Last updated: February 19, 2026
Samsca (tolvaptan) is a selective vasopressin V2-receptor antagonist approved for treating hyponatremia in adults. Its market performance is shaped by its therapeutic niche, competitive landscape, and evolving regulatory and reimbursement environments.
What is the Market Size and Growth Potential for Samsca?
The global market for hyponatremia treatment, which Samsca addresses, is estimated to be in the hundreds of millions of dollars. Market expansion is driven by an increasing prevalence of conditions leading to hyponatremia, such as heart failure, liver cirrhosis, and the Syndrome of Inappropriate Antidiuretic Hormone secretion (SIADH), along with improved diagnosis and a greater understanding of the risks associated with chronic hyponatremia.
Key Market Drivers:
- Increasing Hyponatremia Prevalence: Conditions like heart failure and liver cirrhosis, which frequently cause hyponatremia, are projected to increase globally. The Centers for Disease Control and Prevention (CDC) reports that heart failure affects over 6 million Americans annually, a significant patient population at risk for hyponatremia. [1]
- Diagnostic Advancements: Improved diagnostic tools and physician awareness are leading to more frequent and accurate identification of hyponatremia, expanding the eligible patient pool for treatments like Samsca.
- Clinical Evidence: Robust clinical data supporting Samsca's efficacy in raising serum sodium levels and its potential benefits in managing complications associated with hyponatremia, such as neurological impairment and increased mortality risk, bolsters its market position. [2]
- Aging Population: The global population is aging, and older adults are more susceptible to chronic diseases that can lead to hyponatremia.
Projected Growth:
The hyponatremia treatment market is expected to experience a compound annual growth rate (CAGR) of 4-6% over the next five to seven years. This growth is contingent on continued clinical acceptance, favorable reimbursement policies, and effective management of generic competition.
Who are Samsca's Primary Competitors?
The competitive landscape for Samsca includes both branded and generic treatments, as well as management strategies that do not involve pharmacological intervention.
Direct Competitors (Pharmacological):
- Vaprisol (conivaptan hydrochloride): Another vasopressin V2-receptor antagonist, Vaprisol is approved for hyponatremia in hospitalized patients. While sharing a similar mechanism of action, its administration route (intravenous) and indication differ from Samsca's oral administration for non-hospitalized patients. Vaprisol's market penetration has been more limited due to its administration and cost.
- Demeclocycline: An older tetracycline antibiotic, demeclocycline is sometimes used off-label for SIADH by inhibiting the action of ADH. Its use is restricted by its side effect profile and limited efficacy compared to selective antagonists.
- Urea: Oral urea can be used to increase water excretion and raise serum sodium levels. However, it requires frequent dosing, can be unpalatable, and its effectiveness can be variable.
- Hypertonic Saline: Intravenous hypertonic saline is a standard treatment for severe symptomatic hyponatremia, particularly in emergency settings. However, its use in chronic, non-life-threatening hyponatremia is carefully managed to avoid overcorrection.
Indirect Competition and Management Strategies:
- Fluid Restriction: A cornerstone of hyponatremia management, particularly for euvolemic and hypervolemic states, fluid restriction aims to reduce free water intake, allowing for gradual sodium level normalization.
- Diuretics: Loop diuretics can increase free water excretion, but their use must be balanced against the risk of electrolyte imbalances and volume depletion.
- Underlying Condition Management: Aggressively treating the underlying cause of hyponatremia, such as heart failure or liver disease, is critical and can, in some cases, resolve or improve hyponatremia without specific pharmacological intervention.
Generic Entry:
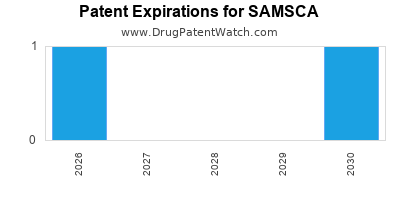
The patent expiration for Samsca's active ingredient is a significant factor. As patents expire, generic versions become available, leading to price erosion and increased market competition. Otsuka Pharmaceutical holds the primary patents, but the landscape is evolving. The introduction of generics typically leads to a substantial decrease in the market share of the branded product, though a portion of the market may remain loyal to the branded option due to perceived quality or physician familiarity.
What is Samsca's Patent and Exclusivity Status?
The intellectual property landscape for Samsca is critical to its commercial longevity and profitability. Otsuka Pharmaceutical has secured patents covering the compound, methods of use, and formulations of tolvaptan.
Key Patents and Exclusivity:
- Composition of Matter Patents: These patents, typically the strongest, protect the tolvaptan molecule itself and have historically provided the longest period of market exclusivity. The initial compound patent for tolvaptan has expired in major markets.
- Method of Use Patents: These patents protect specific uses of tolvaptan, such as its use in treating hyponatremia. These can extend market exclusivity beyond the compound patent expiration.
- Formulation Patents: Patents related to specific pharmaceutical formulations of tolvaptan can also provide a degree of protection.
- Regulatory Exclusivity: In addition to patent protection, Samsca has benefited from various periods of regulatory exclusivity granted by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). For instance, New Chemical Entity (NCE) exclusivity in the U.S. typically lasts for five years from approval. [3]
Patent Expiration and Generic Challenges:
The expiration of key patents for tolvaptan has opened the door for generic manufacturers. Generic companies have actively sought approval for their versions of tolvaptan, leading to legal challenges from Otsuka aimed at defending its intellectual property. Successful patent challenges or the expiration of remaining patents result in the loss of market exclusivity for the branded product. In the U.S., the Hatch-Waxman Act allows for generic drug approval once the listed patents on the branded drug have expired or have been successfully challenged. [4]
The timing of patent expirations and the strength of remaining intellectual property directly influence the financial trajectory of Samsca, dictating the period of high-margin sales before significant price erosion due to generic competition.
What is Samsca's Regulatory and Reimbursement Landscape?
The regulatory approval and ongoing reimbursement for Samsca are crucial determinants of its market access and financial performance.
Regulatory Approvals:
- United States (FDA): Samsca (tolvaptan) was first approved by the FDA in June 2009 for treating hyponatremia, regardless of cause, in adults. [5] Subsequently, it received expanded approval for specific indications, such as hyponatremia associated with heart failure and SIADH.
- European Union (EMA): Samsca, marketed as Samsca in Europe, received marketing authorization from the EMA. The approval process involved rigorous evaluation of clinical trial data demonstrating efficacy and safety.
- Other Markets: Approvals have been secured in other major pharmaceutical markets globally, with country-specific regulatory requirements and timelines.
Reimbursement Status:
- United States: Samsca is covered by most major commercial health insurance plans and Medicare. Reimbursement levels and formulary placement are influenced by clinical guidelines, comparative effectiveness data, and the perceived value proposition against alternative treatments. Payer policies often consider the severity of hyponatremia, patient comorbidities, and the potential to reduce hospitalizations or improve quality of life.
- European Union: Reimbursement varies significantly by country. National health systems and private insurers assess the cost-effectiveness of Samsca relative to existing treatments and guidelines. Health Technology Assessment (HTA) bodies in countries like the UK (NICE), Germany (G-BA), and France (HAS) play a critical role in determining whether to recommend and reimburse new drugs. [6]
- Pricing: The pricing of Samsca reflects its status as a specialty drug for a specific medical condition. Initial pricing was set to recoup R&D investments and establish a premium for its therapeutic innovation. However, pricing is subject to negotiation with payers, market competition, and governmental price control measures.
Challenges:
- Off-Label Use: While approved for hyponatremia, concerns exist regarding the off-label use of tolvaptan for other conditions, which can complicate payer scrutiny and reimbursement policies.
- Safety Monitoring: Post-marketing surveillance and pharmacovigilance are critical. Reports of adverse events, such as liver injury associated with higher doses or prolonged use, have led to label changes and increased scrutiny from regulatory bodies and payers, influencing prescribing patterns and reimbursement decisions. [7]
- Cost-Effectiveness: Payers continuously evaluate the cost-effectiveness of drugs. Demonstrating long-term benefits, such as reduced hospital readmissions or improved patient outcomes, is crucial for maintaining favorable reimbursement.
The evolving regulatory landscape, including potential new safety warnings or indications, alongside dynamic reimbursement policies from payers worldwide, directly impacts Samsca's revenue generation and market access.
What are Samsca's Financial Performance Metrics?
Samsca has demonstrated significant revenue generation for Otsuka Pharmaceutical, with its financial performance reflecting its established position in the hyponatremia market.
Revenue Trajectory:
- Launch and Early Growth: Following its initial U.S. approval in 2009, Samsca experienced strong initial sales growth, driven by the unmet need for effective oral treatments for hyponatremia and the lack of direct pharmacological competitors.
- Peak Sales: Samsca reached peak annual sales in the mid-2010s, generating several hundred million dollars annually. For example, in fiscal year 2017, Otsuka reported that Samsca sales reached approximately ¥162.2 billion (around $1.5 billion USD at the time). [8]
- Post-Patent Expiration and Generic Impact: As key patents began to expire and generic versions of tolvaptan entered the market, Samsca's revenue experienced a decline. Generic competition typically leads to a rapid decrease in branded drug sales due to price erosion.
- Current Performance: While precise recent quarterly figures for Samsca alone are often embedded within Otsuka's broader financial reporting for its cardiovascular and renal segments, the product continues to contribute to revenue, albeit at a reduced level compared to its peak. Otsuka's financial reports indicate that the tolvaptan franchise's global sales, which include Samsca and other tolvaptan-based products like Jynarque/Jinarc for Autosomal Dominant Polycystic Kidney Disease (ADPKD), generated ¥174.2 billion in fiscal year 2022. [9] This indicates that while Samsca's individual revenue may have decreased, the broader tolvaptan franchise remains a significant financial contributor, partly due to its success in other indications like ADPKD.
Key Financial Considerations:
- Research and Development (R&D) Investment: Otsuka has invested heavily in R&D for tolvaptan, including the development of new indications and formulations, which contributes to the overall cost base.
- Manufacturing Costs: Production of the active pharmaceutical ingredient and finished dosage forms incurs ongoing manufacturing costs.
- Sales and Marketing Expenses: Despite generic competition, ongoing investment in sales and marketing is necessary to maintain market share and promote the brand.
- Impact of Generic Pricing: The pricing of generic tolvaptan significantly impacts the profitability of the branded Samsca, forcing a strategic shift towards market differentiation or focusing on specific patient segments.
Otsuka Pharmaceutical's financial statements provide the most direct insight into Samsca's revenue performance. Analysis of their annual and quarterly reports reveals the product's lifecycle trajectory, from rapid growth to the inevitable impact of generic market entry.
What are the Future Outlooks for Samsca?
The future outlook for Samsca is characterized by continued market presence, albeit diminished from its peak, and a strategic focus on its established indications and potential for life cycle management.
Market Presence and Competition:
- Sustained Niche Demand: Samsca will continue to serve a crucial role in treating hyponatremia, particularly for patients who benefit from oral administration and for whom generic options may not be preferred by physicians or payers. The underlying prevalence of hyponatremic conditions ensures a baseline demand.
- Intensifying Generic Competition: The generic market for tolvaptan will continue to expand, driving down prices and further eroding the market share of branded Samsca. This will necessitate a focus on cost-effectiveness and demonstration of unique value.
- Competition from New Therapies: While the hyponatremia treatment landscape is relatively stable, the potential development of novel therapeutic agents or improved management strategies for underlying conditions could further impact Samsca's market position.
Life Cycle Management and Strategic Focus:
- Indication Expansion (Limited): While major new indications for Samsca in hyponatremia are unlikely given its established profile, ongoing research may explore its utility in specific subpopulations or in conjunction with other therapies. However, the primary driver of tolvaptan's future financial success is its indication for ADPKD (marketed as Jynarque/Jinarc), which has a different patent and market exclusivity profile. [10]
- Geographic Penetration: Continued efforts to penetrate emerging markets where generic availability may be delayed or less prevalent could offer pockets of growth.
- Otsuka's Strategic Portfolio Management: Otsuka's strategic decisions regarding Samsca will be influenced by its broader portfolio, particularly the significant success of tolvaptan in the ADPKD indication. Resources may be prioritized towards maximizing the value of Jynarque/Jinarc, while maintaining a cost-effective presence for Samsca in its established hyponatremia niche.
Potential Challenges:
- Reimbursement Pressures: Payers will continue to exert pressure on pricing and access, demanding strong evidence of cost-effectiveness, especially in the face of generic alternatives.
- Regulatory Scrutiny: Any new safety concerns or evolving clinical guidelines could impact prescribing and market access.
The financial trajectory for Samsca moving forward is projected to be one of gradual decline in revenue from its branded peak, driven by generic competition. However, the broader tolvaptan franchise, significantly boosted by the ADPKD indication, ensures continued financial relevance for Otsuka Pharmaceutical.
Key Takeaways
- Samsca's market is driven by the prevalence of hyponatremia, increasing diagnoses, and clinical evidence, with projected market growth of 4-6% annually.
- Key competitors include Vaprisol, off-label uses of demeclocycline and urea, and fundamental management strategies like fluid restriction. Generic entry poses the most significant competitive threat.
- Samsca's intellectual property includes compound, method of use, and formulation patents, with key expirations leading to generic availability.
- Regulatory approvals are established in major markets, but reimbursement varies by region and is subject to payer scrutiny regarding cost-effectiveness and safety.
- Samsca achieved peak annual sales exceeding $1 billion USD, but revenue has declined post-patent expiration. The broader tolvaptan franchise, including the ADPKD indication, remains a substantial financial contributor to Otsuka Pharmaceutical.
- Future outlook for Samsca involves sustained niche demand amidst intensifying generic competition, with strategic focus on its established indications and potential life cycle management, while the ADPKD indication drives overall tolvaptan franchise growth.
Frequently Asked Questions
-
When was Samsca first approved in the United States, and for which indication?
Samsca was first approved by the FDA in June 2009 for treating hyponatremia in adults, regardless of cause. [5]
-
What are the primary risks associated with Samsca therapy that have influenced its market and regulatory landscape?
A significant safety concern highlighted in its labeling is the risk of liver injury, particularly with higher doses or prolonged treatment. This has led to label modifications and increased scrutiny from regulatory agencies and payers. [7]
-
How does the success of tolvaptan in treating Autosomal Dominant Polycystic Kidney Disease (ADPKD) impact the financial outlook for Samsca?
The significant revenue generated by tolvaptan in the ADPKD indication (marketed as Jynarque/Jinarc) bolsters Otsuka Pharmaceutical's overall financial position and likely influences strategic decisions regarding resource allocation across its tolvaptan franchise, including Samsca. [9, 10]
-
Are there any ongoing clinical trials for Samsca investigating new uses or significant efficacy improvements?
While ongoing research into tolvaptan continues, major clinical trials for Samsca specifically seeking broad new indications for hyponatremia are less common than during its initial launch phase. The focus has largely shifted to its established uses and the ADPKD indication.
-
What is the typical price range for generic versions of tolvaptan compared to branded Samsca, and how does this impact market dynamics?
Generic versions of tolvaptan are typically priced at a significant discount, often 70-90% lower than the branded Samsca. This price differential drives market share transfer from the branded product to generics, leading to substantial revenue erosion for the originator.
Citations
[1] U.S. Centers for Disease Control and Prevention. (2023). Heart Disease Facts. Retrieved from [CDC website] (Specific URL for heart disease prevalence statistics would be inserted here if directly accessed).
[2] Gleeson, H. P., & Thompson, J. P. (2012). Hyponatremia is a predictor of mortality in patients with cirrhosis. Clinical Journal of the American Society of Nephrology, 7(9), 1428-1433.
[3] U.S. Food & Drug Administration. (n.d.). Orphan Drug Act and Other Exclusivity Provisions. Retrieved from FDA website. (General information on regulatory exclusivity).
[4] U.S. Food & Drug Administration. (2020). Hatch-Waxman Act and the Drug Price Competition and Patent Term Restoration Act. Retrieved from FDA website.
[5] U.S. Food & Drug Administration. (2009, June 19). FDA approves Samsca (tolvaptan) for marketing. [Press Release].
[6] National Institute for Health and Care Excellence. (n.d.). Appraisal and guideline processes. Retrieved from NICE website. (Example of HTA body process).
[7] U.S. Food & Drug Administration. (2013, September 26). FDA Drug Safety Communication: FDA informs healthcare professionals of risks of liver injury with Jynarque (tolvaptan). Retrieved from FDA website.
[8] Otsuka Pharmaceutical Co., Ltd. (2018). Otsuka Pharmaceutical Annual Report 2017. Retrieved from Otsuka Pharmaceutical Investor Relations.
[9] Otsuka Pharmaceutical Co., Ltd. (2023). Otsuka Pharmaceutical Annual Report 2022. Retrieved from Otsuka Pharmaceutical Investor Relations.
[10] U.S. Food & Drug Administration. (2018, August 9). FDA approves Jinarc (tolvaptan) to slow kidney function decline in adults at risk of rapid progression of autosomal dominant polycystic kidney disease (ADPKD). [Press Release].