Acetaminophen - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for acetaminophen and what is the scope of freedom to operate?
Acetaminophen
is the generic ingredient in one hundred and eight branded drugs marketed by Ortho Mcneil Pharm, Rising, Aspiro, B Braun Medical, Baxter Hlthcare Corp, Caplin, Eugia Pharma, Fresenius Kabi Usa, Gland, Hikma, Inforlife, Mylan, Sandoz, Wockhardt Bio Ag, Zydus Pharms, Mallinckrodt Hosp, Cosette, Able, Acino Prods, Perrigo New York, Taro, Polymedica, J And J Consumer Inc, Aurobindo Pharma, Granules, Heritage, Marksans Pharma, Ohm Labs, Perrigo, Sun Pharm Inds Ltd, Kenvue Brands, Aurobindo Pharma Ltd, Haleon Us Holdings, Mikart, Scherer Labs, Zevra Therap, Forest Pharms, Mallinckrodt, Dr Reddys Labs Sa, Valeant, Dunhall, Pharmobedient, Alvogen, Anda Repository, Halsey, Lgm Pharma, Ne Rx Pharma, Quagen, Senores Pharms, Watson Labs, Mayrand, Shire, Aurolife Pharma Llc, Gilbert Labs, Graham Dm, Ingenus Pharms Llc, Key Therap, Lannett Co Inc, Nuvo Pharms Inc, Us Chem, Genus, Abhai Llc, Actavis Labs Ut Inc, Hikma Pharms, Mirror Pharms Llc, Nesher Pharms, Specgx Llc, Strides Pharma, Strides Pharma Intl, Sun Pharm Industries, Vintage Pharms, Hikma Intl Pharms, Wraser Pharms Llc, Pharm Res Assoc, Leitner Pharms, Boca Pharma Llc, West-ward Pharm Corp, Novartis, Teva, Robins Ah, Solvay, Actavis Mid Atlantic, Chartwell, Chartwell Molecular, Dava Pharms Inc, Genus Lifesciences, Pai Holdings, Valeant Pharms Llc, Am Therap, Amneal Pharms Ny, Ani Pharms, Duramed Pharms Barr, Elite Labs Inc, Everylife, Fosun Pharma, Hibrow Hlthcare, Kv Pharm, Lederle, Mutual Pharm, Puracap Pharm, Purepac Pharm, Rhodes Pharms, Roxane, Superpharm, Usl Pharma, Valeant Pharm Intl, Vitarine, Warner Chilcott, Watson Labs Florida, Whiteworth Town Plsn, Carnrick, Glaxosmithkline, Vangard, Janssen Pharms, Schering Plough, Cent Pharms, Ivax Pharms, Mallinckrodt Inc, Pharm Assoc, Tris Pharma Inc, Wes Pharma Inc, Cypress Pharm Inc, Ucb Inc, Ascher, Actavis Labs Fl Inc, Amneal Pharms, Apil, Ascent Pharms Inc, Barr, Caraco, Epic Pharma Llc, Novel Labs Inc, Ph Health, Prinston Inc, Ranbaxy, Ranbaxy Labs Ltd, Sanaluz, Sun Pharm Inds Inc, Upsher Smith Labs, Vintage Pharms Llc, Abana, Abbott, Abbvie, Bionpharma, Dr Reddys, Glenmark Pharms Ltd, L Perrigo Co, Lnk Intl Inc, Aft Pharms Us, Actavis Elizabeth, Sankalp Lifecare, Bristol Myers Squibb, Endo Operations, Gavis Pharms, Sanofi Aventis Us, Aaipharma Llc, Xanodyne Pharm, Cornerstone, Ivax Sub Teva Pharms, Mirror Pharms, Mylan Pharms Inc, Wockhardt Ltd, Alkem Labs Ltd, Chartwell Rx, Graviti Pharms, Macleods Pharms Ltd, Micro Labs Ltd India, Mpp Pharma, and Zydus Pharms Usa Inc, and is included in five hundred and thirty-three NDAs. There are thirty-one patents protecting this compound and one Paragraph IV challenge. Additional information is available in the individual branded drug profile pages.Acetaminophen has twenty-one patent family members in thirteen countries.
There are sixty-six drug master file entries for acetaminophen. Ninety-two suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for acetaminophen
| International Patents: | 21 |
| US Patents: | 31 |
| Tradenames: | 108 |
| Applicants: | 169 |
| NDAs: | 533 |
| Drug Master File Entries: | 66 |
| Finished Product Suppliers / Packagers: | 92 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 1,433 |
| Patent Applications: | 7,093 |
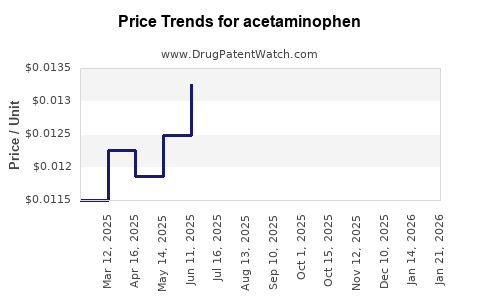
| Drug Prices: | Drug price trends for acetaminophen |
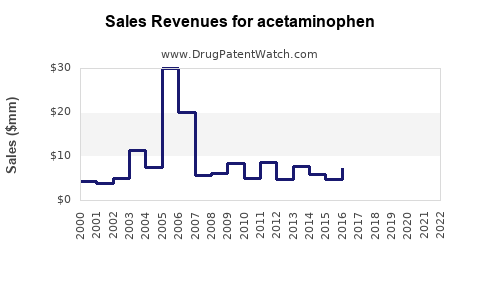
| Drug Sales Revenues: | Drug sales revenues for acetaminophen |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for acetaminophen |
| What excipients (inactive ingredients) are in acetaminophen? | acetaminophen excipients list |
| DailyMed Link: | acetaminophen at DailyMed |
Recent Clinical Trials for acetaminophen
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Brigham and Women's Hospital | PHASE1 |
| National Institutes of Health (NIH) | PHASE1 |
| American Association of Hip and Knee Surgeons | PHASE3 |
Generic filers with tentative approvals for ACETAMINOPHEN
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Start Trial | ⤷ Start Trial | 325MG; 7.5MG | TABLET, EXTENDED RELEASE;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Medical Subject Heading (MeSH) Categories for acetaminophen
Paragraph IV (Patent) Challenges for ACETAMINOPHEN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OFIRMEV | Injection | acetaminophen | 650 mg/65 mL (10 mg/mL) | 022450 | 1 | 2024-07-31 |
| OFIRMEV | Injection | acetaminophen | 1000 mg/100 mL (10 mg/mL) | 022450 | 1 | 2011-04-07 |
US Patents and Regulatory Information for acetaminophen
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Actavis Labs Fl Inc | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 206470-002 | Jun 2, 2016 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amneal Pharms | TRAMADOL HYDROCHLORIDE AND ACETAMINOPHEN | acetaminophen; tramadol hydrochloride | TABLET;ORAL | 090485-001 | Dec 9, 2009 | AB | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Mutual Pharm | HYDROCODONE BITARTRATE AND ACETAMINOPHEN | acetaminophen; hydrocodone bitartrate | TABLET;ORAL | 040236-001 | Sep 25, 1997 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Aurobindo Pharma | ACETAMINOPHEN | acetaminophen | TABLET, EXTENDED RELEASE;ORAL | 207229-001 | Nov 9, 2016 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for acetaminophen
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Kenvue Brands | TYLENOL | acetaminophen | TABLET, EXTENDED RELEASE;ORAL | 019872-001 | Jun 8, 1994 | ⤷ Start Trial | ⤷ Start Trial |
| Kenvue Brands | TYLENOL | acetaminophen | TABLET, EXTENDED RELEASE;ORAL | 019872-001 | Jun 8, 1994 | ⤷ Start Trial | ⤷ Start Trial |
| Kenvue Brands | TYLENOL | acetaminophen | TABLET, EXTENDED RELEASE;ORAL | 019872-001 | Jun 8, 1994 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for acetaminophen
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Denmark | 2421522 | ⤷ Start Trial | |
| Hungary | E026823 | ⤷ Start Trial | |
| Japan | 2012524738 | ⤷ Start Trial | |
| Portugal | 2421522 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Acetaminophen: Market Dynamics and Financial Trajectory
More… ↓