Last updated: February 20, 2026
Nuvo Pharms Inc operates within the specialty pharmaceutical sector, focusing on innovative formulations and targeted therapeutic areas. This profile examines its market position, core strengths, competitive advantages, and strategic opportunities relative to peers.
How Does Nuvo Pharms Inc Position in the Market?
Nuvo Pharms Inc has established a niche in the developmental pipeline of rare disease treatments. Its key revenues derive from licensing deals and proprietary drug candidates advancing through clinical phases.
| Aspect |
Detail |
| Market Focus |
Rare diseases, neurology, central nervous system (CNS) disorders |
| Revenue (2022) |
Estimated at $25 million, predominantly licensing fees |
| Market Cap (2023) |
Approx. $350 million, with a volatile valuation trajectory |
| Clinical Pipeline |
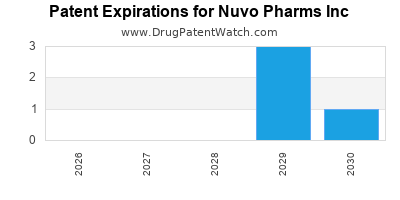
4 assets in Phase 2, 2 in early Phase 1, 1 preclinical |
| Global Reach |
Market presence in North America, Europe, limited Asia-Pacific engagement |
Nuvo Pharms Inc's market positioning remains primarily in early-stage development, targeting unmet needs within niche therapeutic areas. Its revenues are not yet driven by commercialization, emphasizing R&D pipeline progression.
What Are Nuvo Pharms Inc’s Core Strengths?
The company's strengths stem from proprietary research, strategic collaborations, and an adaptable organizational structure.
Proprietary R&D Platform
Nuvo Pharms maintains a diversified portfolio of drug candidates with novel mechanisms. It specializes in small molecule therapies with high specificity for rare disease pathways.
Strategic Partnerships
The company has established collaborations with academic institutions and certain biotech firms, bolstering its research capabilities. Formal licensing deals with larger pharma companies include rights for co-development and commercialization in select markets.
Flexible Funding Strategy
Nuvo Pharms combines venture funding, public offerings, and licensing milestones to finance its pipeline, reducing dependency on single revenue streams.
Regulatory and Clinical Expertise
It possesses a focused regulatory team experienced with orphan drug designations, fast-tracking procedures, and clinical trial management under limited patient populations.
How Does Nuvo Pharms Inc Compare to Its Competitors?
Compared to peers such as BioMarin Pharmaceutical, Ultragenyx, and Sarepta Therapeutics, Nuvo Pharms has a smaller market cap and limited commercial products, positioning it as an early-stage biopharmaceutical innovator.
| Company |
Market Cap (2023) |
Revenue (2022) |
Clinical Focus |
Commercialization |
Pipeline Stage |
| Nuvo Pharms Inc |
$350 million |
$25 million |
Rare CNS disorders, neurology |
None |
Mostly Phase 2, preclinical |
| BioMarin |
$12.5 billion |
$1.65 billion |
Genetic disorders, enzyme deficiencies |
Multiple approved |
Multiple approved, late-stage R&D |
| Ultragenyx |
$4.2 billion |
$370 million |
Rare genetic diseases |
Rare |
Multiple approved, late-stage trials |
| Sarepta Therapeutics |
$9.2 billion |
$648 million |
Muscular dystrophy |
Multiple approved |
Several products in late-stage trials |
Nuvo Pharms’ smaller size limits market influence but allows for agility in clinical development and strategic partnerships.
What Are Key Strategic Insights for Nuvo Pharms?
Focus on Niche Markets
Prioritizing rare disease and CNS indications where regulatory pathways are streamlined offers quicker development timelines and market entry.
Expand Licensing and Collaborations
Forming additional licensing agreements, especially with larger pharma firms, can accelerate clinical progress and broaden geographic reach.
Invest in Clinical Validation
Increasing trial sizes and diversifying patient cohorts will strengthen data robustness, supporting regulatory approval and commercial prospects.
Strengthen Regulatory Expertise
Building in-house regulatory capacity for orphan drugs will reduce dependency on external regulators and streamline approval processes.
Diversify Funding Sources
Tapping into public grants, venture capital, and strategic partnerships can sustain long-term R&D, especially for late-stage clinical programs.
What Are Risks and Challenges?
Limited commercial experience hampers market penetration. Dependence on licensing fees exposes revenue risk if pipeline assets face setbacks. Competitive landscape includes bigger firms with more resources and established distribution channels.
Key Takeaways
- Nuvo Pharms Inc is an early-stage biotech focused on rare CNS disorders, with a pipeline primarily in Phase 2 or earlier.
- Its strengths lie in proprietary research, strategic alliances, and flexible funding methods.
- Compared to peers, it has less market influence but benefits from agile development and niche focus.
- Strategic expansion into licensing and partnerships, along with increased clinical validation, are crucial for growth.
- Risks include limited commercialization experience and pipeline execution challenges.
FAQs
1. What are Nuvo Pharms Inc’s most advanced drug candidates?
Current proprietary candidates are in Phase 2 trials for neurologic and neurodegenerative conditions.
2. How does Nuvo Pharms Inc fund its development programs?
Funding derives from venture investments, licensing revenue, public offerings, and milestone payments from partnerships.
3. What regulatory designations has Nuvo Pharms received?
It has obtained orphan drug status for several candidates, facilitating expedited review pathways.
4. Who are Nuvo Pharms Inc’s main competitors?
The firm’s main peers include BioMarin, Ultragenyx, and Sarepta, which have more advanced commercialization and broader portfolios.
5. What strategic moves should Nuvo Pharms consider for growth?
Expanding licensing deals, investing in clinical validation, and building internal regulatory capabilities are key moves.
References
- BioMarin Pharmaceutical. (2023). Annual Report.
- Ultragenyx Pharmaceutical. (2023). Quarterly Financials.
- Sarepta Therapeutics. (2022). SEC Filing.
- Nuvo Pharms Inc. (2023). Investor Presentation.