The global biopharmaceutical industry is currently situated at a critical juncture, navigating a structural transformation defined by the convergence of massive revenue exposure and a tightening regulatory landscape. Between 2025 and 2030, the sector faces a “patent cliff” of unprecedented magnitude, with nearly 200 drugs—including approximately 70 blockbuster products—set to lose exclusivity.1 This looming transition puts an estimated to
in annual revenue at risk, representing a staggering transfer of value from innovator firms to generic and biosimilar competitors.4 For regulatory and legal service firms, this environment creates a critical business development imperative: the delivery of sophisticated lifecycle extension (LCE) audits that go beyond traditional legal counsel to provide strategic, data-driven frameworks for revenue preservation and market dominance.
The magnitude of this impending patent cliff is approximately three times the size of the previous major wave seen in the mid-2010s.5 This financial reckoning is not merely a localized event for a few firms; it is a systemic shift affecting one-sixth of the total annual revenue of the entire pharmaceutical industry.5 The concentration of risk is particularly acute among the top-tier global pharmaceutical companies, many of whom derive over half of their revenue from a single asset or a small cluster of products nearing loss of exclusivity (LOE). For these organizations, the expiration of a primary patent is not just a commercial challenge but an existential threat that can cause market share to plummet by as much as within months of a key patent expiring.8
The Macroeconomic Landscape of the 2025-2030 Patent Cliff
The pharmaceutical industry operates on a cycle of innovation and commoditization, where the period of market exclusivity serves as the economic engine for the entire sector.9 This period is the reward for a decade or more of high-risk, capital-intensive research and development (R&D) that costs, on average, over to
per approved medicine.9 However, the reality of the patent system often dictates that by the time a drug reaches the pharmacy shelf, a significant portion of its twenty-year patent life has already eroded during the discovery, preclinical, and clinical trial phases.11
Table 1: High-Exposure Assets and Revenue at Risk (2025-2030)
| Brand Name | Active Ingredient | Innovator Company | Estimated US Expiry | Peak Annual Sales (Est.) | Primary Strategy/Signal |
| Keytruda | Pembrolizumab | Merck & Co. | 2028 | SC Reformulation (Qlex) 5 | |
| Eliquis | Apixaban | BMS / Pfizer | 2026-2029 | Settlements/Extensions 4 | |
| Opdivo | Nivolumab | Bristol-Myers Squibb | 2028 | Combination therapies 5 | |
| Stelara | Ustekinumab | J&J | 2025 | Biosimilar settlements 4 | |
| Eylea | Aflibercept | Regeneron / Bayer | 2025-2026 | Device/Dosing innovation 6 | |
| Enbrel | Etanercept | Amgen | 2025 | Biosimilar defense 12 | |
| Ozempic | Semaglutide | Novo Nordisk | 2026 | Undisclosed | Metabolic franchise defense 6 |
| Humira | Adalimumab | AbbVie | 2023 (Active LOE) | Pipeline migration (Skyrizi/Rinvoq) 2 |
The financial impact of these expirations is immediate and catastrophic for the unprepared. Branded drugs typically lose to
of their market share within the first 12 to 24 months of generic entry.6 For small-molecule drugs, the decline is often described as a “cliff,” while for biologics, the complexity of manufacturing and the “highly similar” nature of biosimilars transform the revenue decline into a more manageable “patent slope”.9 This distinction is critical for service firms when pitching LCE audits, as the tactics required to defend a small molecule differ fundamentally from those used to protect a biologic franchise.
This upcoming wave of expirations is forcing companies to aggressively restock their pipelines and master the art of lifecycle management to survive.9 Financial markets are forward-looking, and a looming, unaddressed patent cliff is a massive red flag for investors and analysts, leading to a discounting of a company’s future earnings potential long before the actual expiration date.9 Consequently, the strategic extension of a drug’s commercial life is no longer optional; it is a critical determinant of market leadership and financial stability.13
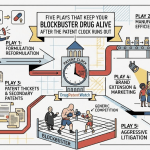
The Strategic Framework of Lifecycle Extension Audits
Modern Lifecycle Management (LCM) is defined as a proactive, holistic discipline designed to optimize a product’s clinical and commercial profile at every stage of its journey.8 It is a fusion of science, law, and marketing aimed at maximizing value through both therapeutic and commercial strategies.8 For service firms, the pitch for an LCE audit must be framed as a “high-yield insurance policy” that protects the massive R&D investments inherent in modern drug development.14
The LCE audit is essentially a strategic triage. It identifies “white space” for new patents, uncovers vulnerabilities in the existing portfolio, and provides a decision-making compass for C-suite executives who must balance commercial strategy with medical responsibility.8 The audit must evaluate the product through three fundamental pillars: Developmental & Clinical, Commercial & Market-Facing, and Legal & Regulatory.9
Developmental and Clinical Strategies
Innovators can extend their market presence by enhancing the core asset itself. This involves identifying new therapeutic uses, improving delivery mechanisms, or optimizing dosing to increase patient adherence and safety. These strategies often focus on reducing the “friction points” that patients experience in real-world settings.9
- Indication Expansion: This is a high-ROI strategy often deployed mid-lifecycle. By treating additional conditions or entering new patient populations, the firm can expand the total addressable market (TAM) while securing new patents for specific uses.9 Oncology and immunology are primary areas for expansion, as a single molecule like pembrolizumab can be tested across dozens of solid tumor indications.3
- Next-Generation Formulations: Moving from immediate-release (IR) to modified-release (MR) or extended-release (ER) versions creates new patent-protected lines. For example, Pfizer transitioned a twice-daily 5-mg tablet (tofacitinib citrate) into an 11-mg once-daily modified-release tablet (Xeljanz XR).9
- Dosing Frequency Optimization: Enhancing convenience for the patient is a powerful defensive tool. In the osteoporosis market, drugs like Boniva were successfully reformulated from a once-daily tablet to a once-monthly oral treatment, and eventually to a quarterly intravenous injection to accommodate specific patient needs.9
- Route of Administration Changes: Transitioning a drug from intravenous (IV) to subcutaneous (SC) injection can dramatically improve the patient experience. Merck’s development of Keytruda Qlex is a prime example, turning a 30-minute IV infusion every three weeks into a 2-minute SC injection every six weeks.2 This reformulation cost between
and
but is expected to preserve
to
in annual revenue, representing an ROI of over
.5
- Fixed-Dose Combinations (FDCs): Combining two or more proven active pharmaceutical ingredients (APIs) into a single dosage form reduces “pill burden” and can offer synergistic therapeutic benefits. Novartis’s Entresto, which combines valsartan and sacubitril into a supramolecular co-crystal, leveraged the complementary mechanisms of both compounds to extend exclusivity beyond the original patents of its components.15
Legal and Regulatory Strategies
The legal pillar focuses on the intellectual property (IP) fortress and the regulatory pathways that govern market entry. The audit must scrutinize the “IP Onion,” starting from the core composition of matter patent and moving outward to secondary patents covering formulations, methods of use, and manufacturing processes.17
- Patent Term Extension (PTE): Under 35 U.S.C. § 156, firms can reclaim up to five years of patent life lost during the lengthy FDA regulatory review process.11 The calculation is based on the sum of the “approval period” (time FDA spends reviewing the NDA) plus one-half of the “testing period” (time from IND effectiveness to NDA filing).11 However, this is subject to a strict 14-year cap from the date of drug approval, making the selection of the “champion” patent for extension a critical tactical decision.11
- Patent Thicketing and Evergreening: Building a “formidable fortress” of interconnected secondary patents can delay competitors by years. AbbVie’s Humira strategy involved 247 patent applications (132 granted), with
filed after original FDA approval to protect manufacturing and treatment methods.5
- The 505(b)(2) Arbitrage: This regulatory pathway allows firms to rely on existing safety data from a reference drug while introducing improvements. It offers a
to
reduction in development costs compared to a new molecular entity (NME), with a faster timeline to market.16
- Orphan Drug Exclusivity (ODE): Treating rare diseases affecting fewer than 200,000 people in the U.S. grants seven years of market exclusivity, blocking the approval of the same drug for the same indication.11
The Impact of the Inflation Reduction Act (IRA) on Lifecycle Management
Service firms must account for the seismic shift introduced by the Inflation Reduction Act of 2022. The IRA grants the Centers for Medicare & Medicaid Services (CMS) the authority to “negotiate” (effectively set) prices for high-expenditure drugs covered under Medicare Part B and Part D.23 This legislation alters the economics of the prescription drug market, reducing prices for some brand-name drugs while potentially discouraging drug development.23
The Small Molecule vs. Biologic Dichotomy
The IRA introduces a “pill penalty” that unfairly discriminates against small-molecule medicines, which typically come in pill form. These drugs are subject to government price setting after only 9 years of market pricing, whereas biologics are allowed 13 years.24 This four-year gap has immediate and long-lasting consequences for biopharma R&D investment levels and portfolio strategy.27
Table 2: Comparative Lifecycle Impact of the IRA
| Metric | Small Molecule (Pill) | Biologic (Large Molecule) | Implications for Strategy |
| Negotiation Trigger | 9 Years Post-Launch | 13 Years Post-Launch | Shorter window to recoup R&D 24 |
| Pre-Negotiation Period | Shorter than historic average | Stable | Shifts R&D toward biologics 27 |
| Early-Stage Funding Change | Decrease since 2021 | Potential Increase | Stifles innovation for diseases of aging 24 |
| Medicare Exposure Risk | Persistent over time | Variable | Higher risk for elderly-focused drugs 23 |
| Post-Approval Research | Disincentivized | Moderately affected | Fewer new indications pursued 25 |
The IRA also requires drug companies to discount medications if prices rise faster than inflation.24 For Medicare Part D plan sponsors, the act increases the proportion of drug costs they are responsible for in the catastrophic coverage phase from to
, creating a powerful incentive for them to seek more significant rebates from manufacturers.27 An LCE audit in this era must include a “Medicare Exposure Analysis” to determine how early price negotiations might truncate the traditional patent-driven lifecycle.23
USPTO Regulatory Shifts and Patent Thicket Vulnerabilities
The legal landscape is further complicated by evolving policies at the United States Patent and Trademark Office (USPTO). In 2024, the USPTO proposed a rule that would have fundamentally altered the use of terminal disclaimers to overcome “obviousness-type” double patenting rejections.28 Terminal disclaimers are used to ensure that the term of a new patent does not extend beyond an existing duplicative patent, but they enable the issuance of patents with overlapping claims—a practice prevalent in “patent thickets”.28
The proposed rule would have required patent applicants to agree that the patent would be enforceable only if it were not tied to another patent containing a claim held invalid by a federal court or the PTAB.28 Essentially, an entire patent family could have been wiped out if just one claim in one patent was found invalid.30 Although the USPTO withdrew the proposed rule in December 2024 due to resource constraints and heavy industry opposition, the strategic risk remains.29 Service firms must counsel clients to reevaluate their continuation application practices to avoid unnecessary terminal disclaimers that could create systemic risk for an entire portfolio.30
The Anatomy of an LCE Audit: Technical Triage and Methodology
A comprehensive LCE audit for biopharma clients is a multi-phase technical endeavor that requires a deep dive into chemical structures, manufacturing protocols, and competitive landscapes. The objective is to identify obvious threats early in the development lifecycle before significant capital is committed.14
Phase 1: Freedom-to-Operate (FTO) Analysis
The core of any audit is the FTO analysis, which answers the critical question: “Can we bring this product to market legally and profitably?”.14 A preliminary FTO is a “scouting mission” that identifies blocking patents held by competitors. It is important to remember that a patent is a “negative right” (the power to exclude others) rather than a “positive right” (the right to practice the invention).14
- Small Molecules: Analysis must deconstruct the precise chemical structure, including salts, polymorphs, enantiomers, prodrugs, and excipients. It must also evaluate the manufacturing process for specific reagents or intermediates.14
- Biologics: Technical focus shifts to amino acid/nucleotide sequences, host cells, expression systems, and purification processes. The final formulation and the delivery device (e.g., auto-injector) are also scrutinized.14
The FTO analysis must also check for “red flags” in academic spin-outs, such as incomplete inventorship, myopic filing (failure to file in key international markets), and “reach-through” claims from Material Transfer Agreements (MTAs).14
Phase 2: Competitive Intelligence and Patent Landscape Mapping
Using patent data as an “offensive weapon,” firms can map the density of filings in a therapeutic area to identify “white space” opportunities.13 Visualization of these landscapes reveals gaps that might represent overlooked biological targets or mechanisms of action.13 Systematic monitoring of competitor filings can signal an impending “product hop” (e.g., a cluster of filings for an ER formulation) years before execution, allowing for adjusted strategies.13
Phase 3: Regulatory Pathway Optimization (505(b)(2))
The 505(b)(2) pathway represents a unique regulatory arbitrage, allowing a sponsor to “borrow” an innovator’s billion-dollar data set to support an improved version of a drug.21 The audit must evaluate the feasibility of this path, balancing innovation with capital efficiency.
Table 3: Economic Comparison of Development Pathways
| Stage of Development | 505(b)(1) NDA (NCE) | 505(b)(2) Hybrid | 505(j) ANDA (Generic) |
| Typical Dev. Cost | |||
| Timeline to Market | |||
| Clinical Requirement | Full Phase 1, 2, and 3 | Bridging Studies (PK/BE) | Bioequivalence only |
| Risk Level | High (Wet lab failures) | Moderate (CMC delays) | Low (Commodity) |
| Innovation Type | New Molecular Entity | Improved/Modified Drug | Replication |
Despite the seductive promise of low costs, the 505(b)(2) pathway is fraught with technical risks. Chemistry, Manufacturing, and Controls (CMC) deficiencies are the leading cause of “Complete Response Letters” (rejections) from the FDA, with of second-cycle delays attributed to “formulation gymnastics,” such as trying to force an insoluble drug into a liquid form or stable patch.16
Business Development: Pitching the LCE Audit to the C-Suite
Service firms must pivot their business development (BD) strategies from transactional legal tasks to high-value strategic consulting. The pitch should focus on “The Art of the Second Act”—the strategic extension of a drug’s commercial life as a critical determinant of long-term survival.13
The “IP Onion” Pitch Strategy
The LCE audit should be framed as a process of “architecting a durable, multi-layered patent estate”.17 Instead of selling a single patent application, firms should sell the “IP Onion”:
- The Core: Composition of matter patents on the API or biologic.17
- Layer 2: Formulations, dosing regimens, and delivery systems.5
- Layer 3: Methods of use for new indications.17
- Layer 4: Manufacturing and purification processes.5
Quantifying ROI and Strategic Value
Pitching the value of advanced analytics and automated tools is essential. Firms that adopt advanced patent analytics and AI-driven competitive intelligence (CI) can achieve measurable improvements in profitability and efficiency.
- EBITDA Improvement: McKinsey projects that advanced analytics can boost pharmaceutical EBITDA by
to
within five years.33
- Cost Reduction: AI can reduce preclinical development costs by
to
and decrease clinical trial costs by up to
through data-driven patient selection.33
- Time-to-Value: Efficient Contract Lifecycle Management (CLM) can slash contract cycle times by
, potentially accelerating trial initiation and drug development timelines by months.34
Table 4: Key Service Providers and Market Specializations
| Firm / Category | Key Focus / Capabilities | Strategic Role in LCE Audit |
| McKinsey / BCG / Bain | Global Strategy | R&D productivity, launch factories 35 |
| Deloitte / PwC / KPMG | Professional Services | ESG, regulatory & tax advisory, supply chain 35 |
| Finnegan | IP Strategy & Litigation | Hatch-Waxman leader, patent strategy 36 |
| Latham & Watkins | High-Stakes IP Litigation | Brand-on-brand biologic disputes, ITC wins 37 |
| Goodwin Procter | Full Lifecycle Advisory | Ranking leader in regulatory, corporate, and IP 38 |
| IQVIA / ZS Associates | Data & Analytics | Real-world data, commercial strategy, sales effectiveness 35 |
| The FDA Group | Ex-FDA Consulting | Regulatory strategy, safety, CMC specialists 39 |
| ProPharma Group | Compliance & Auditing | GxP audits, PAI readiness, supplier oversight 40 |
Operational Excellence and Digital Transformation
The future of lifecycle management belongs to firms that operate at the frontier of biology, technology, and data.42 Winning organizations will run on adaptive, AI-enabled operating systems that automate transactional work and augment analytical decision-making.42
- Harnessing Breakthrough Science: Companies must translate advances in genomics, synthetic biology, and AI-driven discovery into therapies that cure or prevent disease.42
- Designing Novel Patient Experiences: Moving beyond transactional prescriptions to create “branded health journeys” with digital companions and personalized support builds long-term loyalty.42
- End-to-End Digital Integration: Dissolving silos across R&D, manufacturing, commercial, and compliance compresses cycle times and reduces the cost to operate.42
Failure Analysis: Case Studies and Lessons Learned
A robust LCE audit must include “failure analysis” to identify patterns that lead to premature generic entry or regulatory rejection. Successful litigation often hinges on the “Anatomy of a Victory,” while failures often stem from scientific, operational, or strategic risks.10
- Tecfidera (dimethyl fumarate): Biogen faced challenges from generic makers like Mylan.43
- Vascepa (icosapent ethyl): Amarin saw its patents challenged by Hikma and Dr. Reddy’s.43
- Linhaliq: A 505(b)(2) program that failed due to product quality and in vitro release method issues, proving that a “known drug” does not guarantee an “easy formulation”.16
- Operational Failures: Common pitfalls include poor protocol design and recruitment shortfalls, which lead to costly delays.10
Nuanced Conclusions and Strategic Recommendations
The 2025-2030 patent cliff represents both an existential threat and an unprecedented opportunity for biopharma companies and their service providers. The transition from a “patent cliff” to a “patent slope” for biologics, and the aggressive implementation of reformulation strategies, are the primary defenses against revenue erosion.
Actionable Recommendations for Service Firms
- Integrated Medicare Risk Modeling: Service firms must integrate IRA price-setting projections into every LCE audit. The 9-year vs. 13-year window is now a primary driver of R&D resource allocation.24
- Prioritizing CMC Readiness in 505(b)(2) Pitches: Given that
of delays are CMC-related, audits must emphasize manufacturing stability and analytical method validation over clinical safety.16
- Leveraging Real-World Evidence (RWE): Audits should identify opportunities to use RWE for continuous post-market surveillance and label expansions, reducing the need for costly new trials.44
- Dismantling “Junk” Patent Risks: With increased scrutiny on Orange Book listings and terminal disclaimers, firms should conduct “defensive audits” to remove weak patents that could trigger FTC investigation or systemic portfolio failure.18
The complexity of the current regulatory and legal environment requires a multi-disciplinary approach that combines deep scientific knowledge with legal acumen and sophisticated data analytics. Regulatory and legal service firms that can deliver these high-insight LCE audits will become indispensable partners to the biopharmaceutical industry as it navigates the most severe patent cliff in its history.
Works cited
- 3 critical moves for Biopharma patent cliff survival – YouTube, accessed March 1, 2026, https://m.youtube.com/watch?v=VEr2hUn3zXg
- Biopharma patent cliff survival strategy: 3 critical moves – Alcimed, accessed March 1, 2026, https://www.alcimed.com/en/insights/patent-cliff/
- Patent Cliff Looms – Can Kv1.3 Take the Baton for the Next Wave of Drug Licensing?, accessed March 1, 2026, https://www.unibestpharm.com/Patent-Cliff-Looms-Can-Kv1-3-Take-the-Baton-for-the-Next-Wave-of-Drug-Licensing-id47702696.html
- When Blockbusters Fall, Business Development Rises: Win the …, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/when-blockbusters-fall-business-development-rises-win-the-patent-cliff/
- $300 Billion in Pharma Revenue Loses Patent Protection by 2030 …, accessed March 1, 2026, https://deepceutix.com/insights/patent-cliff-reformulation
- The Drug Patent Cliff Portfolio: A Strategic Guide to Identifying and Investing in Companies Facing Major Expiries – DrugPatentWatch, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/the-drug-patent-cliff-portfolio-a-strategic-guide-to-identifying-and-investing-in-companies-facing-major-expiries/
- Patent Cliff Strategy: Role of Competitive Intelligence Software | IntuitionLabs, accessed March 1, 2026, https://intuitionlabs.ai/articles/competitive-intelligence-patent-cliff
- The Art of the Evergreening: A Deep Dive into Drug Life Cycle Management Tactics and How to Challenge Them – DrugPatentWatch, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/the-art-of-the-evergreening-a-deep-dive-into-drug-life-cycle-management-tactics-and-how-to-challenge-them/
- Beyond the Patent Cliff: 15 Strategies for Pharmaceutical Lifecycle …, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/beyond-the-patent-cliff-15-strategies-for-pharmaceutical-lifecycle-management/
- A Strategic Investor’s Guide to Pharmaceutical Portfolio Risk Assessment, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/a-strategic-investors-guide-to-pharmaceutical-portfolio-risk-assessment/
- The Math of Monopoly: Why a 20-Year Drug Patent Lasts Only 12 Years – DrugPatentWatch, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/the-math-of-monopoly-why-a-20-year-drug-patent-lasts-only-12-years/
- Top 10 Blockbuster Drugs Facing U.S. Patent Expirations (2025–2029) – Clival Database, accessed March 1, 2026, https://clival.com/blog/top-10-blockbuster-drugs-facing-us-patent-expirations
- The Evergreening Gambit: A Strategic Guide to Pharmaceutical …, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/the-evergreening-gambit-a-strategic-guide-to-pharmaceutical-patent-lifecycle-management/
- A Pharma Exec’s Guide to Preliminary Freedom-to-Operate Analysis …, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/a-pharma-execs-guide-to-preliminary-freedom-to-operate-analysis/
- Essential Patent Protection Areas for Pharma Startups – Secondary Patents – JD Supra, accessed March 1, 2026, https://www.jdsupra.com/legalnews/essential-patent-protection-areas-for-8499212/
- Review of Drugs Approved via the 505(b)(2) Pathway: Uncovering Drug Development Trends and Regulatory Requirements – DrugPatentWatch, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/review-of-drugs-approved-via-the-505b2-pathway-uncovering-drug-development-trends-and-regulatory-requirements/
- The Patent-Powered Pitch: A Founder’s Guide to Securing Biotech Funding, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/the-patent-powered-pitch-a-founders-guide-to-securing-biotech-funding/
- How to keep your drug on the market for five extra years – DrugPatentWatch, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/how-to-keep-your-drug-on-the-market-for-five-extra-years/
- How does patenting protect pharmaceutical products? – Efpia, accessed March 1, 2026, https://www.efpia.eu/news-events/the-efpia-view/blog-articles/121128-the-degree-to-which-patenting-and-in-particular-secondary-patenting-protect-pharmaceutical-products-during-their-lifecycle-is-often-misconstrued/
- Authorized Generics: Mastering a Controversial Strategy for Pharmaceutical Patent Lifecycle Management – DrugPatentWatch – Transform Data into Market Domination, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/authorized-generics-mastering-a-controversial-strategy-for-pharmaceutical-patent-lifecycle-management/
- The 505(b)(2) Playbook: Scale Your Pipeline Without the Billion-Dollar Gamble, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/the-505b2-playbook-scale-your-pipeline-without-the-billion-dollar-gamble/
- The Real Cost of a 505(b)(2) — What It Takes, Why It’s Worth It, and How to Reduce the Risk – SyneticX, accessed March 1, 2026, https://www.syneticx.com/blog/TheRealCostofa505(b)(2).html
- Research and development intensity and the Inflation Reduction Act’s prescription drug provisions | Brookings, accessed March 1, 2026, https://www.brookings.edu/articles/research-and-development-intensity-and-the-inflation-reduction-acts-prescription-drug-provisions/
- The Inflation Reduction Act Is Negotiating the United States Out of Drug Innovation | ITIF, accessed March 1, 2026, https://itif.org/publications/2025/02/25/the-inflation-reduction-act-is-negotiating-the-united-states-out-of-drug-innovation/
- Inflation Reduction Act | PhRMA, accessed March 1, 2026, https://phrma.org/policy-issues/government-price-setting/inflation-reduction-act
- Mitigating the Inflation Reduction Act’s Adverse Impacts on the Prescription Drug Market, accessed March 1, 2026, https://schaeffer.usc.edu/research/mitigating-the-inflation-reduction-acts-potential-adverse-impacts-on-the-prescription-drug-market/
- Pharma’s New Normal: How the Inflation Reduction Act Will Impact the Biopharmaceutical Industry – L.E.K. Consulting, accessed March 1, 2026, https://www.lek.com/sites/default/files/PDFs/inflation-reduction.pdf
- FTC Submits Comment Supporting Proposed USPTO Terminal Disclaimer Rule, accessed March 1, 2026, https://www.ftc.gov/news-events/news/press-releases/2024/07/ftc-submits-comment-supporting-proposed-uspto-terminal-disclaimer-rule
- USPTO changes course on controversial proposed rule for terminal disclaimers, accessed March 1, 2026, https://www.nixonpeabody.com/insights/alerts/2024/12/16/uspto-changes-proposed-rule-for-terminal-disclaimers
- The USPTO’s Proposed Terminal Disclaimer Rule Change: It’s Radical, But Is It Legal?, accessed March 1, 2026, https://www.iptechblog.com/2024/05/the-usptos-proposed-terminal-disclaimer-rule-change-its-radical-but-is-it-legal/
- USPTO Drops Proposed Rules on Terminal Disclaimers | Foley & Lardner, accessed March 1, 2026, https://www.foley.com/insights/publications/2024/12/uspto-drops-proposed-rules-terminal-disclaimers/
- Uspto Withdraws Consideration of Controversial Change to Terminal Disclaimer Practice, accessed March 1, 2026, https://lifesciences.mofo.com/topics/uspto-withdraws-consideration-of-controversial-change-to-terminal-disclaimer-practice
- Data Analytics in Pharmaceutical Industry: 7 Use Cases Driving ROI in 2026, accessed March 1, 2026, https://sranalytics.io/blog/data-analytics-pharmaceutical-industry/
- AI CLM Software for Biotech: A Guide to Leading Platforms | IntuitionLabs, accessed March 1, 2026, https://intuitionlabs.ai/articles/ai-clm-software-biotech
- Top Pharmaceutical Consulting Firms | IntuitionLabs, accessed March 1, 2026, https://intuitionlabs.ai/articles/top-pharmaceutical-consulting-firms
- Finnegan Wins IP Boutique Firm of the Year, Patent Strategy Firm of the Year, and Hatch-Waxman Litigation Firm of the Year – Branded at the 2025 LMG Life Sciences Americas Awards, accessed March 1, 2026, https://www.finnegan.com/en/firm/news/finnegan-wins-ip-boutique-firm-of-the-year-patent-strategy-firm-of-the-year-and-hatch-waxman-litigation-firm-of-the-year-branded-at-the-2025-lmg-life-sciences-americas-awards.html
- Life Sciences IP Litigation Lawyers | Latham & Watkins LLP | Global Law Firm, accessed March 1, 2026, https://www.lw.com/en/practices/intellectual-property-litigation/life-sciences-ip-litigation
- Goodwin Named an LMG Life Sciences Life Cycle Firm for the Fourteenth Consecutive Year | News & Events, accessed March 1, 2026, https://www.goodwinlaw.com/en/news-and-events/news/2025/10/announcements-lifesciences-goodwin-named-lmg-life-sciences-life-cycle-firm
- Ex-FDA Consultants | The FDA Group, accessed March 1, 2026, https://www.thefdagroup.com/ex-fda-consultants
- Quality & Compliance Services for Pharma and Medical Devices – ProPharma Group, accessed March 1, 2026, https://www.propharmagroup.com/services/quality-and-compliance/
- Top 10 Life Sciences Service Providers for Audit & Supplier, accessed March 1, 2026, https://biobostonconsulting.com/top-10-future-ready-life-sciences-service-providers-elevating-audit-strength-supplier-oversight-compliance-risk-management/
- Future of Pharma: Breakthroughs at Scale – PwC, accessed March 1, 2026, https://www.pwc.com/us/en/industries/pharma-life-sciences/pharmaceutical-industry-trends.html
- Uncovering the Success Patterns in Modern Paragraph IV Litigation – DrugPatentWatch, accessed March 1, 2026, https://www.drugpatentwatch.com/blog/uncovering-the-success-patterns-in-modern-paragraph-iv-litigation/
- The Long Game: Lifecycle of Drug Development | by Saad Ahmad – Medium, accessed March 1, 2026, https://saad-ahmad-umn.medium.com/from-molecule-to-market-the-complete-journey-of-biologic-drug-development-6afb18bf4e68