1. The Economics of Time in Biopharma

Biopharmaceutical asset valuation rests on a single, volatile variable: the duration of market exclusivity. In an industry where a top-performing asset generates daily revenues exceeding $27 million, the precise date of generic or biosimilar entry dictates capital allocation, merger and acquisition valuations, and long-term portfolio strategy. A difference of six months in exclusivity—often the result of a pediatric extension or a specific patent term adjustment—can represent a revenue swing of over $4 billion for a mega-blockbuster.1
The calculation of this date is rarely straightforward. The nominal twenty-year patent term provided by the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) bears little resemblance to the commercial reality of a drug’s life. Regulatory requirements consume a substantial portion of the patent term before the product reaches the market. To correct this imbalance and maintain incentives for innovation, major jurisdictions have established mechanisms to restore lost time. The United States operates under the framework of the Drug Price Competition and Patent Term Restoration Act of 1984, commonly known as the Hatch-Waxman Act. The European Union utilizes the Supplementary Protection Certificate (SPC) system, governed by Regulation (EC) No 469/2009.2
These two systems, while sharing a conceptual goal, diverge radically in their mathematical formulas, legal constraints, and strategic applications. The resulting “Effective Patent Life” (EPL) for a single molecule can differ by years across the Atlantic. For instance, AbbVie’s Humira faced biosimilar competition in Europe in 2018, four years prior to its loss of exclusivity in the United States. Such discrepancies are not accidents of market timing but direct consequences of divergent statutory architectures regarding patent term restoration, patent linkage, and the permissibility of patent thickets.4
This report provides a granular analysis of these differences. It examines the specific calculations of the Hatch-Waxman Patent Term Extension (PTE) versus the European SPC, the strategic implications of regulatory caps like the US “14-year rule” and the EU “15-year limit,” and the disruptive impact of new legislation such as the US Inflation Reduction Act (IRA) and the EU Unitary Patent package.
2. The US Framework: Hatch-Waxman and Patent Term Extension
The United States patent system for pharmaceuticals is defined by a legislative compromise intended to balance the need for innovation incentives with the public interest in affordable generic medicines. The Hatch-Waxman Act introduced the Abbreviated New Drug Application (ANDA) to facilitate generic entry while simultaneously creating the Patent Term Extension (PTE) to compensate innovators for regulatory delays.6
2.1 Statutory Basis and Legislative Intent
Under 35 U.S.C. § 156, the United States Patent and Trademark Office (USPTO) works in concert with the Food and Drug Administration (FDA) to calculate extensions. The legislative intent was to restore a portion of the patent life lost during the “Regulatory Review Period” (RRP). Unlike a simple addition of time, the statute employs a weighted formula that distinguishes between the testing phase and the administrative approval phase.6
2.2 The Regulatory Review Period (RRP) Calculation
The RRP is the foundational metric for US PTE. It is divided into two distinct phases:
- The Testing Phase: Begins on the effective date of an Investigational New Drug (IND) application and ends when a New Drug Application (NDA) or Biologics License Application (BLA) is submitted.
- The Approval Phase: Begins on the date of NDA/BLA submission and ends on the date of FDA approval.2
The statute permits the patent holder to claim:
- 100% of the time spent in the Approval Phase.
- 50% of the time spent in the Testing Phase.
The logic posits that the applicant has some control over the speed of clinical trials (Testing Phase), and thus should share the “cost” of that time. However, the time spent awaiting FDA review (Approval Phase) is viewed as a bureaucratic delay outside the applicant’s control, warranting full compensation.6
Calculation Formula:
$$PTE = (0.5 \times \text{Testing Phase Days}) + (\text{Approval Phase Days}) – \text{Applicant Delays}$$
“Applicant delays” refer to any period where the applicant did not act with “due diligence,” which can be subtracted from the total. The FDA makes the final determination of the RRP, which the USPTO then uses to calculate the extension.9
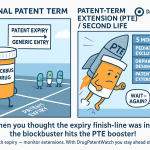
2.3 The 14-Year Rule: A Critical Constraint
The calculated PTE is subject to strict statutory caps. The most misunderstood and financially significant of these is the “14-year rule” found in 35 U.S.C. § 156(c)(3). This provision limits the total remaining patent term (including the extension) to 14 years from the date of FDA approval.
This rule acts as a leveling mechanism. It ensures that no drug, regardless of how much time was lost in development, enjoys more than 14 years of patent-protected market exclusivity. If a drug moves rapidly through development and approval, and the patent still has 15 years of term remaining at the time of launch, the product is ineligible for any PTE. The extension is reduced to zero because the remaining term already exceeds the 14-year statutory target.6
Strategic Implication: This cap disproportionately affects drugs with efficient development timelines. A drug that receives approval seven years after patent filing would have 13 years of nominal term left. The PTE calculation might suggest a 3-year extension, but the 14-year rule would cap the extension at 1 year (13 + 1 = 14). Conversely, a drug that languished in development for 12 years (leaving 8 years of term) could potentially receive a full 5-year extension (8 + 5 = 13), as the total does not breach the 14-year limit.
Industry Insight: “While the five-year cap limits the extension itself, the fourteen-year rule limits the total effective patent life of the product after it has been approved. This is arguably the most critical, and often the most misunderstood, constraint in the entire PTE framework.” — DrugPatentWatch.6
2.4 The Five-Year Cap
Independent of the 14-year rule, no single PTE can exceed five years in length. Even if the calculation yields a restoration period of seven years based on a lengthy RRP, the statute truncates the grant at 60 months. This creates a hard ceiling on the compensation available for extremely protracted regulatory delays.2
2.5 The “One Patent” Limitation
Under US law, an applicant can extend only one patent per approved regulatory review period. This presents a complex strategic choice for intellectual property (IP) counsel. A typical pharmaceutical product is protected by a “patent family” encompassing:
- Composition of matter (the active molecule).
- Method of use (the indication).
- Formulation (the specific delivery vehicle).
Typically, the composition of matter patent is the preferred target for extension as it provides the broadest exclusion rights. However, if the composition patent is nearing expiration or is encumbered by a terminal disclaimer, the applicant may choose to extend a method-of-use patent instead, provided it covers the approved indication. This decision must be made within 60 days of FDA approval, requiring rapid and accurate analysis of the patent portfolio’s relative strength and expiration profile.2
3. The European Framework: Supplementary Protection Certificates
Europe approaches the problem of patent term erosion through a different legal instrument. The Supplementary Protection Certificate (SPC) is not an extension of the patent term itself but a sui generis right that comes into force immediately upon the expiry of the basic patent. While the economic effect is similar—prolonged exclusivity—the calculation methodology creates scenarios where the European expiry date can differ significantly from the US date.3
3.1 The Sui Generis Nature of SPCs
The SPC is a standalone right linked to the basic patent. It protects the specific active ingredient covered by the marketing authorization. Unlike the US PTE, which extends the patent claims relevant to the approved product, the SPC is strictly limited to the product authorized for sale. This distinction becomes critical in infringement cases involving variants or derivatives of the active moiety.10
3.2 The SPC Calculation Formula
The European calculation is notably simpler than the US RRP formula but often less generous for drugs with rapid development timelines. The formula focuses solely on the elapsed time between the patent application and the first marketing authorization in the European Economic Area (EEA).
The Formula:
$$\text{SPC Duration} = (\text{Date of First EEA Marketing Authorization} – \text{Date of Basic Patent Filing}) – 5 \text{ Years}$$
This formula subtracts a standard five-year period from the development timeline. The intent is that the patent holder should bear the first five years of regulatory delay; the SPC compensates for any delay beyond that threshold.3
3.3 The 15-Year Exclusivity Cap
The SPC Regulation (EC) No 469/2009 imposes a 15-year cap on total exclusivity. The combination of the remaining patent term and the SPC cannot exceed 15 years from the date of the first marketing authorization in the EEA. This mirrors the US 14-year rule but provides an additional year of potential exclusivity.3
Additionally, the duration of the SPC itself cannot exceed five years, consistent with the US cap.
3.4 Negative Term SPCs: A European Anomaly
One of the most counterintuitive aspects of the European system is the existence of “negative term” SPCs. The formula $(\text{Date of MA} – \text{Patent Filing}) – 5 \text{ Years}$ can yield a negative result if the development time was less than five years.
For example, if a patent was filed on January 1, 2010, and the Marketing Authorization was granted on January 1, 2014 (4 years later), the calculation is:
$$4 \text{ Years} – 5 \text{ Years} = -1 \text{ Year}$$
Historically, such an SPC would be valueless. However, the introduction of the Pediatric Regulation changed the calculus. A “negative term” SPC acts as a vessel for the six-month pediatric extension. If a company has a calculated SPC of -3 months, applying the +6 month pediatric extension results in a net positive exclusivity of 3 months.
Case Study: Merck’s Januvia (Sitagliptin)
In the Merck Sharp & Dohme case, the Court of Justice of the European Union (CJEU) clarified that an SPC could be granted with a negative term. Merck sought to attach a pediatric extension to an SPC that was mathematically negative. The court ruled that the SPC need not have a positive duration to serve as the basis for a pediatric extension. For a drug generating billions in revenue, a net extension of even a few weeks justifies the administrative cost of filing for a negative SPC.13
4. Pediatric Exclusivity: Divergent Incentives
Both jurisdictions recognize the need to incentivize clinical trials in pediatric populations, which are historically underserved in drug development. However, the mechanisms for reward differ fundamentally, affecting the final expiration date.
4.1 US Pediatric Exclusivity (PREA/BPCA)
In the US, pediatric exclusivity is governed by the Best Pharmaceuticals for Children Act (BPCA). It provides six months of additional market exclusivity. Crucially, this is not an extension of the patent term in the technical sense; rather, it is a period of “pediatric exclusivity” that attaches to all existing exclusivity periods listed in the Orange Book.
This includes:
- The remaining term of the patent.
- Any granted Patent Term Extension (PTE).
- Any regulatory exclusivity (e.g., 5-year New Chemical Entity exclusivity, 7-year Orphan Drug exclusivity).
Strategic Impact: The six-month US extension is additive to the 14-year cap. If a drug hits the 14-year limit via PTE, the pediatric exclusivity is added on top of that, potentially extending the effective life to 14.5 years. This “stacking” effect makes the pediatric program extremely valuable, with estimated returns averaging over $176 million and reaching up to $500 million for blockbusters.8
4.2 EU Paediatric Regulation and SPC Extension
In Europe, the reward for completing an agreed Paediatric Investigation Plan (PIP) is a six-month extension of the SPC. This is a critical distinction: the extension applies to the SPC, not the patent itself.
- Requirement: An SPC must exist (or be applied for) to receive the extension.
- Limitation: If no SPC exists (e.g., because the development was too fast and the result was negative, and the company failed to file for a negative SPC), the pediatric extension cannot be granted.
This structure creates a procedural trap. Companies must foresightfully apply for SPCs, even those with zero or negative terms, to preserve the vehicle for the future pediatric extension. The only exception is for orphan drugs, which receive a separate reward (an extension of the 10-year market exclusivity period) rather than an SPC extension.10
4.3 Strategic Implications of the Six-Month “Bonus”
The financial magnitude of these six-month periods drives intense lifecycle management. For a drug like Keytruda, projected to face competition around 2028, a six-month extension translates to billions in preserved revenue. US analysts estimate that Keytruda’s sales could peak near $36 billion; thus, the pediatric extension alone could be worth nearly $18 billion in gross revenue protection, assuming no other erosion. This incentivizes companies to initiate pediatric trials early, ensuring the data is available to claim the reward before the patent cliff arrives.17
5. Patent Linkage and Market Entry Mechanisms
The calculation of an expiration date is theoretical until tested by the arrival of competition. The mechanism by which generic competitors enter the market—and the barriers innovators can erect to stop them—differs starkly between the US “linkage” system and the European “decoupled” system.
5.1 US Patent Linkage: The Orange Book
The US operates a system of strong patent linkage centered on the Orange Book (Approved Drug Products with Therapeutic Equivalence Evaluations).
- Listing Requirement: Innovators must list patents claiming the drug substance, drug product, or method of use.
- Regulatory Bar: The FDA is statutorily prohibited from approving a generic ANDA if it infringes a valid listed patent, unless specific certifications are made.19
5.2 Paragraph IV Certifications and the 30-Month Stay
When a generic applicant seeks to launch before the expiry of an Orange Book patent, they must file a Paragraph IV certification, asserting that the patent is invalid, unenforceable, or will not be infringed.
- The Trigger: This filing is an artificial act of infringement.
- The 30-Month Stay: If the patent holder sues within 45 days of receiving notice, the FDA automatically stays the approval of the generic application for 30 months.
This 30-month stay provides a guaranteed shield for the innovator. It prevents “at risk” launches and ensures that litigation (or a settlement) occurs before the generic reaches the pharmacy shelf. While intended to resolve disputes early, critics argue it incentivizes “evergreening” by encouraging innovators to list secondary patents to trigger multiple stays.21
5.3 Europe’s Decoupled Regulatory System
Europe generally lacks a patent linkage system. The regulatory approval of a generic medicine by the European Medicines Agency (EMA) or national authorities is based solely on scientific criteria (quality, safety, efficacy). The status of the originator’s patent is legally irrelevant to the granting of the Marketing Authorization (MA).
- No Orange Book: There is no centralized list of patents that regulators consult to block approval.
- Private Enforcement: Innovators must monitor new MA grants and seek preliminary injunctions in national courts to stop the launch. The burden of policing the market falls entirely on the patent holder.19
5.4 Launch at Risk: The European Generic Strategy
Because there is no automatic 30-month stay, European generic companies often employ a “launch at risk” strategy. They obtain regulatory approval and launch their product while the patent is still technically in force, gambling that the patent will be revoked or that the innovator will fail to secure an injunction.
- Risk Profile: If the generic launches and the patent is later upheld, they are liable for significant damages. However, if the patent is weak, this strategy allows for much earlier market entry than in the US.
- Impact: This dynamic fosters a more aggressive generic market in Europe. Combined with the fragmentation of national courts (prior to the UPC), a generic might launch in the UK and Germany while being blocked in France, creating a patchwork of exclusivity loss.19
6. Patent Thickets and Secondary Enforcement
The “effective” life of a drug often extends well beyond the expiration of the primary compound patent due to “patent thickets”—dense clusters of overlapping patents covering formulations, dosage regimens, and manufacturing processes.
6.1 The Mechanics of Thicket Creation
Innovators file secondary patents to create layers of protection. Even if the primary patent expires, a competitor may be blocked by a patent on a specific crystalline form or a method of treating a specific patient sub-population.
- US Continuations: The US system allows for “continuation” applications, enabling applicants to pursue new claims based on an original patent filing years later. This facilitates the construction of extensive thickets around a successful product.25
- EPO Added Matter: The European Patent Office (EPO) applies strict rules against “added matter.” Claims cannot be amended in a way that extends beyond the content of the application as filed. This makes it significantly harder to obtain broad secondary patents in Europe compared to the US.5
6.2 Divergent Patentability Standards: US vs EPO
The divergence in patentability standards leads to different outcomes for the same invention.
- Plausibility: European courts require that the therapeutic effect be “plausible” at the filing date. Speculative broad claims are often revoked.
- Obviousness: The US standard for obviousness can be more permissive for incremental improvements (like new formulations) compared to the EPO’s “problem-solution approach,” which rigorously tests whether an invention involves an inventive step.5
6.3 Case Study: The Humira Divergence
AbbVie’s Humira (adalimumab) provides the definitive case study of this divergence.
- US Outcome: AbbVie constructed a thicket of over 100 patents. While the primary patent expired in 2016, secondary patents on methods of treatment and formulations allowed AbbVie to settle with biosimilar competitors, delaying entry until 2023.
- EU Outcome: The same strategy faced hurdles in Europe. Key secondary patents were revoked or surrendered due to the EPO’s stricter standards. Consequently, biosimilars launched in Europe in October 2018.
- Economic Impact: The five-year gap between EU and US entry allowed AbbVie to generate tens of billions of dollars in additional US revenue, while European healthcare systems reaped the savings of competition. This $100+ billion disparity illustrates the immense value of the US patent thicket strategy.4
7. Commercial Case Studies in Exclusivity Management
Analyzing specific blockbuster drugs reveals how these legal differences manifest in commercial reality.
7.1 Eliquis (Apixaban): Plausibility vs Prediction
The Drug: Eliquis, a leading anticoagulant co-marketed by Bristol Myers Squibb (BMS) and Pfizer.
US Scenario: The primary composition patent (US 6,967,208) was extended via PTE to November 2026. A formulation patent (US 9,326,945) expires in 2031. BMS successfully defended these patents in litigation, and settlements have delayed generic entry until at least April 1, 2028.
EU Scenario: The corresponding European patent (EP 1 427 415) was challenged by Sandoz and Teva. In a landmark decision, the UK High Court revoked the patent on the grounds of plausibility. The court ruled that the patent did not make it plausible that apixaban would be effective across the entire scope of the claim at the time of filing. The Court of Appeal upheld this decision.
Result: Eliquis lost exclusivity in the UK and other markets years earlier than in the US, solely due to the divergent application of patent validity standards.27
7.2 Stelara (Ustekinumab): The Settlement Gap
The Drug: Stelara, an immunology drug by Johnson & Johnson.
US Scenario: Facing the expiry of its primary patent, J&J utilized a thicket of secondary patents to sue biosimilar developers. Settlements were reached pushing US launch to 2025.
EU Scenario: With fewer secondary patents surviving EPO scrutiny, biosimilars were cleared for launch in 2024.
Result: A one-year arbitrage window where the US market remains exclusive while the EU market fragments.31
7.3 Keytruda (Pembrolizumab): The Next Cliff
The Drug: Merck’s Keytruda, the world’s best-selling drug.
The Cliff: Keytruda faces a loss of exclusivity (LOE) around 2028. US sales are projected to peak at $36 billion.
The Defense: Merck is aggressively developing a subcutaneous (SubQ) formulation of Keytruda. This new formulation is protected by fresh patents that could extend exclusivity into the 2030s.
Strategy: In the US, Merck will attempt to switch patients to the SubQ version before 2028. If successful, the 30-month stay and Orange Book linkage will make it difficult for pharmacists to substitute the old IV generic for the new SubQ brand. In Europe, where automatic substitution is less prevalent and linkage is absent, this “product hopping” strategy faces higher barriers to adoption if the payer refuses to reimburse the more expensive SubQ version.17
8. The Impact of the Inflation Reduction Act
The passage of the Inflation Reduction Act (IRA) in 2022 introduced a new variable that threatens to render patent term calculations moot for certain drugs.
8.1 The New Event Horizon for Exclusivity
The IRA authorizes Medicare to negotiate prices for top-selling drugs. The eligibility for negotiation is based on time since FDA approval, not patent status:
- Small Molecules: Negotiable 9 years after approval.
- Biologics: Negotiable 13 years after approval.
Disruption: A small molecule drug might have a valid patent term (including PTE) extending to 14 years. However, under the IRA, Medicare price controls kick in at Year 9. This effectively creates a “regulatory taking” of the final 5 years of patent value for the Medicare segment.
8.2 The Small Molecule vs Biologic Disparity
The 9-year vs 13-year distinction has created a massive incentive distortion.
- Small Molecules: With only 9 years of pricing freedom, the Return on Investment (ROI) for small molecules is compressed. This is shorter than the average 13-14 year effective patent life previously enjoyed.
- Biologics: The 13-year window aligns more closely with traditional patent expiries, making biologics a safer investment.
Evidence: Data from 2024 and 2025 indicates a decline in post-approval clinical trials for small molecules. Companies are cancelling research into new indications for existing small molecules because the 9-year clock does not allow enough time to recoup the investment in secondary trials.35
8.3 Shifts in R&D Allocation
The industry is responding by deprioritizing small molecules in favor of biologics. “Small-molecule drug funding has dropped 70 percent since legislation that would become the IRA’s drug pricing provisions was first drafted,” according to analysis by the Information Technology and Innovation Foundation (ITIF). This shift threatens the pipeline for diseases where small molecules (pills) are preferable to biologics (injections), such as neurological conditions or treatments for the elderly.36
9. The Future of European Exclusivity: The Unitary SPC
Europe is on the cusp of a systemic transformation with the implementation of the Unitary Patent and the proposed Unitary SPC.
9.1 The Unitary Patent System
The Unitary Patent (UP) offers a single patent right covering most EU Member States (currently 17), enforced by a single Unified Patent Court (UPC). This eliminates the need for validating and litigating patents in each country individually.
- Benefit: Reduced administrative costs ($192,000 for EU-wide coverage vs significantly less for UP).
- Risk: A “central attack” at the UPC can revoke the patent for the entire territory in one stroke.37
9.2 The Proposed Unitary SPC Regulation
To complement the UP, the European Commission proposed a Unitary SPC regulation in April 2023. This creates a centralized procedure for granting SPCs.
- Current State: Applicants must file separate SPC applications in each national office, leading to divergent outcomes (e.g., an SPC granted in France but refused in Germany).
- Future State: A single application to the EU Intellectual Property Office (EUIPO) will result in a combined “Unitary SPC” for UP countries and national SPCs for others.
- Timeline: The legislation is moving through the EU Parliament and Council, with implementation expected around 2026.39
9.3 Strategic Implications for Pan-European Enforcement
The Unitary SPC will harmonize the expiration dates across Europe, eliminating the “patchwork” that generics currently exploit. However, it also raises the stakes for validity challenges. A generic company will no longer need to win in German, French, and Italian courts to clear the market; one victory at the UPC will open the entire continent. This binary “all-or-nothing” outcome will likely lead to more aggressive, high-value litigation.42
10. Data-Driven Forecasting and Competitive Intelligence
In this fragmented landscape, relying on the “nominal” expiration date of a patent is a strategic error. Business development teams must employ sophisticated data intelligence to forecast the Effective Patent Life (EPL).
10.1 The Necessity of Effective Patent Life Calculation
Accurate forecasting requires a dynamic model that integrates:
- Statutory Formulas: Calculating US PTE vs EU SPC terms based on specific RRP data.
- Caps and Constraints: Applying the 14-year (US) and 15-year (EU) limits.
- Regulatory Exclusivities: Overlaying NCE (5 years), Orphan (7/10 years), and Pediatric (6 months) periods.
- Litigation Status: Monitoring Paragraph IV filings and 30-month stay expirations.
10.2 Monitoring Global Family Status
Tools like DrugPatentWatch allow users to map global patent families. This is crucial for identifying “weak links.” If a key patent in a family is revoked in the UK due to plausibility (as with Eliquis), it serves as a signal to scrutinize the equivalent patents in other jurisdictions. It also alerts competitors to potential “Launch at Risk” opportunities in Europe while the US market remains locked.44
10.3 The Role of Specialized Intelligence Platforms
Platform data enables the identification of “negative term” SPC filings, which act as early warning signals of a company’s intent to pursue pediatric extensions. Furthermore, tracking formulation patents can reveal “evergreening” strategies—like Merck’s SubQ Keytruda—years before the new product launches.
Industry Insight: “For business development teams, DrugPatentWatch offers critical capabilities… Automatically calculates ‘effective’ expiration dates by integrating patent terms with FDA regulatory exclusivities (NCE, ODE, Pediatric) to predict the true ‘generic entry date’.”.44
11. Key Takeaways
- Divergent Math, Divergent Reality: The US PTE formula (claiming 100% of approval time) and the EU SPC formula (deducting 5 years) yield fundamentally different extension periods. A drug can have a 5-year extension in the US and zero in Europe.
- The 14-Year Cap is the “Silent Killer” of US Exclusivity: The rule limiting total US patent life to 14 years post-approval is the primary reason efficient drug development programs receive zero extension, penalizing speed.
- Patent Thickets Work Better in the US: Due to permissive “continuation” rules and the 30-month stay, US innovators can maintain monopolies years longer than in Europe, where stricter “added matter” rules and the lack of automatic stays facilitate earlier biosimilar entry (e.g., Humira, Stelara).
- Negative SPCs are Strategic Assets: In Europe, filing for a negative term SPC is a necessary maneuver to capture the valuable six-month pediatric extension.
- The IRA Disruption: The US Inflation Reduction Act has effectively introduced a “9-year regulatory cliff” for small molecules, overriding patent protection and shifting industry investment toward biologics.
12. FAQ
Q1: Why is the “14-year rule” in the US considered a “catch-up” mechanism?
A: The 14-year rule ensures that Patent Term Extension is only used to restore lost time, not to extend a monopoly beyond a reasonable period. If a drug is approved quickly and already has 14+ years of patent life remaining, the law deems the innovator has sufficient exclusivity. PTE is thus reserved for drugs that suffered significant regulatory delays that eroded their life below this 14-year baseline.6
Q2: How does the “Launch at Risk” strategy in Europe differ from US Paragraph IV litigation?
A: In the US, a generic filing triggers an automatic 30-month stay, preventing the FDA from approving the generic while litigation proceeds. In Europe, there is no automatic stay. A generic can be approved and launch “at risk” while the patent is still valid. The onus is on the patent holder to seek an injunction. This makes the European market more dynamic but exposes generic companies to high damages if they lose.19
Q3: Can the US Inflation Reduction Act shorten a drug’s commercial life even if the patent is valid?
A: Yes. The IRA allows Medicare to negotiate (effectively cap) prices for small molecules 9 years after approval. Even if a valid patent (with PTE) protects the drug until Year 14, the revenue potential is drastically curtailed at Year 9 for the Medicare population. This decouples “commercial life” from “patent life”.35
Q4: What is the benefit of the Unitary SPC for pharmaceutical companies?
A: Efficiency and consistency. Currently, companies must file and litigate SPCs in 27 different countries, leading to high costs and contradictory legal outcomes. The Unitary SPC will allow a single application to cover most of the EU, streamlining enforcement. However, it also creates the risk of a single, central revocation action.39
Q5: How does the European “plausibility” standard affect patent validity compared to the US?
A: The European standard of “plausibility” requires that the patent application makes it credible that the invention works as claimed at the time of filing. This prevents speculative broad claims. This standard was the key reason the Eliquis patent was revoked in the UK while the equivalent US patent was upheld, leading to earlier generic entry in Europe.28
Works cited
- The End of Exclusivity: Navigating the Drug Patent Cliff for Competitive Advantage, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/the-impact-of-drug-patent-expiration-financial-implications-lifecycle-strategies-and-market-transformations/
- Patent Term Extension (PTE) Under the Hatch-Waxman Act – Azami Global, accessed December 27, 2025, https://azamiglobal.com/blog/patent-term-extension-pte-under-the-hatch-waxman-act/
- The Final Countdown: A Strategic Guide to SPC Expiry in the European Union, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/the-final-countdown-a-strategic-guide-to-spc-expiry-in-the-european-union/
- The Expiry of Humira® Market Exclusivity and the Entry of Adalimumab Biosimilars in Europe: An Overview of Pricing and National Policy Measures – PubMed Central, accessed December 27, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC7839249/
- The Global Patent Thicket: A Comparative Analysis of Pharmaceutical Monopoly Strategies in the U.S., Europe, and Emerging Markets – DrugPatentWatch, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/how-do-patent-thickets-vary-across-different-countries/
- The Billion-Dollar Equation: Mastering Patent Term Extension to Secure Market Dominance, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/calculating-the-regulatory-review-period-for-patent-term-extension/
- Pharmaceutical Patent Term Extension: An Overview – Alacrita Consulting, accessed December 27, 2025, https://www.alacrita.com/whitepapers/pharmaceutical-patent-term-extension-an-overview
- Protection beyond 20 years: data on SPCs and other term extensions for pharmaceutical patents – EPO, accessed December 27, 2025, https://www.epo.org/en/searching-for-patents/helpful-resources/patent-knowledge-news/protection-beyond-20-years-data-spcs
- The Second Life of a Blockbuster: A Comprehensive Guide to Monitoring Drug Patent Term Extensions – DrugPatentWatch, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/the-second-life-of-a-blockbuster-a-comprehensive-guide-to-monitoring-drug-patent-term-extensions/
- Paediatric extensions to Supplementary Protection Certificates in the EU / EEA and UK, accessed December 27, 2025, https://www.jakemp.com/knowledge-hub/paediatric-extensions-to-supplementary-protection-certificates-in-the-eu-eea-and-uk/
- Supplementary Protection Certificates (SPCs): Extending Patent Protection in Europe (2025 Update) – Azami Global, accessed December 27, 2025, https://azamiglobal.com/blog/european-supplementary-protection-certificates-spcs-for-pharmaceuticals-a-practical-guide/
- How to calculate the extension of SPC protection time? – Patsnap Synapse, accessed December 27, 2025, https://synapse.patsnap.com/blog/how-to-calculate-the-extension-of-spc-protection-time
- Negative term SPCs – a positive outcome – D Young & Co, accessed December 27, 2025, https://www.dyoung.com/en/knowledgebank/articles/negativetermspc
- Supplementary Protection Certificates: A Positive Spin on a Negative Term – WILLIAM FRY, accessed December 27, 2025, https://www.williamfry.com/knowledge/supplementary-protection-certificates-a-positive-spin-on-a-negative-term/
- Labeling Changes and Costs for Clinical Trials Performed Under the US Food and Drug Administration Pediatric Exclusivity Extension, 2007 to 2012 – PubMed Central, accessed December 27, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC6248195/
- The need-to-know facts about patent term extensions in Europe – COHAUSZ & FLORACK, accessed December 27, 2025, https://www.cohausz-florack.de/en/blog/article/the-need-to-know-facts-about-patent-term-extensions-in-europe-1/
- Merck’s Keytruda Dependency: A Growth Story With An Expiration Date | Nasdaq, accessed December 27, 2025, https://www.nasdaq.com/articles/mercks-keytruda-dependency-growth-story-expiration-date
- Keytruda’s patent wall lengthens its monopoly – I-MAK, accessed December 27, 2025, https://www.i-mak.org/wp-content/uploads/2021/05/I-MAK-Keytruda-deck-2021-05-03F.pdf
- A Strategic Guide to International Considerations in U.S. Paragraph IV Challenge Planning, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/a-strategic-guide-to-international-considerations-in-u-s-paragraph-iv-challenge-planning/
- Legal Updates & Publications – Drew & Napier LLC, accessed December 27, 2025, https://www.drewnapier.com/publications/Comparing-Patent-Linkage-Systems-in-the-US,-China,
- The timing of 30‐month stay expirations and generic entry: A cohort study of first generics, 2013–2020 – NIH, accessed December 27, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC8504843/
- Patent Certifications and Suitability Petitions – FDA, accessed December 27, 2025, https://www.fda.gov/drugs/abbreviated-new-drug-application-anda/patent-certifications-and-suitability-petitions
- Intricacies of the 30-Month Stay in Pharmaceutical Patent Cases | Articles – Finnegan, accessed December 27, 2025, https://www.finnegan.com/en/insights/articles/intricacies-of-the-30-month-stay-in-pharmaceutical-patent-cases.html
- The New EU “Pharma Package”: The “Bolar” exemption – A comparison of Commission/Parliament/Council positions | Crowell & Moring LLP, accessed December 27, 2025, https://www.crowell.com/en/insights/client-alerts/the-new-eu-pharma-package-the-bolar-exemption-a-comparison-of-commissionparliamentcouncil-positions
- Why Pharmaceutical Patent Thickets Are Unique – Rutgers University, accessed December 27, 2025, https://scholarship.libraries.rutgers.edu/esploro/fulltext/journalArticle/Why-Pharmaceutical-Patent-Thickets-Are-Unique/991032166117504646?repId=12778612660004646&mId=13778612650004646&institution=01RUT_INST
- Biological patent thickets and delayed access to biosimilars, an American problem – PMC, accessed December 27, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC9439849/
- Overpatented, Overpriced 2025: A Data Brief on Medicare-Negotiated Drugs – I-MAK, accessed December 27, 2025, https://www.i-mak.org/overpatented/
- Update on Plausibility in UK Patent Law | Mason Hayes Curran, accessed December 27, 2025, https://www.mhc.ie/latest/insights/bristol-myers-squibbs-spc-is-found-not-plausible
- Sandoz and Teva invalidate blockbuster apixaban patent and SPC, accessed December 27, 2025, https://www.juve-patent.com/cases/sandoz-and-teva-invalidate-blockbuster-apixaban-patent-and-spc/
- The Bristol Myers Squibb-Pfizer Alliance is pleased with the decision by the U.S. Court of Appeals for the Federal Circuit upholding the Eliquis® Patents, accessed December 27, 2025, https://news.bms.com/news/details/2021/The-Bristol-Myers-Squibb-Pfizer-Alliance-is-pleased-with-the-decision-by-the-U.S.-Court-of-Appeals-for-the-Federal-Circuit-upholding-the-Eliquis-Patents/default.aspx
- FDA Approves First Stelara Biosimilar, Wezlana, accessed December 27, 2025, https://www.centerforbiosimilars.com/view/fda-approves-first-stelara-biosimilar-wezlana
- How the U.S. Compares to Europe on Biosimilar Approvals and Products In the Pipeline – Updated January 13, 2025, accessed December 27, 2025, https://www.biosimilarsip.com/2025/01/14/how-the-u-s-compares-to-europe-on-biosimilar-approvals-and-products-in-the-pipeline-updated-january-13-2025/
- When do the patents on STELARA expire, and when will biosimilar STELARA be available? – Drug Patent Watch, accessed December 27, 2025, https://www.drugpatentwatch.com/p/biologics/tradename/STELARA
- Half of Merck’s sales are in jeopardy. Can Keytruda’s sequel save the day?, accessed December 27, 2025, https://www.biopharmadive.com/news/merck-keytruda-subcutaneous-cancer-sales-drug-delivery/801889/
- Early impact of the Inflation Reduction Act on small molecule vs biologic post-approval oncology trials – PMC – NIH, accessed December 27, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC12392883/
- The Inflation Reduction Act Is Negotiating the United States Out of Drug Innovation | ITIF, accessed December 27, 2025, https://itif.org/publications/2025/02/25/the-inflation-reduction-act-is-negotiating-the-united-states-out-of-drug-innovation/
- The potential impact of the unitary Supplementary Protection Certificate on access to health technologies – European Parliament, accessed December 27, 2025, https://www.europarl.europa.eu/RegData/etudes/STUD/2023/753104/IPOL_STU(2023)753104_EN.pdf
- Unitary supplementary protection certificate for plant protection products – European Parliament, accessed December 27, 2025, https://www.europarl.europa.eu/RegData/etudes/BRIE/2024/762281/EPRS_BRI(2024)762281_EN.pdf
- EC Pharma (and Patent) Package on the home straight? – Taylor Wessing, accessed December 27, 2025, https://www.taylorwessing.com/en/insights-and-events/insights/2024/12/ec-pharma-package
- SPCs in Europe: what’s round the corner? – Dyoung, accessed December 27, 2025, https://www.dyoung.com/en/knowledgebank/articles/spcs-patents-europe-faq-2025
- BREAKING NEWS: Draft EU legislation on unitary SPCs and new centralized SPC examination procedure unveiled | Kluwer Patent Blog, accessed December 27, 2025, https://legalblogs.wolterskluwer.com/patent-blog/breaking-news-draft-eu-legislation-on-unitary-spcs-and-new-centralized-spc-examination-procedure-unveiled/
- Unitary SPC in Sight – European Commission Published Legislation Proposal | Finnegan, accessed December 27, 2025, https://www.finnegan.com/en/insights/blogs/european-ip-blog/unitary-spc-in-sight-european-commission-published-legislation-proposal.html
- The Unitary Patent and UPC – Implications for the pharmaceutical sector – PubMed, accessed December 27, 2025, https://pubmed.ncbi.nlm.nih.gov/36195203/
- When Do Drug Patents Expire: Understanding the Lifecycle of Pharmaceutical Innovations, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/when-do-drug-patents-expire/
- Implementing Patent-Expiry Forecasting: A 12-Step Checklist for Competitive Advantage, accessed December 27, 2025, https://www.drugpatentwatch.com/blog/implementing-patent-expiry-forecasting-a-12-step-checklist-for-competitive-advantage/
- The Impact of the Inflation Reduction Act on the Economic Lifecycle of a Pharmaceutical Brand – IQVIA, accessed December 27, 2025, https://www.iqvia.com/locations/united-states/blogs/2024/09/impact-of-the-inflation-reduction-act
- Teva Pharma. USA, Inc. v. Sandoz, Inc. | 574 U.S. 318 (2015), accessed December 27, 2025, https://supreme.justia.com/cases/federal/us/574/318/