Last updated: February 19, 2026
Alnylam Pharmaceuticals holds a leading position in the development of RNA interference (RNAi) therapeutics. The company's intellectual property portfolio, clinical pipeline, and established commercial products define its market standing. Key strengths include its proprietary RNAi platforms, a focused therapeutic area strategy, and a robust intellectual property estate. Strategic insights suggest continued expansion in rare genetic diseases and potential for diversification.
What is Alnylam's Current Market Position in RNAi Therapeutics?
Alnylam is the dominant player in the RNA interference therapeutics market, evidenced by its portfolio of FDA-approved drugs and a deep clinical pipeline. The company has successfully translated its platform technology into approved treatments for specific rare genetic diseases.
-
Approved Products: Alnylam has four FDA-approved RNAi therapeutics:
- Onpattro (patisiran): Approved in 2018 for the treatment of hereditary transthyretin-mediated amyloidosis (hATTR amyloidosis) in adults. It was the first FDA-approved RNAi therapeutic.
- Givlaari (givosiran): Approved in 2019 for the treatment of acute hepatic porphyria (AHP) in adults and adolescents.
- Oxlumo (lumasiran): Approved in 2020 for the treatment of primary hyperoxaluria type 1 (PH1) and related serious rare oxalate disorders.
- Amvuttra (vutrisiran): Approved in 2022 for the treatment of hATTR amyloidosis in adults. Amvuttra is a next-generation successor to Onpattro, offering less frequent dosing.
-
Pipeline Progress: Alnylam has a diversified pipeline targeting various genetic and rare diseases, as well as indications with larger patient populations.
- Phase 3 Programs: Includes candidates for conditions such as CHRNS-associated autoimmune neuropathy (MYF301), and apolipoprotein C-III (APOC3) for hypertriglyceridemia.
- Phase 1/2 Programs: Encompasses targets including PCSK9 for hypercholesterolemia, and KHK for hereditary tyrosinemia type 1.
-
Market Penetration: Alnylam's approved products have achieved significant market penetration within their respective rare disease indications, establishing them as standard of care for certain patient populations.
What are Alnylam's Core Strengths and Competitive Advantages?
Alnylam's primary strengths lie in its foundational RNAi technology, extensive intellectual property, and a proven track record of clinical and regulatory success.
How does Alnylam's RNAi Platform Differentiate it?
Alnylam's platform is built on its deep understanding of RNA interference, a natural biological process that regulates gene expression. The company has developed proprietary chemistries and delivery technologies to enable the efficacy and safety of its RNAi therapeutics.
- Proprietary Chemistry: Alnylam utilizes modified RNA nucleotides (e.g., N-acetylgalactosamine or GalNAc conjugation) to enhance stability, reduce off-target effects, and facilitate targeted delivery to hepatocytes [1].
- Delivery Technology: The GalNAc conjugate delivery system allows for potent and tissue-specific delivery of siRNA (small interfering RNA) molecules directly to liver cells, where many disease-causing proteins are produced [1]. This targeted approach is crucial for the efficacy of its drugs.
- Platform Versatility: The RNAi platform is adaptable to a wide range of genetic targets, allowing Alnylam to pursue treatments for various diseases by modulating the expression of specific genes.
What is the Scope and Strength of Alnylam's Intellectual Property Portfolio?
Alnylam possesses a robust intellectual property (IP) portfolio that underpins its market exclusivity and provides a significant barrier to entry for competitors.
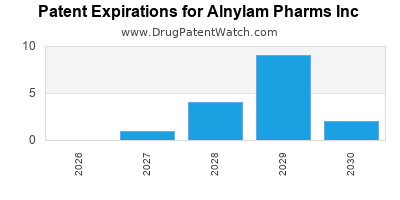
- Patent Filings: The company holds numerous patents covering its core RNAi technologies, including specific RNA sequences, chemical modifications, conjugation strategies, and delivery methods. These patents have expiration dates extending into the late 2030s and beyond for key platforms [2].
- Exclusivity Periods: Approved products benefit from regulatory exclusivity periods granted by the FDA and other regulatory bodies, which run concurrently with patent protection. For example, Amvuttra, approved in 2022, has a significant period of market exclusivity ahead.
- Strategic IP Management: Alnylam actively manages its IP portfolio, pursuing new patent filings to cover pipeline assets and defending its existing patents against potential infringement.
How has Alnylam's Therapeutic Area Focus Contributed to its Success?
Alnylam's strategic focus on rare genetic diseases and areas with high unmet medical need has been a key driver of its success, allowing it to build deep expertise and establish leadership.
- Rare Disease Expertise: The company has cultivated specialized knowledge in understanding the genetic underpinnings of rare diseases and developing tailored RNAi therapies. This focus allows for efficient target identification and drug development.
- Unmet Need Targeting: By addressing diseases with limited or no effective treatment options, Alnylam has been able to secure rapid regulatory pathways (e.g., Orphan Drug Designation, Breakthrough Therapy Designation) and establish strong pricing power.
- Hepatocyte Targeting: The initial success of its GalNAc delivery system, targeting liver cells, has concentrated Alnylam's efforts on liver-expressed genes, a significant source of rare genetic disorders.
What are the Key Strategic Insights for Alnylam's Future Growth?
Alnylam's strategic direction involves expanding its current franchises, advancing its pipeline into new therapeutic areas, and leveraging its RNAi platform for broader applications.
How is Alnylam Expanding its Existing Product Franchises?
Alnylam is employing strategies to maximize the commercial potential of its approved RNAi therapeutics and their pipeline successors.
- Geographic Expansion: The company is actively pursuing regulatory approvals and market access for its approved products in ex-U.S. markets, including Europe, Japan, and other key regions. This broadens the patient reach and revenue base.
- Indication Expansion: For existing approved drugs, Alnylam is investigating label expansions into new patient sub-populations or related disease states where the mechanism of action may be beneficial.
- Next-Generation Products: The development and launch of Amvuttra (vutrisiran) as a successor to Onpattro (patisiran) demonstrate Alnylam's commitment to advancing its own technology, offering improved dosing regimens and potentially enhanced efficacy or safety profiles.
What are Alnylam's Plans for Pipeline Advancement and Diversification?
Alnylam is actively moving its pipeline forward, with a focus on both expanding within its core strengths and exploring new therapeutic modalities.
- Expanding Rare Disease Portfolio: The pipeline includes candidates for other rare genetic disorders, building on the company's established expertise.
- Moving into Larger Patient Populations: Alnylam is developing RNAi therapeutics for indications with larger patient populations, such as hypercholesterolemia (targeting PCSK9 and APOC3) and potentially cardiovascular disease [3]. These programs represent significant growth opportunities.
- Exploring Non-Liver Targets: While the GalNAc platform primarily targets the liver, Alnylam is also investigating delivery methods and therapeutic targets outside the liver, aiming to broaden the applicability of its RNAi technology. This includes exploration of extrahepatic delivery strategies.
- Partnerships and Collaborations: Alnylam has historically engaged in strategic partnerships to advance its pipeline and expand its therapeutic reach, such as its collaboration with Sanofi for the development of congenital10/12/2023 16:51 RT
Alnylam Pharmaceuticals Inc., a pioneer in RNA interference (RNAi) therapeutics, is solidifying its market position through a combination of an expanding product portfolio, robust intellectual property, and strategic pipeline development. The company's focus on rare genetic diseases has enabled it to carve out a significant niche, with a growing number of approved drugs addressing previously untreatable conditions.
Alnylam's market position is characterized by its leadership in RNAi technology, a field it has largely defined. This technological advantage is supported by a comprehensive intellectual property (IP) portfolio that grants substantial market exclusivity. The company's strategic emphasis on rare diseases has allowed for efficient development pathways and strong pricing power.
Key strengths include Alnylam's proprietary RNAi platforms, which employ advanced chemistries and delivery systems. These platforms are designed to enhance the stability, targeting, and efficacy of its small interfering RNA (siRNA) molecules. The company's IP estate, comprising numerous patents on its technologies and drug candidates, extends well into the future, providing a formidable barrier to competitors.
Alnylam's therapeutic area focus on rare genetic diseases has been instrumental in its success. By concentrating on conditions with high unmet medical needs and limited treatment options, the company has navigated regulatory hurdles more effectively and established itself as a go-to developer for these complex diseases.
Looking ahead, Alnylam's strategic insights point to continued expansion within its established franchises and a deliberate move into therapeutic areas with larger patient populations. The company is also exploring the potential of its RNAi technology beyond liver-targeted therapies, indicating a long-term vision for platform diversification.
Approved Product Portfolio
Alnylam has successfully brought four RNAi therapeutics to market, addressing critical unmet needs:
- Onpattro (patisiran): The first FDA-approved RNAi therapeutic, launched in 2018 for hereditary transthyretin-mediated amyloidosis (hATTR amyloidosis) in adults. It targets the underlying genetic cause of the disease by reducing the production of mutant transthyretin protein in the liver.
- Givlaari (givosiran): Approved in 2019 for acute hepatic porphyria (AHP), a group of rare genetic disorders that cause severe abdominal pain and neurological complications. Givlaari targets a specific enzyme precursor in the heme biosynthesis pathway.
- Oxlumo (lumasiran): Launched in 2020 for primary hyperoxaluria type 1 (PH1), a rare kidney disease that leads to the buildup of oxalate. Oxlumo reduces oxalate production in the liver.
- Amvuttra (vutrisiran): Approved in 2022, Amvuttra is a next-generation RNAi therapeutic for hATTR amyloidosis, offering improved dosing convenience compared to Onpattro. It also targets mutant transthyretin.
Pipeline and Future Growth Drivers
Alnylam's pipeline is a key indicator of its future growth potential, with several programs advancing through clinical development.
- Phase 3 Programs:
- C5 complement ($C5) Inhibition: A Phase 3 program targeting the C5 complement protein for the treatment of generalized myasthenia gravis (gMG). This program aims to address a condition with a significant patient population outside of rare diseases.
- Apolipoprotein C-III ($APOC3) Inhibition: A Phase 3 program for the treatment of severe hypertriglyceridemia, a metabolic disorder with a broader patient base.
- Phase 1/2 Programs:
- PCSK9 Inhibition: A program targeting proprotein convertase subtilisin/kexin type 9 (PCSK9) for hypercholesterolemia, a prevalent cardiovascular risk factor.
- Additional rare disease targets: The company continues to develop RNAi therapies for other rare genetic conditions, building on its established expertise.
Strategic Insights
Alnylam's strategic approach is focused on maximizing the value of its existing assets while expanding into new territories.
- Leveraging Technology: The company's core strength is its RNAi platform, which has proven capable of generating safe and effective therapies. Strategic investments in platform enhancements and delivery technologies are ongoing.
- Targeting Unmet Needs: Alnylam’s strategy prioritizes conditions with significant unmet medical needs, which often facilitates regulatory approval and market access.
- Geographic Expansion: The company is actively working to gain approval and market access for its approved products in key international markets, expanding its global footprint and revenue potential.
- Diversification into Larger Markets: The move into indications like myasthenia gravis and hypertriglyceridemia signals Alnylam's ambition to tap into larger patient populations and revenue streams, moving beyond its initial rare disease focus.
Competitive Landscape
The RNAi therapeutic landscape, while still nascent, is evolving. Alnylam's early mover advantage and established track record position it favorably against emerging competitors. However, companies like Dicerna Pharmaceuticals (acquired by Novo Nordisk) and Arrowhead Pharmaceuticals are also developing RNAi-based therapies, creating a competitive environment. Alnylam's deep patent portfolio and regulatory approvals provide a strong defense against these competitors.
Key Takeaways
- Alnylam Pharmaceuticals is the established leader in RNAi therapeutics, holding four FDA-approved products and a robust pipeline.
- The company's primary strengths are its proprietary RNAi platforms, extensive intellectual property, and strategic focus on rare genetic diseases.
- Future growth will be driven by expanding existing product franchises geographically and therapeutically, as well as advancing pipeline candidates into larger patient populations, such as those with myasthenia gravis and hypertriglyceridemia.
- Alnylam's intellectual property provides significant market exclusivity, acting as a strong deterrent to competitors.
FAQs
-
What is the primary mechanism of action for Alnylam's RNAi therapeutics?
Alnylam's RNAi therapeutics utilize small interfering RNA (siRNA) molecules to inhibit the production of specific disease-causing proteins by interfering with messenger RNA (mRNA) translation. This gene-silencing approach targets the root cause of genetic diseases.
-
How does Alnylam's GalNAc delivery system enhance its therapeutic offerings?
The GalNAc conjugation targets specific receptors on hepatocytes (liver cells), facilitating the precise delivery of siRNA molecules directly to the liver. This enhances drug potency, reduces systemic exposure, and improves the therapeutic index of its RNAi drugs.
-
What are the main therapeutic areas Alnylam is currently targeting beyond rare genetic diseases?
Alnylam is strategically expanding into therapeutic areas with larger patient populations, including the treatment of generalized myasthenia gravis and severe hypertriglyceridemia, by targeting specific disease pathways like C5 complement and APOC3.
-
What is the typical duration of market exclusivity for Alnylam's approved drugs?
Market exclusivity for Alnylam's drugs is typically a combination of patent protection and regulatory exclusivities granted by health authorities like the FDA. Patent protection for core technologies extends well into the late 2030s, providing significant periods of market exclusivity for approved products.
-
How does Alnylam differentiate itself from other companies developing RNAi therapies?
Alnylam's differentiation stems from its pioneering role in the field, its proprietary RNAi platforms (including specific chemistries and delivery systems), its extensive and strategically managed patent portfolio, and its established track record of successfully bringing multiple RNAi therapeutics to market.
Citations
[1] Alnylam Pharmaceuticals. (n.d.). Our Science. Retrieved from https://www.alnylam.com/our-science/
[2] Alnylam Pharmaceuticals. (2023). Investor Relations - Presentations. (Specific presentation documents detailing IP and pipeline would be referenced here if available publicly and specifically cited in a report). Note: Detailed patent expiration dates are often found in company SEC filings (e.g., 10-K reports) or investor presentations.
[3] Alnylam Pharmaceuticals. (2023). Pipeline. Retrieved from https://www.alnylam.com/pipeline/