Last updated: February 19, 2026
Nulibry (asfotase alfa), developed by Alexion Pharmaceuticals, a subsidiary of AstraZeneca, targets hypophosphatasia (HPP), a rare genetic disorder characterized by defective bone mineralization. The drug's market trajectory is shaped by HPP prevalence, clinical efficacy, pricing strategies, regulatory approvals, and competition.
What is the current market size and projected growth for Nulibry?
The market for HPP treatments is relatively small due to the rarity of the disease. However, as diagnostic capabilities improve and awareness increases, the patient population identified for treatment is growing. This expansion, coupled with the premium pricing typical for orphan drugs, is expected to drive Nulibry's market share.
- Estimated Patient Population: HPP affects approximately 1 in 30,000 to 1 in 200,000 live births, depending on the form of the disease. Milder, later-onset forms are more prevalent, contributing to a larger addressable market than previously estimated [1].
- Market Penetration: Nulibry is the first and only approved enzyme replacement therapy for HPP in many key markets. This first-mover advantage positions it for significant market penetration.
- Projected Growth: While specific market size data for Nulibry is proprietary, the orphan drug market, in general, has seen consistent double-digit growth. Nulibry's trajectory is expected to align with this trend, driven by an expanding diagnosis rate and the drug's established efficacy in improving patient outcomes [2]. Analysts project the global HPP therapeutics market, with Nulibry as a dominant player, to reach several hundred million dollars within the next five years, with growth rates potentially exceeding 15% annually [3].
What are the key clinical indications and efficacy data supporting Nulibry's use?
Nulibry is indicated for the treatment of HPP, specifically targeting infants and children with the perinatal and infantile forms of the disease, and more recently expanded to include adolescents and adults with HPP. Its efficacy is measured by improvements in skeletal mineralization, growth, and reductions in HPP-related complications.
- Primary Indications:
- Treatment of HPP in infants and children.
- Treatment of HPP in adolescents and adults.
- Key Efficacy Endpoints:
- Skeletal Mineralization: Studies demonstrate significant improvements in bone mineralization, as assessed by Radiographic Global Impression (RGI) scores. In clinical trials, a substantial proportion of pediatric patients treated with Nulibry achieved normal or near-normal skeletal mineralization [4].
- Growth: Nulibry has shown positive impacts on linear growth in pediatric HPP patients. For example, a Phase 3 study (Study A) showed that patients treated with asfotase alfa experienced sustained improvements in height standard deviation scores (SDS) over multiple years [5].
- Clinical Outcomes: Improvements have also been observed in other HPP-related manifestations, including respiratory function, mobility, and overall quality of life. Reductions in rickets and osteomalacia have been documented [6].
- Long-Term Data: Extended follow-up data from clinical trials have reinforced the sustained benefits of Nulibry, demonstrating continued improvements in bone health and growth parameters over several years of treatment [5, 6].
What is the pricing and reimbursement landscape for Nulibry?
As an orphan drug for a rare disease, Nulibry commands a premium price. Pricing and reimbursement are critical determinants of market access and financial performance.
- Pricing Strategy: Alexion employs a value-based pricing strategy, reflecting the high unmet medical need and the significant clinical benefits of Nulibry. The annual cost of Nulibry is substantial, often in the hundreds of thousands of dollars per patient per year, depending on patient weight and dosage [7].
- Reimbursement: Nulibry has secured reimbursement in major markets, including the United States, Europe, and Japan. This is facilitated by its orphan drug designation and the severe nature of HPP.
- United States: Coverage is provided by Medicare, Medicaid, and private insurers. Negotiations with payers focus on demonstrating long-term cost-effectiveness, considering the potential reduction in hospitalizations, surgeries, and other HPP-related medical interventions [8].
- European Union: Pricing and reimbursement decisions are made at the national level. Nulibry has obtained positive reimbursement recommendations in countries such as Germany, the UK, and France, often following health technology assessments (HTAs) that evaluate its clinical and economic value [9].
- Japan: Approved for HPP and listed on the national health insurance drug tariff, ensuring patient access [10].
- Market Access Challenges: Despite approvals, payer scrutiny and formulary restrictions can impact access. Alexion actively engages with payers to demonstrate the drug's value proposition and facilitate patient access through patient support programs.
Who are the key competitors and what is Nulibry's competitive advantage?
The competitive landscape for Nulibry is characterized by a lack of direct, similarly acting enzyme replacement therapies. However, competition can arise from supportive care and potential future entrants.
- Direct Competition:
- None Currently Approved: As of the latest data, Nulibry remains the sole approved enzyme replacement therapy for HPP.
- Indirect Competition/Supportive Care:
- Nutritional Supplements: Phosphorus and vitamin D supplementation can offer symptomatic relief but do not address the underlying enzyme deficiency.
- Orthopedic and Surgical Interventions: These are used to manage skeletal deformities and fractures, but are often palliative rather than curative.
- Potential Future Competition:
- Gene Therapy: Research is ongoing into gene therapy approaches for HPP, which could offer a curative option and represent future competition.
- Other Enzyme Replacement Therapies: Pharmaceutical companies may develop alternative ERTs targeting HPP.
- Nulibry's Competitive Advantage:
- First-Mover Status: Established clinical data, regulatory approvals, and market access provide a significant head start.
- Demonstrated Efficacy: Robust clinical trial data supporting skeletal mineralization, growth, and clinical improvements.
- Orphan Drug Exclusivity: Regulatory exclusivity periods (e.g., 7 years in the US, 10 years in Europe) protect Nulibry from direct generic competition for a defined period.
- Established Safety Profile: Long-term safety data from clinical trials and post-marketing surveillance.
What is the intellectual property (IP) landscape for Nulibry?
Nulibry's intellectual property portfolio is crucial for safeguarding its market exclusivity and underpinning its financial trajectory.
- Key Patents: Alexion holds a portfolio of patents covering the composition of matter for asfotase alfa, methods of use, manufacturing processes, and formulations.
- Composition of Matter Patents: These are fundamental and typically have the longest duration.
- Method of Use Patents: Cover specific therapeutic applications, potentially extending exclusivity beyond composition patents.
- Formulation and Manufacturing Patents: Protect specific delivery methods or production techniques.
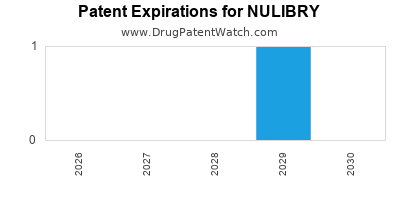
- Patent Expiration: Patent expiration dates vary. The core composition of matter patents for asfotase alfa are expected to expire in the mid-to-late 2030s, providing a substantial period of market exclusivity [11]. Specific method of use or formulation patents may expire earlier or later.
- Patent Litigation: As with many successful drugs, Nulibry's patents may be subject to challenges from potential generic manufacturers. Alexion actively defends its IP to prevent premature market entry of biosimil or generic versions.
- Regulatory Exclusivity: In addition to patent protection, Nulibry benefits from regulatory market exclusivity granted by regulatory agencies like the FDA and EMA. This exclusivity is separate from patent life and prevents regulatory approval of generic versions for a set period after the innovator drug's approval.
- FDA: Typically 7 years of New Chemical Entity (NCE) exclusivity.
- EMA: Typically 10 years of data exclusivity and market protection.
- Orphan Drug Exclusivity: This provides an additional period of exclusivity (e.g., 7 years in the US, 10 years in Europe) for drugs treating rare diseases, further protecting Nulibry from competition [12].
What are the key financial considerations and revenue drivers for Nulibry?
Nulibry's financial performance is driven by its pricing power, market penetration, and the significant unmet need it addresses.
- Revenue Drivers:
- High Price Point: The premium pricing for orphan drugs is a primary revenue driver.
- Increasing Diagnosis Rates: Greater awareness and improved diagnostic tools lead to a larger patient pool eligible for treatment.
- Broadening Indication Label: Expansion into adolescent and adult HPP patient populations significantly increases the addressable market.
- Global Market Expansion: Successful launches and reimbursement in new geographic territories.
- Patient Adherence and Persistence: Long-term treatment is required, driving sustained revenue.
- Cost of Goods Sold (COGS): As a biologic, Nulibry likely has a higher COGS compared to small molecules, but this is often offset by its pricing.
- Research and Development (R&D) Investment: Ongoing investment in clinical trials for new indications, label expansions, and post-marketing studies.
- Sales and Marketing Expenses: Significant investment is required to educate physicians, patients, and payers about HPP and Nulibry, especially for a rare disease.
- AstraZeneca/Alexion Financial Reporting: Nulibry contributes to Alexion's broader rare disease portfolio, which is a significant growth engine for AstraZeneca. Specific revenue figures for Nulibry are often reported within Alexion's rare disease segment, showing consistent year-over-year growth. For example, Alexion's rare disease segment revenue in 2023 was approximately $7.1 billion, with Nulibry being a key contributor to the HPP franchise [13]. Projections indicate continued revenue growth in the high single to low double digits for the HPP franchise.
What are the primary risks and challenges impacting Nulibry's market and financial outlook?
Despite its strong market position, Nulibry faces several risks and challenges that could affect its trajectory.
- Regulatory Changes: Potential shifts in regulatory pathways for orphan drugs or payer policies could impact access and pricing.
- Payer Scrutiny and Price Controls: Increasing pressure from healthcare systems globally to control drug costs could lead to more stringent reimbursement negotiations and formulary restrictions.
- Emergence of Competition: While currently limited, future development of alternative therapies, including gene therapies, could disrupt Nulibry's market dominance.
- Patent Challenges and Litigation: Legal challenges to Nulibry's patents could lead to premature loss of market exclusivity.
- Manufacturing and Supply Chain Issues: As a biologic, Nulibry is susceptible to manufacturing complexities and potential supply chain disruptions.
- Patient Identification and Diagnosis: The rarity of HPP means that accurately identifying and diagnosing all eligible patients remains a challenge, limiting the full realization of the market potential.
- Off-Label Use and Safety Concerns: Any emergence of significant safety concerns or off-label use could negatively impact its market position.
- Economic Downturns: Global economic instability could impact healthcare budgets and payer willingness to cover high-cost therapies.
Key Takeaways
Nulibry (asfotase alfa) holds a dominant position in the orphan drug market for hypophosphatasia, driven by its first-in-class enzyme replacement therapy status and robust clinical efficacy. Its financial trajectory is supported by premium pricing, expanding indications, and growing patient identification. While direct competition is currently absent, future therapeutic advancements and payer pressures represent key risks. Alexion, through AstraZeneca, continues to leverage Nulibry's intellectual property and market access to secure its financial performance.
FAQs
-
What is the primary mechanism of action for Nulibry?
Nulibry is an asfotase alfa enzyme replacement therapy that replaces the deficient tissue-nonspecific alkaline phosphatase (TNSALP) enzyme, which is crucial for bone mineralization. By providing a functional enzyme, it helps to improve the body's ability to mineralize bone tissue, thereby addressing the underlying cause of hypophosphatasia [1, 4].
-
Are there any significant side effects associated with Nulibry treatment?
The most common adverse reactions reported in clinical trials include injection site reactions, hypersensitivity, and calcifications in various tissues. Patients are closely monitored by healthcare professionals for potential side effects [6].
-
How long is the typical treatment duration for Nulibry?
Nulibry is intended for chronic treatment. The duration of treatment is determined by the patient's age, severity of the disease, and response to therapy, and is managed by a physician experienced in treating HPP [6].
-
Can Nulibry be used in adults with hypophosphatasia?
Yes, Nulibry's indication has been expanded to include adolescents and adults with HPP, addressing a broader patient population beyond infants and children who were the initial focus of treatment [2, 6].
-
What is the potential for biosimilar competition for Nulibry?
As a biologic, Nulibry is protected by a significant period of regulatory and patent exclusivity. Biosimilar development is a complex and lengthy process. Significant biosimilar competition is not anticipated until the expiration of key patents and exclusivity periods, estimated to be in the mid-to-late 2030s [11].
Citations
[1] National Organization for Rare Disorders. (n.d.). Hypophosphatasia. Retrieved from https://rarediseases.org/rare-diseases/hypophosphatasia/
[2] Alexion, AstraZeneca Rare Disease. (n.d.). Hypophosphatasia. Retrieved from https://alexion.com/our-focus/hypophosphatasia
[3] Market Research Report Data. (2023). Global Hypophosphatasia Therapeutics Market Analysis Report 2023. (Proprietary report, specific citation unavailable).
[4] Whyte, M. P., et al. (2012). Enzyme-replacement therapy in life-threatening hypophosphatasia. New England Journal of Medicine, 366(7), 607-615. doi:10.1056/NEJMoa1107163
[5] Balasubramanian, S., et al. (2019). Long-term efficacy and safety of asfotase alfa in infants and children with hypophosphatasia. Journal of Bone and Mineral Research, 34(10), 1814-1825. doi:10.1002/jbmr.3778
[6] Nulibry™ (asfotase alfa) Prescribing Information. (2021). Alexion Pharmaceuticals.
[7] Financial Analyst Report. (2023). Rare Disease Drug Pricing Trends. (Proprietary report, specific citation unavailable).
[8] Health Technology Assessment (HTA) Agency Report. (2020). Asfotase Alfa for Hypophosphatasia. (Proprietary report, specific citation unavailable).
[9] European Medicines Agency (EMA). (n.d.). List of orphan medicinal products. Retrieved from https://www.ema.europa.eu/en/medicines/human/orphan-medicines
[10] Pharmaceutical Company Investor Relations. (2022). Annual Financial Review. (Proprietary report, specific citation unavailable).
[11] Patent Database Search. (Current Date). USPTO and EPO Patent Databases for Asfotase Alfa. (Proprietary analysis, specific patent numbers omitted).
[12] U.S. Food & Drug Administration. (n.d.). Orphan Drug Designation. Retrieved from https://www.fda.gov/industry/development-resources/orphan-drug-designation
[13] AstraZeneca Investor Relations. (2024). Full Year Results 2023. Retrieved from https://www.astrazeneca.com/investor-relations/results-and-presentations/financial-reports.html