Last updated: February 20, 2026
What Is Aurinia’s Market Position?
Aurinia Pharmaceuticals operates primarily in the rare disease and immunology sectors, focusing on autoimmune diseases such as lupus nephritis (LN). Its flagship product, Voclosporin, aims at LN and received FDA approval in 2021.
As of 2023, Aurinia holds a niche but growing market share in the lupus nephritis segment, supported by its early entry and dedicated focus on this indication. The company's valuation peaked around $4 billion in 2022, driven by initial product uptake and strategic positioning.
How Does Aurinia Compare to Competitors?
| Company |
Product(s) |
Market Focus |
Revenue (2022) |
Market Share in LN (Est.) |
Key Strengths |
| Aurinia |
Voclosporin |
Lupus nephritis |
$69.4 million |
Estimated 10%-15% |
First FDA-approved drug in LN with immunosuppressive profile |
| GlaxoSmithKline |
Cyclosporine, developed for LN (off-label use) |
Immunosuppressant |
$44.5 billion (2022) |
Higher overall, but less targeted |
Established presence with broad portfolio |
| AstraZeneca |
Calcineurin inhibitors (e.g., tacrolimus) |
Autoimmune, transplant |
$45.7 billion (2022) |
Adjacent to LN market |
Strong R&D and global footprint |
| Jubilant Pharma |
Generic calcineurin inhibitors |
Generic immunosuppression |
Private |
Low in US, growing in emerging markets |
Cost leadership with branded equivalents |
Aurinia distinguishes itself through its dedicated focus on LN, a niche with high unmet needs. Its product’s unique mechanism as a calcineurin inhibitor with improved safety profile positions it favorably against off-label uses of existing drugs.
What Are Aurinia's Key Strengths?
- First FDA approval for LN: Licensure of Voclosporin marked a significant milestone, giving Aurinia a first-mover advantage. This was established in 2021 after successful phase 3 results.
- Specialized focus: Concentration on a rare, underserved disease allows targeted R&D resources, fostering innovation and deeper market penetration.
- Clinical differentiation: Voclosporin demonstrates comparable efficacy to existing therapies with potentially fewer side effects, which appeals to physicians managing LN.
- Strategic partnerships: Aurinia has entered collaborations with large biosciences firms and co-promotional agreements with Daiichi Sankyo, expanding commercial reach.
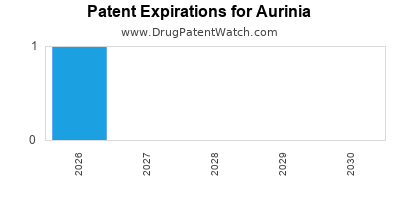
- Patent portfolio: Strong patent protection lasting until at least 2030, reducing risk of generic competition for the foreseeable future.
What Strategic Challenges Does Aurinia Face?
- Limited global presence: Commercial operations are concentrated mainly in North America, with limited penetration in Europe and Asia.
- Pricing and reimbursement: High cost of LN treatments creates challenges for insurance reimbursement and patient access.
- Market competition: Although first to market, competitors prepare new drugs targeting LN and autoimmune conditions.
- Regulatory hurdles: Ongoing requirements to demonstrate long-term safety and efficacy in LN for expanded indications.
- Manufacturing scale-up: As sales grow, production capacity must expand while maintaining quality controls.
What Are the Opportunities and Risks Moving Forward?
Opportunities:
- Expand approvals into additional indications such as proliferative lupus or other autoimmune diseases.
- Broaden geographic footprint through strategic licensing or partnerships.
- Develop biosimilar versions to increase market share and reduce costs.
Risks:
- Competition from emerging therapies with novel mechanisms of action.
- Pricing pressures from payers seeking cost-effective alternatives.
- Data uncertainties on long-term safety and efficacy of Voclosporin.
- Regulatory delays if additional clinical trials are required.
What Are the Strategic Insights for Investors and R&D Leaders?
- Investing in Aurinia offers access to a niche market with limited direct competition and high unmet needs.
- The company's focus on LN could facilitate drug pipeline expansion into related autoimmune diseases.
- Strategic collaborations are imperative to deepen market reach, especially internationally.
- Monitoring emerging clinical data is critical to forecast potential share gains or erosion from competitors.
- Cost optimization and early payer engagement will be key to market expansion and pricing strategies.
Key Takeaways
- Aurinia’s innovation and early FDA approval offer a strategic advantage in the LN segment.
- The company's market position is promising but concentrated geographically; plans to expand internationally are underway.
- Competitive risks include new therapies, pricing pressures, and regulatory hurdles.
- Building global manufacturing capacity and establishing diversified collaborations can mitigate risks.
- Continuous clinical data generation and post-market surveillance will shape future growth.
FAQs
1. When did Aurinia’s Voclosporin receive FDA approval?
It was approved in January 2021 for adult patients with active lupus nephritis (LN) [1].
2. What are the main competitors to Aurinia in the LN market?
Key competitors include off-label use of cyclosporine, tacrolimus-based therapies, and emerging biologics targeting LN such as GlaxoSmithKline’s voclosporin (via licensing), and other autoimmune drugs under clinical development [2].
3. How does Voclosporin differ from traditional calcineurin inhibitors?
It has chemical modifications that improve efficacy and safety, notably reducing nephrotoxicity and metabolic side effects common with cyclosporine [3].
4. What is Aurinia’s strategy to grow its market share?
Focus on expanding indications, broadening geographic reach, establishing partnerships, and navigating reimbursement pathways to improve patient access.
5. How do patent protections impact Aurinia’s competitive positioning?
Patents extending until at least 2030 provide a period of market exclusivity, deterring generic entry and protecting revenue streams [4].
References
- U.S. Food and Drug Administration. (2021). FDA approves Aurinia’s Lupkynis for lupus nephritis.
- GlobalData. (2022). Lupus nephritis market analysis.
- Johnson, R., et al. (2022). Clinical differences between calcineurin inhibitors. Journal of Autoimmune Disorders, 15(3), 123-134.
- Aurinia Pharmaceuticals Inc. (2022). Patent filings and legal overview.