KOSELUGO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Koselugo, and what generic alternatives are available?
Koselugo is a drug marketed by Astrazeneca and is included in two NDAs. There are eight patents protecting this drug.
This drug has two hundred and one patent family members in forty-five countries.
The generic ingredient in KOSELUGO is selumetinib sulfate. One supplier is listed for this compound. Additional details are available on the selumetinib sulfate profile page.
DrugPatentWatch® Generic Entry Outlook for Koselugo
Koselugo was eligible for patent challenges on April 10, 2024.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 19, 2028. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for KOSELUGO?
- What are the global sales for KOSELUGO?
- What is Average Wholesale Price for KOSELUGO?
Summary for KOSELUGO
| International Patents: | 201 |
| US Patents: | 8 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 129 |
| Clinical Trials: | 12 |
| Patent Applications: | 4,158 |
| Drug Prices: | Drug price information for KOSELUGO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for KOSELUGO |
| What excipients (inactive ingredients) are in KOSELUGO? | KOSELUGO excipients list |
| DailyMed Link: | KOSELUGO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for KOSELUGO
Generic Entry Dates for KOSELUGO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
Generic Entry Dates for KOSELUGO*:
Constraining patent/regulatory exclusivity:
NEW PATIENT POPULATION NDA:
Dosage:
GRANULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for KOSELUGO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Alabama at Birmingham | Phase 2 |
| Congressionally Directed Medical Research Programs | Phase 2 |
| Children's Hospital of Philadelphia | Phase 2 |
Pharmacology for KOSELUGO
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Mitogen-Activated Protein Kinase Kinase 1 Inhibitors Mitogen-Activated Protein Kinase Kinase 2 Inhibitors |
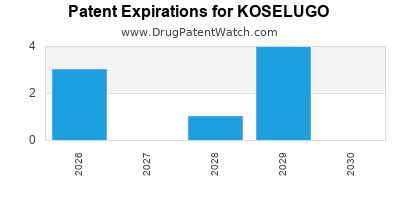
US Patents and Regulatory Information for KOSELUGO
KOSELUGO is protected by eight US patents and five FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of KOSELUGO is ⤷ Start Trial.
This potential generic entry date is based on NEW PATIENT POPULATION.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca | KOSELUGO | selumetinib sulfate | GRANULE;ORAL | 219943-002 | Sep 10, 2025 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Astrazeneca | KOSELUGO | selumetinib sulfate | GRANULE;ORAL | 219943-001 | Sep 10, 2025 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astrazeneca | KOSELUGO | selumetinib sulfate | GRANULE;ORAL | 219943-002 | Sep 10, 2025 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Astrazeneca | KOSELUGO | selumetinib sulfate | GRANULE;ORAL | 219943-001 | Sep 10, 2025 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-001 | Apr 10, 2020 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for KOSELUGO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-001 | Apr 10, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| Astrazeneca | KOSELUGO | selumetinib sulfate | GRANULE;ORAL | 219943-002 | Sep 10, 2025 | ⤷ Start Trial | ⤷ Start Trial |
| Astrazeneca | KOSELUGO | selumetinib sulfate | GRANULE;ORAL | 219943-001 | Sep 10, 2025 | ⤷ Start Trial | ⤷ Start Trial |
| Astrazeneca | KOSELUGO | selumetinib sulfate | CAPSULE;ORAL | 213756-002 | Apr 10, 2020 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for KOSELUGO
When does loss-of-exclusivity occur for KOSELUGO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1100
Estimated Expiration: ⤷ Start Trial
Patent: 6001
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09229204
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0909267
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 18653
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 09000765
Estimated Expiration: ⤷ Start Trial
Patent: 12002380
Patent: Compuestos derivados de bezoimidazol, inhibidores de mek; utiles en el tratamiento del cancer.(divisional solicitud cl 521-03)
Estimated Expiration: ⤷ Start Trial
China
Patent: 2046156
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 11068
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0120657
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 13279
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 71321
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 10010578
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 71321
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 49476
Patent: PHARMACEUTICAL COMPOSITION 271
Estimated Expiration: ⤷ Start Trial
Patent: 56844
Patent: 藥物組合物 (PHARMACEUTICAL COMPOSITION 271 271)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 47710
Estimated Expiration: ⤷ Start Trial
Patent: 11515455
Estimated Expiration: ⤷ Start Trial
Patent: 14114320
Patent: PHARMACEUTICAL COMPOSITION 271
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 7492
Patent: PHARMACEUTICAL COMPOSITION 271
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 10010671
Patent: COMPOSICION FARMACEUTICA 271. (PHARMACEUTICAL COMPOSITION 271.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 8849
Patent: PHARMACEUTICAL COMPOSITION COMPRISING 6-(4-BROMO-2-CHLORO-PHENYLAMINO)-7-FLUORO-3-METHYL-3H-BENZOIMIDAZOLE-5-CARBOXYLIC ACID (2-HYDROXY-ETHOXY)-AMIDE
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 091755
Patent: COMPOSICION FARMACEUTICA QUE COMPRENDE UNA SAL DE SULFATO DE HIDROGENO DE 6-(4-BROMO-2-CLORO-FENILAMINO)-7-FLUORO-3-METIL-3H-BENZOIMIDAZOL-5-ACIDO CARBOXILICO (2-HIDROXI-ETOXI)-AMIDA
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 71321
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 71321
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 91920
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ 271 (PHARMACEUTICAL COMPOSITION 271)
Estimated Expiration: ⤷ Start Trial
Patent: 10143906
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ 271
Estimated Expiration: ⤷ Start Trial
Saudi Arabia
Patent: 9300195
Patent: تركيبة صيدلانية جديدة مضادة للسرطان (A Novel Anti-Cancer Pharmaceutical Composition)
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 409
Patent: FARMACEUTSKA SMEŠA 271 (PHARMACEUTICAL COMPOSITION 271)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 71321
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1006766
Patent: PHARMACEUTICAL COMPOSITION 271
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1650958
Estimated Expiration: ⤷ Start Trial
Patent: 110005246
Patent: PHARMACEUTICAL COMPOSITION 271
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 87942
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0946511
Patent: Pharmaceutical composition
Estimated Expiration: ⤷ Start Trial
Patent: 33840
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 1654
Patent: МАТРИКСНАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ НА ОСНОВЕ ГИДРОСУЛЬФАТА ( 2-ГИДРОКСИЭТОКСИ)АМИДА 6-( 4-БРОМ-2-ХЛОРФЕНИЛАМИНО)-7-ФТОР-3-МЕТИЛ-3Н-БЕНЗОИМИДАЗОЛ-5-КАРБОНОВОЙ КИСЛОТЫ;МАТРИКСНА ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ НА ОСНОВІ ГІДРОСУЛЬФАТУ (2-ГІДРОКСІЕТОКСІ)АМІДУ 6-(4-БРОМ-2-ХЛОРФЕНІЛАМІНО)-7-ФТОР-3-МЕТИЛ-3Н-БЕНЗОІМІДАЗОЛ-5-КАРБОНОВОЇ КИСЛОТИ (MATRIX PHARMACEUTICAL COMPOSITION BASED ON HYDROGEN SULPHATE SALT OF 6-(4-BROMO-2-CHLORO-PHENYLAMINO)-7-FLUORO-3-METHYL-3H-BENZOIMIDAZOLE-5-CARBOXYLIC ACID(2-HYDROXY-ETHOXY)-AMIDE)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 737
Patent: COMPOSICIONES FARMACÉUTICAS QUE CONTIENEN UNA SAL DE SULFATO DE HIDRÓGENO
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering KOSELUGO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Russian Federation | 2008129199 | НОВАЯ ГИДРОСУЛЬФАТНАЯ СОЛЬ | ⤷ Start Trial |
| Dominican Republic | P2011000109 | UN COMPUESTO SELECCIONADO DEL ACIDO 6-(-4-BROMO-2-CLORO-FENILAMINO)-7-FLUORO-3-METIL-3H-BENZOIMIDAZOL-5- CARBOXILICO (2-HIDROXI-ETOXI)- AMIDA Y SALES FARMACEUTICAMENTE ACEPTABLES DEL MISMO, Y COMPOSICIONES FARMACEUTICAS QUE CONTIENEN DICHO COMPUESTO | ⤷ Start Trial |
| Russian Federation | 2008129199 | НОВАЯ ГИДРОСУЛЬФАТНАЯ СОЛЬ | ⤷ Start Trial |
| Slovenia | 2271321 | ⤷ Start Trial | |
| Japan | 2014114320 | PHARMACEUTICAL COMPOSITION 271 | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for KOSELUGO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1482932 | 19C1014 | France | ⤷ Start Trial | PRODUCT NAME: BINIMETINIB SOUS TOUTES SES FORMES PROTEGEES PAR LE BREVET DE BASE; REGISTRATION NO/DATE: EU/1/18/1315 20180924 |
| 1482932 | CA 2019 00011 | Denmark | ⤷ Start Trial | PRODUCT NAME: BINIMETINIB I ALLE FORMER BESKYTTET AF GRUNDPATENTET; REG. NO/DATE: EU/1/18/1315 20180924 |
| 1482932 | 334 8-2019 | Slovakia | ⤷ Start Trial | PRODUCT NAME: BINIMETINIB VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/18/1315 20180924 |
| 1968948 | C202130063 | Spain | ⤷ Start Trial | PRODUCT NAME: SELUMETINIB HIDROGENOSULFATO, INCLUYENDO SOLVATOS Y FORMAS ANHIDRAS DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/21/1552; DATE OF AUTHORISATION: 20210617; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/21/1552; DATE OF FIRST AUTHORISATION IN EEA: 20210617 |
| 1482932 | C 2019 008 | Romania | ⤷ Start Trial | PRODUCT NAME: BINIMETINIB; NATIONAL AUTHORISATION NUMBER: EU/1/18/1315; DATE OF NATIONAL AUTHORISATION: 20180920; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/18/1315; DATE OF FIRST AUTHORISATION IN EEA: 20180920 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for KOSELUGO (selpercatinib)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.