Share This Page
Drug Price Trends for QULIPTA
✉ Email this page to a colleague
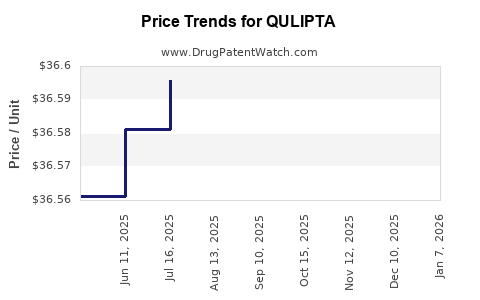
Average Pharmacy Cost for QULIPTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QULIPTA 60 MG TABLET | 00074-7094-30 | 38.47494 | EACH | 2026-01-02 |
| QULIPTA 10 MG TABLET | 00074-7095-30 | 38.36679 | EACH | 2026-01-02 |
| QULIPTA 30 MG TABLET | 00074-7096-30 | 38.40480 | EACH | 2026-01-02 |
| QULIPTA 10 MG TABLET | 00074-7095-30 | 36.53980 | EACH | 2025-12-17 |
| QULIPTA 30 MG TABLET | 00074-7096-30 | 36.57600 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QULIPTA
What is QULIPTA?
QULIPTA (ubrogepant) is a calcitonin gene-related peptide (CGRP) receptor antagonist approved by the FDA in October 2020 for the emergency treatment of migraine with or without aura in adults. It is marketed by AbbVie and is delivered as a daily oral medication.
Market Overview
Migraine Market Size
The global migraine drug market was valued at approximately USD 4.2 billion in 2021. It is projected to reach USD 8.4 billion by 2028, growing at a CAGR of 10.2% (Fortune Business Insights, 2022). The growth is driven by increased migraine prevalence, improved diagnosis, and new therapeutic options.
Key Competitors
- Erenumab (Aimovig): $3.2 billion annual sales, marketed by Amgen and Novartis.
- Eptinezumab (Vyepti): Approximate USD 500 million annually, marketed by Lundbeck.
- Rimegepant (Nurtec ODT): Estimated at USD 1.2 billion sales, marketed by Bristol-Myers Squibb.
- Ubrogepant (QULIPTA): Sales are rising but remain under USD 100 million annually as of 2022 due to recent market entry.
Market Penetration
QULIPTA faces competition from both oral gepants (e.g., rimegepant) and injectable CGRP antibodies. Its oral administration offers advantage for patients preferring non-injectable options, but its market share remains limited as physicians and patients are increasingly adopting CGRP monoclonal antibodies.
Pricing Strategy
Current Pricing
QULIPTA is priced at approximately USD 30–35 per tablet. The recommended dosage is one tablet at onset of migraine, with potential repeat dosing after 2 hours.
Reimbursement and Coverage
Insurance coverage varies by region, with Medicare in the U.S. covering QULIPTA largely through prior authorization. Prices are subject to pharmacy benefit management negotiations, which can reduce out-of-pocket costs. There is no mandatory price regulation, but formulary acceptance influences sales.
Comparative Pricing
| Drug | Delivery Method | Approximate Price per Dose | Monthly Cost (assuming 15 doses) |
|---|---|---|---|
| QULIPTA | Oral | USD 30–35 | USD 450–525 |
| Rimegepant (Nurtec) | Oral | USD 30–35 | USD 450–525 |
| Erenumab | Injection | USD 575 per month | USD 575 |
| Eptinezumab | Injection | USD 400–500 per dose | USD 400–500 per dose |
QULIPTA's pricing aligns with oral gepants but remains less expensive than injectable CGRP antibodies.
Price Projections
Short-term (2023–2025)
- Initial stabilization: Prices will likely stabilize around current levels USD 30–35 per tablet.
- Market penetration: Sales expected to grow as awareness increases, with potential discounts through formulary negotiations, leading to effective prices around USD 25–30 per dose for high-volume payers.
- Regulatory changes: No imminent policy shifts anticipated that would significantly alter pricing structures.
Long-term (2026–2030)
- Product competition: Introduction of generics or biosimilars could decrease prices by 20–40%.
- Market evolution: As more oral options become available, QULIPTA’s price may decline to USD 20–25 per dose.
- Reimbursement policies: Insurance coverage expansion could reduce costs for patients, influencing overall revenue.
Revenue forecasts
- Expected to reach USD 200–300 million globally in 2025, assuming steady market share growth.
- Potential for USD 500 million+ in sales if market expansion accelerates and physician prescribing rates increase.
Conclusion
QULIPTA is positioned as an oral migraine acute treatment, contending in a growing market with established injectables and newer oral options. Its pricing remains competitive but faces downward pressure from increased competition and policy shifts over the coming years.
Key Takeaways
- The global migraine market is expanding, with a CAGR of 10.2%.
- QULIPTA's current price is USD 30–35 per tablet, with likely stabilization in the short term.
- Long-term price declines are probable due to generics, biosimilars, and competitive therapies.
- Its market share depends on physician prescribing patterns and payer negotiations.
- Estimated global sales could reach USD 300 million by 2025, with significant growth potential.
FAQs
1. What are the primary differentiators of QULIPTA compared to other migraine treatments?
QULIPTA offers oral administration for acute migraine relief, with a rapid onset of action and fewer contraindications than some injectable CGRP antagonists.
2. How does insurance coverage impact QULIPTA pricing?
Coverage policies, prior authorization requirements, and negotiations influence patient out-of-pocket costs, affecting overall sales volume.
3. What factors could influence future price reductions?
Entry of generics, biosimilars, increased competition from oral therapies, and policy reforms are key drivers of potential price decreases.
4. How does QULIPTA's reimbursement compare internationally?
Pricing and reimbursement policies vary widely, with the U.S. offering more flexible coverage options, while some countries enforce price controls, limiting profit margins.
5. What is the potential for QULIPTA to expand its indications?
Current approval is limited to acute migraine treatment in adults. Broader indications, such as preventive therapy, would require regulatory approval and potentially impact pricing and market share.
References
[1] Fortune Business Insights. (2022). Migraine Drugs Market Size, Share, Trends & Analysis. Retrieved from https://www.fortunebusinessinsights.com
[2] U.S. Food and Drug Administration. (2020). FDA approves new medication for treatment of migraines. Retrieved from https://www.fda.gov
[3] IQVIA. (2022). Prescription Drug Market Data. IQVIA Institute Reports.
[4] PhRMA. (2021). The Role of Prescription Medicines in Migraine Management. Pharmaceutical Research and Manufacturers of America.
More… ↓
