Last updated: February 19, 2026
Myfembree (relugolix, estradiol, and norethindrone acetate) is a novel oral medication approved by the U.S. Food and Drug Administration (FDA) for the treatment of heavy menstrual bleeding associated with uterine leiomyomas (fibroids) in premenopausal women. The drug combines a gonadotropin-releasing hormone (GnRH) antagonist with estrogen and progestin to reduce fibroid size and associated bleeding. Its market entry and subsequent trajectory are shaped by competition, patent landscape, and physician adoption.
What is the current market landscape for Myfembree?
Myfembree operates within the women's health pharmaceutical market, specifically addressing conditions like uterine fibroids. This market is characterized by a growing need for effective, non-surgical treatment options and an increasing awareness of gynecological health issues.
Key Market Segments & Competition:
- Uterine Fibroid Treatment: This is the primary indication for Myfembree. The competitive landscape includes:
- GnRH Agonists: Medications like leuprolide acetate (Lupron Depot) and goserelin acetate (Zoladex) have historically been used to suppress ovarian function and reduce fibroid growth, but they require injections and can cause significant menopausal symptoms due to estrogen deprivation.
- Oral Contraceptives and Progestins: These are often used for symptom management but may not be as effective in significantly reducing fibroid size.
- Surgical Options: Myomectomy and hysterectomy remain definitive treatments, though Myfembree offers a non-surgical alternative for symptom control.
- Other Emerging Therapies: Research is ongoing for novel small molecules and biologics targeting fibroid growth pathways.
- Menopause Management (Indirect Competition): While not a direct competitor, the estrogen and progestin components of Myfembree also play a role in managing potential side effects associated with GnRH antagonists, indirectly positioning it within the broader context of hormone-based therapies.
Market Size and Growth:
The global uterine fibroids market was valued at approximately \$2.6 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 5.8% from 2023 to 2030, driven by increasing prevalence and demand for minimally invasive treatments [1]. Premenopausal women are the target demographic, representing a significant portion of this market.
Physician Adoption and Prescribing Patterns:
Initial physician adoption is influenced by clinical trial data demonstrating efficacy and safety, formulary access, and educational initiatives from the manufacturer. The oral formulation of Myfembree is a key differentiator from injectable GnRH agonists, potentially improving patient compliance and convenience. However, managing potential side effects like bone mineral density loss and cardiovascular risks remains a consideration for long-term use, influencing prescribing patterns.
What is the patent and exclusivity landscape for Myfembree?
The patent portfolio and regulatory exclusivity are critical determinants of Myfembree's market exclusivity and profitability.
Key Patents and Expiry Dates:
Myfembree's intellectual property is primarily protected by patents covering the compound itself, its formulations, and methods of use.
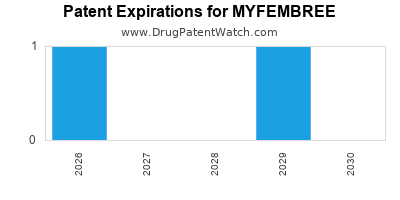
- U.S. Patent No. 8,658,646: This foundational patent covers relugolix as a GnRH antagonist. It is listed in the FDA’s Orange Book and is generally considered to be a key patent for the active pharmaceutical ingredient (API). While exact expiry dates can be complex due to patent term extensions and potential litigation, patents of this nature often extend well into the 2030s.
- Formulation Patents: Additional patents protect the specific combination of relugolix, estradiol, and norethindrone acetate, as well as manufacturing processes. These can also extend exclusivity.
- Method of Use Patents: Patents covering the use of Myfembree for treating heavy menstrual bleeding associated with uterine fibroids.
Regulatory Exclusivity:
- New Chemical Entity (NCE) Exclusivity: Upon FDA approval, Myfembree was granted 5 years of NCE exclusivity, preventing the FDA from approving a generic version of the same drug that relies on the sponsor's preclinical and clinical data for approval. This exclusivity commenced with the FDA approval date of August 10, 2021.
- Orphan Drug Exclusivity (Not Applicable): Uterine fibroids are not typically classified as rare diseases, so orphan drug exclusivity is not a factor.
- Patent Term Extension (PTE): Under the Hatch-Waxman Act, patent terms can be extended to compensate for patent term lost during FDA regulatory review. This can add up to 5 years to the patent life, depending on the length of the review period.
- Hatch-Waxman Exclusivity: For a generic manufacturer to launch a product before the main patent expires, they must either demonstrate non-infringement or invalidate the patent(s). This often leads to "Paragraph IV" challenges.
Projected Market Exclusivity:
Based on typical patent term extensions and the potential for litigation, Myfembree is likely to maintain market exclusivity against generic competition until at least 2031-2033. However, the exact date is subject to the outcomes of any patent challenges.
What is the financial trajectory and revenue forecast for Myfembree?
The financial trajectory of Myfembree is directly linked to its market penetration, pricing, prescription volume, and the costs associated with its commercialization and lifecycle management.
Launch and Initial Sales:
Myfembree was launched in late 2021 by Myovant Sciences, later acquired by Sumitomo Pharma. Initial sales performance is crucial for establishing market presence and recouping R&D investments. The drug’s commercialization strategy, including sales force deployment, marketing campaigns, and patient support programs, significantly impacts early revenue.
Pricing Strategy:
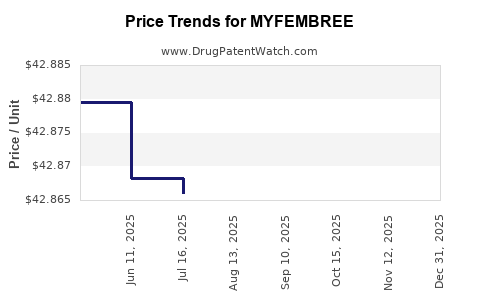
The pricing of Myfembree reflects its innovative nature, clinical benefits, and the value proposition compared to existing treatments. List prices for specialty pharmaceuticals can be high, often subject to negotiations with payers and pharmacy benefit managers (PBMs).
- Estimated Wholesale Acquisition Cost (WAC): While subject to change and payer negotiations, the WAC for a month's supply of Myfembree is in the range of \$900 - \$1,000. This positions it as a premium treatment option.
- Net Price Realization: The actual revenue realized by the manufacturer will be lower than WAC due to rebates, discounts, and chargebacks to payers and PBMs. Net price realization is a critical metric for financial performance.
Sales Performance and Projections:
- 2022 Sales: Myfembree generated approximately \$104 million in net sales in 2022, indicating a solid initial uptake following its launch in late 2021 [2].
- 2023 Performance: First-half 2023 net sales reached \$80.4 million, demonstrating continued growth and exceeding 2022 first-half performance [3]. Full-year 2023 projections suggest sales could exceed \$160 million.
- Future Projections: With continued physician education, expanding payer coverage, and potential label expansions (though none are currently announced for uterine fibroids), Myfembree's sales are projected to grow. Analysts forecast peak sales in the range of \$500 million to \$800 million annually within the next 5-7 years, assuming sustained market share and no significant competitive disruptions.
Factors Influencing Financial Trajectory:
- Payer Access and Formulary Placement: Securing favorable formulary placement is crucial. Restrictions, prior authorizations, and high co-pays can limit patient access and impact sales volume.
- Physician Prescribing Trends: The rate at which gynecologists and obstetricians adopt Myfembree into their treatment algorithms is a primary driver.
- Competition: The introduction of new, highly effective competitors or significant price reductions by existing players could impact Myfembree's market share and pricing power.
- Patient Adherence and Persistence: The long-term effectiveness and tolerability of Myfembree will influence patient persistence, directly affecting refill rates and ongoing revenue.
- Post-Marketing Studies and Real-World Evidence: Data from real-world use can reinforce efficacy and safety, supporting market access and potentially expanding indications.
- Lifecycle Management: The manufacturer's strategy for maximizing the product’s lifecycle, including potential new formulations or combination therapies, can extend revenue streams.
What are the key drivers for Myfembree's market adoption?
The successful adoption of Myfembree by healthcare providers and patients is contingent upon several critical factors.
Clinical Efficacy and Safety Profile:
- Demonstrated Reduction in Heavy Menstrual Bleeding: Clinical trials have shown that Myfembree significantly reduces heavy menstrual bleeding and improves patient quality of life. Data from the SPIRIT program (SPIRIT 1 and SPIRIT 2) demonstrated substantial reductions in menstrual blood loss and improvements in pain associated with uterine fibroids [4].
- Fibroid Shrinkage: The combination therapy also demonstrated a reduction in fibroid volume, offering a tangible physical benefit beyond symptom relief.
- Safety Data: The safety profile, including management of estrogen deficiency symptoms and bone mineral density, is paramount. While the combination therapy mitigates some risks associated with GnRH antagonists alone, long-term safety monitoring remains important.
Convenience and Patient Experience:
- Oral Administration: As an oral pill taken once daily, Myfembree offers a significant convenience advantage over injectable therapies like leuprolide or goserelin, which require clinic visits and can be associated with needle phobia or discomfort.
- Reduced Menopausal Symptoms: Unlike GnRH agonists used alone, the inclusion of estradiol and norethindrone acetate in Myfembree aims to mitigate bone loss and other menopausal symptoms typically associated with prolonged estrogen suppression.
Healthcare Provider Education and Support:
- Awareness Campaigns: Manufacturers invest heavily in educating healthcare providers about the drug's mechanism of action, clinical benefits, and appropriate patient selection.
- Key Opinion Leader (KOL) Engagement: Engaging with leading gynecologists and obstetricians helps to build trust and advocate for the drug's use.
- Data Dissemination: Publication of clinical trial results in peer-reviewed journals and presentation at medical conferences are vital for establishing scientific credibility.
Payer Reimbursement and Access:
- Formulary Inclusion: Obtaining favorable formulary placement from commercial and government payers is essential for patient access. This involves demonstrating the drug’s value proposition relative to its cost.
- Prior Authorization Requirements: Minimizing administrative hurdles like extensive prior authorization requirements can streamline the prescribing process and improve patient adherence.
- Co-pay Assistance Programs: Manufacturer-sponsored patient assistance programs can help offset out-of-pocket costs for patients, improving affordability and uptake.
Unmet Needs in Uterine Fibroid Treatment:
- Demand for Non-Surgical Options: A significant number of women prefer to avoid surgery for uterine fibroids. Myfembree addresses this unmet need by providing an effective medical management option.
- Symptom Burden: The debilitating symptoms of heavy menstrual bleeding, pain, and pressure associated with fibroids significantly impact women's quality of life. Therapies that effectively alleviate these symptoms are highly valued.
What are the potential challenges and risks for Myfembree?
Despite its promising profile, Myfembree faces several challenges that could impact its market performance.
Competition:
- Existing GnRH Antagonists/Agonists: While Myfembree offers oral convenience, established injectable GnRH therapies have a track record. Generic versions of some older GnRH agonists are available, potentially at lower price points.
- Emerging Therapies: The pipeline for uterine fibroid treatments is active. Novel small molecules, targeted therapies, and advanced surgical techniques could disrupt the market.
- Combination Therapies: Competitors may develop their own combination therapies or improved formulations of existing drugs.
Regulatory and Safety Considerations:
- Long-Term Safety Data: While initial data is positive, continued post-marketing surveillance is necessary to fully understand long-term safety, particularly regarding bone mineral density and cardiovascular events. Any emerging safety signals could lead to prescribing restrictions or label changes.
- Off-Label Use: Potential off-label use for other gynecological conditions could lead to regulatory scrutiny or unintended adverse events.
Payer and Market Access Hurdles:
- High Price Point: The drug's premium pricing may lead to stringent utilization management by payers, including prior authorizations, step edits, and high patient co-pays, limiting access for some patients.
- Value-Based Pricing Scrutiny: As healthcare systems increasingly focus on value-based care, demonstrating a clear return on investment for Myfembree compared to less expensive alternatives will be critical.
Physician and Patient Adoption Barriers:
- Physician Inertia: Some physicians may be hesitant to switch from established treatment paradigms to a newer therapy, especially if they have extensive experience with older options.
- Patient Education on Risks: Effectively communicating the potential risks and benefits, including the need for monitoring, to patients is crucial for informed consent and adherence.
- Treatment Duration and Adherence: The long-term effectiveness and tolerability will determine patient persistence. Managing a chronic condition requires sustained adherence, which can be challenging.
Patent Litigation and Generic Entry:
- Patent Challenges: As with any high-value drug, Myfembree is susceptible to patent challenges from potential generic manufacturers seeking to launch their versions of the drug. Successful litigation could significantly shorten its period of market exclusivity and impact profitability.
- Generic Erosion: Even with patent protection, the threat of generic competition looms as patents approach expiration.
Manufacturing and Supply Chain:
- Complex Synthesis: The multi-component nature of Myfembree may present manufacturing complexities, requiring robust quality control and a reliable supply chain. Any disruptions could impact availability and sales.
Key Takeaways
Myfembree is a significant entrant in the women's health market, addressing the unmet need for effective, non-surgical treatment of heavy menstrual bleeding associated with uterine fibroids. Its oral formulation offers a key advantage over existing injectable therapies. The drug has demonstrated strong initial sales, exceeding \$100 million in its first full year of launch, with projections indicating continued growth towards peak sales potentially in the hundreds of millions annually. The patent and regulatory exclusivity landscape is robust, offering a projected market exclusivity period through at least the early 2030s, though this is subject to potential patent litigation. Key drivers of Myfembree's success include its demonstrated clinical efficacy, convenience of oral administration, and effective market access strategies. However, the drug faces challenges from existing and emerging competitors, potential payer restrictions due to its premium pricing, and the ever-present risk of patent challenges and generic erosion. Continued robust clinical data, effective stakeholder engagement, and proactive lifecycle management will be critical for sustaining Myfembree’s financial trajectory.
Frequently Asked Questions
-
What is the primary mechanism of action of Myfembree?
Myfembree is a combination oral contraceptive that contains relugolix, a GnRH antagonist, along with estradiol and norethindrone acetate. Relugolix works by suppressing the pituitary gland's release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH), which in turn reduces ovarian production of estrogen and progesterone. The added estradiol and norethindrone acetate are included to mitigate the bone mineral density loss and menopausal symptoms that can occur with prolonged estrogen suppression.
-
How does Myfembree compare to other treatments for uterine fibroids?
Compared to injectable GnRH agonists (e.g., leuprolide, goserelin), Myfembree offers the significant advantage of oral administration, improving patient convenience and adherence. Unlike GnRH agonists used alone, Myfembree includes estrogen and progestin to help manage estrogen deficiency side effects. It also provides an alternative to surgery for women seeking non-invasive management of heavy menstrual bleeding.
-
What is the expected duration of Myfembree's patent protection and market exclusivity?
Myfembree benefits from a combination of patents and regulatory exclusivities. Key patents are expected to provide protection through the early to mid-2030s, with NCE exclusivity initially granting 5 years from its August 2021 FDA approval. The exact end of exclusivity is subject to patent term extensions and potential litigation outcomes, but a period of exclusivity until at least 2031-2033 is anticipated.
-
What are the main financial risks facing Myfembree?
The primary financial risks include competition from existing and emerging therapies, potential for payer restrictions and pricing pressure due to its high cost, challenges in physician and patient adoption, and the possibility of patent litigation that could lead to earlier generic entry and a reduction in projected revenue.
-
What is the anticipated revenue potential for Myfembree?
Following a strong launch with over \$100 million in net sales in 2022 and continued growth in 2023, analysts project Myfembree could achieve peak annual sales in the range of \$500 million to \$800 million within the next 5-7 years, contingent on sustained market share and favorable market dynamics.
Citations
[1] Grand View Research. (2023). Uterine Fibroids Market Size, Share & Trends Analysis Report By Treatment (Medication, Surgery), By Type (Submucosal, Intramural, Subserosal), By End-use (Hospitals, Clinics), By Region, And Segment Forecasts, 2023 - 2030. https://www.grandviewresearch.com/industry-analysis/uterine-fibroids-market
[2] Myovant Sciences. (2023). Myovant Sciences Reports Fourth Quarter and Full Year 2022 Financial Results and Provides Business Update. https://investors.myovant.com/news-releases/news-release-details/myovant-sciences-reports-fourth-quarter-and-full-year-2022/
[3] Sumitomo Pharma Co., Ltd. (2023). Sumitomo Pharma Announces Financial Results for the First Quarter of Fiscal Year Ending March 31, 2024. https://www.sumitomo-pharma.com/ir/library/financial/results/fy2023_q1.html (Note: While the direct press release link might change, search for Sumitomo Pharma Q1 FY2024 results)
[4] Surrey, E. S., Soliman, A. M., Gunnels, D. J., Zhou, W., & Gallagher, F. (2021). Efficacy and safety of relugolix combination therapy in women with uterine fibroids: results from the Phase 3 SPIRIT program. Contraception, 104(3), 248-255. https://doi.org/10.1016/j.contraception.2021.05.014