NEXTSTELLIS Drug Patent Profile
✉ Email this page to a colleague
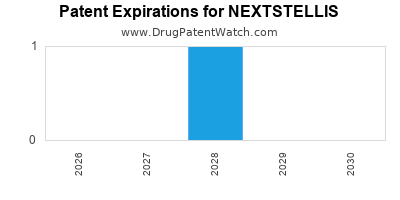
When do Nextstellis patents expire, and when can generic versions of Nextstellis launch?
Nextstellis is a drug marketed by Mayne Pharma and is included in one NDA. There are seven patents protecting this drug.
This drug has two hundred and thirty-five patent family members in fifty-three countries.
The generic ingredient in NEXTSTELLIS is drospirenone; estetrol. There are eleven drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the drospirenone; estetrol profile page.
DrugPatentWatch® Generic Entry Outlook for Nextstellis
Nextstellis was eligible for patent challenges on April 15, 2025.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 17, 2036. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for NEXTSTELLIS?
- What are the global sales for NEXTSTELLIS?
- What is Average Wholesale Price for NEXTSTELLIS?
Summary for NEXTSTELLIS
| International Patents: | 235 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 32 |
| Clinical Trials: | 1 |
| Drug Prices: | Drug price information for NEXTSTELLIS |
| What excipients (inactive ingredients) are in NEXTSTELLIS? | NEXTSTELLIS excipients list |
| DailyMed Link: | NEXTSTELLIS at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for NEXTSTELLIS
Generic Entry Date for NEXTSTELLIS*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for NEXTSTELLIS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| McGill University Health Centre/Research Institute of the McGill University Health Centre | Phase 4 |
US Patents and Regulatory Information for NEXTSTELLIS
NEXTSTELLIS is protected by seven US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of NEXTSTELLIS is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Mayne Pharma | NEXTSTELLIS | drospirenone; estetrol | TABLET;ORAL | 214154-001 | Apr 15, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for NEXTSTELLIS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Estetra SPRL | Lydisilka | estetrol, drospirenone | EMEA/H/C/005382Oral contraception.The decision to prescribe Lydisilka should take into consideration the individual woman’s current risk factors, particularly those for venous thromboembolism (VTE), and how the risk of VTE with Lydisilka compares with other combined hormonal contraceptives (CHCs) (see sections 4.3 and 4.4). | Authorised | no | no | no | 2021-05-19 | |
| Gedeon Richter Plc. | Drovelis | estetrol, drospirenone | EMEA/H/C/005336oral contraceptive | Authorised | no | no | no | 2021-05-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for NEXTSTELLIS
When does loss-of-exclusivity occur for NEXTSTELLIS?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 16280858
Estimated Expiration: ⤷ Start Trial
Patent: 21203265
Estimated Expiration: ⤷ Start Trial
Patent: 22283615
Estimated Expiration: ⤷ Start Trial
Patent: 24201228
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2017027229
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 88495
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 17003209
Estimated Expiration: ⤷ Start Trial
China
Patent: 7787224
Estimated Expiration: ⤷ Start Trial
Patent: 6077455
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 17012670
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 180041
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0200850
Estimated Expiration: ⤷ Start Trial
Patent: 0211968
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 504
Estimated Expiration: ⤷ Start Trial
Patent: 170161
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 23006
Estimated Expiration: ⤷ Start Trial
Patent: 24878
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Patent: 01944
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 017000294
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 17085659
Estimated Expiration: ⤷ Start Trial
El Salvador
Patent: 17005596
Patent: UNIDAD DE DOSIFICACION ORODISPERSABLE QUE CONTIENE UN COMPONENTE ESTETROL
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 5687
Estimated Expiration: ⤷ Start Trial
Patent: 1890085
Estimated Expiration: ⤷ Start Trial
Patent: 2090984
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Patent: 01944
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 0217243
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 54650
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 49232
Estimated Expiration: ⤷ Start Trial
Patent: 57741
Estimated Expiration: ⤷ Start Trial
Patent: 200027
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 6282
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 66560
Estimated Expiration: ⤷ Start Trial
Patent: 40355
Estimated Expiration: ⤷ Start Trial
Patent: 18521985
Estimated Expiration: ⤷ Start Trial
Patent: 21098710
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Patent: 01944
Estimated Expiration: ⤷ Start Trial
Patent: 701944
Estimated Expiration: ⤷ Start Trial
Patent: 2022508
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 6164
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 4904
Estimated Expiration: ⤷ Start Trial
Patent: 17016274
Estimated Expiration: ⤷ Start Trial
Moldova, Republic of
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 728
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 972
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 7945
Estimated Expiration: ⤷ Start Trial
Patent: 6452
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 22020
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 180522
Patent: UNIDAD DE DOSIFICACION ORODISPERSABLE QUE CONTIENE UN COMPONENTE ESTETROL
Estimated Expiration: ⤷ Start Trial
Patent: 231714
Patent: UNIDAD DE DOSIFICACION ORODISPERSABLE QUE CONTIENE UN COMPONENTE ESTETROL
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 017502326
Patent: ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Patent: 01944
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Patent: 01944
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 02000251
Estimated Expiration: ⤷ Start Trial
Patent: 02200102
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 252
Patent: ORODISPERZIBILNA DOZNA JEDINICA KOJA SADRŽI KOMPONENTU ESTETROLA (ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT)
Estimated Expiration: ⤷ Start Trial
Patent: 844
Patent: ORODISPERZIBILNA DOZNA JEDINICA KOJA SADRŽI KOMPONENTU ESTETROLA (ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 10333
Estimated Expiration: ⤷ Start Trial
Patent: 01944
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1708286
Patent: ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT
Estimated Expiration: ⤷ Start Trial
Patent: 2303338
Patent: ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2662025
Estimated Expiration: ⤷ Start Trial
Patent: 2735893
Estimated Expiration: ⤷ Start Trial
Patent: 180019697
Estimated Expiration: ⤷ Start Trial
Patent: 230074847
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 00774
Estimated Expiration: ⤷ Start Trial
Patent: 06228
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 17000497
Patent: ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3099
Patent: ДИСПЕРГОВАНА В ПОРОЖНИНІ РОТА ОДИНИЦЯ ДОЗУВАННЯ, ЯКА МІСТИТЬ ЕСТЕТРОЛЬНИЙ КОМПОНЕНТ (ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT)
Estimated Expiration: ⤷ Start Trial
Patent: 0516
Patent: ЗДАТНА ДО ДИСПЕРГУВАННЯ В ПОРОЖНИНІ РОТА ОДИНИЦЯ ДОЗУВАННЯ, ЯКА МІСТИТЬ ЕСТЕТРОЛЬНИЙ КОМПОНЕНТ (ORODISPERSIBLE DOSAGE UNIT CONTAINING AN ESTETROL COMPONENT)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering NEXTSTELLIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hungary | E037383 | ⤷ Start Trial | |
| Canada | 2476940 | COMPOSITIONS PHARMACEUTIQUES CONTENANT UN OU PLUSIEURS STEROIDES, UN OU PLUSIEURS TETRAHYDROFOLATES ET DE LA VITAMINE B12 (PHARMACEUTICAL COMPOSITIONS COMPRISING ONE OR MORE STEROIDS, ONE OR MORE TETRAHYDROFOLATE COMPONENTS AND VITAMIN B12) | ⤷ Start Trial |
| Japan | 6866560 | ⤷ Start Trial | |
| Spain | 2337129 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for NEXTSTELLIS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2588114 | LUC00227 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: DROSPIRENONE; AUTHORISATION NUMBER AND DATE: 31332 20191022 |
| 3701944 | PA2022508,C3701944 | Lithuania | ⤷ Start Trial | PRODUCT NAME: DROSPIRENONAS DERINYJE SU ESTETROLIU; REGISTRATION NO/DATE: EU/1/21/1547 20210519 |
| 0398460 | 04C0022 | France | ⤷ Start Trial | PRODUCT NAME: ESTRADIOL ANHYDRE DROSPIRENONE; REGISTRATION NO/DATE IN FRANCE: NL 28661 DU 20040316; REGISTRATION NO/DATE AT EEC: RVG 27505 DU 20021211 |
| 3632448 | 202240023 | Slovenia | ⤷ Start Trial | PRODUCT NAME: DROSPIRENONE; NATIONAL AUTHORISATION NUMBER: H/21/02860/001-004; DATE OF NATIONAL AUTHORISATION: 20211217; AUTHORITY FOR NATIONAL AUTHORISATION: SI; FIRST AUTHORISATION IN THE EUROPEAN ECONOMIC AREA: 61678; DATE OF FIRST AUTHORISATION IN THE EUROPEAN ECONOMIC AREA: 20191016; AUTHORITY OF FIRST AUTHORISATION IN THE EUROPEAN ECONOMIC AREA: DK |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for NEXTSTELLIS (Partnership of Stellantis and GSK)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.