KERENDIA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Kerendia, and when can generic versions of Kerendia launch?
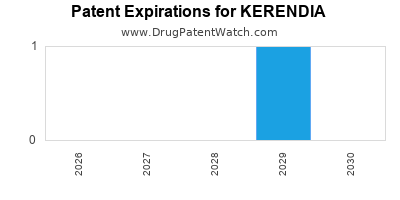
Kerendia is a drug marketed by Bayer Hlthcare and is included in one NDA. There are two patents protecting this drug and two Paragraph IV challenges.
This drug has ninety-eight patent family members in fifty countries.
The generic ingredient in KERENDIA is finerenone. One supplier is listed for this compound. Additional details are available on the finerenone profile page.
DrugPatentWatch® Generic Entry Outlook for Kerendia
Kerendia was eligible for patent challenges on July 9, 2025.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be July 29, 2035. This may change due to patent challenges or generic licensing.
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for KERENDIA?
- What are the global sales for KERENDIA?
- What is Average Wholesale Price for KERENDIA?
Summary for KERENDIA
| International Patents: | 98 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 33 |
| Clinical Trials: | 6 |
| Patent Applications: | 325 |
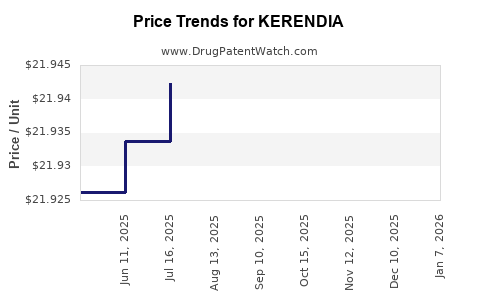
| Drug Prices: | Drug price information for KERENDIA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for KERENDIA |
| What excipients (inactive ingredients) are in KERENDIA? | KERENDIA excipients list |
| DailyMed Link: | KERENDIA at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for KERENDIA
Generic Entry Date for KERENDIA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for KERENDIA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Boehringer Ingelheim | Phase 4 |
| University Medical Center Groningen | Phase 4 |
| University of North Carolina, Chapel Hill | Phase 2 |
Pharmacology for KERENDIA
| Drug Class | Nonsteroidal Mineralocorticoid-Receptor Antagonist |
| Mechanism of Action | Mineralocorticoid Receptor Antagonists |
Paragraph IV (Patent) Challenges for KERENDIA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| KERENDIA | Tablets | finerenone | 40 mg | 215341 | 1 | 2026-02-02 |
| KERENDIA | Tablets | finerenone | 10 mg and 20 mg | 215341 | 9 | 2025-07-09 |
US Patents and Regulatory Information for KERENDIA
KERENDIA is protected by two US patents and three FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of KERENDIA is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bayer Hlthcare | KERENDIA | finerenone | TABLET;ORAL | 215341-001 | Jul 9, 2021 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Bayer Hlthcare | KERENDIA | finerenone | TABLET;ORAL | 215341-002 | Jul 9, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Bayer Hlthcare | KERENDIA | finerenone | TABLET;ORAL | 215341-001 | Jul 9, 2021 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Bayer Hlthcare | KERENDIA | finerenone | TABLET;ORAL | 215341-003 | Jul 11, 2025 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for KERENDIA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Bayer AG | Kerendia | finerenone | EMEA/H/C/005200Kerendia is indicated for the treatment of chronic kidney disease (stage 3 and 4 with albuminuria) associated with type 2 diabetes in adults. | Authorised | no | no | no | 2022-02-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for KERENDIA
When does loss-of-exclusivity occur for KERENDIA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 5774
Patent: PROCEDIMIENTO PARA LA PREPARACIÓN DE (4S)-4-(4-CIANO-2-METOXIFENILO)-5-ETOXI-2,8-DIMETILO-1,4-DIHIDRO-1,6-NAFTIRIDINA-3-CARBOXAMIDA Y SU PURIFICACIÓN PARA SU USO COMO PRINCIPIO ACTIVO FARMACÉUTICO
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 16312904
Patent: Method for the preparation of (4S)-4-(4-cyano-2-methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1-6-naphthyridine-3-carboxamide and the purification thereof for use as an active pharmaceutical ingredient
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 95905
Patent: PROCEDE DE PREPARATION DE (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1,6-NAPHTYRIDINE-3-CARBOXAMIDE ET DE PURIFICATION DE CE DERNIER AFIN DE L'UTILISER EN TANT QUE PRINCIPE ACTIF PHARMACEUTIQUE (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 18000427
Patent: Procedimiento para la preparación de (4s)-4-(4-ciano-2-metoxifenilo)-5-etoxi-2,8-dimetilo-1,4-dihidro-1,6-naftiridina-3-carboxamida y su purificación para su uso como principio activo farmacéutico
Estimated Expiration: ⤷ Start Trial
China
Patent: 7849043
Patent: 用于制备(4S)‑4‑(4‑氰基‑2‑甲氧基苯基)‑5‑乙氧基‑2,8‑二甲基‑1,4‑二氢‑1,6‑萘啶‑3‑甲酰胺的方法及其用于用作药物活性物质的纯化 (METHOD FOR THE PREPARATION OF (4S)-4-(4-cyano-2-methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1-6-naphthyridine-3-carboxamide and the purification thereof for use as an active pharmaceutical ingredient)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 18001466
Patent: Procedimiento para la preparación de (4s)-4-(4-ciano-2-metoxifenilo)-5-etoxi-2,8-dimetilo-1,4-dihidro-1,6-naftiridina-3-carboxamida y su purificación para su uso como principio activo farmacéutico
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0191431
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 37800
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 37800
Patent: PROCÉDÉ DE PRÉPARATION DE (4S)-4-(4-CYANO-2-MÉTHOXYPHÉNYL)-5-ÉTHOXY-2,8-DIMÉTHYL-1,4-DIHYDRO-1,6-NAPHTYRIDINE-3-CARBOXAMIDE ET DE PURIFICATION DE CE DERNIER AFIN DE L'UTILISER EN TANT QUE PRINCIPE ACTIF PHARMACEUTIQUE (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 51565
Patent: 用於製備(4S)-4-(4-氰基-2-甲氧基苯基)-5-乙氧基-2,8-二甲基-1,4-二氫-1,6-萘啶-3-甲酰胺的方法及其用於用作藥物活性物質的純化 (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 44574
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 7371
Patent: שיטה להכנה של (s4)-4-(4-ציאנו-2-מתוקסיפניל)-5-אתוקסי-8,2-דימתיל-4,1-דיהידרו-6,1-נפתירידין-3-קרבוקסאמיד וטיהורו לשימוש כמרכיב רוקחי פעיל (Method for the preparation of (4s)-4-(4-cyano-2-methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1-6-naphthyridine-3-carboxamide and the purification thereof for use as an active pharmaceutical ingredient)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 24247
Estimated Expiration: ⤷ Start Trial
Patent: 18523699
Patent: (4S)−4−(4−シアノ−2−メトキシフェニル)−5−エトキシ−2,8−ジメチル−1,4−ジヒドロ−1,6−ナフチリジン−3−カルボキサミドの調製方法および医薬品有効成分として使用するためのその精製方法
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 0160186
Patent: طريقة لتحضير (4S)-4-(4- سيانو-2- ميثوكسي فينيل)-5-إيثوكسي-8،2- داي ميثيل-4،1- دايهيدرو-6،1- نفثيريدين-3- كربوكساميد وتنقيته للاستخدام كمقوم نشط دوائيا (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 37800
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 5226
Patent: METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 7960
Patent: PROCEDIMIENTO PARA LA PREPARACION DE (4S)-4-(4-CIANO-2-METOXIFENIL O)-5-ETOXI-2,8-DIMETILO-1,4-DIHIDRO-1,6-NAFTIRIDINA-3-CARBOXAMIDA Y SU PURIFICACION PARA SU USO COMO PRINCIPIO ACTIVO FARMACEUTICO. (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5- ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT.)
Estimated Expiration: ⤷ Start Trial
Patent: 18002027
Patent: PROCEDIMIENTO PARA LA PREPARACION DE (4S)-4-(4-CIANO-2-METOXIFENIL O)-5-ETOXI-2,8-DIMETILO-1,4-DIHIDRO-1,6-NAFTIRIDINA-3-CARBOXAMIDA Y SU PURIFICACION PARA SU USO COMO PRINCIPIO ACTIVO FARMACEUTICO. (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5- ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT.)
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 180554
Patent: PROCEDIMIENTO PARA LA PREPARACION DE (4S)-4-(4-CIANO-2-METOXIFENILO)-5-ETOXI-2,8-DIMETILO-1,4-DIHIDRO-1,6-NAFTIRIDINA-3-CARBOXAMIDA Y SU PURIFICACION PARA SU USO COMO PRINCIPIO ACTIVO FARMACEUTICO
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 37800
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 37800
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 43429
Patent: СПОСОБ ПОЛУЧЕНИЯ (4S)-4-(4-ЦИАНО-2-МЕТОКСИФЕНИЛ)-5-ЭТОКСИ-2,8-ДИМЕТИЛ-1,4-ДИГИДРО-1,6-НАФТИРИДИН-3-КАРБОКСАМИДА И ЕГО ОЧИСТКА ДЛЯ ПРИМЕНЕНИЯ В КАЧЕСТВЕ ФАРМАЦЕВТИЧЕСКОГО БИОЛОГИЧЕСКИ АКТИВНОГО ВЕЩЕСТВА (METHOD OF PRODUCING (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1,6-NAPHTHYRIDINE-3-CARBOXAMIDE AND PURIFICATION THEREOF FOR USE AS PHARMACEUTICAL BIOLOGICALLY ACTIVE SUBSTANCE)
Estimated Expiration: ⤷ Start Trial
Patent: 18109761
Patent: СПОСОБ ПОЛУЧЕНИЯ (4S)-4-(4-ЦИАНО-2-МЕТОКСИФЕНИЛ)-5-ЭТОКСИ-2,8-ДИМЕТИЛ-1,4-ДИГИДРО-1,6-НАФТИРИДИН-3-КАРБОКСАМИДА И ЕГО ОЧИСТКА ДЛЯ ПРИМЕНЕНИЯ В КАЧЕСТВЕ ФАРМАЦЕВТИЧЕСКОГО БИОЛОГИЧЕСКИ АКТИВНОГО ВЕЩЕСТВА
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 055
Patent: POSTUPAK ZA PROIZVODNJU (4S)-4-(4-CIJANO-2-METOKSIFENIL)-5-ETOKSI-2,8-DIMETIL-1,4-DIHIDRO-1,6-NAFTIRIDIN-3-KARBOKSAMIDA I NJEGOVO PREČIŠĆAVANJE U SVRHU UPOTREBE KAO FARMACEUTSKI AKTIVNE SUPSTANCE (METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201801110T
Patent: METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 37800
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1801859
Patent: METHOD FOR THE PREPARATION OF (4S)-4-(4-CYANO-2-METHOXYPHENYL)-5-ETHOXY-2,8-DIMETHYL-1,4-DIHYDRO-1-6-NAPHTHYRIDINE-3-CARBOXAMIDE AND THE PURIFICATION THEREOF FOR USE AS AN ACTIVE PHARMACEUTICAL INGREDIENT
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 180041138
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 39904
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1722944
Patent: Process for preparing (4S)-4-(4-cyano-2-methoxyphenyl)-5-ethoxy-2,8-dimethyl-1,4-dihydro-1,6-naphthyridine-3-carboxamide and purificationthereof for use as a pharmaceutical activeingredient
Estimated Expiration: ⤷ Start Trial
Patent: 25045
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 864
Patent: PROCEDIMIENTO PARA LA PREPARACIÓN DE (4S)-4-(4-CIANO-2-METOXIFENILO)-5-ETOXI-2,8-DIMETILO-1,4-DIHIDRO-1,6-NAFTIRIDINA-3-CARBOXAMIDA Y SU PURIFICACIÓN PARA SU USO COMO PRINCIPIO ACTIVO FARMACÉUTICO
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering KERENDIA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Jordan | 3018 | ⤷ Start Trial | |
| Taiwan | I725045 | ⤷ Start Trial | |
| Canada | 2679232 | ⤷ Start Trial | |
| Norway | 2022013 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for KERENDIA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2132206 | 2290018-7 | Sweden | ⤷ Start Trial | PRODUCT NAME: FINERENONE AND ITS SALTS, SOLVATES AND SOLVATES OF THE SALT THEREOF; REG. NO/DATE: EU/1/21/1616 20220217 |
| 2132206 | CA 2022 00025 | Denmark | ⤷ Start Trial | PRODUCT NAME: FINERENONE OG DETS SALTE, SOLVATER OG SOLVATER AF SALTENE DERAF; REG. NO/DATE: EU/1/21/1616 20220217 |
| 2132206 | C202230028 | Spain | ⤷ Start Trial | PRODUCT NAME: FINERENONA Y SUS SALES, SOLVATOS Y SOLVATOS DE LAS SALES; NATIONAL AUTHORISATION NUMBER: EU/1/21/1616; DATE OF AUTHORISATION: 20220216; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/21/1616; DATE OF FIRST AUTHORISATION IN EEA: 20220216 |
| 2132206 | 202240019 | Slovenia | ⤷ Start Trial | PRODUCT NAME: FINERENON AND ITS SALTS, SOLVATES AND SOLVATES ITS SALTS; NATIONAL AUTHORISATION NUMBER: EU/1/21/1616; DATE OF NATIONAL AUTHORISATION: 20220216; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for KERENDIA
More… ↓
DrugChatter Q&A for KERENDIA
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.