943 drugs approved. $15M to $100M in development costs versus $2.6B for an NCE. One regulatory mechanism sits at the intersection of every major force reshaping pharmaceutical IP strategy: the Hatch-Waxman patent dance, IRA price negotiation, PBM formulary gatekeeping, and the collapse of skinny-labeling defenses in federal courts. This is the definitive operational guide to the 505(b)(2) NDA.
What the 505(b)(2) Pathway Actually Is
The 505(b)(2) is a full New Drug Application that permits the applicant to rely, at least in part, on data they did not generate and have no contractual right to reference. The FDA’s prior finding that a Reference Listed Drug (RLD) is safe and effective becomes a shared public asset, available to any sponsor willing to build a scientific bridge to it.
That framing matters. The 505(b)(2) is not a generic application with extra steps, nor is it a full NCE program with a shortcut. From 2003 through 2023, 943 drugs received approval via this pathway. From 2017 to 2023 alone, the U.S. approved 417 505(b)(2) products, compared to 99 via China’s analogous Class 2 NDA route. By 2024-2025, 69 reformulation NDAs received approval in a single FDA review cycle, concentrated in oncology and CNS. The pathway now accounts for roughly 60% of all approved NDAs in recent years.
The practical scope is wide. Approved products span new formulations (Type 5, accounting for 42.4% of approvals from 2010 to 2020), new dosage forms (Type 3, 26.4%), and new combinations (Type 4, 13.1%). What unites them is the central regulatory mechanism: the scientific bridge.
The Bridge: Science, Not Paperwork
The bridge is the data set that justifies the FDA treating the RLD’s safety record as applicable to the new product. Its most common form is a comparative pharmacokinetic study — typically a single-dose, two-period crossover trial in healthy volunteers — demonstrating that the new product’s absorption profile is predictable from the RLD’s known behavior.
When the bridge is solid, the developer inherits the RLD’s toxicology, carcinogenicity, and reproductive safety data. Years of preclinical work collapse into weeks. The Valley of Death between Phase I and Phase III, where most NCE programs fail, becomes a manageable slope.
When the bridge fails, the economics collapse with it. If the new formulation creates meaningfully higher peak plasma concentrations (Cmax) or a shifted time-to-peak (Tmax), the FDA treats those exposures as novel. New toxicology studies follow. The development budget expands, the timeline stretches, and the cost advantage over a 505(b)(1) program shrinks fast. This is not a theoretical risk: a reformulation that turns a once-daily tablet into an extended-release capsule, an intravenous solution into a subcutaneous injection, or a systemic drug into an inhalation product changes the exposure profile in ways the original RLD data cannot cover.
Aradigm learned this directly. Its inhaled ciprofloxacin product, Linhaliq, sought approval for non-cystic fibrosis bronchiectasis by referencing the known safety record of oral ciprofloxacin. The FDA rejected the application and demanded a new two-year Phase III study, citing both product quality failures in vitro release testing and the argument that pulmonary delivery of a fluoroquinolone created a distinct safety profile the existing dataset could not validate. The fact that ciprofloxacin has been in clinical use since 1987 was irrelevant. Route of administration changed the drug.
The Legal Trigger: Why No Right of Reference Creates the Patent Dance
The defining legal characteristic of a 505(b)(2) is what the applicant does not have: the RLD holder’s permission. In a 505(b)(1) program, the sponsor either owns the data or has licensed it. In a 505(b)(2), the applicant exploits the FDA’s published finding without any contractual relationship to the innovator.
The Hatch-Waxman Act’s quid pro quo for that exploitation is mandatory patent certification. The applicant must declare, under 21 U.S.C. § 505(b)(2)(A)(vii), one of four things about each Orange Book-listed patent covering the RLD: the patent has expired (Paragraph I), the patent information has not been submitted (Paragraph II), the applicant will wait for the patent to expire (Paragraph III), or the patent is invalid, unenforceable, or will not be infringed by the new product (Paragraph IV).
A Paragraph IV certification triggers a Notice Letter obligation. The applicant must notify the patent holder and RLD sponsor of the certification and its legal basis. The innovator then has 45 days to sue. If it does, FDA approval is automatically stayed for 30 months. That stay is the innovator’s primary weapon, and it works regardless of litigation merit — the clock runs whether the patent is strong or weak, broad or narrow.
Unlike a 505(j) generic filer, the 505(b)(2) applicant receives no 180-day exclusivity reward for a successful Paragraph IV challenge. That asymmetry shapes litigation strategy fundamentally. Generics fight to invalidate; they win a duopoly. 505(b)(2) applicants are more often trying to design around the patent, negotiate a settlement that allows an early launch date, or time their filing to clear the patent cliff without triggering the stay at all.
Key Takeaways: Regulatory Architecture
The 505(b)(2) is a full NDA, not an abbreviated filing. The bridge is a scientific burden, not a checkbox. No right of reference means mandatory patent certification. A Paragraph IV certification buys a 30-month stay for the innovator at zero cost in litigation merit. The 505(b)(2) applicant gets none of the 180-day exclusivity that rewards generic Paragraph IV challengers.
Development Economics: Where the Cost Advantage Actually Lives
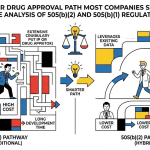
The headline figure — 90% lower development costs than an NCE — requires decomposition to be useful. The savings are not distributed evenly across the development timeline.
Preclinical and Phase I: Where Most of the Money Disappears (or Doesn’t)
NCE preclinical programs cost $200 million to $400 million. Compound screening, medicinal chemistry, ADMET profiling, regulatory toxicology (six-month rat and dog chronic studies, genotoxicity, carcinogenicity), and reproductive toxicity studies collectively consume years and hundreds of millions before a single human sees the drug.
A 505(b)(2) program’s preclinical budget typically runs $2 million to $5 million, covering formulation development work and, where the bridge requires it, short-duration bridging toxicology. If the excipients are novel or the new route creates localized tissue exposure (think intrathecal or pulmonary delivery), that figure climbs, but it rarely approaches the NCE baseline.
Phase I for an NCE is first-in-human dose escalation: multiple cohorts, escalating doses, extended safety monitoring, $25 million to $50 million over 12 to 18 months. A 505(b)(2) Phase I is almost always a single-dose PK crossover in 12 to 24 healthy volunteers. The entire study runs four to eight weeks and costs $2 million to $5 million. The FDA uses the result to confirm the bridge — that the new product’s exposure is predictable — not to establish whether the active moiety is safe at all.
Phase II and Phase III: The Strategic Variable
Phase II is where the 505(b)(2) cost model most diverges from the NCE model. For a new formulation of an approved drug targeting the same indication with a similar PK profile, Phase II is often skipped entirely. The FDA accepts that if the bridge holds and the molecule’s efficacy at therapeutically relevant exposures is established, a new formulation will work. The sponsor goes directly from Phase I to Phase III.
When a Phase III is required — for a new indication, a route change with clinically meaningful PK differences, or a new patient population — it is almost always smaller and shorter than an NCE Phase III. Non-inferiority designs against the RLD, single pivotal studies rather than two, surrogate endpoints where they have regulatory precedent. The result: a 505(b)(2) Phase III costs $10 million to $50 million compared to $200 million to $500 million or more for an NCE.
Total blended 505(b)(2) program cost: $15 million to $100 million. Total blended NCE cost: $800 million to $2.6 billion (with Phase III failures excluded from the average).
The CMC Problem No Financial Model Catches
Chemistry, Manufacturing, and Controls costs are the hidden variable that most investor models underestimate. A 505(b)(2) program is often asking a formulation scientist to do something genuinely hard: take a drug whose physical chemistry was optimized for a tablet and force it into a patch, a nasal spray, a liposomal injection, or an extended-release bead system.
Stability failures account for a disproportionate share of second-cycle review deficiencies. Impurity profiles change when a drug moves from one dosage form to another. In vitro release methods for complex formulations — osmotic pumps, long-acting injectables, inhalation products — require development and validation that the FDA treats as scientifically equivalent to clinical evidence. 73% of second-cycle delays for 505(b)(2) NDAs trace to CMC deficiencies, not clinical safety gaps. This does not appear as a line item in most development cost projections.
The Exclusivity Stack: Building a Defensible Position
Exclusivity is the mechanism by which regulatory approval converts into commercial duration. The 505(b)(2) pathway offers multiple stacking options, and sophisticated developers engineer the stack before the first IND.
Three-year New Clinical Investigation (NCI) exclusivity is the baseline. It requires that the application contain reports of clinical investigations essential to approval — not bioavailability studies alone. It blocks the FDA from approving a competitor’s 505(b)(2) or ANDA using the same clinical data for the same change for three years from the approval date. Critically, it does not block a competitor who generates independent data supporting a different change or who conducts their own clinical program.
Five-year NCE exclusivity applies when the active moiety has never been approved by the FDA in any form. This is the exclusivity class that most investors associate only with truly novel drugs, but it is available to 505(b)(2) applicants when the pathway is used creatively. A prodrug whose active metabolite has never independently been approved, a novel ester or salt that FDA designates as a distinct moiety, or a fixed-dose combination where one component has never previously been approved can each qualify. NCE exclusivity blocks the submission of any ANDA or 505(b)(2) relying on the new moiety for five years.
Seven-year Orphan Drug Exclusivity (ODE) is the most powerful protection in the 505(b)(2) toolkit for the right asset. It requires an Orphan Drug Designation, granted when the indication affects fewer than 200,000 patients in the U.S. or when the developer can demonstrate no reasonable expectation of profitability without the designation. ODE blocks any competitor from marketing the same drug for the same orphan indication for seven years, regardless of their own data. It does not require patent protection and does not expire with patents.
The exclusivity stack that produces the highest IP-to-capital ratio combines NCE or ODE exclusivity with a suite of Orange Book-listed patents covering the formulation, the method of treatment, and where applicable, the device component. When an applicant layers a formulation patent expiring in year 12 post-approval, a method patent expiring in year 15, and three-year NCI exclusivity on a subsequent indication, the effective market exclusivity stretches well beyond what any single protection would provide.
Investment Strategy: Development Economics
For early-stage investors, the economic inflection point in a 505(b)(2) program is the bridging study result, not Phase III. A failed bridge — PK data showing Cmax 40% above the RLD with no dose adjustment possible — can kill a program before it ever reaches pivotal trials. Model the bridge failure probability explicitly, particularly for inhaled products, long-acting injectables, and any formulation requiring gastrointestinal absorption enhancement. Assign a CMC contingency budget of 30% to 50% above the clinical trial line item. CMC overruns are the most common cause of budget breach in this class.
For late-stage investors and M&A analysts, the exclusivity stack is the primary value driver. A 505(b)(2) approved tomorrow with three-year NCI exclusivity is worth materially less than one with NCE exclusivity on a novel active moiety, even with similar clinical data. Map the exclusivity type, duration, and stacking potential before pricing the asset. The BMS acquisition of Karuna Therapeutics at $14 billion in 2023, pre-Cobenfy approval, reflected precisely this: the market was pricing xanomeline’s NCE exclusivity potential into a transaction valued primarily on that IP architecture.
Cobenfy (Xanomeline-Trospium): The 505(b)(2) IP Architecture Case Study
No recent approval illustrates the ceiling of what a 505(b)(2) can achieve better than Cobenfy (xanomeline and trospium chloride), developed by Karuna Therapeutics and now marketed by Bristol-Myers Squibb.
The Regulatory Pathway
The application relied on trospium chloride’s existing safety database for the peripheral anticholinergic component. Trospium had been approved for overactive bladder since 2004 under the brand Sanctura; its absorption, distribution, metabolism, excretion, and safety profile at therapeutic doses were well-characterized. Karuna used that prior finding to satisfy the FDA’s requirement for trospium safety data in the combination, freeing the clinical program to focus on xanomeline’s central activity and the fixed-dose combination’s efficacy in schizophrenia.
Xanomeline itself had been in research for decades, first studied by Eli Lilly in the 1990s for Alzheimer’s disease. It was never approved as a standalone drug, making it a new active moiety for purposes of FDA exclusivity. The combination’s efficacy was established through the EMERGENT clinical program: three placebo-controlled studies, including the Phase 3 EMERGENT-2 and EMERGENT-3 trials, plus two open-label long-term safety studies. In EMERGENT-2, xanomeline-trospium reduced PANSS total scores by 21.2 points from baseline versus 11.6 points for placebo, a least-squares mean difference of 9.6 points (95% CI: -13.9 to -5.2, P < 0.0001). The FDA approved Cobenfy in September 2024.
IP Valuation
Xanomeline’s status as a never-before-approved moiety anchored five-year NCE exclusivity running from September 2024 to September 2029. The combination generated a new Orange Book patent family covering the fixed-dose combination formulation, the specific molar ratio of xanomeline to trospium, and the method of treating schizophrenia with M1/M4 muscarinic agonism. Method-of-treatment patents covering specific subpopulations — treatment-resistant schizophrenia, first-episode psychosis — remain in prosecution. With patent term restoration (PTR) and potential pediatric exclusivity, the effective patent estate extends into the late 2030s in the base case.
The mechanism represents the first genuinely new pharmacological approach to schizophrenia in decades. Where all prior antipsychotics targeted dopamine D2 and serotonin 5-HT2A receptors, Cobenfy operates through M1/M4 muscarinic agonism. That mechanistic novelty is not just a clinical differentiator; it is a patent claim anchor. Competitors cannot design around the mechanism without abandoning the approach entirely.
BMS acquired Karuna for approximately $14 billion before Cobenfy received approval. That valuation implied a peak revenue estimate above $3 billion annually. The number only closes if NCE exclusivity holds through 2029 and the commercial launch meets model assumptions. Investors who priced Karuna below the acquisition price did so by failing to model the NCE exclusivity value of the combination structure. A 505(b)(2) with an unapproved active moiety is not a reformulation play. It is an NCE program dressed in 505(b)(2) economics.
Key Takeaways: IP Valuation
The moiety analysis drives the exclusivity outcome. A fixed-dose combination containing one unapproved active moiety qualifies for five-year NCE exclusivity on the entire combination. Cobenfy demonstrates that a 505(b)(2) can generate $14 billion in acquisition value when the IP architecture is correctly engineered. The patent family for a fixed-dose combination should cover formulation, ratio, co-formulation, method of treatment, device (if applicable), and subpopulation method claims.
Treanda to Bendeka: Lifecycle Management as Commercial Execution
The Problem Teva Had
Treanda (bendamustine), Teva’s lymphoma chemotherapy, generated over $500 million in annual U.S. revenue before its patent cliff. Standard infusion time was 30 to 60 minutes in clinic. Generics were filing ANDAs. The market was about to commoditize.
The 505(b)(2) Response
Eagle Pharmaceuticals formulated Bendeka as a concentrated, ready-to-dilute bendamustine product that reduces infusion time to 10 minutes. Teva licensed the asset. The 505(b)(2) NDA referenced Treanda’s safety data, established the PK bridge, and Eagle secured Orphan Drug Exclusivity for Bendeka in the chronic lymphocytic leukemia indication.
The strategic calculation was not purely regulatory. Teva and Eagle launched Bendeka before Treanda generics achieved meaningful market penetration, then structured contracts with infusion centers that made conversion economically rational for the provider. A 10-minute infusion versus a 60-minute infusion translates directly into throughput for an oncology infusion center: more patient chair time, more procedures per day, more reimbursement per square foot. The clinical change (infusion duration) aligned with the provider’s billing economics. That alignment, not the FDA approval, determined commercial success.
Treanda generics arrived. The market did not revert. Bendeka had converted enough of the formulary to sustain pricing.
Lesson for Portfolio Managers
Lifecycle management via 505(b)(2) requires alignment between the clinical improvement and the economic incentive of the decision-maker who drives adoption. For oncology infusion products, that decision-maker is the infusion center pharmacist and medical director, not the prescribing oncologist. The clinical change (shorter infusion) mattered to the provider’s economics, not just the patient’s experience. Any 505(b)(2) lifecycle program should answer the question: whose P&L does this improve, and by how much?
Yosprala (Aspirin/Omeprazole): The Payer Veto
Clinical Logic, Commercial Failure
Aralez Pharmaceuticals approved Yosprala as a fixed-dose combination of aspirin and omeprazole targeting cardiac patients on long-term aspirin therapy who develop gastric intolerance. The clinical premise was sound: aspirin adherence in secondary cardiovascular prevention is suboptimal, and GI side effects are a leading cause of discontinuation. A single co-formulated pill simplifies the regimen.
The FDA approved it easily under 505(b)(2). Aralez launched in 2016.
The PBM Veto
CVS Caremark, Express Scripts, and other major PBMs reviewed the formulary placement request and reached the same conclusion: Yosprala was a combination of aspirin, available over the counter for pennies, and omeprazole, available as a generic for under $10 per month. The total cost of the components was under $15. Aralez priced Yosprala at brand levels — hundreds of dollars per month.
The PBMs declined to cover it at any tier that would support adoption. They placed it on exclusion lists or restricted tiers, and they were direct about why: a convenience pack of two cheap generics does not justify brand premium pricing. Patients refused out-of-pocket costs when the components were available separately at their local pharmacy for a fraction of the price.
Aralez attempted negotiations, published HEOR data on adherence improvements, and sought step therapy protocols. None of it moved the payers. The company filed for bankruptcy in 2018. Yosprala was a regulatory success and a commercial failure, sequentially.
The Structural Problem
The Yosprala failure exposed a gap that persists in 505(b)(2) commercial strategy. “Convenience” as a value proposition survives payer scrutiny only when it demonstrably reduces total cost of care: fewer ER visits, reduced hospitalizations, lower downstream utilization. Adherence improvements in secondary cardiovascular prevention may produce those outcomes, but the data horizon is long (five to ten years of reduced cardiovascular events) and payers applying one-year or three-year formulary budget horizons will not credit those benefits at launch. A 505(b)(2) targeting an adherence problem must generate HEOR data with a short-horizon economic benefit — cost per quality-adjusted life year within a two-year budget cycle — to survive formulary review. If the components are generic, the payer’s alternative is always the co-pay for two separate generics, and that is the price ceiling.
Emflaza (Deflazacort): The Orphan Pricing Calculation
The Asset
Deflazacort is a corticosteroid available in Europe and the U.K. since the 1980s for Duchenne Muscular Dystrophy. It had never been formally approved by the FDA. Marathon Pharmaceuticals used the 505(b)(2) pathway to obtain the first U.S. approval for DMD in February 2017, referencing European clinical data and conducting bridging studies.
Marathon secured seven-year Orphan Drug Exclusivity. The DMD prevalence in the U.S. is approximately 10,000 to 15,000 patients. The product had a clear clinical rationale, strong patient community advocacy, and an essentially captive market.
The Pricing Decision
Marathon set the U.S. list price at $89,000 per year. In the United Kingdom, deflazacort was available from licensed pharmacies for approximately $1,000 annually. Senators Bernie Sanders and Elijah Cummings wrote to Marathon within weeks of the announcement, calling the pricing “unjustifiable” and threatening legislative action. Eight additional senators followed. The public reaction was swift enough that Marathon paused the commercial launch before it began and sold the asset to PTC Therapeutics.
PTC eventually launched Emflaza at a lower price point, though still at a substantial premium to the U.K. equivalent.
What the Case Clarifies
The Emflaza episode is frequently mischaracterized as a simple pricing scandal. It is more precisely a demonstration of the political ceiling on orphan drug repricing via 505(b)(2). The pathway’s efficiency at converting foreign-approved drugs into FDA-approved products with seven years of ODE creates structural tension: the developer’s capital must be recovered from a small patient population, but the existence of a low-cost international equivalent provides a vivid benchmark that neither the press nor Congress will ignore. Developers using 505(b)(2) to access ODE for well-known compounds must model reputational risk as a commercial variable, not a PR problem. Legislative risk is real; the IRA’s negotiation provisions and the ongoing congressional scrutiny of orphan drug pricing are direct descendants of the Emflaza controversy.
Key Takeaways: Commercial Execution
The 505(b)(2) commercial model requires different validation than an NCE. Three questions determine commercial viability before launch: Does the clinical improvement align with the economic incentive of the formulary decision-maker? Can HEOR data demonstrate near-term cost-of-care reduction, not just adherence improvement? If the components are generic, what is the payer’s alternative, and can the brand price survive comparison? Orphan pathway products must include reputational risk modeling when the international equivalent is well-known and cheap.
The Paragraph IV Litigation Playbook
Mechanics of the 30-Month Stay
The Paragraph IV certification triggers a sequence that every IP team at a brand company has mapped exhaustively. The Notice Letter must include a detailed statement of the factual and legal basis for the certification: claim charts, invalidity arguments, non-infringement positions. The brand holder’s 45-day window to sue is effectively treated as obligatory — the downside of not suing (an uncontested generic or 505(b)(2) launch) is worse than the cost of the litigation. Filing a complaint automatically freezes FDA approval for 30 months unless the court rules earlier.
For the 505(b)(2) applicant, the 30-month stay is a planning parameter, not just a legal risk. Filing the NDA 30 months before the intended commercial launch is standard practice when a Paragraph IV challenge is expected. The litigation period becomes part of the development timeline. Teams that do not model this explicitly end up surprised by a stay they should have predicted.
Design-Around vs. Invalidation: The Strategic Fork
A 505(b)(2) applicant’s litigation strategy typically diverges from a generic’s at the strategic level. The generic’s 180-day exclusivity reward incentivizes fighting hard for invalidation — winning the litigation means winning the market. The 505(b)(2) applicant has no such prize. Its incentive is to reach the market, not to determine patent validity for all future competitors.
This produces two common approaches. The first is non-infringement on design-around grounds: the 505(b)(2) product is different enough from the RLD that the claims do not read on it. The second is commercial settlement: the applicant negotiates a launch date with the innovator, often a few months before patent expiry, in exchange for dropping the Paragraph IV litigation. These settlements are subject to FTC scrutiny under the Actavis framework for reverse payments, but pure date settlements without cash payments are generally permissible.
The Skinny Label Trap: GSK v. Teva and Amarin v. Hikma
Section viii statement practice — “skinny labeling” — allows an applicant to carve out a patented method of use from the product label while seeking approval only for unpatented indications. The theory is that a label explicitly excluding the patented use cannot induce infringement of that use.
The theory has been under attack since at least 2020.
In GSK v. Teva (Coreg), the Federal Circuit found Teva liable for induced infringement of GSK’s heart failure patent despite a skinny label explicitly omitting the heart failure indication. The court’s reasoning looked beyond the label to Teva’s press releases and product communications, which described carvedilol as “AB-rated” to Coreg and promoted it as a “generic equivalent.” In a market with automatic substitution, the court held, those communications encouraged pharmacists and physicians to dispense the drug for the patented indication even without an explicit direction to do so.
The Amarin v. Hikma case (2024) applied similar reasoning to Vascepa generics. Hikma launched a skinny-labeled icosapentaenoic acid product carving out Amarin’s REDUCE-IT cardiovascular risk reduction indication. Amarin’s approved indication for severe hypertriglyceridemia was off-patent; Hikma sought approval only for that use. The Federal Circuit reversed a district court dismissal in June 2024, ruling that Hikma’s website language — describing the product in terms that echoed Vascepa’s overall profile — could plausibly constitute induced infringement. The court did not find infringement; it found the claim plausible enough to survive dismissal.
The operational consequence for 505(b)(2) developers is material. Any product launched with a carved-out label while sharing a marketing presentation with the brand’s patented indication faces induced infringement exposure. Commercial and legal teams must develop a shared review protocol for every external communication: press releases, conference presentations, sales force materials, website copy, and pharmacist-facing resources. The label carve-out is a legal position that requires consistent reinforcement across every touchpoint with the market. One press release describing the product as “equivalent” to the brand while carrying a carve-out can create a damages exposure that dwarfs the revenue the carve-out was meant to protect.
Investment Strategy: Litigation
For biotech investors evaluating a 505(b)(2) asset’s litigation risk, the primary questions are: How many Orange Book-listed patents does the RLD carry, and what is the remaining term on the broadest claims? Has the brand holder demonstrated willingness to sue (prior litigation history)? Is the intended product launch dependent on a skinny label carve-out that requires aggressive market messaging to drive adoption? That last combination — carve-out label plus heavy physician/pharmacist marketing — is the induced infringement risk profile to avoid.
The IRA’s Small Molecule Penalty and What It Does to 505(b)(2) Economics
The Nine-Year Cliff
The Inflation Reduction Act’s Medicare Drug Price Negotiation Program allows CMS to set Maximum Fair Prices for high-expenditure drugs. The statute draws an explicit distinction by drug type: small molecules become eligible for negotiation nine years after approval; biologics become eligible 13 years after approval. Most 505(b)(2) products are small molecules. The four-year gap is not incidental — it was a deliberate legislative choice that reflects the political economy of the IRA, and it has direct consequences for the 505(b)(2) business model.
A 505(b)(2) small molecule with three-year NCI exclusivity, approved in 2020, has its exclusivity expire in 2023. A generic or competing 505(b)(2) could theoretically launch by 2024. But if no competitor does — perhaps because the CMC complexity deters generic entry — the drug reaches IRA negotiation eligibility in 2029. The brand may still hold method-of-use patents through 2032 and have no generic competition. IRA negotiation risk and patent exclusivity duration now interact as coupled variables rather than independent ones. Financial models that treat them separately understate the combined risk.
The Orphan Exemption and Its Limits
Orphan drugs with a single approved indication are exempt from IRA price negotiation. That exemption is the primary reason orphan-indication 505(b)(2) programs have attracted disproportionate capital since 2022.
The exemption has a structural limit: it applies only while the drug is approved solely for the orphan indication. If the developer seeks a second approval — for a common disease, or even for a second rare disease with prevalence above the orphan threshold — the drug loses its exemption and becomes negotiation-eligible if it meets the expenditure and years-on-market criteria. This creates a perverse incentive to avoid developing additional indications for orphan-approved products, suppressing the standard lifecycle management strategy of indication expansion. A 505(b)(2) developer who secures ODE and then successfully pursues a second indication may be destroying IRA exemption value equal to or greater than the revenue gain from the new indication.
Capital Allocation Consequences
Capital has moved. Small molecule 505(b)(2) programs targeting Medicare-heavy populations — primarily the therapeutic areas of cardiovascular, CNS, and metabolic disease — have seen funding compress as investors re-run the revenue models with IRA negotiation risk included. Products with clear path to negotiation eligibility before patent expiry are raising rounds at lower pre-money valuations than the same programs would have commanded pre-IRA.
The reallocation is toward biologics (13-year protection), gene therapies (potentially exempt as single-administration products), and single-indication orphan programs with demonstrated ability to sustain ODE for the commercial life of the product. For specialty pharmaceutical companies whose business model is built on small molecule 505(b)(2) reformulations across broad populations, the IRA is a structural headwind, not a one-time event.
Key Takeaways: IRA
The nine-year small molecule negotiation window is shorter than the typical exclusivity-plus-patent effective market life of a successful 505(b)(2). Model IRA eligibility and patent protection as interacting, not independent, variables. The orphan exemption survives only with a single approved indication — indication expansion destroys it. Capital is repricing small molecule 505(b)(2) risk accordingly.
Complex Formulations: Where Defensible Moats Still Exist
The New Competitive Frontier
The 505(b)(2) market for simple oral solids — standard tablets and capsules — is saturated. Development costs are low enough that multiple sponsors often identify the same opportunity simultaneously, the clinical program is straightforward enough that regulatory risk is minimal, and the resulting product typically commands a modest premium for a limited time. The margin compression timeline on simple oral solids is three to five years post-launch.
Complex formulations present a different picture. The CMC barriers to entry for long-acting injectables, inhalation products, transdermal patches, and liposomal or nanoparticle-based systems are high enough to deter casual competition even when the underlying molecule is off-patent. The FDA cannot approve a generic version of a complex 505(b)(2) product via ANDA until a generic developer demonstrates that their version is pharmaceutically and therapeutically equivalent — a standard that requires solving the same formulation problem independently. That problem can take years and tens of millions of dollars, during which the 505(b)(2) holder maintains pricing power.
Long-Acting Injectables
Long-acting injectable (LAI) reformulations of approved small molecules represent one of the most capital-efficient 505(b)(2) categories. Converting a daily oral pill to a monthly or quarterly injection requires solving polymer encapsulation, controlled-release kinetics, and injectability without creating dose-dumping or particle aggregation. These are tractable but non-trivial formulation problems. Approval requires a Phase I PK bridge establishing that the target steady-state plasma concentrations of the LAI formulation fall within acceptable bounds relative to the oral RLD. If the steady-state profile is equivalent, the efficacy and safety database of the oral product applies.
The commercial logic is compelling for psychiatry and HIV prophylaxis: monthly injections solve the adherence problem that daily pills cannot. Providers can confirm administration. Payers, who understand the cost of non-adherence in these populations, are generally willing to pay a premium for verified dosing.
Inhalation Products
Inhaled drug products are among the highest-risk 505(b)(2) categories from a CMC perspective, but among the most defensible commercially when approval is achieved. The FDA’s product-specific guidance for inhalation products requires device-formulation equivalence: not just comparable drug deposition in the lungs, but reproducible performance across clinically representative use conditions. A dry powder inhaler (DPI) referencing an old metered-dose inhaler (MDI) must demonstrate equivalent regional lung deposition, equivalent device robustness across humidity and temperature ranges, and equivalent patient use characteristics.
Generic developers face the same hurdles. The result is that inhalation products maintain price competition resistance for longer than any other non-biologic dosage form. Teva’s generic Advair program required years of development and multiple FDA complete response letters before receiving approval. A 505(b)(2) inhalation product with a clinically distinct delivery mechanism — different particle size distribution, different device resistance profile — can sustain a pricing premium even when the active moiety is long off-patent.
AI-Assisted Formulation Development
Machine learning models trained on PK datasets can now predict with meaningful accuracy whether a proposed formulation change will achieve an acceptable PK bridge to the RLD, reducing the probability of a bridge failure before committing to the clinical study. Stability degradation models allow developers to identify high-risk excipient combinations and manufacturing conditions before stability batch failures occur. Impurity prediction algorithms flag potential degradation products that would require new toxicology studies — the formulation trap that catches the majority of CMC-delayed applications.
These tools do not eliminate formulation risk, but they shift it earlier in the development timeline, where it is cheaper to resolve. A CMC failure identified by computational modeling during pre-formulation costs $200,000 to fix. The same failure identified in a 12-month stability study, six months before NDA submission, costs the program two years.
Key Takeaways: Formulation Strategy
Simple oral solid 505(b)(2) programs face rapid margin compression. Complex formulations — long-acting injectables, inhalation products, transdermal systems — offer CMC-based moats that outlast patent protection. Investing in computational formulation tools at the pre-development stage reduces the 73% CMC delay rate. The IND meeting with FDA is the moment to explicitly address CMC complexity and agree on the acceptable bridge parameters before committing capital to the Phase I study.
Taxonomy of 505(b)(2) Applications: What Gets Approved and Why
Type 3: New Dosage Form
The largest single category by volume after new formulations. Route of administration changes (oral to intravenous, oral to transdermal) and presentation changes (tablet to suspension, lyophilized to ready-to-dilute) fall here. The FDA’s Type 3 classification requires the applicant to justify why the new dosage form’s clinical performance can be predicted from the RLD’s data, and where it cannot, to generate new data.
The Linhaliq case is the cautionary example: inhaled ciprofloxacin is a Type 3 application whose bridge did not hold because pulmonary pharmacokinetics and local tissue concentrations at the site of infection are sufficiently distinct from systemic oral pharmacokinetics that the FDA treated the in vitro and in vivo data requirements as equivalent to a new drug program.
Type 4: New Combination
Fixed-dose combinations (FDCs) are the category where 505(b)(2) creates the most complex IP architecture and generates both the highest commercial upside (Cobenfy) and the most predictable commercial failure (Yosprala). The regulatory burden for a Type 4 product includes demonstrating the rationale for the combination (each component contributes to the claimed effects, or one component reduces adverse effects of the other), establishing that the combination’s PK can be predicted from the individual components’ known profiles, and generating the bridging data to establish those properties.
The IP opportunity in a Type 4 product is the combination patent: a patent claiming the specific combination and its ratio, which cannot be infringed by either component alone. When one component is a new active moiety, the entire combination qualifies for NCE exclusivity.
Type 5: New Formulation or New Manufacturer
Type 5 products — reformulations of approved drugs using different excipients, particle sizes, or manufacturing processes — account for the largest share of 505(b)(2) approvals by volume (42.4% from 2010 to 2020). They are also the category most vulnerable to PBM price compression when the clinical benefit is incremental and the components are not novel.
Successful Type 5 programs establish a clinical or pharmacokinetic improvement that translates into measurable outcomes: faster onset (for rescue medications), improved tolerability (for long-term therapies), extended duration of action (reducing dosing frequency), or reduced food effect (improving predictability of exposure). Programs that cannot translate a formulation improvement into one of these outcomes face formulary exclusion risk regardless of FDA approval.
Orange Book Patent Strategy: How to List, What to List, and When
The Listing Obligation
NDA holders must submit patent information for patents that claim the drug substance, the drug product, or the method of use. The obligation is not discretionary. Failure to list a qualifying patent forfeits the ability to trigger the 30-month stay on a subsequent Paragraph IV challenge. Listing a patent that does not qualify — a manufacturing process patent, for example, or a metabolite patent claiming a compound not present in the drug product — exposes the NDA holder to a counterclaim for improper listing, which carries attorney’s fees under certain circumstances.
What the Orange Book Does Not Capture
The Orange Book lists patents but provides no information about their vulnerability. A patent can be listed with a 2035 expiry date and be invalid on its face, subject to a pending IPR proceeding, or narrowly drafted enough that a design-around is trivial. The Orange Book creates the 30-month stay mechanism; it does not guarantee that the listed patents will survive challenge.
Platforms that track P-IV litigation outcomes, IPR decisions, and settlement terms provide the intelligence layer that the Orange Book lacks. For a 505(b)(2) developer evaluating a potential RLD, knowing that the brand’s formulation patents have been invalidated in three prior ANDA litigations is more operationally relevant than knowing their expiry dates.
The “Patent Thicket” and Evergreening Mechanics
Originators have developed systematic approaches to extending effective exclusivity beyond the original compound patent: adding formulation patents after the initial NDA approval, seeking method-of-use patents for newly identified patient subpopulations, obtaining pediatric exclusivity extensions (six months, but relevant when layered with other protections), and filing continuation applications to obtain claims that read specifically on the commercial product rather than the original discovery.
A 505(b)(2) developer approaching a densely patented RLD must evaluate not just the expiry date of the primary compound patent, but the full family across all formulation, method-of-use, and device claims. An RLD with a 2024 compound patent expiry and 2031 formulation patents, 2033 method claims, and a pending pediatric exclusivity application is not a 2024 opportunity. It is a 2033 opportunity at the earliest, unless the 505(b)(2) developer can design an application that avoids the formulation and method claims entirely.
Investment Strategy: Patent Intelligence
The patent expiry date is the floor of the competitive timeline, not the ceiling. Build the full patent family map before committing to an RLD. Include IPR outcomes, litigation history, and continuation application activity in the analysis. Use patent analytics platforms that track claim scope and prior art citations, not just expiry dates. The gap between compound patent expiry and formulation/method patent expiry is where 505(b)(2) competitors get surprised.
Regulatory Intelligence: How to Select the Right RLD
The RLD Selection Decision
The choice of Reference Listed Drug is the most consequential regulatory decision in a 505(b)(2) program. It determines the available safety bridge, the patent certification obligation, the applicable exclusivity periods, the competing applications already in the queue, and the FDA review division that will evaluate the application. No other decision downstream of the IND has the same breadth of consequence.
The ideal RLD has a well-characterized safety and efficacy profile sufficient to support the bridge, a patent estate that is expired, expiring, or designable around, no 505(b)(2) or ANDA with three-year exclusivity for the same change, and a review history with the FDA that establishes the safety data requirements clearly. Finding all four characteristics simultaneously requires systematic competitive intelligence, not intuition.
Identifying Stealth Competitors
A material risk specific to the 505(b)(2) pathway is the “silent” competitor: another developer who has identified the same RLD, the same modification type, and the same clinical program, and who is six to twelve months ahead in development. If the competitor files their NDA first and receives three-year NCI exclusivity, the second filer is blocked from approval for three years regardless of their own data quality. There is no 180-day race to the patent finish line that rewards the first filer; the reward is simply FDA approval, and the exclusivity blocks others from receiving it.
Systematic tracking of global clinical trial registries, patent application publications, and FDA pre-IND meeting request disclosures can identify stealth competitors before they reach NDA submission. This is not speculative intelligence work; it is a standard component of competitive diligence for any 505(b)(2) program that takes more than 24 months to develop.
FAQ: Operational Questions for IP and Regulatory Teams
Can a 505(b)(2) product receive an “AB” therapeutic equivalence rating?
Rarely. AB-rating requires pharmaceutical equivalence (same active ingredient, dosage form, strength, route) and demonstrated bioequivalence. 505(b)(2) products are typically different in one or more of those dimensions — different dosage form, different strength, different formulation — which makes them pharmaceutically inequivalent and therefore ineligible for AB-rating. Most receive no therapeutic equivalence code, or a BX code indicating that equivalence data are insufficient for a rating. The commercial consequence is that pharmacists cannot automatically substitute a 505(b)(2) product for the RLD: a prescription for the brand must be filled with the brand, and a prescription for the 505(b)(2) product must be filled with that product. The 505(b)(2) holder needs a sales force to drive prescriptions, not a dispensing substitution mechanism.
How does the 30-month stay calculus differ between 505(b)(2) and ANDA contexts?
The mechanism is identical. The strategic logic is not. A 505(j) ANDA filer fighting for the 180-day first-filer exclusivity has a multi-hundred-million-dollar reason to fight the patent to final judgment. A 505(b)(2) applicant with no 180-day prize typically has a stronger incentive to settle on a launch date or design around the patent rather than litigate to invalidation. Brand companies know this, and their litigation strategy with 505(b)(2) filers often differs: they may be willing to settle sooner, on terms the brand prefers, because the 505(b)(2) applicant’s tolerance for a prolonged fight is lower than a generic’s.
Does a 505(b)(2) approval reset the IRA negotiation clock?
The IRA’s negotiation eligibility is tied to the “approval date” of the “qualifying single source drug” — essentially, the date of the approved NDA for the product as a single-source drug. If the 505(b)(2) product is deemed to contain a distinct active moiety from older approved drugs (e.g., a true new moiety or a substantially different FDC), it may have its own nine-year clock running from its approval date. If CMS determines the active moiety is substantially the same as an already-negotiated drug, the clock does not reset. CMS guidance on this question remains incomplete. Developers of 505(b)(2) products with high Medicare exposure should model both scenarios.
What triggers a complete response letter (CRL) in a 505(b)(2) application?
The three most common triggers are CMC deficiencies (inadequate stability data, unresolved impurity questions, unvalidated in vitro release methods), clinical deficiencies (inadequate bridging data showing the PK profile differs too substantially from the RLD to justify reliance on its safety record), and labeling deficiencies (failure to adequately characterize the differences between the new product and the RLD, or labeling claims the FDA considers inadequately supported). CMC deficiencies are the most common cause of second-cycle review. Planning for a 12-month stability dataset at NDA submission rather than a six-month dataset reduces CRL risk materially.
The Honest State of the 505(b)(2) Market in 2025
The pathway has matured from a niche regulatory mechanism into an industry standard. 943 approvals from 2003 to 2023. 69 reformulations in the 2024-2025 cycle. 60% of all NDA approvals now route through 505(b)(2). The easy wins — simple oral reformulations, straightforward FDCs of two generic components, Rx-to-OTC switches — are almost entirely competed away.
What remains defensible is complex. Long-acting injectables for populations with documented adherence failures, where the payer has quantified the cost of non-adherence. Inhalation products where device-formulation co-optimization creates a CMC barrier that generics cannot clear quickly. FDCs where one component is a new active moiety, generating NCE exclusivity and a new patent family. Orphan-indication repurposing of well-characterized molecules, managed with reputational awareness and pricing restraint.
The litigation environment is more hostile than it was five years ago. Skinny labeling is not dead, but it requires discipline at every commercial touchpoint. The IRA has compressed the revenue runway for small molecule programs. The PBM gatekeeping problem has not abated — if anything, consolidation in the PBM market has concentrated the formulary decision in fewer hands with greater leverage.
For developers who can navigate those constraints, the 505(b)(2) pathway remains the most capital-efficient route to a branded pharmaceutical market position. The economics have not changed. The competitive and regulatory environment around them has.
Key Takeaways: 2025 Market Assessment
Simple oral solid 505(b)(2) programs face commoditization dynamics. The pathway’s durable value is in complex formulations, new active moiety FDCs, and correctly structured orphan programs. Post-IRA, small molecule valuation requires integrated modeling of patent exclusivity and negotiation eligibility as coupled variables. The skinny label carve-out strategy requires a commercial execution protocol that prevents any external communication from implying therapeutic equivalence to the brand. CMC investment at pre-formulation is the highest-return risk mitigation available in the development budget.
Copyright notice: This analysis is an original work. All drug names, company names, litigation matters, and regulatory events cited are real. Data points are sourced from FDA approval records, published clinical trial registries, federal court opinions, and publicly available regulatory guidance.