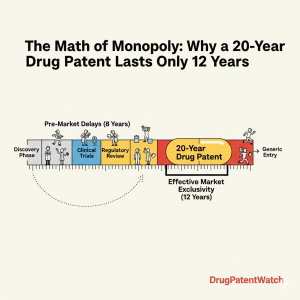
The 20-year patent term is a nominal legal baseline that rarely reflects the commercial reality of a pharmaceutical product. While the United States Patent and Trademark Office (USPTO) grants a statutory monopoly of two decades from the date of filing, the actual window for recouping research and development (R&D) costs—the effective patent life—is significantly shorter.1 For business development and intellectual property (IP) teams, the discrepancy between the legal clock and the commercial calendar is the most critical calculation in the industry. Statutory protections exist to incentivize innovation by allowing companies to charge prices that reflect the immense risk and cost of drug discovery, which now averages over $2.23 billion per successful asset.3 However, the mandatory phases of clinical trials and regulatory review consume between five and 10 years of that granted term before a single dollar of revenue is generated.1 The result is a compressed commercial window, often lasting just seven to 12 years, during which a brand must maximize returns before the “patent cliff” triggers a rapid collapse in market share.2
The Statutory Baseline and the 20-Year Illusion
The foundational legal principle governing patent duration in the United States is rooted in 35 U.S.C. § 154(a)(2). Following the implementation of the Uruguay Round Agreements Act in 1995, the U.S. moved from a system that granted 17 years of protection from the date of issuance to the current standard: 20 years from the earliest effective filing date.1 This change, mandated by the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS), was intended to harmonize global IP standards and prevent patents that could emerge unexpectedly after years of pendency.2 For the life sciences, this shift created a structural disadvantage. Pharmaceutical companies must file for patent protection early in the discovery phase, often before a molecule has even entered preclinical testing. The 20-year clock begins ticking while the drug is still a laboratory hypothesis. Consequently, the period known as “patent pendency”—the time between filing and the actual grant of the patent—averages 3.8 years for new chemical entities (NCEs).1 In some complex cases, this pendency can exceed eight years, effectively eroding the term before the inventor even has the legal right to exclude others.1
The Mechanics of Effective Patent Life (EPL)
How Long Does a Pharmaceutical Patent Last in Practice?
The journey from a patent filing to a pharmacy shelf is a marathon of attrition. A drug candidate must progress through discovery, preclinical research, and three phases of human clinical trials. Discovery and preclinical research alone can take four to seven years, followed by another six to seven years for clinical testing.2 Once the clinical data is compiled, the manufacturer submits a New Drug Application (NDA) or Biologics License Application (BLA) to the Food and Drug Administration (FDA), which then takes an average of six months to two years to review the data.2 This attrition is the primary driver of the pharmaceutical industry’s focus on lifecycle management. Because the effective patent life is so short, every day of market exclusivity for a blockbuster drug can represent millions in revenue. For instance, a single day of exclusivity for a top-selling medication can generate upwards of $8.2 million.2 When the core patent finally expires, the transition is not a gentle decline but a catastrophic financial drop where innovators may lose 90% of their revenue to generic competitors within months.1
| Development Phase | Typical Duration | Patent Life Erosion |
| Discovery & Preclinical | 4–7 Years | Direct Loss |
| Clinical Trials (Phases I-III) | 6–7 Years | Direct Loss |
| FDA Regulatory Review | 0.5–2 Years | Direct Loss |
| Total Pre-Market Time | 10–15 Years | Significant Erosion |
Calculating Patent Term Adjustment (PTA)
To mitigate the erosion caused by bureaucratic delays at the USPTO, Congress established Patent Term Adjustment (PTA) under 35 U.S.C. § 154(b). PTA is an additive mechanism that restores time lost due to specific administrative failures.1 Unlike other extensions, PTA has no statutory maximum duration and is added directly to the end of the 20-year term.1 While PTA ensures the inventor receives the full benefit of a 20-year term despite office inefficiency, it does not address the time lost during FDA review. This distinction is critical: PTA compensates for the time it takes to get the patent, while a separate mechanism—Patent Term Extension—compensates for the time it takes to get the drug to market.
The Three Triggers of PTA: A, B, and C Delays
PTA is calculated based on three categories of USPTO-related delays. A-Delay occurs if the USPTO fails to issue a first Office action within 14 months of filing or fails to respond to an applicant’s reply within four months.1 B-Delay guarantees that a patent will be issued within three years of its filing date, provided the applicant does not file a Request for Continued Examination (RCE).7 C-Delay accounts for time lost to interferences, secrecy orders, or successful appeals.1
Hatch-Waxman and the Restoration of Time
The Drug Price Competition and Patent Term Restoration Act of 1984, commonly known as the Hatch-Waxman Act, represents the grand compromise of the industry.10 It created an expedited path for generic drugs in exchange for allowing innovators to reclaim a portion of the patent life lost during the FDA’s regulatory review process.2 This restoration, known as Patent Term Extension (PTE) under 35 U.S.C. § 156, is subject to strict limitations and caps.
The Calculus of Patent Term Extension (PTE)
The calculation of PTE is a precise exercise in regulatory accounting. It is generally equal to the sum of the “approval period” (the time the FDA spends reviewing the NDA) plus one-half of the “testing period” (the time from the Investigational New Drug application becoming effective until the NDA is filed), less any time the applicant failed to act with due diligence.1
The mathematical formula for the period of extension is:
$$PTE = RRP – PGRRP – DD – \frac{1}{2}(TP – PGTP)$$
Where $RRP$ is the total regulatory review period, $PGRRP$ is the portion of that period occurring before the patent was granted, $DD$ is any period of lack of due diligence by the applicant, $TP$ is the testing phase, and $PGTP$ is the testing phase occurring before the patent grant.12
Statutory Caps and the 14-Year Ceiling
Critical statutory caps prevent this restoration from becoming indefinite. First, the total extension granted cannot exceed five years.1 Second, the “effective patent life cap” stipulates that the total remaining patent term after the extension is applied cannot exceed 14 years from the date of the drug’s approval.1 This 14-year ceiling establishes the federal government’s intended limit for post-approval market protection.
FDA Regulatory Exclusivities: The Parallel Fortress
While patents are property rights granted by the USPTO, the FDA grants a parallel form of protection known as regulatory exclusivity. These exclusivities act as a bar, preventing the FDA from accepting or approving generic applications for a specified period, regardless of whether a patent is in force.1 They are designed to incentivize development in areas that may not be sufficiently protected by patents alone, such as rare diseases or new chemical entities.2
“Patents and exclusivity work in a similar fashion but are distinct from one another… Patents can be issued or expire at any time regardless of the drug’s approval status. Exclusivity attaches upon approval.” 13
New Chemical Entities (NCE) and the 5-Year Barrier
The primary types of regulatory exclusivity in the U.S. include NCE Exclusivity, which provides five years of data exclusivity for drugs containing a never-before-approved active moiety. For four of these five years, the FDA is prohibited from even accepting an ANDA.2
The Power of Orphan Drug Exclusivity (ODE)
Orphan Drug Exclusivity (ODE) grants seven years of market exclusivity for drugs treating rare diseases affecting fewer than 200,000 people in the U.S. This is a powerful barrier that blocks the approval of the same drug for the same indication.2
Biologics and the 12-Year Exclusivity Shield
Under the Biologics Price Competition and Innovation Act (BPCIA), new biologics receive 12 years of market exclusivity. This longer period reflects the greater complexity and higher R&D costs of large-molecule drugs.2
The Six-Month Pediatric Multiplier
One of the most valuable strategic tools in the innovator’s arsenal is Pediatric Exclusivity (PED). Under the Best Pharmaceuticals for Children Act, if a manufacturer conducts clinical studies in pediatric populations at the FDA’s written request, they are awarded an additional six months of market protection.1 The power of PED lies in its nature as a multiplier. It is not a standalone exclusivity but an extension added to the end of all existing patents and exclusivities listed in the Orange Book for that active moiety.1
Strategic Lifecycle Management: Layering and Thickets
Sophisticated pharmaceutical teams do not rely on a single patent to protect their assets. Instead, they construct a multi-layered web of protection known as a “patent thicket”.17 This involves filing dozens, sometimes hundreds, of secondary patents that cover every imaginable aspect of a drug beyond its core chemical structure.17
Secondary Patents: Formulations, Uses, and Processes
This layering allows a company to extend the effective monopoly far beyond the expiration of the original compound patent. By the time a primary patent expires, there may be several secondary patents still in force, often covering the exact formulation or dose that represents the majority of the market.2
| Patent Type | Target of Protection | Strategic Purpose |
| Primary (Composition) | The active pharmaceutical ingredient (API) | The foundational, “crown jewel” protection.2 |
| Secondary (Formulation) | Specific dosage forms, coatings, or carriers | Prevents bioequivalent “copycat” generics.17 |
| Method of Use | New therapeutic indications or dosing regimens | Forces generics to wait for expiration.22 |
| Process/Manufacturing | The unique method of synthesizing the drug | Creates technical hurdles for generic manufacturing.20 |
The 30-Month Stay and Serial Litigation
Under the Hatch-Waxman Act, the mere act of an innovator suing an ANDA applicant triggers a statutory 30-month stay on FDA approval of the generic product.22 This stay is a unique U.S. provision that essentially weaponizes litigation. Even if a secondary patent is perceived as weak, a lawsuit provides an automatic 30-month extension of market exclusivity while the parties litigate.17 For top-selling drugs, this has led to “serial litigation” where innovators stagger the listing of secondary patents to create a perpetual litigation overhang.22
Case Study: The Humira Thicket and Settlement Outcomes
AbbVie’s defense of Humira (adalimumab) redefined the “patent thicket” for the biologic era. AbbVie filed over 250 patent applications and secured more than 130 granted patents on Humira, the majority of which were filed after the drug was already on the market.17 This thicket created a litigation landscape so complex and costly that every major biosimilar competitor eventually chose to settle rather than fight. These settlements delayed biosimilar entry in the U.S. until 2023, seven years after the primary patent had expired.22 This delay cost the U.S. healthcare system an estimated $80 billion to $100 billion, while Humira continued to generate daily revenues of roughly $50 million during the extended monopoly period.17
| Drug Brand | Manufacturer | Total Patents (Approx.) | Impact of Thicket |
| Humira | AbbVie | 136 | Delayed entry by 7 years post-primary expiry.17 |
| Keytruda | Merck | 53–78 | Extension of monopoly until at least 2036.17 |
| Eliquis | BMS/Pfizer | 27 | Delayed generic entry from 2022 to 2028.24 |
Case Study: The Lipitor 180-Day War
Pfizer’s defense of Lipitor (atorvastatin) provides a masterclass in managing the transition from monopoly to competition. Lipitor was a pharmaceutical goliath, with peak annual sales of $12.9 billion.6 As the main U.S. patent approached its November 30, 2011 expiration, Pfizer did not simply surrender. Instead, they executed an “180-Day War” strategy to retain market share.6 Before the patent expired, Pfizer engaged in “direct-to-consumer” marketing to build brand loyalty and pursued a settlement with Ranbaxy. Post-expiration, Pfizer launched its own authorized generic and used aggressive rebate programs to keep the brand on preferred formularies.26 Despite the patent cliff, Pfizer’s strategy allowed it to retain approximately 30% of the market share at the end of the 180-day exclusivity period.26
The Tectonic Shift: Inflation Reduction Act and 2025-2030
The 2022 Inflation Reduction Act (IRA) has introduced a shift in how pharmaceutical patent life is valued. The law grants the federal government the power to negotiate prices for top-selling drugs under Medicare.2 Crucially, the timeline for negotiation eligibility differs by drug type. Small-molecule drugs are eligible for negotiation nine years after approval, while biologics are eligible 13 years after approval.2 This four-year gap has profoundly altered R&D priorities. Because biologics offer a longer window of pricing before government intervention, investors and pharmaceutical companies are shifting capital away from small molecules and toward biologics.2
The European Context: Unified Patent Court (UPC) and Unitary SPCs
While the U.S. remains the most significant market for pharmaceutical returns, Europe is undergoing its own transformation with the launch of the Unified Patent Court (UPC) and the Unitary Patent system.28 Two years into its operation, the UPC is beginning to reshape cross-border enforcement strategies. Originator companies are now adopting “layered” IP strategies in Europe, blending Unitary Patents for broad injunction potential in 18 EU member states with traditional national patents in jurisdictions where they want more procedural flexibility.30 The court has demonstrated a commitment to speed, often resolving cases on the merits within a year.32
Leveraging Patent Data for Competitive Advantage
In an environment where a single day of exclusivity is worth millions, the ability to accurately forecast the loss of exclusivity (LOE) is a core commercial competency.16 Market access, business development, and investment teams can no longer rely on simple expiration dates. They must audit the entire “patent estate” and regulatory landscape. Platforms like DrugPatentWatch have become essential tools for this mandate. By tracking global patent portfolios across 130 countries, monitoring Paragraph IV challenges, and analyzing litigation outcomes, these platforms allow teams to forecast LOE with precision and identify white space in therapeutic areas with high unmet need but limited patent activity.3
The Economics of R&D and Return on Investment
The financial pressure on pharmaceutical firms is mounting as the average cost to develop a single drug asset has climbed to $2.23 billion in 2024.3 This rising cost environment makes the preservation of every month of patent life a matter of corporate survival. Despite these headwinds, the industry has seen a recovery in its internal rate of return (IRR), which rose to 5.9% in 2024, up from a record low of 1.2% in 2022.3 This turnaround is largely attributed to a surge in high-value products in obesity and diabetes entering late-stage pipelines. However, the progress is fragile, as clinical trial complexity and regulatory requirements continue to lengthen development cycles.4
| Financial Metric | 2022 Value | 2024 Value | Trend |
| Average R&D Cost per Asset | $2.28 Billion | $2.23 Billion | Plateauing 3 |
| Internal Rate of Return (IRR) | 1.2% | 5.9% | Improving 3 |
| Average Forecast Peak Sale | $389 Million | $510 Million | Increasing 4 |
Navigating the 2025-2030 Patent Cliff
Between 2025 and 2030, an estimated $236 billion to $400 billion in annual global branded sales are at risk.2 This includes blockbuster drugs like Keytruda and Eliquis.2 For analysts tracking these transitions, granular data is necessary to predict when these walls will crumble. The revenue cliff forces innovators to replenish pipelines through heavy R&D investment and aggressive M&A, with an emphasis on novel mechanisms of action which, although comprising only 23.5% of the pipeline, are projected to generate 37.3% of future revenue.4
The Rise of Patent Pools and Collaborative Discovery
In response to the productivity crisis and the complexity of patent thickets, some organizations are turning to collaborative models. Public health patent pools, such as the Medicines Patent Pool (MPP), act as a one-stop-shop where multiple patent holders voluntarily license their IP to a central entity.36 This reframes the patent system from a purely exclusionary tool into a collective management structure that can accelerate the production of affordable medicines in low- and middle-income countries. Participation in these pools can mitigate reputational risks and help shape markets for future innovative products.36
The Global Dimension: Harmonization and Divergence
While international agreements like TRIPS provide a baseline of 20 years from filing, national implementation varies significantly. In China, recent patent law amendments have introduced patent term extensions similar to the U.S. system, incentivizing companies to prioritize Chinese regulatory submissions to maximize patent life in that market.2 Meanwhile, the European EPO’s “added matter” doctrine and stricter prohibition on double-patenting act as structural barriers against the same types of thickets seen in the U.S..17 This regulatory arbitrage means that a drug’s monopoly period often expires in Europe years before it does in the United States.22
Product Hopping and the Switch Strategy
A final pillar of lifecycle management is “product hopping.” As a core patent nears expiration, a manufacturer may launch a reformulated version of the drug—such as an extended-release tablet or a subcutaneous injection—and use marketing or financial incentives to switch the patient population to the new, patent-protected version.20 A “hard switch” involves removing the original product from the market, forcing patients to choose the new version, while a “soft switch” leaves the original on the market but focuses all marketing efforts on the new entrant.37 This strategy effectively renders the generic version of the old drug obsolete before it even enters the market.
Key Takeaways
- Effective Life vs. Nominal Life: While the statutory term is 20 years from filing, the effective patent life is typically only seven to 12 years due to R&D and regulatory erosion.2
- Restoration Caps: Patent Term Extension (PTE) can restore up to five years of lost time but is strictly capped at a total of 14 years of post-approval market life.1
- The Parallel Path: FDA regulatory exclusivities provide independent protection that can bar generic entry even if patents have expired.2
- Strategic Layering: Success in lifecycle management depends on “patent thickets”—layering secondary patents (formulation, use, process) to protect the market long after the core compound patent expires.17
- The IRA Paradigm: The Inflation Reduction Act has created a new urgency, with small molecules facing price negotiation years earlier than biologics, reshaping R&D investment flows.2
- Digital Intelligence: Mastery of patent data through platforms like DrugPatentWatch is no longer a peripheral task but a central strategic mandate for biopharma business development teams.16
FAQ
1. Can a pharmaceutical patent be extended beyond 20 years? Yes. Through Patent Term Adjustment (PTA) for USPTO delays and Patent Term Extension (PTE) for FDA review delays, a patent’s life can exceed the 20-year window. However, PTE is capped at a maximum of five additional years and cannot extend a drug’s market life beyond 14 years post-approval.1
2. What is the difference between a “patent” and “exclusivity”? Patents are granted by the USPTO and protect the invention; exclusivity is granted by the FDA upon drug approval and prevents the approval of generic competitors. They are governed by different statutes and can run concurrently or independently.13
3. Why do biologics have a 12-year exclusivity while small molecules only have five? The 12-year period reflects the greater technical complexity, manufacturing costs, and clinical trial risks associated with biologics. It aims to provide a sufficient incentive for innovation in large-molecule medicine.2
4. How does “Pediatric Exclusivity” work? If a company completes FDA-requested studies in children, the FDA adds a six-month “bonus” to the end of every existing patent and exclusivity listed for that drug in the Orange Book. This is a powerful multiplier for blockbuster products.1
5. What is a “patent thicket”? A patent thicket is a dense, overlapping web of dozens or hundreds of patents filed around a single drug. By patenting formulations, manufacturing methods, and specific uses, companies make it prohibitively expensive and legally risky for generic competitors to enter the market.17
Works cited
- How Long Does a Patent Last for Drugs? A Comprehensive Guide …, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/how-long-does-a-patent-last-for-drugs/
- A Strategic Investor’s Guide to Pharmaceutical Patent Expiration …, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/a-strategic-investors-guide-to-pharmaceutical-patent-expiration/
- Maximizing ROI on Drug Development by Monitoring Competitive Patent Portfolios, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/maximizing-roi-on-drug-development-by-monitoring-competitive-patent-portfolios/
- Measuring the return from pharmaceutical innovation 2024 | Deloitte …, accessed January 31, 2026, https://www.deloitte.com/us/en/Industries/life-sciences-health-care/articles/measuring-return-from-pharmaceutical-innovation.html
- How Long Do Drug Patents Last? – Trademarkia, accessed January 31, 2026, https://www.trademarkia.com/news/patents/how-long-do-drug-patents-last
- The End of Exclusivity: Navigating the Drug Patent Cliff for Competitive Advantage – DrugPatentWatch, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/the-impact-of-drug-patent-expiration-financial-implications-lifecycle-strategies-and-market-transformations/
- Term of patent in the United States – Wikipedia, accessed January 31, 2026, https://en.wikipedia.org/wiki/Term_of_patent_in_the_United_States
- How to calculate patent expiration date? – Law Stack Exchange, accessed January 31, 2026, https://law.stackexchange.com/questions/98731/how-to-calculate-patent-expiration-date
- How Drug Life-Cycle Management Patent Strategies May Impact Formulary Management, accessed January 31, 2026, https://www.ajmc.com/view/a636-article
- Patent Term Extensions and the Last Man Standing | Yale Law & Policy Review, accessed January 31, 2026, https://yalelawandpolicy.org/patent-term-extensions-and-last-man-standing
- Patent Term Extension – Sterne Kessler, accessed January 31, 2026, https://www.sternekessler.com/news-insights/insights/patent-term-extension-2025/
- Not So Simple Math: Calculating the Regulatory Review Period for Patent Term Extension, accessed January 31, 2026, https://www.troutman.com/insights/not-so-simple-math-calculating-the-regulatory-review-period-for-patent-term-extension/
- Frequently Asked Questions on Patents and Exclusivity – FDA, accessed January 31, 2026, https://www.fda.gov/drugs/development-approval-process-drugs/frequently-asked-questions-patents-and-exclusivity
- Patents and Exclusivity | FDA, accessed January 31, 2026, https://www.fda.gov/media/92548/download
- Patents vs. Market Exclusivity: Why Does it Take so Long to Bring Generics to Market?, accessed January 31, 2026, https://www.raps.org/news-and-articles/news-articles/2016/8/patents-vs-market-exclusivity-why-does-it-take-s
- Checking Drug Patent Status: A Strategic Guide for Pharmaceutical …, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/checking-drug-patent-status-a-strategic-guide-for-pharmaceutical-market-access-teams/
- Unveiling the Secrets Behind Big Pharma’s Patent Thickets …, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/unveiling-the-secrets-behind-big-pharmas-patent-thickets/
- Optimizing Your Drug Patent Strategy: A Comprehensive Guide for Pharmaceutical Companies – DrugPatentWatch, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/optimizing-your-drug-patent-strategy-a-comprehensive-guide-for-pharmaceutical-companies/
- Patent Portfolios Protecting 10 Top-Selling Prescription Drugs | Request PDF, accessed January 31, 2026, https://www.researchgate.net/publication/380549756_Patent_Portfolios_Protecting_10_Top-Selling_Prescription_Drugs
- Pharmaceutical Patenting Practices: A Legal Overview – Congress.gov, accessed January 31, 2026, https://www.congress.gov/crs-product/IF11561
- The Evolution of Patent Claims in Drug Lifecycle Management – DrugPatentWatch, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/the-evolution-of-patent-claims-in-drug-lifecycle-management/
- When One Lawsuit Isn’t Enough: Serial Patent Litigation and Its …, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/when-one-lawsuit-isnt-enough-serial-patent-litigation-and-its-impact-on-generic-timelines/
- Humira: The First $20 Billion Drug – AJMC, accessed January 31, 2026, https://www.ajmc.com/view/humira-the-first-20-billion-drug
- Overpatented, Overpriced 2025: A Data Brief on Medicare-Negotiated Drugs – I-MAK, accessed January 31, 2026, https://www.i-mak.org/overpatented/
- Advanced Models for Predicting Pharma Stock Performance in the Face of Patent Expiration, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/advanced-models-for-predicting-pharma-stock-performance-in-the-face-of-patent-expiration/
- Pfizer’s 180-Day War for Lipitor – PM360, accessed January 31, 2026, https://pm360online.com/pfizers-180-day-war-for-lipitor/
- Managing the challenges of pharmaceutical patent expiry: a case study of Lipitor, accessed January 31, 2026, https://www.researchgate.net/publication/309540780_Managing_the_challenges_of_pharmaceutical_patent_expiry_a_case_study_of_Lipitor
- What’s being litigated: Technology trends at the Unified Patent Court, accessed January 31, 2026, https://clarivate.com/intellectual-property/blog/technology-trends-at-the-unified-patent-court/
- Unitary Patent & Unified Patent Court | epo.org, accessed January 31, 2026, https://www.epo.org/en/applying/european/unitary
- The Unified Patent Court 2025 – Munich Local Division – Chambers Global Practice Guides, accessed January 31, 2026, https://practiceguides.chambers.com/practice-guides/the-unified-patent-court-2025/munich-local-division/trends-and-developments/O21444
- More Than a Feeling: UPC, Rewriting the Score for Pharma at BIO 2025 – Wolters Kluwer, accessed January 31, 2026, https://legalblogs.wolterskluwer.com/patent-blog/more-than-a-feeling-upc-rewriting-the-score-for-pharma-at-bio-2025/
- Key Takeaways from the UPC Litigation Forum and Pharma …, accessed January 31, 2026, https://www.hsfkramer.com/notes/lifesciences/2025-posts/key-takeaways-from-the-upc-litigation-forum-and-pharma-biotech-patent-lit-2025-ls
- Examining Developments and Trends at the Unified Patent Court in 2025 – IPWatchdog.com, accessed January 31, 2026, https://ipwatchdog.com/2025/12/22/upc-developments-trends-2025/
- Measuring the return from pharmaceutical innovation 2025 | Deloitte Switzerland, accessed January 31, 2026, https://www.deloitte.com/ch/en/Industries/life-sciences-health-care/research/measuring-return-from-pharmaceutical-innovation.html
- Drug Patents Expiring in 2026: A Comprehensive Guide – IntuitionLabs, accessed January 31, 2026, https://intuitionlabs.ai/articles/drug-patent-expirations-2026
- A Strategic Guide to Pharmaceutical Patent Pool Participation – DrugPatentWatch, accessed January 31, 2026, https://www.drugpatentwatch.com/blog/a-strategic-guide-to-pharmaceutical-patent-pool-participation/
- How Drugmakers Use the Patent Process to Keep Prices High | Commonwealth Fund, accessed January 31, 2026, https://www.commonwealthfund.org/publications/explainer/2025/nov/how-drugmakers-use-patent-process-keep-prices-high