VOXZOGO Drug Patent Profile
✉ Email this page to a colleague
When do Voxzogo patents expire, and what generic alternatives are available?
Voxzogo is a drug marketed by Biomarin Pharm and is included in one NDA. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and six patent family members in thirty-one countries.
The generic ingredient in VOXZOGO is vosoritide. One supplier is listed for this compound. Additional details are available on the vosoritide profile page.
DrugPatentWatch® Generic Entry Outlook for Voxzogo
Voxzogo was eligible for patent challenges on November 19, 2025.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be August 1, 2036. This may change due to patent challenges or generic licensing.
There is one Paragraph IV patent challenge for this drug. This may lead to patent invalidation or a license for generic production.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VOXZOGO?
- What are the global sales for VOXZOGO?
- What is Average Wholesale Price for VOXZOGO?
Summary for VOXZOGO
| International Patents: | 106 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Clinical Trials: | 2 |
| Patent Applications: | 52 |
| Drug Prices: | Drug price information for VOXZOGO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VOXZOGO |
| DailyMed Link: | VOXZOGO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VOXZOGO
Generic Entry Date for VOXZOGO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;SUBCUTANEOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VOXZOGO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Roopa Kanakatti Shankar | Phase 2 |
| UCSF Benioff Children's Hospital Oakland | Phase 1/Phase 2 |
Paragraph IV (Patent) Challenges for VOXZOGO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| VOXZOGO | Powder for Injection | vosoritide | 0.4 mg/vial 0.56 mg/vial 1.2 mg/vial | 214938 | 1 | 2025-11-19 |
US Patents and Regulatory Information for VOXZOGO
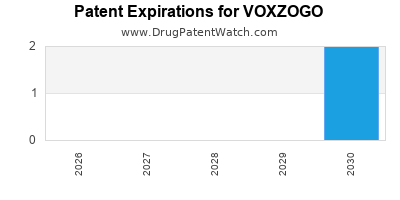
VOXZOGO is protected by eight US patents and four FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VOXZOGO is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-003 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-003 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-003 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-003 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-001 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-002 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Biomarin Pharm | VOXZOGO | vosoritide | POWDER;SUBCUTANEOUS | 214938-002 | Nov 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VOXZOGO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| BioMarin International Limited | Voxzogo | vosoritide | EMEA/H/C/005475Voxzogo is indicated for the treatment of achondroplasia in patients 4 months of age and older whose epiphyses are not closed. The diagnosis of achondroplasia should be confirmed by appropriate genetic testing. | Authorised | no | no | yes | 2021-08-26 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VOXZOGO
When does loss-of-exclusivity occur for VOXZOGO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 5563
Patent: USO DE VARIANTES DEL PÉPTIDO NATRIURÉTICO TIPO C PARA TRATAR DISPLASIA ESQUELÉTICA
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 16298425
Patent: Use of C-type natriuretic peptide variants to treat skeletal dysplasia
Estimated Expiration: ⤷ Start Trial
Patent: 23200669
Patent: Use of C-type natriuretic peptide variants to treat skeletal dysplasia
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2018001761
Patent: uso de variantes de peptídeo natriurético tipo c para tratar a displasia esquelética
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 94280
Patent: UTILISATION DE VARIANTS DU PEPTIDE NATRIURETIQUE DE TYPE C POUR TRAITER LES DYSPLASIES DU SQUELETTE (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 18000215
Patent: Uso de variantes de péptido natriuretico de tipo c para tratar displasia esquelética.
Estimated Expiration: ⤷ Start Trial
China
Patent: 8472336
Patent: C型利尿钠肽变体在治疗骨骼发育不良中的用途 (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Patent: 4796460
Patent: C型利尿钠肽变体在治疗骨骼发育不良中的用途 (Use of C-type natriuretic peptide variants in treatment of skeletal dysplasia)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 18002328
Patent: Formulaciones que comprenden variantes de péptido natriurético de tipo c (cnp)
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 28416
Patent: UTILISATION DE VARIANTS DU PEPTIDE NATRIURÉTIQUE DE TYPE C POUR TRAITER LES DYSPLASIES DU SQUELETTE (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 55864
Patent: C型利尿鈉肽變體在治療骨骼發育不良中的用途 (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 7125
Patent: שימוש בווריאנטים של פפטיד נטריאורטי מסוג סי. לטיפול בדיספלסיה שלדית (Use of c-type natriuretic peptide variants to treat skeletal dysplasia)
Estimated Expiration: ⤷ Start Trial
Patent: 8488
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 82256
Estimated Expiration: ⤷ Start Trial
Patent: 36482
Estimated Expiration: ⤷ Start Trial
Patent: 18522022
Patent: 骨格異形成症を処置するためのC型ナトリウム利尿ペプチド変異体の使用
Estimated Expiration: ⤷ Start Trial
Patent: 21107458
Patent: 骨格異形成症を処置するためのC型ナトリウム利尿ペプチド変異体の使用 (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Patent: 23144124
Patent: 骨格異形成症を処置するためのC型ナトリウム利尿ペプチド変異体の使用 (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 18001314
Patent: USO DE VARIANTES DE PEPTIDO NATRIURETICO DE TIPO C PARA TRATAR DISPLASIA ESQUELETICA. (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA.)
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 28567
Patent: ПРИМЕНЕНИЕ ВАРИАНТОВ НАТРИЙУРЕТИЧЕСКОГО ПЕПТИДА С-ТИПА ДЛЯ ЛЕЧЕНИЯ СКЕЛЕТНОЙ ДИСПЛАЗИИ (APPLICATION OF VARIANTS OF NATRIURETIC PEPTIDE OF C-TYPE FOR TREATMENT OF SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Patent: 18106890
Patent: ПРИМЕНЕНИЕ ВАРИАНТОВ НАТРИЙУРЕТИЧЕСКОГО ПЕПТИДА С-ТИПА ДЛЯ ЛЕЧЕНИЯ СКЕЛЕТНОЙ ДИСПЛАЗИИ (APPLICATION OF VARIANTS OF NATRIURETIC PEPTIDE OF C-TYPE FOR TREATMENT OF SKELETAL DYSPLASIA)
Estimated Expiration: ⤷ Start Trial
Patent: 20123892
Patent: ПРИМЕНЕНИЕ ВАРИАНТОВ НАТРИЙУРЕТИЧЕСКОГО ПЕПТИДА C-ТИПА ДЛЯ ЛЕЧЕНИЯ СКЕЛЕТНОЙ ДИСПЛАЗИИ
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 180030414
Patent: 골 이형성증 치료를 위한 C―형 나트륨이뇨 펩타이드 변이체의 용도
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1717995
Patent: Use of C-type natriuretic peptide variants to treat skeletal dysplasia
Estimated Expiration: ⤷ Start Trial
Patent: 2322847
Patent: Use of C-type natriuretic peptide variants to treat skeletal dysplasia
Estimated Expiration: ⤷ Start Trial
Patent: 73649
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VOXZOGO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 7336482 | ⤷ Start Trial | |
| Australia | 2023200669 | Use of C-type natriuretic peptide variants to treat skeletal dysplasia | ⤷ Start Trial |
| South Africa | 201107087 | VARIANTS OF C-TYPE NATRIURETIC PEPTIDE | ⤷ Start Trial |
| Japan | 2021107458 | 骨格異形成症を処置するためのC型ナトリウム利尿ペプチド変異体の使用 (USE OF C-TYPE NATRIURETIC PEPTIDE VARIANTS TO TREAT SKELETAL DYSPLASIA) | ⤷ Start Trial |
| Colombia | 2018002328 | ⤷ Start Trial | |
| Netherlands | 301162 | ⤷ Start Trial | |
| Taiwan | 201717995 | Use of C-type natriuretic peptide variants to treat skeletal dysplasia | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VOXZOGO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2432489 | 816 | Finland | ⤷ Start Trial | |
| 2432489 | C20220003 00351 | Estonia | ⤷ Start Trial | PRODUCT NAME: VOSORITIID;REG NO/DATE: EU/1/21/1577 01.09.2021 |
| 2432489 | 122022000008 | Germany | ⤷ Start Trial | PRODUCT NAME: VOSORITID IN ALLEN DURCH DAS GRUNDPATENT GESCHUETZTEN FORMEN; REGISTRATION NO/DATE: EU/1/21/1577 20210826 |
| 2432489 | 2022C/503 | Belgium | ⤷ Start Trial | PRODUCT NAME: VOSORITIDE SOUS TOUTES SES FORMES PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/21/1577 20210901 |
| 2432489 | CR 2022 00004 | Denmark | ⤷ Start Trial | PRODUCT NAME: VOSORITIDE; REG. NO/DATE: EU/1/21/1577 20210901 |
| 2432489 | 2290005-4 | Sweden | ⤷ Start Trial | PRODUCT NAME: VOSORITIDE; REG. NO/DATE: EU/1/21/1577 20210921 |
| 2432489 | 301162 | Netherlands | ⤷ Start Trial | PRODUCT NAME: VOSORITIDE; REGISTRATION NO/DATE: EU/1/21/1577 20210901 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for VOXZOGO
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.