Last updated: February 19, 2026
Supernus Pharmaceuticals holds a significant position in the neurology market, focusing on the development and commercialization of novel treatments for central nervous system (CNS) disorders. The company's strategy centers on leveraging its established platforms and pipeline to address unmet medical needs, particularly in epilepsy and Parkinson's disease. Its competitive advantage is rooted in proprietary drug delivery technologies and a disciplined approach to R&D.
What is Supernus Pharmaceuticals' Core Therapeutic Focus?
Supernus Pharmaceuticals primarily targets central nervous system (CNS) disorders. Its key therapeutic areas include:
- Epilepsy: The company has developed and commercialized multiple epilepsy treatments, including Oxtellar XR (oxcarbazepine extended-release) and Trodelvy (trospium chloride extended-release) for overactive bladder, and Qelbree (trileptal extended-release) for generalized onset seizures and partial onset seizures.
- Parkinson's Disease: Supernus is actively developing treatments for Parkinson's disease, with a focus on addressing both motor and non-motor symptoms. Its pipeline includes SPN-812 for ADHD.
- Other CNS Disorders: While epilepsy and Parkinson's are the mainstays, the company's expertise in CNS drug development positions it to explore other related indications.
What are Supernus Pharmaceuticals' Key Commercialized Products?
Supernus Pharmaceuticals has a portfolio of commercialized products, primarily in the epilepsy space:
- Oxtellar XR (oxcarbazepine extended-release): Approved for adjunctive treatment of partial-onset seizures in patients 6 years of age and older. It utilizes the company's proprietary extended-release technology.
- Trodelvy (trospium chloride extended-release): Approved for the treatment of overactive bladder (OAB). This product highlights Supernus's ability to adapt its delivery platforms beyond epilepsy.
- Qelbree (trileptal extended-release): Approved for adjunctive treatment of partial-onset seizures in patients 6 years of age and older. It is a novel, non-hormonal, once-daily therapy.
How Does Supernus Pharmaceuticals Differentiate its Products?
Supernus Pharmaceuticals differentiates its products through several key mechanisms:
- Proprietary Extended-Release Technology: The company's core strength lies in its ability to reformulate existing drugs or develop new ones with improved pharmacokinetic profiles. This includes twice-daily to once-daily dosing, reduced peak-to-trough fluctuations, and potentially improved tolerability.
- Addressing Unmet Needs: Supernus focuses on indications with significant unmet medical needs, aiming to provide differentiated treatment options for patients who may not respond adequately to existing therapies or experience undesirable side effects.
- Targeted R&D Strategy: The company maintains a disciplined R&D approach, prioritizing projects with a high probability of success and clear differentiation from existing treatments. This includes leveraging established mechanisms of action with improved delivery.
- Focus on CNS Disorders: By concentrating its efforts within the complex CNS therapeutic area, Supernus builds specialized knowledge and expertise, fostering internal synergies and a deeper understanding of the patient population and prescriber landscape.
What is the Competitive Landscape for Supernus's Key Products?
The competitive landscape for Supernus's products is dynamic and populated by both branded and generic pharmaceutical companies.
Epilepsy Market
The epilepsy market is characterized by a wide range of antiepileptic drugs (AEDs), including both older, off-patent generics and newer, branded medications.
- Oxtellar XR and Qelbree: These products compete against a broad spectrum of established AEDs.
- Generic Competition: Older AEDs such as carbamazepine, valproic acid, lamotrigine, and levetiracetam are available as low-cost generics, representing significant price competition. These generics often have established efficacy and safety profiles.
- Branded Competition: Newer branded AEDs from companies like UCB, Lundbeck, Eisai, and Pfizer offer various mechanisms of action and delivery systems. Examples include Brivaracetam (Briviact) from UCB, Perampanel (Fycompa) from Eisai, and Lacosamide (Vimpat) from UCB. These competitors often emphasize novel mechanisms, improved tolerability, or specific seizure type coverage.
- Dosing and Delivery: Supernus's extended-release formulations aim to differentiate by offering once-daily dosing convenience and potentially improved tolerability through reduced pharmacokinetic variability. However, other companies also offer extended-release versions of older drugs or employ novel delivery technologies.
- Market Dynamics: Prescriber preference for epilepsy treatments is influenced by efficacy in specific seizure types, tolerability profiles, drug-drug interaction potential, and physician familiarity. The availability of generic options places pressure on pricing for branded products.
Overactive Bladder (OAB) Market
The OAB market is mature and highly competitive, with numerous treatment options.
- Trodelvy: Competes against a wide array of anticholinergic medications, beta-3 adrenergic agonists, and other therapeutic modalities.
- Generic Anticholinergics: Oxybutynin, tolterodine, and solifenacin are widely available as generics, offering significant cost advantages.
- Branded Competition: Mirabegron (Myrbetriq) from Astellas Pharma, a beta-3 adrenergic agonist, offers an alternative mechanism of action to anticholinergics. Other branded options with extended-release formulations also exist.
- Dosing and Formulation: Trodelvy's extended-release formulation aims for convenient once-daily dosing. However, the market is crowded with various extended-release versions of existing drugs, and the therapeutic benefit of the extended-release technology must be clearly demonstrated against these established options.
- Market Dynamics: The OAB market is driven by patient compliance, tolerability, and efficacy. Generic availability significantly impacts market share and pricing.
What is Supernus Pharmaceuticals' R&D Pipeline and Strategy?
Supernus Pharmaceuticals maintains a focused R&D pipeline designed to build upon its core competencies and expand its presence in CNS disorders.
- Pipeline Overview: The pipeline includes product candidates for Parkinson's disease and other CNS indications.
- Parkinson's Disease: Supernus is developing therapies targeting both motor and non-motor symptoms. Specific candidates may aim to improve motor control, reduce dyskinesias, or address non-motor symptoms like psychosis or sleep disturbances. The company aims to leverage its drug delivery expertise to optimize existing therapeutic approaches or introduce novel mechanisms.
- ADHD: SPN-812 (viloxazine extended-release) is a non-stimulant treatment for ADHD. It is a selective norepinephrine reuptake inhibitor. Approval for this indication would broaden Supernus's CNS portfolio.
- R&D Strategy:
- Leveraging Existing Platforms: The company prioritizes projects that can utilize its established drug delivery technologies, particularly extended-release formulations. This reduces development risk and time-to-market.
- Addressing Unmet Medical Needs: R&D efforts are directed towards areas with significant patient populations and limited therapeutic options or where existing treatments have substantial drawbacks.
- Disciplined Capital Allocation: Supernus employs a rigorous process for evaluating R&D projects, focusing on those with a high probability of technical and commercial success.
- Potential for In-Licensing/Acquisition: While not explicitly a primary strategy, Supernus may pursue in-licensing or acquisition opportunities that align with its therapeutic focus and complement its existing pipeline or commercial infrastructure.
What are the Strengths of Supernus Pharmaceuticals?
Supernus Pharmaceuticals possesses several key strengths that underpin its market position:
- Established Drug Delivery Technology: The company's proprietary extended-release technology is a significant asset, enabling the development of differentiated products with improved dosing profiles and potentially enhanced tolerability.
- Demonstrated Commercial Success: Supernus has a proven track record of successfully bringing CNS therapies to market and building commercial infrastructure, particularly in the epilepsy segment.
- Focused Therapeutic Area Expertise: Concentration in CNS disorders allows for the development of deep scientific, clinical, and commercial expertise within this complex therapeutic category.
- Experienced Management Team: The company is led by a management team with extensive experience in pharmaceutical development, regulatory affairs, and commercialization.
- Cash Position and Financial Stability: Supernus has historically maintained a solid financial position, providing the resources to fund its R&D pipeline and commercial operations.
What are the Potential Risks and Challenges for Supernus Pharmaceuticals?
Supernus Pharmaceuticals faces several inherent risks and challenges within the pharmaceutical industry:
- Pipeline Risk: The success of any pharmaceutical company is heavily dependent on its R&D pipeline. Clinical trial failures, regulatory setbacks, or the emergence of superior competing therapies can significantly impact future growth.
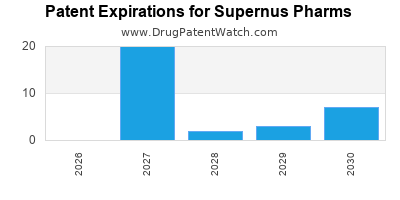
- Market Exclusivity and Generic Competition: As patents expire, Supernus faces intense pressure from generic manufacturers, which can lead to rapid declines in revenue for its commercialized products.
- Pricing and Reimbursement Pressures: The healthcare environment is subject to increasing scrutiny on drug pricing. Payers and government entities continually seek to control healthcare costs, which can impact product launch prices and long-term reimbursement levels.
- Regulatory Hurdles: The drug development and approval process is lengthy, costly, and uncertain. Navigating complex regulatory requirements from agencies like the FDA is a constant challenge.
- Competition: The CNS market is highly competitive, with numerous companies developing and marketing treatments for epilepsy, Parkinson's disease, and other neurological conditions.
- Execution Risk: Successfully commercializing new products, managing manufacturing and supply chains, and adapting to market dynamics require strong operational execution.
What are Supernus Pharmaceuticals' Strategic Insights and Outlook?
Supernus Pharmaceuticals' strategic outlook is shaped by its established strengths and the dynamics of the CNS market.
- Continued Focus on CNS: The company is likely to maintain its deep commitment to neurology, leveraging its existing infrastructure and expertise to pursue opportunities within this therapeutic area.
- Pipeline Advancement as Key Growth Driver: The success of its current R&D pipeline, particularly in Parkinson's disease and ADHD, will be critical for future revenue diversification and growth.
- Maximizing Existing Product Value: Supernus will continue to focus on maximizing the commercial performance of its existing products, Oxtellar XR, Trodelvy, and Qelbree, through lifecycle management and market penetration strategies, while acknowledging the eventual impact of generic competition.
- Strategic Partnerships and Collaborations: The company may explore strategic partnerships, co-development agreements, or in-licensing opportunities to enhance its pipeline or expand its therapeutic reach.
- Adaptation to Market Access Challenges: Navigating evolving reimbursement landscapes and payer demands will require a proactive and adaptable market access strategy to ensure patient access to its therapies.
- Diversification within CNS: While epilepsy and Parkinson's are current focal points, opportunities may arise to explore adjacent CNS indications where its technological capabilities and market understanding can be applied.
Key Takeaways
- Supernus Pharmaceuticals is a neurology-focused company with a strong foundation in extended-release drug delivery technology.
- Its commercial portfolio is primarily in epilepsy, facing significant competition from generics and novel branded therapies.
- The company's R&D pipeline centers on Parkinson's disease and ADHD, representing key future growth opportunities.
- Key strengths include its proprietary technology, CNS expertise, and commercial track record.
- Risks include pipeline failures, generic erosion, and market access challenges.
- Future strategy likely involves continued CNS focus, pipeline execution, and adaptation to market access complexities.
Frequently Asked Questions
-
What is the primary mechanism of action for Supernus's epilepsy drugs?
Supernus's epilepsy drugs utilize established mechanisms. Oxtellar XR is oxcarbazepine, which blocks voltage-sensitive sodium channels. Qelbree is trileptal, also a sodium channel blocker.
-
How does Supernus plan to differentiate its Parkinson's disease pipeline candidates?
Differentiation will likely involve optimizing pharmacokinetic profiles through advanced drug delivery, targeting specific symptom clusters (motor and non-motor), and potentially exploring novel therapeutic targets or combinations.
-
What is the typical lifecycle of a branded drug that faces generic competition, and how does this impact Supernus?
Branded drugs typically see peak sales in the years following launch. Upon loss of market exclusivity and the introduction of generics, sales often decline sharply, sometimes by over 80-90% within the first year. This necessitates Supernus's continuous R&D investment to replace declining revenue streams.
-
What is the significance of Supernus's extended-release technology in a crowded market?
The extended-release technology allows for once-daily dosing, which can improve patient compliance and convenience. It can also lead to smoother drug concentration levels in the bloodstream, potentially reducing side effects associated with peak drug levels. In a market saturated with drugs, such improvements can be a key differentiator.
-
Beyond epilepsy and Parkinson's, are there other CNS areas Supernus is exploring?
While epilepsy and Parkinson's are the primary current focuses, Supernus's core competency in CNS drug development means it is positioned to evaluate and potentially pursue opportunities in other neurological and psychiatric conditions if they align with its strategic objectives and pipeline development capabilities.
Citations
[1] Supernus Pharmaceuticals. (n.d.). Our Science. Retrieved from https://www.supernus.com/our-science
[2] Supernus Pharmaceuticals. (n.d.). Products. Retrieved from https://www.supernus.com/products
[3] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Specific drug approvals can be searched here).
[4] Supernus Pharmaceuticals. (n.d.). Pipeline. Retrieved from https://www.supernus.com/pipeline