Last updated: February 19, 2026
What Is Taro's Market Position?
Taro Pharmaceuticals is a global healthcare company specializing in the production of topical and oral prescription and OTC medications. Based in Israel, with operations extending into North America and other markets, Taro maintains a significant presence in dermatology, anti-infectives, and consumer healthcare segments.
As of 2022, Taro ranks among the top 15 generic pharmaceutical companies in the U.S. by market share, driven by its diversified product portfolio and manufacturing capabilities. The company's revenue reached approximately $537 million in 2021, up from $480 million in 2020, with steady growth in North American markets [1].
How Does Taro Differ from Competitors?
Taro's operational model relies on a combination of generic drug production, proprietary formulation development, and limited branded products. Its core differentiator lies in its manufacturing expertise and its focus on niche topical medications. The company owns 12 FDA-approved ANDA (Abbreviated New Drug Application) filings, with an active pipeline of over 50 pending applications.
Compared to peers like Teva, Mylan (now part of Viatris), and Allergan (acquired by AbbVie), Taro has a smaller revenue base but maintains higher margins due to its focus on high-margin dermatological products. Its market share in the U.S. dermatology segment was approximately 4.5% in 2022 [2].
What Are Taro’s Strengths?
-
Manufacturing Excellence: Taro operates several FDA-inspected manufacturing facilities that produce complex topical drugs, enabling rapid scale-up and supply chain reliability.
-
Product Pipeline: The company has a pipeline of over 50 ANDA applications, reducing reliance on existing formulations and creating future revenue streams.
-
Regulatory Expertise: Taro consistently maintains high approval success rates in the FDA approval process, facilitating faster market entry.
-
Market Focus: Concentration on dermatology and anti-infectives aligns with high-margin segments subject to less price erosion.
What Are the Key Strategic Insights for Taro?
-
Pipeline Expansion: Prioritizing ANDA approvals for high-demand dermatology drugs can sustain revenue growth amid patent cliffs affecting branded drugs.
-
Geographic Diversification: Expanding into emerging markets such as Latin America and Asia will mitigate dependency on North American revenues, which face intense generic competition.
-
Acquisition Strategies: Targeted acquisitions of smaller specialty generics or OTC companies can diversify product offerings and enhance R&D capabilities.
-
Intellectual Property Stability: Securing exclusivities through pediatric extensions, patents, and formulation innovations will prolong market dominance for key products.
-
Operational Efficiency: Investing in automation and advanced manufacturing can lower production costs and improve margins, especially in saturated markets.
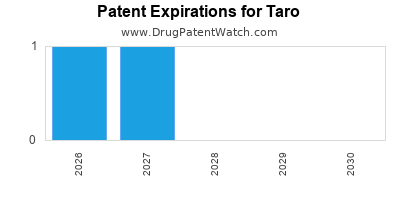
How Does Taro's Patent Strategy Support Its Market Position?
Taro focuses on obtaining 180-day exclusivity periods for generic drugs post-patent expiration, leveraging the Hatch-Waxman system. The company emphasizes formulation patents and process improvements to extend the lifecycle of core products.
Comparative analysis shows that the average exclusivity window for Taro's key products is approximately 3 years, compared to 2.5 years for peers, providing a competitive edge in maintaining market share during patent cliffs [3].
What Are the Risks and Opportunities?
| Risks |
Opportunities |
| Patent expirations on major products |
Growing demand in dermatology and OTC sectors |
| Intense generic competition in mature markets |
Strategic acquisitions to expand pipeline |
| Regulatory delays or rejections |
Expansion into emerging markets with growing healthcare needs |
| Supply chain disruptions |
Innovation in formulation technologies for patent extensions |
Key Takeaways
Taro has built a positioning rooted in manufacturing quality, product pipeline development, and strategic focus on high-margin therapeutic niches. Expansion into emerging markets and acquisition initiatives represent growth avenues. However, patent expirations and fierce competition in mature markets require vigilant pipeline management and operational efficiency.
FAQs
1. What are Taro’s primary revenue drivers?
Dermatology topical medications and anti-infectives account for the majority of Taro’s revenue, leveraging its manufacturing expertise and targeted pipeline.
2. How does Taro protect its market share post-patent expiration?
By securing additional patent protections, leveraging exclusivity periods, and developing formulation innovations that extend the lifecycle of existing drugs.
3. What markets are strategic for Taro’s growth?
North America remains core, but expansion into Latin America, Asia, and emerging markets presents significant growth potential.
4. How does Taro’s R&D approach differ from larger competitors?
Taro focuses on formulation patents and process improvements for niche products rather than broad-spectrum R&D. Its pipeline emphasizes quick regulatory approval and market entry.
5. What are the major challenges facing Taro?
Patent cliffs, high generic market saturation, regulatory delays, and supply chain risks pose ongoing challenges.
References
[1] Taro Pharmaceuticals. (2022). Annual Report.
[2] IQVIA. (2022). Market Share Data – Dermatology Segment.
[3] US Food and Drug Administration. (2022). ANDA Approval Data.