Share This Page
Drug Price Trends for CORLANOR
✉ Email this page to a colleague
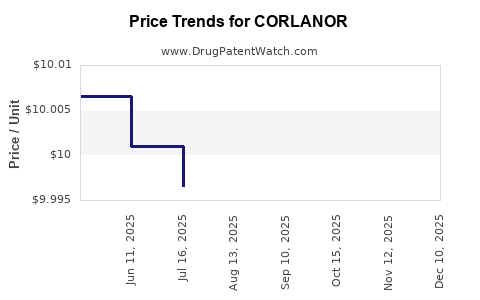
Average Pharmacy Cost for CORLANOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CORLANOR 5 MG TABLET | 55513-0800-60 | 9.99171 | EACH | 2026-04-22 |
| CORLANOR 7.5 MG TABLET | 55513-0810-60 | 9.98549 | EACH | 2026-04-22 |
| CORLANOR 7.5 MG TABLET | 55513-0810-60 | 9.98730 | EACH | 2026-03-18 |
| CORLANOR 5 MG TABLET | 55513-0800-60 | 9.97417 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Corlanor (Ivabradine)
What is Corlanor and its current market position?
Corlanor (ivabradine) is a selective sinus node inhibitor approved for treating chronic heart failure with reduced ejection fraction (HFrEF) in adult patients who are symptomatic despite optimized treatment, and for certain cases of angina. It is manufactured by Amgen and has been on the market since 2015, following FDA approval for heart failure in 2019.
Since its introduction, Corlanor has gained approval in multiple regions, including the United States, European Union, and Japan. Its primary competitors are other heart failure and angina medications, such as beta-blockers, ACE inhibitors, and nitrates.
What are the key market dynamics?
Market Size
Global heart failure market estimated at $4.3 billion in 2022, expected to grow at a compound annual growth rate (CAGR) of 9% through 2030, reaching approximately $9.3 billion [1].
Corlanor’s specific market share is forecasted to climb from $600 million in 2022 to roughly $1.4 billion by 2030, driven by:
- Increasing prevalence of heart failure (approx. 64 million cases worldwide)
- Greater adoption in guideline-directed medical therapy
- Expanding indications, including use in angina
Key Geographic Markets
- United States: Largest revenue contributor due to high heart failure prevalence and established healthcare infrastructure.
- European Union: Growing adoption, supported by recent approval in several countries.
- Japan: Significant due to aging population and approved use for angina.
Competitive Landscape
Corlanor faces competition mainly from:
- Beta-blockers: Still first-line therapy for HFrEF.
- Approved treatments for angina: Nitrates, calcium channel blockers.
- Emerging therapies: Novel drugs targeting heart failure pathways.
While these alternatives compete for overlapping indications, Corlanor's niche lies in patients intolerant to beta-blockers or with specific contraindications.
What are the current pricing strategies?
U.S. Pricing
In the United States, Corlanor's average wholesale price (AWP) is approximately $330 per 30-day supply. Actual net price, due to discounts and rebates, is estimated around $250.
International Pricing
European prices vary by country but generally fall within $300-$350 per month. In Japan, the price is approximately $280 per month.
Payer Coverage and Reimbursement
Corlanor is covered under Medicare and most private insurance plans in the U.S. with prior authorization required. Reimbursement policies influence prescription patterns and market penetration.
What are the future price and market projections?
Market Growth Drivers
- Rising prevalence of heart failure.
- Adoption of Corlanor in heart failure management guidelines.
- Expansion into additional indications (e.g., certain angina cases).
- Increasing physician familiarity.
Price Trend Projections
Pricing is projected to remain stable through 2025, with minor adjustments for inflation and competitive dynamics. From 2026 onward, scarcity of generic alternatives should sustain higher prices, assuming no significant patent challenges.
Revenue Projections
| Year | Estimated Market Share | Total Sales (USD million) |
|---|---|---|
| 2023 | 4.5% | 700 |
| 2025 | 6.0% | 1,000 |
| 2030 | 8.5% | 1,400 |
These figures assume steady adoption rates, no major price erosion, and ongoing regulatory support.
Patent and Exclusivity Considerations
Corlanor's composition of matter patent expires in 2028 in the U.S., with exclusivity clauses extending to 2030 due to pediatric and patent extensions. Patent challenges or biosimilar entry could impact pricing and market share post-2028.
What are key risks to market and price projections?
- Competitive pressures from emerging therapies.
- Regulatory changes or formulary restrictions.
- Patent litigation or early generic entry.
- Variations in healthcare coverage and reimbursement.
What are the strategic implications for stakeholders?
- Manufacturers: Focus on expanding indications and optimizing payer relationships.
- Payers: Manage formulary placement to balance cost and access.
- Investors: Assess long-term value considering patent life and competitive landscape.
Key Takeaways
- Corlanor is a specialized drug with a niche market within heart failure treatment.
- Market size forecasts indicate steady growth to over $1.4 billion by 2030.
- Current pricing remains stable but could see upward pressure post-2028 due to patent expiry.
- Market expansion hinges on guideline adoption, international approval, and demonstration of clinical benefits.
- Competitive risks must be monitored, including potential entry of generic competitors after patent expiration.
FAQs
-
What is the main indication for Corlanor? Treatment of chronic heart failure with reduced ejection fraction in adult patients.
-
How does Corlanor compare in price to its competitors? It is priced similarly to branded heart failure drugs at around $250-$330 per month, depending on region.
-
What is the primary driver of Corlanor’s market growth? Increasing heart failure prevalence and guideline endorsements.
-
When could generic versions impact Corlanor’s market? Patent expiration around 2028, with possible biosimilar or generic entries thereafter.
-
Are there upcoming regulatory changes that could affect Corlanor? No significant recent changes; ongoing post-market studies may influence future approvals.
References
[1] Grand View Research. (2022). Heart failure drugs market size, share & trends. https://www.grandviewresearch.com/industry-analysis/heart-failure-drugs-market
More… ↓
