VERQUVO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Verquvo, and when can generic versions of Verquvo launch?
Verquvo is a drug marketed by MSD and is included in one NDA. There are six patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and fourteen patent family members in fifty countries.
The generic ingredient in VERQUVO is vericiguat. One supplier is listed for this compound. Additional details are available on the vericiguat profile page.
DrugPatentWatch® Generic Entry Outlook for Verquvo
Verquvo was eligible for patent challenges on January 19, 2025.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 19, 2031. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are two tentative approvals for the generic drug (vericiguat), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VERQUVO?
- What are the global sales for VERQUVO?
- What is Average Wholesale Price for VERQUVO?
Summary for VERQUVO
| International Patents: | 214 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 52 |
| Clinical Trials: | 2 |
| Patent Applications: | 281 |
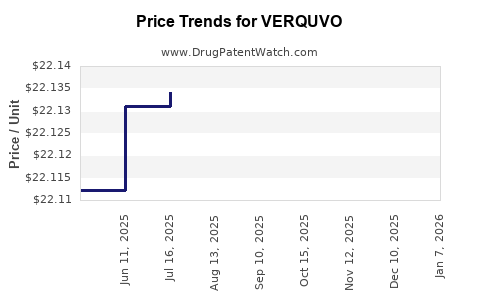
| Drug Prices: | Drug price information for VERQUVO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VERQUVO |
| What excipients (inactive ingredients) are in VERQUVO? | VERQUVO excipients list |
| DailyMed Link: | VERQUVO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VERQUVO
Generic Entry Date for VERQUVO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VERQUVO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Johns Hopkins University | Phase 2 |
| Merck Sharp & Dohme LLC | Phase 2 |
| Josef Stehlik | Phase 4 |
Pharmacology for VERQUVO
| Drug Class | Soluble Guanylate Cyclase Stimulator |
| Mechanism of Action | Guanylate Cyclase Stimulators |
Paragraph IV (Patent) Challenges for VERQUVO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| VERQUVO | Tablets | vericiguat | 5 mg and 10 mg | 214377 | 4 | 2025-01-21 |
US Patents and Regulatory Information for VERQUVO
VERQUVO is protected by six US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VERQUVO is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Msd | VERQUVO | vericiguat | TABLET;ORAL | 214377-001 | Jan 19, 2021 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Msd | VERQUVO | vericiguat | TABLET;ORAL | 214377-003 | Jan 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Msd | VERQUVO | vericiguat | TABLET;ORAL | 214377-003 | Jan 19, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VERQUVO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Bayer AG | Verquvo | vericiguat | EMEA/H/C/005319Treatment of symptomatic chronic heart failure | Authorised | no | no | no | 2021-07-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VERQUVO
When does loss-of-exclusivity occur for VERQUVO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1628
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 11257335
Patent: Substituted 5-fluoro-1H-pyrazolopyridines and use thereof
Estimated Expiration: ⤷ Start Trial
Patent: 11257336
Patent: The use of sGC stimulators, sGC activators, alone and combinations with PDE5 inhibitors for the treatment of systemic sclerosis (SSc).
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012030010
Patent: 5-flúor-1h-pirazolopiridinas substituídas e seu uso
Estimated Expiration: ⤷ Start Trial
Patent: 2020023470
Patent: PROCESSO PARA PREPARAÇÃO DE 5-FLÚOR-1H-PIRAZOLOPIRIDINAS SUBSTITUÍDAS
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 00697
Patent: 5-FLUORO-1H-PYRAZOLOPYRIDINES SUBSTITUEES ET LEUR UTILISATION (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 00709
Patent: UTILISATION DE STIMULATEURS DE LA SGC, D'ACTIVATEURS DE LA SGC, SEULS ET EN ASSOCIATION AVEC DES INHIBITEURS DE LA PDE5 EN VUE DU TRAITEMENT DE LA SCLERODERMIE SYSTEMIQUE (THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC))
Estimated Expiration: ⤷ Start Trial
Patent: 55143
Patent: UTILISATION DE STIMULATEURS DE LA SGC, D'ACTIVATEURS DE LA SGC, SEULS ET EN ASSOCIATION AVEC DES INHIBITEURS DE LA PDE5 EN VUE DU TRAITEMENT DE LA SCLERODERMIE (THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC).)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 12003226
Patent: Compuestos derivados de 5-fluoro-1h-pirazolopiridina ; procedimiento de preparacion de estos; composicion framaceutica que los comprende; y su uso en el tratamiento de enfermedades cardiovasculares, sexuales y metabolicas.
Estimated Expiration: ⤷ Start Trial
Patent: 12003281
Patent: Uso de compuestos específicos estimuladores y/o activadores de sgc; formulacion farmaceutica que comprende dichos compuestos; kit que comprende los compuestos, y uso para la prevencion y/o tratamiento de la esclerosis sistematica.
Estimated Expiration: ⤷ Start Trial
China
Patent: 2939289
Patent: Substituted 5-fluoro-1h-pyrazolopyridines and use thereof
Estimated Expiration: ⤷ Start Trial
Patent: 3038232
Patent: The use of sGC stimulators, sGC activators, alone and combinations with PDE5 inhibitors for the treatment of systemic sclerosis (SSc)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 40226
Patent: 5-fluoro-1h-pirazolipiridinas sustituidas y su uso
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 120596
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Patent: 120597
Patent: EL USO DE ESTIMULADORES DE LA sGC, ACTIVADORES DE LA sGC, SOLOS Y EN COMBINANCIÓN CON INHIBIDORES DE LA PDE5 PARA EL TRATAMIENTO DE ESCLEROSIS SISTÉMICA (EcS)
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0150987
Estimated Expiration: ⤷ Start Trial
Patent: 0160370
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 086
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS
Estimated Expiration: ⤷ Start Trial
Patent: 120161
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16703
Estimated Expiration: ⤷ Start Trial
Patent: 17372
Estimated Expiration: ⤷ Start Trial
Patent: 21021
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 76547
Estimated Expiration: ⤷ Start Trial
Patent: 76548
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 012000299
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 12012310
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 3631
Patent: ЗАМЕЩЕННЫЕ 5-ФТОР-1H-ПИРАЗОЛОПИРИДИНЫ И ИХ ПРИМЕНЕНИЕ (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 0735
Patent: ПРИМЕНЕНИЕ sGC СТИМУЛЯТОРОВ ДЛЯ ЛЕЧЕНИЯ СИСТЕМНОГО СКЛЕРОЗА (SSc) (USE OF sGC STIMULATORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSc))
Estimated Expiration: ⤷ Start Trial
Patent: 1291309
Patent: ПРИМЕНЕНИЕ sGC СТИМУЛЯТОРОВ, sGC АКТИВАТОРОВ, ОДНИХ И В КОМБИНАЦИИ С PDE5 ИНГИБИТОРАМИ, ДЛЯ ЛЕЧЕНИЯ СИСТЕМНОГО СКЛЕРОЗА (SSc)
Estimated Expiration: ⤷ Start Trial
Patent: 1291336
Patent: ЗАМЕЩЕННЫЕ 5-ФТОР-1Н-ПИРАЗОЛОПИРИДИНЫ И ИХ ПРИМЕНЕНИЕ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 76547
Patent: 5-FLUORO-1H-PYRAZOLOPYRIDINES SUBSTITUÉES ET LEUR UTILISATION (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 76548
Patent: UTILISATION DE STIMULATEURS DE LA sGC, D'ACTIVATEURS DE LA sGC, SEULS ET EN ASSOCIATION AVEC DES INHIBITEURS DE LA PDE5 EN VUE DU TRAITEMENT DE LA SCLÉRODERMIE SYSTÉMIQUE (THE USE OF sGC STIMULATORS, sGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSc))
Estimated Expiration: ⤷ Start Trial
France
Patent: C1038
Estimated Expiration: ⤷ Start Trial
Germany
Patent: 2010021637
Patent: Substituierte 5-Fluor-1H-Pyrazolopyridine und ihre Verwendung
Estimated Expiration: ⤷ Start Trial
Guatemala
Patent: 1200313
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 82112
Patent: 取代的 -氟- -吡唑並吡啶類化合物及其用途 (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF 5--1H-)
Estimated Expiration: ⤷ Start Trial
Patent: 83868
Patent: 單獨的和與 抑制劑相組合的 刺激劑、 活化劑用於治療系統性硬化症 的用途 (THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC). PDE5 SGC SGC (SSC))
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 25162
Estimated Expiration: ⤷ Start Trial
Patent: 26912
Estimated Expiration: ⤷ Start Trial
Patent: 100032
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 3050
Patent: 5- פלואורו - 1h - פיראזולופירידינים מותמרים והשימוש בהם (Substituted 5 -fluoro-1h- pyrazolopyridines and use thereof)
Estimated Expiration: ⤷ Start Trial
Patent: 3128
Patent: שימוש במגרי sgc, מפעילי sgc לבד וצירופים עם מעכבי 5pde לטיפול בטרשת מערכתית (Use of sgc stimulators, sgc activators. alone and in combinations with pde5 inhibitors for the treatment of systemic sclerosis (ssc))
Estimated Expiration: ⤷ Start Trial
Patent: 9669
Patent: 5-פלואורו-h1-פיראזולופירידינים מותמרים והשימוש בהם (Substituted 5-fluoro-1h-pyrazolopyridines and use thereof)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 83852
Estimated Expiration: ⤷ Start Trial
Patent: 96991
Estimated Expiration: ⤷ Start Trial
Patent: 13526598
Estimated Expiration: ⤷ Start Trial
Patent: 13530150
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 99
Patent: مركبات 5- فلورو-1H- بيرازولوبيريدينات مستبدلة واستخدامها (Substituted 5-fluoro-1H-pyrazolopyridines and their use)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 576547
Estimated Expiration: ⤷ Start Trial
Patent: 2021518
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0221
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 4356
Patent: SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND THEIR USE
Estimated Expiration: ⤷ Start Trial
Patent: 0094
Patent: THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC).
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 12013324
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO. (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF.)
Estimated Expiration: ⤷ Start Trial
Patent: 12013574
Patent: EL USO DE ESTIMULADORES DE LA SGC, ACTIVADORES DE LA SGC, SOLOS Y EN COMBINACION CON INHIBIDORES DE LA PDE5 PARA EL TRATAMIENTO DE ESCLEROSIS SISTEMICA (ECS). (THE USE OF sGC STIMULATORS, sGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSc).)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 207
Patent: UPOTREBA sGC STIMULATORA, sGC AKTIVATORA, POJEDINAČNO I U KOMBINACIJI SA PDE5 INHIBITORIMA ZA TRETMAN SISTEMSKE SKLEROZE (SSc) (THE USE OF sGC STIMULATORS, sGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS)
Estimated Expiration: ⤷ Start Trial
Patent: 393
Patent: SUPSTITUISANI 5-FLUOR-1H-PIRAZOLOPIRIDINI I NJIHOVA PRIMJENA (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 248
Patent: 5-FLUORO-1H-PYRAZOLOPYRIDINES SUBSTITUÉES ET LEUR UTILISATION
Estimated Expiration: ⤷ Start Trial
Patent: 249
Patent: UTILISATION DE STIMULATEURS DE LA SGC, D'ACTIVATEURS DE LA SGC, SEULS ET EN ASSOCIATION AVEC DES INHIBITEURS DE LA PDE5 EN VUE DU TRAITEMENT DE LA SCLÉRODERMIE SYSTÉMIQUE
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 1146
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3799
Patent: The use of sgc stimulators, sgc activators, alone and combinations with pde5 inhibitors for the treatment of systemic sclerosis (ssc).
Estimated Expiration: ⤷ Start Trial
Patent: 3800
Patent: Substituted 5-fluoro-1h-pyrazolopyridines and use thereof
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 21032
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 130402
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 012502320
Patent: SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 012502322
Patent: THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 76547
Estimated Expiration: ⤷ Start Trial
Patent: 76548
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 76548
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 261
Patent: UPOTREBA SGC STIMULATORA, SGC AKTIVATORA, POJEDINAČNO I U KOMBINACIJI SA PDE5 INHIBITORIMA ZA TRETMAN SISTEMSKE SKLEROZE (SSC) (THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC))
Estimated Expiration: ⤷ Start Trial
Patent: 704
Patent: SUPSTITUISANI 5-FLUOR-1H-PIRAZOLOPIRIDINI I NJIHOVA PRIMENA (SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 5460
Patent: SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 5690
Patent: THE USE OF sGC STIMULATORS, sGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSc).
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 76547
Estimated Expiration: ⤷ Start Trial
Patent: 76548
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1208824
Patent: THE USE OF SGC STIMULATORS,SGC ACTIVATORS,ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC)
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1862420
Estimated Expiration: ⤷ Start Trial
Patent: 1881174
Estimated Expiration: ⤷ Start Trial
Patent: 130116000
Patent: SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 130116001
Patent: THE USE OF SGC STIMULATORS, SGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSC)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 49979
Estimated Expiration: ⤷ Start Trial
Patent: 67795
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1210592
Patent: Substituted 5-fluoro-1H-pyrazolopyridines and their use
Estimated Expiration: ⤷ Start Trial
Patent: 16263
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 12000549
Patent: SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES AND USE THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 12000550
Patent: THE USE OF sGC STIMULATORS, sGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSc).
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 9660
Estimated Expiration: ⤷ Start Trial
Patent: 6521
Patent: ЗАСТОСУВАННЯ SGC-СТИМУЛЯТОРІВ, SGC-АКТИВАТОРІВ ОКРЕМО І В КОМБІНАЦІЇ З ІНГІБІТОРАМИ ФДЕ5 ДЛЯ ЛІКУВАННЯ СИСТЕМНОЇ СКЛЕРОДЕРМІЇ (SSC) (THE USE OF sGC STIMULATORS, sGC ACTIVATORS, ALONE AND COMBINATIONS WITH PDE5 INHIBITORS FOR THE TREATMENT OF SYSTEMIC SCLEROSIS (SSc))
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 396
Patent: 5-FLUORO-1H-PIRAZOLOPIRIDINAS SUSTITUIDAS Y SU USO
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VERQUVO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2011147810 | ⤷ Start Trial | |
| Malaysia | 170094 | ⤷ Start Trial | |
| South Korea | 20190018021 | 치환된 5-플루오로-1H-피라졸로피리딘의 제조 방법 (5--1H- METHOD FOR PRODUCING SUBSTITUTED 5-FLUORO-1H-PYRAZOLOPYRIDINES) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VERQUVO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2576547 | 132021000000137 | Italy | ⤷ Start Trial | PRODUCT NAME: VERICIGUAT(VERQUVO); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/21/1561, 20210720 |
| 2576547 | 301146 | Netherlands | ⤷ Start Trial | PRODUCT NAME: VERICIGUAT; REGISTRATION NO/DATE: EU/1/21/1561 20210720 |
| 2576547 | 2190031-1 | Sweden | ⤷ Start Trial | PRODUCT NAME: VERICIGUAT AND ITS SALTS, SOLVATES AND SOLVATES OF THE SALTS; REG. NO/DATE: EU/1/21/1561 20210720 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for VERQUVO (sacitoclax)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.