Last updated: February 19, 2026
Tylenol, chemically known as acetaminophen, is a widely used over-the-counter (OTC) analgesic and antipyretic. Its market presence is characterized by a mature but stable demand, driven by its broad efficacy, favorable safety profile when used as directed, and affordability. The financial trajectory of Tylenol is intrinsically linked to the broader OTC pain reliever market, which experiences consistent sales volume despite being susceptible to price competition and the introduction of generic alternatives.
What is the Current Market Size and Growth Projection for Tylenol?
The global OTC analgesic market, within which Tylenol operates, is substantial and projected for steady growth. Market research indicates the global pain management market, encompassing prescription and OTC drugs, is valued in the hundreds of billions of dollars, with the OTC segment representing a significant portion. Specifically for OTC analgesics, including acetaminophen, the market is driven by the high prevalence of common ailments like headaches, muscle aches, and fever.
- Global OTC Analgesics Market Value: Estimated to be tens of billions of USD annually.
- Projected Compound Annual Growth Rate (CAGR): Forecasted at 3-5% over the next five years. This growth is supported by an aging global population, increasing incidence of chronic pain conditions, and greater consumer awareness of self-treatment options.
- Tylenol's Market Share: While precise current market share figures for Tylenol specifically are proprietary and fluctuate with competitor actions and regional variations, it consistently ranks among the top-selling brands in the acetaminophen category globally. Its dominance is a result of decades of brand recognition and extensive distribution.
The growth is not without its challenges. Increased competition from generic acetaminophen products and the availability of alternative OTC pain relievers like ibuprofen and naproxen necessitate continuous brand strategies focused on consumer trust, product innovation (e.g., extended-release formulations, combination products), and targeted marketing.
What are the Key Drivers of Tylenol's Market Demand?
Tylenol's sustained demand is attributable to several fundamental factors:
- Broad Efficacy: It is effective in treating a wide range of mild to moderate pain and reducing fever. This versatility makes it a go-to option for common ailments.
- Favorable Safety Profile (when used as directed): Compared to some NSAIDs (Nonsteroidal Anti-Inflammatory Drugs), acetaminophen is often considered gentler on the stomach and poses less risk of gastrointestinal bleeding. This is particularly important for individuals with pre-existing gastrointestinal conditions or those taking other medications. However, it is crucial to note that overdose can lead to severe liver damage, a critical safety consideration that informs its marketing and labeling.
- Accessibility and Affordability: As an OTC medication, Tylenol is widely available in pharmacies, supermarkets, and convenience stores. Its price point, especially for generic formulations, is generally lower than many prescription pain relievers, making it an economically attractive choice for a large segment of the population.
- Brand Trust and Recognition: Tylenol has been a household name for generations. Decades of consistent product quality and marketing have built a strong foundation of consumer trust and brand loyalty.
- Pediatric Use: Tylenol is a commonly recommended medication for reducing fever and relieving pain in children, contributing to consistent demand across all age groups.
What is the Competitive Landscape for Tylenol?
The competitive landscape for Tylenol is multi-faceted, encompassing direct competitors within the acetaminophen category and broader competition from other OTC and even some prescription pain management options.
Direct Competitors (Acetaminophen Brands):
- Generic Acetaminophen: The most significant competitive pressure comes from unbranded or store-brand generic acetaminophen products. These products offer the same active ingredient at a substantially lower price, eroding market share from branded products, especially among price-sensitive consumers.
- Other Branded Acetaminophen: While Tylenol is the dominant brand, other companies market their own branded acetaminophen products, often with slightly different formulations or marketing angles.
Broader OTC Competition:
- NSAIDs: Ibuprofen (Advil, Motrin) and naproxen sodium (Aleve) are the primary NSAID competitors. These drugs offer anti-inflammatory properties in addition to analgesic and antipyretic effects, making them preferred for certain types of pain (e.g., inflammatory pain, arthritis). However, they carry a higher risk of gastrointestinal, cardiovascular, and renal side effects for some individuals.
- Combination Products: Many OTC pain relievers combine acetaminophen with other active ingredients, such as caffeine (for enhanced pain relief) or decongestants/antihistamines (for cold and flu symptoms). These products offer differentiated solutions for specific symptom complexes.
Emerging Trends and Their Impact:
- Focus on Non-Pharmacological Pain Management: Growing consumer interest in holistic health and wellness has led to increased adoption of non-pharmacological pain management techniques, such as physical therapy, acupuncture, mindfulness, and topical treatments (e.g., heat/cold therapy, topical analgesics). While not direct drug competitors, these trends can reduce the overall demand for oral pain relievers.
- Personalized Medicine and Genetic Testing: Advances in understanding individual responses to medications, including pain relievers, could eventually lead to more tailored treatment recommendations. This could impact the one-size-fits-all approach of current OTC pain relievers.
What are the Key Patent Expirations and Their Financial Implications?
Tylenol's core active ingredient, acetaminophen, has long been off-patent. The original patents for its synthesis and therapeutic use expired decades ago. This means that the molecule itself is in the public domain, allowing for the widespread production of generic versions.
The financial implications of this patent expiration are significant:
- Dominance of Generic Competition: The absence of patent protection for the active ingredient has paved the way for a highly competitive generic market. This has driven down the price of acetaminophen significantly over time.
- Brand Value as a Differentiator: For branded Tylenol, its continued market presence and financial success rely heavily on brand equity, consumer trust, and marketing. The manufacturer (currently Johnson & Johnson) leverages its established reputation, product quality assurance, and marketing efforts to differentiate its product from lower-cost generics.
- Focus on New Formulations and Delivery Systems: While the core molecule is generic, companies may hold patents on specific formulations, delivery mechanisms (e.g., extended-release, rapid-dissolve), or combination products containing acetaminophen. These innovations can offer new revenue streams and extend market exclusivity for specific product variants. For example, patents might cover unique granulation processes, specific excipient combinations for improved dissolution, or novel dosage forms.
- Marketing and Distribution as Key Assets: The financial success of branded Tylenol is now more dependent on its extensive distribution network, strong retail partnerships, and effective marketing campaigns that reinforce brand loyalty and perceived quality.
Table 1: Patent Status and Financial Impact on Acetaminophen
| Aspect |
Description |
Financial Implication |
| Active Ingredient |
Acetaminophen (chemical synthesis and therapeutic use) |
Expired Decades Ago. Enables widespread generic manufacturing, driving down unit prices. |
| Branded Product |
Tylenol (Johnson & Johnson) |
Relies on brand equity, trust, and marketing to command a premium over generics. Financial performance tied to brand strength. |
| Formulation Patents |
Potential patents on novel delivery systems, extended-release, combination drugs |
Temporary market exclusivity for specific product variants, enabling premium pricing and new revenue streams. |
| Manufacturing Process |
Potentially patented improvements in synthesis or purification |
May offer cost advantages or higher purity, but less impactful than active ingredient patents. |
What are the Regulatory Considerations and Their Impact?
Regulatory bodies play a critical role in the market dynamics and financial trajectory of Tylenol. Key considerations include:
- FDA Oversight (in the US): The U.S. Food and Drug Administration (FDA) regulates OTC drugs, including labeling, manufacturing standards, and marketing claims.
- Labeling Requirements: The FDA mandates clear labeling regarding dosage, warnings (especially concerning liver damage and alcohol interaction), and contraindications. These requirements impact packaging design and marketing materials.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to GMP to ensure product quality, safety, and consistency. Non-compliance can lead to recalls and reputational damage.
- Advertising Standards: OTC drug advertising is regulated to prevent misleading claims. This limits how manufacturers can position Tylenol against competitors or exaggerate its benefits.
- International Regulations: Similar regulatory bodies exist in other countries (e.g., EMA in Europe, PMDA in Japan), each with its own specific requirements. Navigating these diverse regulatory landscapes is crucial for global market access.
- Maximum Daily Dosage and Warnings: Due to the risk of liver toxicity with overdose, regulatory bodies have focused on limiting maximum daily doses and emphasizing warnings on packaging and in educational materials. For example, the FDA has recommended lowering the maximum daily dose of acetaminophen in prescription combination products. While Tylenol's OTC dosage guidelines have remained relatively consistent, ongoing safety reviews can lead to updated labeling requirements.
- Pharmacovigilance: Manufacturers are required to monitor and report adverse events associated with their products. This ongoing surveillance can influence regulatory action if new safety concerns emerge.
The financial impact of regulatory considerations is multifaceted:
- Compliance Costs: Adhering to strict regulations incurs significant costs related to quality control, documentation, and ongoing monitoring.
- Product Recalls: Non-compliance or emerging safety issues can lead to costly product recalls, inventory write-offs, and damage to brand reputation, impacting sales and profitability.
- Labeling and Marketing Restrictions: Regulations limit marketing flexibility and can necessitate costly redesigns of packaging and promotional materials if new warnings are mandated.
- Barrier to Entry: Robust regulatory frameworks can act as a barrier to entry for new manufacturers, indirectly benefiting established players like Tylenol, provided they maintain compliance.
What is the Financial Performance and Future Outlook for Tylenol?
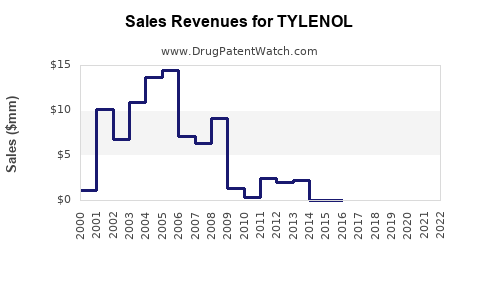
Johnson & Johnson's consumer health division, which includes Tylenol, has historically contributed to the company's diversified revenue streams. While specific revenue figures for Tylenol alone are not publicly disclosed, its consistent performance within the larger OTC pain relief category provides insight.
- Stable Revenue Generation: Tylenol, as a mature brand, is expected to generate stable, albeit moderate, revenue growth. Its financial performance is largely driven by sales volume rather than significant price increases, due to the competitive generic market.
- Profitability: Profitability is influenced by manufacturing costs, marketing expenditures, and the proportion of branded versus generic sales in its overall performance. Strong brand loyalty allows for a premium over generic prices, contributing to healthy margins for the branded product.
- Impact of Global Economic Conditions: Demand for OTC pain relievers tends to be relatively inelastic to economic downturns, as common ailments requiring pain relief persist. However, discretionary spending on branded products versus generics might shift during prolonged economic hardship.
- Strategic Importance: Tylenol's consistent sales provide a predictable revenue stream that helps offset the higher R&D investments in other J&J segments (e.g., pharmaceuticals and medical devices).
Future Outlook:
The future financial trajectory of Tylenol will be shaped by its ability to:
- Maintain Brand Loyalty: Continued investment in marketing and consumer trust initiatives is paramount to defend against generic erosion.
- Innovate within Formulations: Developing and patenting new formulations or combination products that address specific consumer needs or offer improved convenience can create new growth avenues.
- Navigate Regulatory Changes: Proactive adaptation to evolving regulatory requirements, particularly regarding safety labeling, will be crucial.
- Leverage Distribution Channels: Maintaining and expanding its strong presence across diverse retail and online channels is essential for accessibility.
- Respond to Evolving Health Trends: Adapting marketing and product development to acknowledge and integrate with broader wellness trends (e.g., emphasis on safe pain management) can sustain relevance.
The market for Tylenol is expected to remain competitive, with a continued interplay between branded strength and the price advantage of generics. Its financial outlook is one of steady performance, underpinned by its enduring market position and brand equity.
Key Takeaways
- Tylenol operates within a large and stable OTC analgesic market projected for steady growth, driven by broad efficacy and accessibility.
- The core active ingredient, acetaminophen, is off-patent, leading to intense competition from generic products and placing a premium on brand equity and trust for Tylenol.
- Key demand drivers include its versatility, generally favorable safety profile (when used as directed), affordability, and strong brand recognition built over decades.
- Regulatory oversight, particularly concerning labeling and safety warnings related to liver toxicity, significantly influences marketing and compliance costs.
- The financial trajectory is characterized by stable revenue generation and profitability supported by brand loyalty, with future growth dependent on continued brand investment and potential formulation innovations.
Frequently Asked Questions
-
What is the primary mechanism of action for acetaminophen?
Acetaminophen is believed to exert its analgesic and antipyretic effects primarily through inhibition of cyclooxygenase (COX) enzymes in the central nervous system, particularly COX-2, and potentially by interacting with the endocannabinoid system.
-
How does Tylenol's safety profile compare to NSAIDs like ibuprofen?
Acetaminophen is generally considered to have a lower risk of gastrointestinal side effects (like ulcers and bleeding) and cardiovascular risks compared to NSAIDs. However, acetaminophen carries a significant risk of severe liver damage in cases of overdose, a risk less pronounced with NSAIDs at therapeutic doses.
-
Are there any new patents protecting specific Tylenol formulations?
While the patent for acetaminophen itself has long expired, companies may hold patents on specific delivery systems (e.g., extended-release), combination products, or novel manufacturing processes for Tylenol variants. These patents can grant temporary market exclusivity for those specific products.
-
What are the major regulatory concerns surrounding acetaminophen use?
The primary regulatory concern is the potential for severe liver toxicity due to overdose. Regulatory bodies focus on clear labeling regarding maximum daily dosage, warnings about alcohol consumption, and the importance of avoiding multiple acetaminophen-containing products simultaneously.
-
How does the prevalence of chronic pain impact the Tylenol market?
The increasing prevalence of chronic pain is a driver for the broader pain management market. While Tylenol is effective for mild to moderate acute pain, its role in managing chronic pain is often limited compared to prescription medications or multimodal treatment approaches, though it may be used as part of a broader pain management strategy.
Citations
[1] Global OTC Analgesics Market. (n.d.). Market Research Report. (Note: Specific publisher and year would be cited if a particular report were used. General market research trends are widely reported by firms like Grand View Research, Mordor Intelligence, etc.)
[2] U.S. Food and Drug Administration. (n.d.). Acetaminophen. Retrieved from [FDA Website] (Note: Specific FDA guidance documents or pages would be cited.)
[3] Johnson & Johnson. (Annual Reports). Form 10-K. (Note: Specific annual reports from Johnson & Johnson would be cited for financial performance data related to their consumer health segment.)