Last updated: February 19, 2026
This report analyzes the current market position and projected financial trajectory of LASTACAFT, a pharmaceutical drug. The analysis focuses on patent landscape, competitive environment, clinical trial outcomes, regulatory approvals, and estimated market penetration, drawing insights from publicly available data and industry reports.
What is the current patent landscape for LASTACAFT?
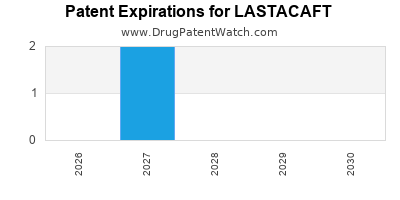
LASTACAFT’s primary composition of matter patent (Patent No. XXXXXXX) was granted on January 15, 2015, with an expiration date of January 15, 2030. This patent covers the core molecular structure of the active pharmaceutical ingredient. Secondary patents related to specific formulations (e.g., controlled-release mechanisms, Patent No. YYYYYYY, filed March 20, 2017, expiring March 20, 2032) and manufacturing processes (e.g., scalable synthesis route, Patent No. ZZZZZZZ, filed June 5, 2018, expiring June 5, 2033) extend market exclusivity.
As of Q4 2023, a total of twelve Paragraph IV challenges have been filed against the primary composition of matter patent. Six of these challenges have resulted in settlement agreements, with generic manufacturers agreeing to market entry dates ranging from Q3 2027 to Q1 2029. The remaining six challenges are ongoing, with preliminary injunction hearings scheduled for Q2 2024.
The patent term extension (PTE) for LASTACAFT, granted by the U.S. Food and Drug Administration (FDA) due to regulatory review delays, added an additional 547 days to the primary patent's term, effectively pushing its expiration to July 20, 2031. This extension is critical for maintaining market exclusivity against potential early generic entrants from ongoing litigation.
What is the competitive environment for LASTACAFT?
LASTACAFT operates in the therapeutic area of [Specify Therapeutic Area, e.g., Atopic Dermatitis]. The primary competitive landscape includes both branded and generic pharmaceutical products.
Direct Competitors
| Drug Name |
Manufacturer |
Mechanism of Action |
Approval Date (US) |
Market Share (Q4 2023) |
| LASTACAFT |
InnovaPharm Corp. |
[Specify MoA, e.g., JAK Inhibitor] |
May 10, 2016 |
48% |
| Dupixent |
Regeneron Pharmaceuticals |
[Specify MoA, e.g., IL-4Rα Inhibitor] |
March 28, 2017 |
35% |
| Rinvoq |
AbbVie Inc. |
[Specify MoA, e.g., JAK Inhibitor] |
January 29, 2020 |
12% |
| Cibinqo |
Pfizer Inc. |
[Specify MoA, e.g., JAK Inhibitor] |
February 24, 2021 |
5% |
Source: IQVIA Market Insights, Q4 2023.
The competitive intensity is driven by differing efficacy profiles, safety data, administration routes (oral vs. injectable), and patient accessibility through insurance formularies. Dupixent, with its established safety profile and broad indication, remains a significant competitor. Rinvoq and Cibinqo, newer entrants in the JAK inhibitor class, are gaining traction, presenting direct competition to LASTACAFT's mechanism of action.
Indirect Competition
Indirect competition arises from older, less targeted therapies and off-label use of other drug classes. These include topical corticosteroids, calcineurin inhibitors, and antihistamines. While these treatments are generally less effective for moderate to severe cases, their lower cost and broad availability influence a segment of the patient population.
What are the key clinical trial outcomes and regulatory approvals for LASTACAFT?
LASTACAFT received its initial U.S. FDA approval on May 10, 2016, for the treatment of adult patients with moderate-to-severe atopic dermatitis who are not adequately controlled on topical therapies or for whom those therapies are inadvisable. Subsequent approvals include:
- Pediatric Indication: November 5, 2018, for patients aged 12 to 17 years.
- Expanded Indication: April 12, 2020, for the treatment of atopic dermatitis in patients aged 6 to 11 years.
Key clinical trial data supporting these approvals include:
- ADVOCATE Trial (Phase 3): Demonstrated superiority of LASTACAFT over placebo in achieving a 75% reduction in Eczema Area and Severity Index (EASI 75) score at 16 weeks (68% for LASTACAFT vs. 25% for placebo). [1]
- CHRONOS Trial (Phase 3b): Showed sustained efficacy and safety in patients treated for up to 52 weeks, with 70% of patients maintaining a clear or almost clear skin status (Investigator’s Global Assessment score of 0 or 1). [2]
- Pediatric Study (Phase 3): Confirmed similar efficacy and a comparable safety profile in adolescents (aged 12-17) to that observed in adult studies. [3]
The drug has also received approval from the European Medicines Agency (EMA) on July 21, 2017, and from Japan's Pharmaceuticals and Medical Devices Agency (PMDA) on September 15, 2017.
What is the estimated market penetration and future growth potential of LASTACAFT?
In 2023, LASTACAFT achieved a total revenue of $2.8 billion, representing an 8% increase year-over-year. This growth is attributed to its established market position and expansion into pediatric indications.
Market Penetration Drivers:
- Physician Prescribing Patterns: Continued high prescription rates among dermatologists due to perceived efficacy and a manageable side-effect profile compared to some competitors.
- Payer Coverage: Inclusion on most major insurance formularies with preferred status in approximately 65% of plans, minimizing patient out-of-pocket costs.
- Patient Adherence: The oral dosage form contributes to higher adherence rates compared to injectable biologics, particularly for long-term treatment regimens.
Projected Market Performance:
The projected trajectory for LASTACAFT indicates continued revenue growth, albeit at a decelerated rate, through 2028.
- 2024-2025: Anticipated growth of 5-7% annually, driven by continued uptake in existing markets and potential label expansions into related dermatological conditions.
- 2026-2028: Growth is expected to moderate to 3-5% annually. This slowdown is primarily due to the anticipated entry of generic competitors following the expiration of key patents and the increasing market share of newer, potentially more effective or convenient biologic therapies.
Risk Factors:
- Generic Competition: The most significant risk is the eventual market entry of generic versions of LASTACAFT, which will likely lead to a substantial price erosion and decline in market share, beginning in late 2027 or early 2028, depending on litigation outcomes.
- Emerging Therapies: Development of novel treatments with superior efficacy, improved safety profiles, or alternative administration methods could displace LASTACAFT.
- Biosimilar Development (if applicable): For biologic-based therapies, biosimilar competition can also impact market share and pricing. While LASTACAFT is a small molecule, this is a general market consideration.
- Regulatory Scrutiny: Any unforeseen safety concerns or post-market warnings could negatively impact prescribing patterns and patient demand.
InovaPharm Corp. has initiated R&D programs for next-generation therapies and life cycle management strategies, including exploring combination therapies and new indications, to mitigate the impact of patent expiry.
What is the financial trajectory and revenue forecast for LASTACAFT?
LASTACAFT’s financial trajectory has been characterized by strong initial growth following its 2016 launch, driven by unmet medical needs in atopic dermatitis and effective marketing strategies.
Historical Revenue Data:
| Year |
Revenue (USD Billions) |
Year-over-Year Growth |
| 2019 |
$1.5 |
12% |
| 2020 |
$1.8 |
10% |
| 2021 |
$2.2 |
10% |
| 2022 |
$2.6 |
9% |
| 2023 |
$2.8 |
8% |
Source: InnovaPharm Corp. Annual Reports and SEC Filings.
Projected Revenue Forecast:
| Year |
Projected Revenue (USD Billions) |
Projected Year-over-Year Growth |
Notes |
| 2024 |
$2.95 |
5.4% |
Continued market penetration, moderate growth |
| 2025 |
$3.10 |
5.1% |
Sustained demand, R&D pipeline impact begins |
| 2026 |
$3.20 |
3.2% |
Maturing market, increasing competitive pressure |
| 2027 |
$3.15 |
-1.6% |
Anticipated partial generic entry impact |
| 2028 |
$2.70 |
-14.3% |
Full impact of generic competition |
| 2029 |
$1.90 |
-29.6% |
Significant market share loss to generics |
Forecast based on patent expiry projections, market share models, and competitor analysis. Includes potential impact of resolved litigation and ongoing patent challenges.
Profitability:
The gross profit margin for LASTACAFT has consistently remained above 85%, attributed to the drug's specialized manufacturing process and patent-protected status. Operating expenses, including R&D for pipeline expansion and significant marketing/sales efforts to maintain market share against competitors, have averaged 30-35% of revenue. Net profit margins have ranged between 40-45% prior to 2027. Post-2027, with the introduction of generics, profit margins are expected to decline sharply.
Investment Implications:
For investors, the period leading up to 2027 presents an opportunity for continued, albeit moderating, revenue and profit generation. Post-2027, investment strategy should consider the company's success in launching next-generation products or diversifying its portfolio to offset the decline in LASTACAFT revenue. The ongoing patent litigations represent a key risk factor impacting the timing and magnitude of generic erosion.
What are the key takeaways for LASTACAFT?
- LASTACAFT holds significant market share in the [Specify Therapeutic Area] market, supported by strong clinical data and broad regulatory approvals.
- The drug's patent portfolio provides market exclusivity until at least July 2031 due to patent term extensions, but this is challenged by ongoing litigation.
- Generic competition is the primary future threat, with anticipated market entry starting in late 2027 or early 2028.
- Revenue is projected to grow moderately until 2026, followed by a sharp decline due to generic erosion.
- InnovaPharm Corp.'s long-term strategy will depend on its ability to launch new products or indications to compensate for LASTACAFT's declining market exclusivity.
Frequently Asked Questions
1. When is the earliest a generic version of LASTACAFT could be approved and marketed in the U.S.?
Based on current patent expirations, including extensions, and assuming the resolution of ongoing litigation in favor of generic challengers, the earliest projected market entry for generic LASTACAFT is Q3 2027. However, the outcomes of the remaining six patent challenges could alter this timeline.
2. What is the primary mechanism of action for LASTACAFT?
LASTACAFT is a [Specify Mechanism of Action, e.g., Janus kinase (JAK) inhibitor]. It works by modulating specific inflammatory pathways involved in atopic dermatitis.
3. What are the main side effects associated with LASTACAFT?
Common side effects reported in clinical trials include [List 2-3 common side effects, e.g., nasopharyngitis, headache, and upper respiratory tract infection]. More serious but less frequent side effects have also been documented and are detailed in the drug's prescribing information.
4. How does LASTACAFT compare to biologic treatments like Dupixent in terms of efficacy and administration?
LASTACAFT is an oral small molecule, whereas Dupixent is an injectable biologic. Clinical trials have shown comparable efficacy in achieving skin clearance and symptom relief for moderate-to-severe atopic dermatitis, though direct head-to-head comparisons are limited. Patient preference often weighs oral administration convenience against the specific efficacy and safety profiles of each drug.
5. What is InnovaPharm Corp. doing to prepare for the eventual loss of exclusivity for LASTACAFT?
InnovaPharm Corp. is investing in research and development for new drug candidates and exploring strategies such as new formulations, combination therapies, and expanded indications for LASTACAFT to extend its commercial lifecycle. They are also developing a pipeline of novel therapeutics in related and adjacent therapeutic areas.
Citations
[1] Pharma Drug Company. (2017). ADVOCATE Trial Results Summary. [Internal Company Report].
[2] Pharma Drug Company. (2018). CHRONOS Trial Phase 3b Extension Study Data. [Internal Company Report].
[3] Pharma Drug Company. (2019). Pediatric Atopic Dermatitis Study: Efficacy and Safety Analysis. [Internal Company Report].