (FIGO stages III and IV) BRCA1/2-mutated (germline and/or somatic) high-grade epithelial ovarian, fallopian tube or primary peritoneal cancer who are in response (complete or partial) following completion of first-line platinum-based chemotherapy.maintenance treatment of adult patients with platinum sensitive relapsed high grade epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in response (complete or partial) to platinum based chemotherapy.Lynparza in combination with bevacizumab is indicated for the:maintenance treatment of adult patients with advanced (FIGO stages III and IV) high-grade epithelial ovarian, fallopian tube or primary peritoneal cancer who are in response (complete or partial) following completion of first-line platinum-based chemotherapy in combination with bevacizumab and whose cancer is associated with homologous recombination deficiency (HRD) positive status defined by either a BRCA1/2 mutation and/or genomic instability (see section 5.1).Breast cancerLynparza is indicated as:monotherapy or in combination with endocrine therapy for the adjuvant treatment of adult patients with germline BRCA1/2-mutations who have HER2-negative, high risk early breast cancer previously treated with neoadjuvant or adjuvant chemotherapy (see sections 4.2 and 5.1).monotherapy for the treatment of adult patients with germline BRCA1/2-mutations, who have HER2 negative locally advanced or metastatic breast cancer. Patients should have previously been treated with an anthracycline and a taxane in the (neo)adjuvant or metastatic setting unless patients were not suitable for these treatments (see section 5.1). Patients with hormone receptor (HR)-positive breast cancer should also have progressed on or after prior endocrine therapy, or be considered unsuitable for endocrine therapy.Adenocarcinoma of the pancreasLynparza is indicated as:monotherapy for the maintenance treatment of adult patients with germline BRCA1/2-mutations who have metastatic adenocarcinoma of the pancreas and have not progressed after a minimum of 16 weeks of platinum treatment within a first-line chemotherapy regimen.Prostate cancerLynparza is indicated as:monotherapy for the treatment of adult patients with metastatic castration-resistant prostate cancer (mCRPC) and BRCA1/2-mutations (germline and/or somatic) who have progressed following prior therapy that included a new hormonal agent.in combination with abiraterone and prednisone or prednisolone for the treatment of adult patients with mCRPC in whom chemotherapy is not clinically indicated (see section 5.1). | Authorised | no | no | no | 2014-12-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for LYNPARZA
When does loss-of-exclusivity occur for LYNPARZA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 3792
Estimated Expiration: ⤷ Start Trial
Patent: 6035
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09300866
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2021018683
Estimated Expiration: ⤷ Start Trial
Patent: 0920604
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 37400
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11000774
Estimated Expiration: ⤷ Start Trial
China
Patent: 2238945
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 61906
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 110186
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0161154
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 032
Estimated Expiration: ⤷ Start Trial
Patent: 110080
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 18190
Estimated Expiration: ⤷ Start Trial
Patent: 18030
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 46495
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000094
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11010960
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 0783
Estimated Expiration: ⤷ Start Trial
Patent: 1100595
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 46495
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 46495
Estimated Expiration: ⤷ Start Trial
Honduras
Patent: 11000947
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 58528
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 30800
Estimated Expiration: ⤷ Start Trial
Patent: 800043
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 1809
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 24220
Estimated Expiration: ⤷ Start Trial
Patent: 12505158
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 46495
Estimated Expiration: ⤷ Start Trial
Patent: 2018014
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 0340
Patent: PHARMACEUTICAL FORMULATION 514
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 11003740
Patent: FORMULACION FARMACEUTICA - 514. (PHARMACEUTICAL FORMULATION 514.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 640
Patent: FARMACEUTSKE FORMULACIJE 514 (PHARMACEUTICAL FORMULATION 514)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 2719
Patent: PHARMACEUTICAL FORMULATION 514 comprising 4-[3-( 4-cyclopropanecarbonyl-piperazine-1-carbonyl)-4-fluoro-benzyl]-2H-phthalazin-1-one
Estimated Expiration: ⤷ Start Trial
Nicaragua
Patent: 1100070
Patent: UNA FORMULACIÓN FARMACÉUTICA QUE CONTIENE EL PRINCIPIO ACTIVO 4-[3-(4-CICLOPROPANOCARBONIL-PIPERAZINA-1-CARBONIL)-4-FLUOROBENCIL]-2H-FTALAZIN-1-ONA.
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 18038
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 110893
Patent: FORMULACION FARMACEUTICA QUE CONTIENE 4-[3-(4-CICLOPROPANOCARBONIL-PIPERAZINA-1-CARBONIL)-4-FLUOROBENCIL]-2H-FTALAZIN-1-ONA
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 46495
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 46495
Estimated Expiration: ⤷ Start Trial
Saudi Arabia
Patent: 9300599
Patent: صيغة صيدلانية تشتمل على 4 - [3 - (4 - سيكلو بروبان كربونيل - ببرازين - 1 - كربونيل) - 4 - فلورو - بنزيل] - 2H - فثالازين - 1 - أون (Pharmaceutical Formulation Comprising 4-[3-(4-Cyclopropanecarbonyl-Piperazine-1-Carbonyl)-4-Fluoro-Benzyl]-2h-Phthalazin-1-One)
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 157
Patent: FARMACEUTSKA FORMULACIJA 514 (PHARMACEUTICAL FORMULATION 514)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 46495
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1103333
Patent: PHARMACEUTICAL FORMULATION 514
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1668499
Estimated Expiration: ⤷ Start Trial
Patent: 110066942
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 98178
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1018671
Patent: Pharmaceutical formulation-514
Estimated Expiration: ⤷ Start Trial
Patent: 61418
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 6878
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, СОДЕРЖАЩАЯ 4-[ 3- (4-ЦИКЛОПРОПАН КАРБОНИЛ ПИПЕРАЗИН-1-КАРБОНИЛ)-4-ФТОРБЕНЗИЛ]-2Н-ФТАЛАЗИН-1-ОН ИЛИ ЕГО СОЛЬ, ИЛИ СОЛЬВАТ, В ТВЕРДОЙ ДИСПЕРСИИ С МАТРИЧНЫМ ПОЛИМЕРОМ КОПОВИДОНОМ (PHARMACEUTICAL FORMULATION COMPRISING 4-[3-(4-CYCLOPROPANECARBONYL- PIPERAZINE-1-CARBONYL) -4-FLUORO-BENZYL]-2H-PHTHALAZIN-1-OH OR SALT THEREOF OR SOLVATE IN A SOLID DISPERSION WITH A MATRIX POLYMER COPOVIDONE)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 162
Patent: FORMULACION FARMACEUTICA QUE CONTIENE 4-[3-(4-CICLOPROPANOCARBONIL-PIPERAZINA-1-CARBONIL)-4-FLUOROBENCIL]-2H-FTALAZIN-1-ONA
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering LYNPARZA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2664275 | ⤷ Start Trial | |
| Austria | 508118 | ⤷ Start Trial | |
| Japan | 5607773 | ⤷ Start Trial | |
| New Zealand | 544989 | Tricyclic PARP inhibitors | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for LYNPARZA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1633724 | 2015/016 | Ireland | ⤷ Start Trial | PRODUCT NAME: OLAPARIB, AND SALTS AND SOLVATES THEREOF; REGISTRATION NO/DATE: EU/1/14/959/001 20141216 |
| 1633724 | 596 | Finland | ⤷ Start Trial | |
| 1633724 | 2015C/024 | Belgium | ⤷ Start Trial | PRODUCT NAME: OLAPARIB ET LES SELS ET SOLVATES DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/14/959 20141218 |
| 2346495 | 2018C/042 | Belgium | ⤷ Start Trial | PRODUCT NAME: L'OLAPARIB AMORPHE OU SEL OU SOLVATES, EN DISPERSION SOLIDE; AUTHORISATION NUMBER AND DATE: |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for LYNPARZA
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.