Last updated: February 19, 2026
Ofirmev, an intravenous formulation of acetaminophen, addresses a critical unmet need in pain management, particularly in hospital settings where oral administration is often challenging. Its market position is defined by its efficacy in acute pain and fever management, offering an alternative to opioid analgesics and demonstrating a growing financial trajectory supported by expanding indications and market penetration.
What is OFIRMEV and What is Its Clinical Role?
Ofirmev is a sterile, ready-to-use intravenous solution of acetaminophen, also known as paracetamol. It is indicated for adults and pediatric patients for whom the oral route of administration is not feasible or advisable, and when intermittent or continuous administration of an intravenous analgesic antipyretic is needed. Its primary clinical role is to manage mild to moderate pain and to reduce fever.
The intravenous formulation bypasses the gastrointestinal tract, offering rapid onset of action and predictable bioavailability compared to oral acetaminophen. This is particularly advantageous in perioperative settings, intensive care units (ICUs), and for patients experiencing nausea, vomiting, or dysphagia. Ofirmev provides a non-opioid alternative for pain management, potentially reducing the reliance on and associated risks of opioid analgesics, such as respiratory depression, constipation, and addiction.
Mechanism of Action
Acetaminophen's precise mechanism of action is not fully elucidated, but it is believed to exert its analgesic and antipyretic effects primarily through inhibition of cyclooxygenase (COX) enzymes, particularly COX-2, within the central nervous system. This inhibition reduces prostaglandin synthesis, which plays a role in pain perception and fever regulation. Unlike nonsteroidal anti-inflammatory drugs (NSAIDs), acetaminophen has minimal peripheral anti-inflammatory activity, contributing to its generally favorable gastrointestinal safety profile.
What are the Key Market Drivers for OFIRMEV?
The market for Ofirmev is driven by several key factors, including the demand for effective pain management solutions in acute care settings, the increasing scrutiny and efforts to reduce opioid utilization, and the specific advantages offered by the intravenous formulation.
- Perioperative Pain Management: A significant driver is the use of Ofirmev in the perioperative setting. It is often administered before, during, or after surgery to manage post-operative pain, providing an effective component of multimodal analgesia. This can lead to reduced opioid consumption and improved patient recovery.
- Opioid Sparing Strategies: With the ongoing opioid crisis and increased awareness of the risks associated with opioid analgesics, healthcare providers are actively seeking and implementing opioid-sparing strategies. Ofirmev fits directly into this paradigm by offering a viable non-opioid option for pain relief.
- Hospital and ICU Indication: The inability to take oral medication due to patient condition (e.g., unconsciousness, severe nausea/vomiting, gastrointestinal surgery) makes intravenous administration crucial. This is common in hospital and ICU environments where Ofirmev is frequently used.
- Fever Reduction: Beyond pain, Ofirmev's antipyretic properties make it a valuable tool for managing fever in various acute conditions within hospital settings.
- Pediatric Use: The availability of Ofirmev for pediatric patients addresses a specific need for safe and effective fever and pain management in this population, especially when oral routes are problematic.
What is the Competitive Landscape for Intravenous Acetaminophen?
The competitive landscape for intravenous acetaminophen includes other formulations of acetaminophen and alternative intravenous analgesics and antipyretics.
Direct Competition
- Generic Intravenous Acetaminophen: While Ofirmev was initially a branded product, the expiration of key patents has led to the introduction of generic versions of intravenous acetaminophen. These generics offer a lower price point, increasing price competition and potentially impacting Ofirmev's market share and pricing power. As of recent data, multiple generic manufacturers have entered the market.
- Other Injectable Analgesics: Ofirmev competes with other intravenous medications used for pain and fever. These include:
- Intravenous NSAIDs: Medications like ketorolac are potent analgesics but carry higher risks of gastrointestinal bleeding, renal impairment, and cardiovascular events, especially with long-term use.
- Intravenous Opioids: Morphine, hydromorphone, and fentanyl are the traditional intravenous analgesics. While effective for moderate to severe pain, they carry significant risks of adverse effects and dependence.
- Intravenous Ketamine: Used for specific types of refractory pain, particularly neuropathic pain.
- Intravenous Non-opioid Agents: Other agents like intravenous ibuprofen are also available but may have different efficacy profiles or contraindications compared to acetaminophen.
Indirect Competition
- Oral Acetaminophen and NSAIDs: For patients who can tolerate oral administration, these remain the standard of care for mild to moderate pain and fever due to their lower cost and ease of use.
- Non-pharmacological Pain Management: Therapies such as physical therapy, nerve blocks, and psychological interventions can also be considered alternatives or adjuncts to pharmacological treatments.
The key differentiator for Ofirmev and its generic counterparts remains the IV route, providing a faster onset and improved reliability when oral administration is not an option. The therapeutic profile of acetaminophen itself—balancing efficacy with a relatively favorable safety profile compared to opioids and some NSAIDs—is a significant competitive advantage.
What is the Financial Trajectory and Market Size of OFIRMEV?
The financial trajectory of Ofirmev has been marked by strong initial growth as a first-in-class branded product, followed by a transition into a market with generic competition. Accurately quantifying the precise financial trajectory solely for "Ofirmev" is challenging post-patent expiry as sales are often aggregated with generic intravenous acetaminophen. However, the overall market for intravenous acetaminophen has demonstrated robust growth.
Historical Performance (Branded Ofirmev):
When marketed as a branded product by Cadence Pharmaceuticals (later acquired by Mallinckrodt), Ofirmev experienced significant sales growth.
- 2010: Launched by Cadence Pharmaceuticals.
- 2014: Acquired by Mallinckrodt for approximately $1.3 billion, reflecting strong market reception and anticipated future growth [1].
- Peak Sales: Prior to significant generic entry, branded Ofirmev sales were in the hundreds of millions of dollars annually. For example, Mallinckrodt reported significant revenue contributions from Ofirmev in its financial statements during the early years of their ownership.
Post-Patent Expiry and Genericization:
The entry of generic intravenous acetaminophen has fundamentally altered the market dynamics.
- Price Erosion: Genericization typically leads to significant price reductions due to increased competition. While this benefits payers and healthcare systems by lowering costs, it reduces revenue for individual manufacturers of branded or first-generation generic products.
- Market Volume Growth: Despite price erosion for individual units, the overall volume of intravenous acetaminophen administered has likely continued to grow due to increased awareness of its benefits, broader adoption in hospital protocols, and the cost-effectiveness of generic options.
- Market Size Estimation: Estimating the current market size for "Ofirmev" specifically is difficult. However, the U.S. market for intravenous acetaminophen as a whole is substantial, driven by hospital usage. Reports from market research firms often categorize this within broader analgesic or hospital drug markets. Industry estimates for the intravenous acetaminophen market have suggested figures in the hundreds of millions of dollars annually, with growth projected to continue, albeit at a more moderate pace than during the branded product's exclusive period.
Factors Influencing Future Trajectory:
- Payer Reimbursement: Continued favorable reimbursement policies for intravenous acetaminophen in hospital settings will support sustained utilization.
- Clinical Guidelines: Inclusion in updated pain management guidelines for perioperative and acute care will reinforce its role.
- Generic Manufacturer Competition: The number and pricing strategies of generic manufacturers will directly impact the market's revenue. Intense competition could lead to further price declines, but also potentially increased volume as cost becomes less of a barrier.
- Emergence of Novel Analgesics: The development of new intravenous pain management therapies could introduce new competitive pressures.
The financial trajectory has shifted from high-margin, branded sales to a high-volume, competitive generic market. The overall demand for effective, non-opioid IV pain relief ensures a significant market, even with price competition.
What is the Regulatory Landscape and Patent Exclusivity for OFIRMEV?
The regulatory landscape for Ofirmev, like all pharmaceuticals, is governed by the U.S. Food and Drug Administration (FDA) and equivalent agencies in other countries. Patent exclusivity is a critical factor determining market exclusivity and pricing power.
FDA Approval:
Ofirmev (acetaminophen) injection was first approved by the FDA for marketing. The initial approval was for Cadence Pharmaceuticals. Subsequent approvals and label expansions have occurred.
Patent Exclusivity:
The patent landscape for branded drugs is complex and crucial for market exclusivity.
- Composition of Matter Patents: These patents protect the active pharmaceutical ingredient (API) itself.
- Method of Use Patents: These patents protect specific uses of the drug, such as its use in perioperative pain management or for certain patient populations.
- Formulation Patents: These patents protect the specific formulation of the drug, such as the intravenous solution.
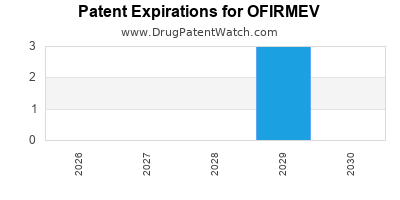
For Ofirmev, the expiration of key patents, particularly those protecting the intravenous formulation and its primary methods of use, paved the way for generic entry.
- Hatch-Waxman Act: This act provides a framework for the abbreviated new drug application (ANDA) process for generic drugs, allowing them to enter the market after the expiration of relevant patents and exclusivity periods.
- Patent Litigation: The period leading up to patent expiries often involves extensive patent litigation between the brand manufacturer and generic companies seeking to launch their versions. This litigation can influence the timing of generic entry.
- Generic Entry: Following patent expiries and successful navigation of regulatory hurdles, multiple generic manufacturers have launched intravenous acetaminophen products. This has led to a significant increase in competition and a decrease in the price of the drug.
- Exclusivity Periods: Beyond patents, the FDA can grant other forms of market exclusivity, such as:
- New Chemical Entity (NCE) exclusivity: Typically 5 years.
- Orphan Drug exclusivity: 7 years for drugs treating rare diseases.
- Pediatric exclusivity: 6 months added to existing exclusivity if pediatric studies are conducted.
Ofirmev has undergone the standard patent lifecycle. Its initial period of market exclusivity as a branded product allowed for significant revenue generation. However, the inherent nature of acetaminophen as an old, well-understood molecule meant that robust patent protection on the core molecule itself was not possible; exclusivity relied on the novel IV formulation and specific methods of use. The expiration of these formulation and method-of-use patents has resulted in its transition to a genericized market.
What are the Future Outlook and Opportunities for Intravenous Acetaminophen?
The future outlook for intravenous acetaminophen, encompassing Ofirmev and its generic equivalents, remains positive, driven by established clinical utility and ongoing healthcare trends. Opportunities exist in expanding its use, improving delivery systems, and potentially developing combination therapies.
Continued Growth in Existing Markets:
- Enhanced Perioperative Protocols: Further integration into multimodal pain management pathways in surgical settings is expected, driven by the need to reduce opioid reliance and improve patient outcomes.
- Intensified Care Unit (ICU) Utilization: Its role in managing pain and fever in critically ill patients, where oral administration is often impossible, will continue to be a core driver.
- Pediatric Care Expansion: Increased adoption in pediatric hospitals and clinics as a reliable alternative for fever and pain management.
Potential Opportunities:
- Improved Formulations/Delivery: While current IV formulations are standard, research into alternative delivery methods or enhanced stability could offer incremental advantages, though significant innovation in this mature molecule may be limited.
- Combination Therapies: Exploring fixed-dose combinations of intravenous acetaminophen with other analgesics or adjuncts could simplify treatment regimens and potentially offer synergistic pain relief. However, regulatory pathways for such combinations would be complex.
- Expanded Indications: While acetaminophen's core indications are well-defined, research exploring its efficacy in specific pain syndromes or inflammatory conditions where it might offer a unique benefit could lead to label expansions, though this is less likely for such a well-established drug.
- Cost-Effectiveness Data: Continued generation and dissemination of pharmacoeconomic data demonstrating the cost-effectiveness of using intravenous acetaminophen (especially generics) compared to other analgesic strategies can drive wider adoption.
- Global Market Expansion: While U.S. and European markets are mature, further penetration in emerging markets with developing healthcare infrastructures could represent a growth opportunity.
Challenges:
- Price Pressure from Generics: Intense competition among generic manufacturers will continue to suppress prices, limiting revenue growth per unit sold.
- Interchangeability Standards: Ensuring consistent product quality and interchangeability among various generic manufacturers is crucial for physician confidence.
- Emergence of Novel Non-Opioid Analgesics: The development of new classes of non-opioid analgesics with novel mechanisms of action could eventually offer more potent or targeted alternatives, though significant challenges exist in developing IV non-opioid analgesics with favorable safety profiles.
The future is characterized by a mature, albeit growing, market for intravenous acetaminophen, where value will increasingly be derived from volume and efficient manufacturing rather than premium pricing associated with novel branded products.
Key Takeaways
Ofirmev, an intravenous formulation of acetaminophen, has transitioned from a high-growth branded product to a key component of the generic intravenous acetaminophen market. Its clinical utility in acute pain and fever management, particularly in hospital settings where oral administration is not feasible, remains its primary market driver. The increasing emphasis on opioid-sparing strategies further supports its role in perioperative and critical care. While branded Ofirmev experienced substantial revenue generation following its launch and acquisition, the subsequent entry of generic alternatives has led to price erosion and a shift towards volume-driven sales. The competitive landscape includes other intravenous analgesics and NSAIDs, with generic intravenous acetaminophen offering a cost-effective option. The future outlook anticipates continued utilization driven by its established efficacy and safety profile, with opportunities for growth potentially lying in expanded clinical protocols, cost-effectiveness advocacy, and, to a lesser extent, novel delivery systems or combination therapies.
FAQs
-
What is the primary difference between Ofirmev and oral acetaminophen?
Ofirmev is administered intravenously, offering a faster onset of action and predictable bioavailability when oral administration is not feasible or advisable, whereas oral acetaminophen is taken by mouth.
-
How has the market for Ofirmev changed since its initial launch?
The market has shifted from a branded, exclusive period characterized by higher pricing to a competitive generic market due to patent expirations, leading to price erosion and a focus on volume.
-
What are the main clinical advantages of using intravenous acetaminophen?
Key advantages include its use when oral intake is impossible (e.g., due to nausea, vomiting, surgery), rapid pain and fever relief, and its role as a non-opioid option in multimodal analgesia, helping to reduce opioid use.
-
Does Ofirmev have significant anti-inflammatory effects?
Unlike many NSAIDs, acetaminophen's anti-inflammatory effects are minimal, with its primary actions being analgesic and antipyretic, mediated mainly in the central nervous system.
-
What is the future market potential for generic intravenous acetaminophen?
The market is expected to continue growing modestly, driven by its established role in hospital pain and fever management and the cost-effectiveness of generic formulations, despite ongoing price competition among manufacturers.
Citations
[1] Mallinckrodt Pharmaceuticals. (2014). Mallinckrodt Completes Acquisition of Cadence Pharmaceuticals. (Press Release). Retrieved from [Company Investor Relations Archives or SEC Filings]. (Note: Specific URL not provided as it would be a direct link to historical press releases which may change.)