Introduction
China’s pharmaceutical sector completed a structural transformation in less than a decade. The country that was processing the world’s generics backlog in 2015 is now the source of billion-dollar biologic assets acquired by AstraZeneca, Merck, and Johnson & Johnson. The combined value of China’s out-licensing deals reached approximately $46 billion in 2024. Thirty percent of all licensing deals conducted by major global pharmaceutical companies now involve a Chinese biotech counterparty. These are not gradual market shifts. They are the output of a deliberate, state-engineered industrial policy executed at a pace that most Western regulatory systems could not replicate.
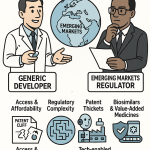
The strategic case for tracking China’s pharmaceutical ecosystem has never been more direct for IP teams and institutional investors. Patent portfolios are now the primary collateral in China’s biopharma capital markets. The Hong Kong Stock Exchange allows pre-revenue biotechs to list on the basis of their pipeline IP alone. Shanghai’s STAR Market uses a similar framework. When a Chinese biotech files a PD-1/VEGF bispecific antibody patent, that filing is not an administrative formality; it is the valuation anchor for a potential nine-figure licensing transaction. Understanding how those patents are constructed, how they interact with the NMPA’s accelerated approval pathways, and how they hold up in Chinese IP courts is the core analytical competency that separates informed participants from passive observers in this market.
“China’s share of the global R&D pipeline now rivals that of the United States. Its biotech hubs are producing a disproportionate number of innovations in ADCs, bispecific antibodies, and cell and gene therapies. The question for Western investors is no longer whether to engage, but how to do so with an accurate model of the risks.”DrugPatentWatch Analytical Framework, 2026
The New Architecture of Chinese Pharma: From Generics Factory to Patent-Generating Engine
Three forces built China’s current pharmaceutical innovation architecture: a top-down national policy mandate, an overhaul of the drug regulatory system that cut approval timelines to near-FDA parity, and a capital market restructuring that made patent portfolios the primary basis of biotech valuation. Each force amplified the others. Policy provided the direction and the political protection for capital allocation. Regulatory reform reduced the timeline between filing an Investigational New Drug (IND) application and generating Phase I/II data to the point where Chinese companies could outpace Western peers in early-stage proof-of-concept generation. And the capital markets, restructured by HKEX’s 2018 Chapter 18A amendment and Shanghai’s STAR board, rewarded those companies that could translate regulatory speed into IP-backed clinical assets. The interaction of these three forces produced the ecosystem that global MNCs are now licensing from.
1.1 The “Healthy China 2030” Policy Superstructure and Its IP Implications
“Healthy China 2030” is the governing strategic document for China’s pharmaceutical sector. Published in 2016, it is not a health ministry budget plan. It is a national industrial policy with explicit targets for domestic drug innovation, an innovation-friendly regulatory environment, and the integration of China into global pharmaceutical value chains. The document designates a robust domestic pharmaceutical sector as a component of national strategic security, placing it in the same category as semiconductors and advanced manufacturing in terms of state priority. That classification has direct consequences for patent policy, capital allocation, and the speed at which bureaucratic obstacles get resolved.
The operationalization of “Healthy China 2030” into patent strategy is visible in the “Pharmaceutical Industry High-Quality Development Action Plan (2023-2025).” The plan provides concrete guidance for converting China’s scientific output into protected IP. It directs state-owned enterprises and research institutions to build patent portfolios that can withstand international challenge, and it targets an increase in the number of first-in-class drug candidates originating from Chinese labs. For IP teams analyzing Chinese counterparties, this policy context is material: companies receiving state guidance and funding under this framework face pressure to file broadly and aggressively, which affects both the density of their patent portfolios and the likelihood they will litigate to defend them.
The government’s incentive structure for innovation explicitly uses the country’s large patient population as a policy tool. China’s 1.4 billion residents generate a volume of clinical trial recruitment that compresses Phase I and Phase II timelines by months relative to North American or European sites. State policy amplifies this natural advantage by funding hospital networks as trial sites and by establishing regulatory pathways that accept data from Chinese sites as globally valid. The result is a system where the raw material for patent-generating clinical data is abundant, centrally organized, and politically prioritized.
Policy Note for IP Teams: Companies operating under the “High-Quality Development Action Plan” are required to disclose patent filing activity as part of state reporting. This creates a tracking opportunity. DrugPatentWatch’s NMPA filing database allows analysts to monitor IND submissions alongside Chinese National Intellectual Property Administration (CNIPA) patent filings for the same molecule, identifying the gap between patent claim date and clinical entry — a key metric for assessing the effective patent life remaining at potential approval.
1.2 The Regulatory Revolution: NMPA Reform, Accelerated Pathways, and the IND Timeline Compression
China’s National Medical Products Administration (NMPA) executed one of the fastest regulatory modernizations in pharmaceutical history between 2015 and 2025. The starting point was severe: a 20,000-application backlog, a review staff of 150, and approval timelines that left Chinese patients waiting five to eight years after US or EU launch for access to new medicines. The end point, a decade later, is a system where the NMPA proposed in June 2025 to reduce IND review times from 60 to 30 working days, directly matching the FDA’s standard. That compression is not simply a bureaucratic efficiency gain. It changes the patent clock arithmetic for every drug developed in China.
The NMPA cleared its backlog by expanding its Center for Drug Evaluation (CDE) review staff from 150 to over 700 full-time reviewers by 2018, supplemented by hundreds of contracted external experts. Once the backlog was resolved, the agency introduced the suite of accelerated pathways that now defines the system: Priority Review, Breakthrough Therapy Designation, and Conditional Approval on surrogate endpoints. The share of new drug applications granted Priority Review grew from 14% in 2016 to 77% by 2019, reflecting both the policy intent and the changing composition of submissions as Chinese biotechs filed an increasing volume of oncology assets targeting well-validated mechanisms.
China’s 2017 accession to the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) was the structural prerequisite for integrating Chinese clinical data into global drug programs. ICH membership allowed the NMPA to accept foreign clinical data for new drug applications, which eliminated the costly requirement for standalone China-specific trials. For Chinese companies developing assets for global out-licensing, it meant they could design their trials to satisfy both the NMPA and the FDA simultaneously, compressing the timeline between Chinese approval and the delivery of globally valid clinical data to a licensing partner.
Technology Roadmap: NMPA Accelerated Pathway Eligibility by Modality (2025)
How key drug modalities map to available NMPA accelerated review pathways
Priority Review
ADCs & Bispecifics
Antibody-drug conjugates targeting novel epitopes and bispecific antibodies with dual-mechanism rationale qualify for Priority Review as drugs addressing significant unmet clinical needs. Review target: 130 working days for NDA.
Breakthrough Therapy
Cell & Gene Therapy
CAR-T and gene-editing therapeutics are the primary beneficiaries of Breakthrough Therapy designation under the revised 2021 guidelines. This designation triggers rolling review, reducing effective NDA clock by 30–50%.
Conditional Approval
Oncology Biologics
PD-1/L1 inhibitors and other immuno-oncology agents can secure Conditional Approval on surrogate endpoints (ORR, PFS). Post-marketing confirmatory trials are then required to convert conditional approval to full registration.
Standard Review
mRNA Platforms
Prophylactic mRNA vaccines and therapeutic mRNA constructs currently follow standard 200-working-day NDA review unless granted Priority status. The NMPA is developing modality-specific guidance expected in 2026.
The Marketing Authorization Holder (MAH) system, introduced as a pilot in 2016 and fully implemented nationally by 2019, was the structural reform that enabled the current biotech startup ecosystem. Before MAH, a marketing authorization was inseparable from a manufacturing license. A research-stage company without its own GMP-certified facility could not hold an NDA. The MAH system decoupled these two functions, allowing a biotech to develop a drug, hold the marketing authorization, and contract manufacturing to a specialized CDMO. This single reform lowered the capital requirement for clinical-stage biotech formation by removing the need to build or acquire manufacturing assets before reaching commercialization. The downstream effect is visible in the pipeline density: China went from a handful of IND-stage startups in 2016 to hundreds by 2024, all operating on a CRO/CDMO outsourcing model enabled by MAH.
1.3 IP Valuation as a Core Strategic Asset: Patent Portfolio Construction and the China Patent Linkage System
In China’s restructured biopharma capital markets, a company’s patent portfolio is not a defensive legal instrument. It is the primary source of enterprise valuation for pre-revenue biotechs and the central negotiating leverage in out-licensing transactions. Understanding how Chinese companies build, protect, and monetize their patent estates is essential for any analyst modeling deal risk or intrinsic value.
China’s patent linkage system, introduced under the 2021 Patent Law amendments and modeled on the US Hatch-Waxman framework, is the mechanism through which innovator patents interact with generic drug approvals. When a generic company files an application with the NMPA for a drug listed on the patent registry, the innovator has 45 days to challenge the application. A successful challenge triggers a nine-month stay on the generic approval. This stay mechanism gives innovators a procedural tool to defend their market exclusivity. For IP teams tracking Chinese assets, the quality of a compound’s patent linkage registry listing — the breadth of the claims, the number of listed patents, and the expiry dates of each — is a direct input into the commercial exclusivity timeline and, consequently, the net present value of any licensing deal.
Chinese pharma companies now file patents across multiple dimensions of a single drug’s intellectual property: the compound patent itself, formulation patents covering specific salt forms and delivery vehicles, manufacturing process patents protecting proprietary synthesis routes, and secondary patents on dosing regimens and new indications. This layered approach constitutes the evergreening toolkit available under Chinese patent law, and it mirrors the strategy long deployed by Western innovators. Jiangsu Hengrui’s selpercatinib analog programs, for example, are protected by compound patents with primary expiry dates, supplemented by formulation and process patents that extend effective commercial exclusivity by three to five years. Analysts pricing a Hengrui asset in a licensing context must map this full patent stack, not only the lead compound patent, to accurately model the exclusivity window that the licensing partner is acquiring.
| Year | Reform | Mechanism | IP / Commercial Implication |
|---|---|---|---|
| 2015 | Backlog clearance program | CDE staff expansion from 150 to 700+ | Reduced queue from 20,000 applications; cleared path for accelerated pathways to function |
| 2016 | MAH system pilot | Decoupled marketing authorization from manufacturing license | Enabled asset-light biotech formation; patent holder now the commercial anchor, not the factory |
| 2017 | ICH accession | Acceptance of foreign clinical data; Chinese sites eligible for global MRCTs | Single trial design satisfies NMPA and FDA; compresses timeline to globally licensable data |
| 2018 | Priority Review expansion | 77% of NDAs granted Priority by 2019, up from 14% in 2016 | Effective patent life at approval extends when approval clock shortens; higher NPV for assets |
| 2019 | MAH national rollout | Full national implementation across all provinces | Standardized asset-transfer mechanism; enabled clean IP transfer in licensing deals |
| 2021 | Patent Law 4th Amendment | Patent linkage system; punitive damages up to 5x; shifted burden of proof for damages | Stronger deterrent against infringement; linkage registry becomes key due diligence document |
| 2025 | Proposed IND review reduction | 60 working days to 30 working days for eligible innovative drug clinical trial applications | Direct FDA parity; reduces IP clock erosion during pre-clinical to IND phase |
The 2021 Patent Law amendment introduced punitive damages of up to five times the compensatory amount for willful patent infringement, a meaningful escalation from the prior regime. It also raised the cap on statutory damages and, critically for pharmaceutical cases, shifted the burden of proof for damages calculation in cases where evidence is held by the defendant. These changes upgraded the practical deterrent value of a Chinese patent against generic or biosimilar entry. For an MNC evaluating a Chinese asset as the target of a licensing deal, the enforceability of the innovator’s patent estate is a direct input into the exclusivity premium embedded in the deal price. A portfolio protected by broad compound claims with high linkage registry coverage, filed before the priority date of any known competitor program, commands a materially higher licensing premium than a narrowly claimed portfolio with generic challenge vulnerability.
1.4 The Capital Architecture: HKEX Chapter 18A, the STAR Market, and the Biotech Valuation Equation
China’s biotech capital market restructuring between 2018 and 2021 created the financing infrastructure that the ecosystem required to scale. The Hong Kong Stock Exchange’s 2018 amendment to Chapter 18A of its listing rules allowed pre-revenue biotechnology companies to list on the basis of their pipeline and IP assets alone, without meeting the standard revenue or profitability thresholds. Shanghai’s STAR Market, launched in 2019, applied a similar framework. These two changes gave Chinese biotechs access to deep public equity markets at a stage where their primary asset was a patent-protected clinical pipeline, not a revenue stream.
The financial result was rapid and concentrated. The combined market value of Chinese pharmaceutical innovation companies listed on Nasdaq, HKEX, and the STAR Market grew from $3 billion in 2016 to over $380 billion by July 2021. In 2020 alone, HKEX and the STAR Market hosted 21 of 23 Chinese biotech IPOs. The pipeline assets underlying these listings were valued on a probability-weighted NPV basis, where the patent expiry date of the lead compound, the breadth of the composition-of-matter claims, and the scope of the linkage registry coverage were direct inputs into the analyst model. This means that CNIPA filing data is now a market-moving dataset for any analyst tracking Chinese biotech equities.
The private funding environment reinforced this valuation framework. Abogen Biosciences raised $700 million in a Series C in 2021, one of the largest private biotech financings on record, with the valuation anchored to its proprietary mRNA platform IP and its manufacturing process patents on lipid nanoparticle delivery systems. Avistone Biotechnology secured over $200 million in its Series A and a further $140 million in its Series B on the basis of its oncology pipeline patent portfolio. These are not venture capital anomalies; they reflect a systematic investor consensus that proprietary platform IP is the primary source of value in the Chinese biotech sector, and that the NMPA’s accelerated pathways translate directly into a longer effective commercial exclusivity window for the most valuable assets.
Key Takeaways — Section 1
- The NMPA’s reduction of IND review times from 60 to 30 working days — now matching the FDA’s standard — has a direct effect on the effective patent life of Chinese drug assets at approval. Every month shaved from the regulatory clock is a month of protected commercial revenue added to the patent estate’s NPV.
- The MAH system is the structural prerequisite for China’s asset-light biotech model. It made a company’s patent portfolio, rather than its manufacturing footprint, the core valuation anchor — a shift that aligned Chinese biotech capital market logic with that of US and EU biotech markets.
- China’s patent linkage registry, introduced in 2021, is now a primary due diligence document for any licensing deal. The number of listed patents, the breadth of their claims, and their expiry dates define the commercial exclusivity window being priced into deal terms.
- The HKEX Chapter 18A and STAR Market frameworks created a public equity market that prices Chinese biotech companies on pipeline IP value. CNIPA filing activity is therefore a leading indicator for secondary equity market movements, not just a legal record.
- ICH membership transformed the Chinese Phase I/II trial from a domestic regulatory formality into a globally valid data asset. A successful Phase II readout from a Chinese site now carries the same weight in an FDA submission as data generated in the US or EU — a change that fundamentally alters the deal-making calculus for MNC business development teams.
IP Team Guidance: Mapping Chinese Patent Estates for Deal Valuation
Before entering due diligence on any Chinese biotech asset, IP teams should construct a complete patent stack: the lead composition-of-matter patent (CNIPA filing date, priority date, and projected expiry including any term adjustment), all formulation and process patents listed in the NMPA linkage registry, and any pending PCT applications that could generate US or EU counterpart protection. The gap between the CNIPA priority date and the projected approval date determines the remaining exclusivity window, which is the central variable in deal NPV modeling.
Investor Signal: Reading the Regulatory Clock as a Patent Clock
For institutional investors tracking Chinese biotech equities, the practical analytical tool is to monitor CDE Priority Review grant announcements alongside CNIPA publication data for the same compound. A Priority Review grant that reduces the approval timeline from 200 to 130 working days adds roughly three to four months to the drug’s commercial exclusivity window. For a drug projecting $500 million in peak annual China sales, that temporal extension is worth $125 million to $170 million in additional NPV at a 10% discount rate. This calculation should be a standard line in any Chinese biotech valuation model.
Opportunities in a Transformed Market: Domestic Scale, License-Out Economics, and Platform IP
China’s pharmaceutical market is the world’s second largest, and its growth is driven by demographic pressures that are not cyclical. The country records approximately 5 million new cancer cases annually — roughly a quarter of the global total. Its diabetic population exceeds 96 million patients. The population aged over 65 is growing faster than the healthcare infrastructure can absorb. These are structural demand conditions that guarantee sustained commercial opportunity for any drug developer with meaningful clinical efficacy in oncology, metabolic disease, or cardiovascular indications. For IP analysts and investors, the question is not whether the demand exists. The question is which IP positions are defensible enough to capture it.
2.1 The Domestic Demand Dynamo: Market Scale, Disease Burden, and the NRDL Access Equation
The China pharmaceutical market is projected to grow at a CAGR of 7.5% between 2024 and 2032. That growth rate, sustained over eight years across a market already measured in hundreds of billions of dollars, generates a cumulative revenue expansion that makes China the single largest organic growth opportunity in global pharma. The market’s structure, however, imposes a specific commercial logic. Access to the majority of the patient population runs through the National Reimbursement Drug List (NRDL). Without NRDL inclusion, a drug is limited to the private-pay market at premium pricing — a segment that covers roughly 10% to 15% of the commercially viable patient population. With NRDL inclusion, the drug reaches the full insured population, but only at the price negotiated with the National Healthcare Security Administration (NHSA), which historically imposes cuts of 30% to 70% relative to the launch price.
For IP teams, this market access equation reframes what “value” means in a Chinese patent estate. The commercial exclusivity window protected by a patent is not the period during which the innovator can maximize price. In the NRDL system, price is administratively determined at NRDL entry and renegotiated at each subsequent annual NRDL review. What patent exclusivity actually protects is the period during which no biosimilar or generic can enter the NRDL tender process and undercut the innovator’s volume. The relevant strategic calculation is therefore the projected date of biosimilar or generic NMPA approval relative to the patent expiry date — and the linkage registry stay mechanism is the tool that creates the gap between those two dates.
Oncology accounts for 62% of China’s R&D pipeline, a concentration that directly reflects the disease burden figures. China’s global share of new cancer diagnoses runs at approximately 25%, a patient pool that creates deep Phase II and Phase III recruitment capacity for oncology developers. The government’s “Healthy China 2030” targets for reducing cancer mortality by 2030 have translated into specific policy incentives: accelerated NMPA review pathways for oncology drugs, a dedicated NRDL negotiation track for anticancer medicines, and preferential hospital formulary access for NMPA-approved cancer therapies. For any company with an oncology asset targeting a high-prevalence Chinese indication — lung cancer, gastric cancer, hepatocellular carcinoma — these structural incentives compress both the clinical development timeline and the time-to-reimbursement.
| Indication | Patient Population | Global Share | NMPA Accelerated Pathway | NRDL Status / Negotiation Dynamics |
|---|---|---|---|---|
| Non-small cell lung cancer | ~900,000 new cases/yr | ~37% of global NSCLC | Priority Review; Breakthrough for novel targets | EGFR/ALK inhibitors fully listed; PD-1 agents listed with 40–60% cuts; bispecifics in negotiation |
| Hepatocellular carcinoma | ~400,000 new cases/yr | ~50% of global HCC | Priority Review standard for all HCC submissions | Sorafenib/lenvatinib listed; atezolizumab/bevacizumab combination listed 2023 |
| Type 2 diabetes | >96 million patients | ~25% of global T2D | Standard Review; GLP-1 agonists under enhanced scrutiny | GLP-1 market contested: Novo Nordisk accepted 50%+ cuts; domestic analogs entering NRDL tender |
| Gastric cancer | ~500,000 new cases/yr | ~44% of global gastric cancer | Priority Review for HER2+ and MSI-H indications | Trastuzumab listed; CLDN18.2-targeting agents in Phase III and NRDL pipeline |
| B-cell lymphoma / CLL | ~100,000 new cases/yr | Growing; historically undercounted | Breakthrough for BTK inhibitor follow-ons; novel mechanisms Priority | BeiGene’s zanubrutinib listed domestically; ibrutinib generics in VBP pipeline |
2.2 The License-Out Boom: Deal Economics, IP Valuation Mechanics, and the Global Pipeline Restock
The reversal of the innovation flow from China outward to global markets is now the defining commercial narrative in biopharma dealmaking. For most of the prior two decades, the standard China model was “In China, For China”: multinationals licensed their approved global drugs into the Chinese market, negotiating local development or co-promotion rights. The current market runs on the inverse logic. Chinese biotechs generate Phase I and Phase II proof-of-concept data on novel mechanisms, then license global rights to MNCs that need to fill patent cliff-driven pipeline gaps. The $46 billion in 2024 out-licensing deal value is not a temporary anomaly. It is a market in equilibrium, serving both sides of a structural need.
The supply side of this market is Chinese biotechs with regulatory speed and cost-efficient clinical operations. The demand side is Western MNCs facing patent expirations on blockbuster drugs without sufficient internal pipeline to replace the revenue. AbbVie faces the post-Humira revenue gap. Bristol Myers Squibb faces the post-Revlimid and post-Eliquis cliff. Pfizer, Merck, and Johnson & Johnson each have large-molecule biologics approaching loss of exclusivity between 2025 and 2030. The internal R&D productivity of these companies has not kept pace with the scale of the replacement requirement. Chinese biotechs, delivering de-risked clinical assets with Phase I/II human data at a fraction of the cost of equivalent Western development, are the most efficient solution available.
The deal structures in this market have become increasingly sophisticated. Early deals were straightforward: an upfront payment for ex-China rights, a royalty stream on net sales, and milestone payments tied to clinical and regulatory events. The current generation of deals embeds more complex IP-sharing provisions. Many transactions grant the Chinese biotech co-development rights in specified territories (typically Asia-Pacific excluding China), revenue sharing in the co-development territories, and rights of first negotiation for next-generation assets in the same platform. The IP-sharing dimension is the part of the term sheet that most requires specialist due diligence. When a Chinese biotech grants “ex-China global rights” to an MNC licensee, the underlying question for both parties’ IP teams is the completeness of the Chinese patent protection that is being transferred. A gap in the CNIPA registration — an unprotected crystalline polymorph, a missing formulation claim for the specific delivery format the licensee intends to commercialize — can reopen the door to generic challenge in markets where the MNC does not hold independent filings.
Novartis licensed ex-China commercialization rights. FDA-approved second-generation BTK inhibitor. Compound patent expiry 2034 in the US. Brukinsa is the strongest evidence that a Chinese-developed small molecule can achieve global best-in-class designation — it outperformed ibrutinib on PFS in a head-to-head CLL trial.
The IP valuation mechanics that drive these deal prices follow a consistent framework. Analysts and business development teams on both sides of the transaction model the net present value of the protected revenue stream in the licensed territories, probability-weighted by phase of clinical development. The key variables are the strength of composition-of-matter claim breadth (whether the patent covers the active moiety broadly or only a narrow salt/polymorph), the expiry date adjusted for any potential term extension, the geographic coverage of the patent family (CNIPA plus PCT counterparts in the US, EU, and Japan at minimum), and the vulnerability to Paragraph IV challenge or equivalent generic opposition in each licensed territory. A Chinese bispecific antibody with broad composition-of-matter coverage in all major markets, validated Phase II data in a high-prevalence indication, and no identified composition challenge trajectory commands a deal premium of 1.5 to 2x the NPV of a comparable asset with narrow claims or pending patent office rejections in the US or EU.
2.3 Domestic Champions and Their IP Portfolios: Hengrui, CSPC, BeiGene, and Akeso
Jiangsu Hengrui Pharmaceuticals — The Vertically Integrated Innovator
Hengrui began as a generics manufacturer and executed a decade-long pivot to become China’s highest-revenue innovative pharma company. Its R&D spend reached RMB 6.15 billion in 2023, representing 26% of total revenue — a ratio that exceeds the R&D intensity of many Western mid-cap pharma companies. The company received approval for three Class 1 innovative drugs and four Class 2 new drugs in 2023 alone, building out its patent estate across oncology, cardiovascular disease, and CNS indications.
Hengrui’s IP strategy follows a layered filing model. Its lead compound patents are supplemented by formulation patents covering the specific crystalline forms used in its approved products, manufacturing process patents for the synthesis routes that achieve GMP-compliant yield at commercial scale, and dosing regimen patents protecting combination therapy protocols. This multi-layer structure is designed to extend effective commercial exclusivity three to five years beyond the primary compound patent expiry through legitimate secondary patent protection — the standard evergreening approach, executed at high volume across a broad therapeutic portfolio. The company’s GLP-1 portfolio out-licensing at a total potential value of $6 billion reflects both the commercial scale of the obesity and diabetes markets and the perceived strength of its CNIPA and PCT patent coverage in those territories.
CSPC Pharmaceutical Group — The FDA Registration Pioneer
CSPC’s strategic differentiation is its pursuit of US FDA approval for domestically developed compounds, a path most Chinese companies have delegated to licensing partners. Xuanning, its antihypertensive small molecule, received full FDA approval — the first novel small molecule developed entirely by a Chinese company to clear that bar. This regulatory achievement is commercially significant beyond the single product: it demonstrates that CSPC’s internal chemistry, manufacturing, and controls (CMC) capability meets FDA standards without a Western manufacturing partner, which expands the scope of IP it can independently protect in the US market.
CSPC’s pipeline of over 130 innovative drug projects generates a patent filing volume that makes it one of the most active CNIPA filers among Chinese innovative pharma companies. The breadth of the pipeline is the company’s primary strategic asset: rather than concentrating IP in a narrow therapeutic focus, CSPC is building diversified coverage across oncology, CNS, and cardiovascular indications. This diversification reduces the risk that VBP or NRDL pricing pressure in any single area will materially impair the overall portfolio value.
BeiGene — The Global-First IP Architecture
BeiGene is the clearest example of a Chinese company that built its IP architecture for global registration from the outset, rather than designing for the Chinese market and licensing outward post-approval. Zanubrutinib (Brukinsa) holds US, EU, and Chinese approvals for multiple B-cell malignancy indications. Its US compound patent runs through 2034, and its PCT filing strategy generated counterparts in the EU and Japan with equivalent coverage. The drug’s head-to-head superiority over ibrutinib in a randomized CLL trial is reflected in the strength of the patent estate’s commercial position: a patent that protects a clinically superior product commands a longer effective market exclusivity period than its nominal expiry date implies, because physicians and payers resist switching to a generic that has never been tested head-to-head against the molecule they know.
BeiGene’s Novartis out-licensing deal for ex-China Brukinsa rights at over $650 million upfront was priced in part on this clinical differentiation premium embedded in the patent estate. For IP teams modeling comparable Chinese biotech assets, BeiGene’s deal structure is the benchmark: upfront payment reflecting current patent estate value, milestone payments tied to regulatory approvals in each territory, and royalty tiers calibrated to net sales thresholds that account for the probability of generic entry after the primary patent expiry.
2.4 Technology Platform IP Roadmaps: ADCs, Bispecific Antibodies, and mRNA
Three technology platforms account for the majority of high-value out-licensing deal activity in Chinese biopharma: antibody-drug conjugates, bispecific antibodies, and messenger RNA therapeutics. Each platform has a distinct IP architecture, a different stage of global regulatory validation, and a different risk profile for licensing partners. IP teams need a working model of each.
Technology Roadmap: Antibody-Drug Conjugate (ADC) IP Architecture
China’s ADC ecosystem has developed faster than any other therapeutic modality in the country’s recent history. Chinese companies have filed more than 400 ADC-related patent applications with CNIPA in the past five years, covering linker chemistry, payload selection, antibody scaffold composition, and conjugation site engineering. The IP landscape in ADCs is dense and complex, because the core composition-of-matter patent covers not just the antibody but the complete conjugate structure — the antibody target specificity, the linker’s cleavage mechanism, and the payload’s cytotoxic mechanism must all be claimed together to achieve full protection. A patent that claims only the antibody component without the linker-payload system is vulnerable to design-around by competitors using alternative linker chemistry with the same payload class.
Composition-of-matter patents on the antibody moiety, including epitope specificity, CDR sequences, and any engineered Fc modifications. This is the broadest and most defensible layer; the hardest to design around.
Proprietary cleavable or non-cleavable linker chemistry. Cleavable linkers (cathepsin-sensitive, pH-sensitive) are the current standard; novel cleavage mechanisms represent the highest-value secondary patent layer, blocking competitor use of the same payload delivery mechanism.
Platform IP Layer 3
Payload Patents
MMAE, DM1, DXd, and SN-38 are the most commonly deployed payloads; most are off-patent or licensed. Novel payloads — topoisomerase I inhibitors with improved tolerability profiles — represent a new patent opportunity layer being pursued by Daiichi-licensed Chinese programs.
Platform IP Layer 4
Manufacturing Process Patents
Proprietary conjugation site engineering (site-specific vs. random conjugation) and drug-antibody ratio (DAR) control processes. These patents protect the manufacturing advantage that produces ADCs with defined homogeneity — a key differentiator in regulatory submissions and a barrier to biosimilar replication.
The PD-1 x VEGF bispecific antibody category has attracted the most licensing deal value among Chinese bispecific programs. Akeso’s ivonescimab, licensed to Summit Therapeutics for global ex-China rights at over $5 billion in total potential value, is the defining transaction. The bispecific’s composition-of-matter patent covers the dual-variable domain structure that simultaneously engages PD-1 and VEGF, blocking both the immunological checkpoint and the angiogenic signal. This dual-mechanism design is the core IP novelty: blocking PD-1 alone (as pembrolizumab does) leaves the tumor’s vascular escape route open. Blocking both pathways simultaneously — in a single molecule — is the clinical and patent hypothesis. The Phase III head-to-head against pembrolizumab in NSCLC is the clinical test of that hypothesis, and its outcome will either validate or deflate the bispecific antibody class’s premium valuation in the license-out market.
The mRNA platform IP landscape is dominated by manufacturing process and delivery system patents rather than sequence-level protection. Therapeutic mRNA sequences for a given protein target are relatively straightforward to redesign around a composition-of-matter claim. The durable competitive moat in mRNA is in the lipid nanoparticle (LNP) delivery system: the specific lipid formulations, ionizable lipid chemistry, and particle size distribution methods that determine biodistribution, transfection efficiency, and tolerability. Abogen Biosciences, which raised $700 million in its Series C round, has built its valuation on a proprietary LNP platform with CNIPA filings covering its ionizable lipid structures and its formulation process. For licensing partners evaluating an mRNA asset from a Chinese company, the operative due diligence question is not the mRNA sequence — it is whether the LNP delivery system is independently patented, whether those LNP patents avoid Freedom to Operate (FTO) conflicts with the Moderna and BioNTech/Acuitas portfolios, and whether the manufacturing scale-up process is covered by process patents that would prevent a contract manufacturer from replicating the formulation without a license.
Key Takeaways — Section 2
- The $46 billion in 2024 China out-licensing deal value reflects a structural market equilibrium: Western MNCs need pipeline assets to fill patent cliff-driven revenue gaps, and Chinese biotechs have the clinical development speed and cost efficiency to deliver de-risked Phase I/II assets faster and cheaper than internal Western programs can.
- Deal pricing in the license-out market follows a consistent NPV framework. The key variables are composition-of-matter claim breadth, patent family geographic coverage (CNIPA plus PCT counterparts in US, EU, Japan), remaining exclusivity window, and vulnerability to Paragraph IV or equivalent challenge. IP teams must model all four variables to arrive at a defensible deal premium estimate.
- Hengrui, CSPC, BeiGene, and Akeso each represent a distinct IP portfolio architecture. Hengrui is a vertically integrated multi-layer filer; CSPC is a diversified high-volume pipeline builder; BeiGene is a global-first architect with simultaneous US/EU/China registration; Akeso is a bispecific platform specialist. Licensing deal structures should be modeled to the specific architecture of the counterparty, not to a generic Chinese biotech template.
- ADC IP protection requires a four-layer patent stack covering the antibody scaffold, linker chemistry, payload, and manufacturing process. A deal that acquires only composition-of-matter rights on the antibody moiety is materially less defensible than one that includes proprietary linker and site-specific conjugation process patents.
- In the mRNA space, the LNP delivery system patents — not the mRNA sequence itself — are the primary source of durable competitive protection. FTO analysis against the Moderna and BioNTech/Acuitas LNP patent families is the mandatory first step in any mRNA licensing due diligence, regardless of the Chinese licensor’s stated novelty claims.
Section 3: Navigating the Headwinds: Critical Obstacles and Systemic Risks
Despite the immense opportunities, China’s pharmaceutical innovation landscape is fraught with significant obstacles and systemic risks. The very policies designed to catalyze innovation have simultaneously created a high-pressure, commercially challenging environment. Navigating this landscape requires a clear-eyed understanding of the formidable headwinds, which range from severe pricing pressures and a persistent gap between research and commercialization to intellectual property uncertainties and intense market competition. These challenges temper the optimistic narrative and demand sophisticated, risk-aware strategies from all market participants.
3.1 The Price-Volume Paradox: The Squeeze of VBP and NRDL
A central paradox of China’s pharmaceutical market is that the government is simultaneously encouraging high-risk, high-cost innovation while implementing aggressive cost-containment policies that severely erode the commercial value of those innovations. This tension is primarily manifested through two powerful mechanisms: the national Volume-Based Procurement (VBP) program for off-patent drugs and the National Reimbursement Drug List (NRDL) negotiations for innovative medicines.
The VBP program, initiated in 2018, is a centralized procurement system primarily for generic drugs that have passed a quality consistency evaluation.25 It operates on a “winner-takes-all” or “winner-takes-most” basis, where the company offering the lowest price is guaranteed a massive volume of sales across public medical institutions in a given region.26 This has led to brutal price competition and dramatic price reductions. On average, VBP has driven price cuts of 52-54%, with the price of some drugs plummeting by as much as 96%.26 The policy effectively squeezes the profit margins out of the low-end generic market, creating a powerful incentive for companies to either exit the market or invest in innovation to escape the price war.25
For innovative drugs, the primary hurdle is the NRDL negotiation process. Gaining inclusion on the NRDL is critical for achieving broad market access, as it ensures reimbursement coverage for the vast majority of the population covered by state medical insurance.28 However, this access comes at a steep cost. To be included, manufacturers must agree to substantial, often deep, price cuts. According to a McKinsey survey, pricing pressure from NRDL negotiations is a major concern for 81% of industry participants.4 Even market leaders are not immune; Novo Nordisk, for instance, had to accept price cuts of up to 50% on its insulin portfolio to maintain its market position after inclusion in VBP-like schemes.3
This creates a difficult strategic dilemma for manufacturers of innovative drugs. They can choose to forgo NRDL listing and attempt to sell their product in the small, private-pay market at a premium price, severely limiting patient access and sales volume. Alternatively, they can trade significant margin for volume by accepting a deep price cut to get on the NRDL.28 This price-volume trade-off fundamentally alters the commercial calculus for launching a new drug in China. The government has explicitly linked these two pressures through a policy known as “vacating the cage to change the bird,” where the cost savings generated from VBP for generics are used to create budget space to fund NRDL coverage for new, innovative drugs.25 This system, while effective at controlling overall healthcare spending, creates a high-pressure commercial environment that can make it difficult to recoup the substantial costs of R&D.
| Table 3: The VBP/NRDL Pricing Gauntlet: Summary of Pressures |
| Mechanism |
| Target Drugs |
| Mechanism of Action |
| Observed Price Impact |
| Strategic Implication for Companies |
This pricing system acts as a powerful, double-edged policy tool. On one hand, the VBP system brutally punishes companies that remain in the commoditized generics space, creating a strong “push” factor that forces them to invest in innovation to survive.25 On the other hand, the NRDL system, while rewarding innovation with market access, does so at a price that can make the economics of “me-too” or incremental innovation challenging. The logical strategy to navigate this environment is to develop a drug quickly, get it approved, and maximize revenue in the private-pay market for as long as possible before entering the inevitable NRDL negotiations. This logic inherently favors “fast-follower” or “best-in-class” drugs where the biological target and clinical pathway are already validated, as this is a much faster and less risky path than pursuing a truly novel, first-in-class target. Consequently, the pricing regime, while designed to foster innovation, may inadvertently be creating a “me-too trap,” incentivizing incremental advances over breakthrough science and thus contributing to the R&D-to-commercialization gap for truly novel medicines.
3.2 The R&D-to-Commercialization Chasm: A Gap Between Quantity and Quality
A persistent and fundamental challenge for China’s pharmaceutical sector is the significant gap between its massive R&D inputs and its output of globally recognized, truly innovative products. Despite skyrocketing R&D investment, a world-leading volume of scientific publications, and a surge in patent filings, China’s capacity to consistently translate this activity into first-in-class medicines remains a key weakness.11
A primary cause of this “commercialization chasm” is a structural imbalance in R&D funding. The system is heavily skewed towards later-stage experimental development, which accounted for 83.5% of R&D expenditure in a 2011 analysis, while neglecting foundational basic research, which received only 4.7%.11 This is in stark contrast to the US, where basic research received 19% of funding. This chronic underinvestment in basic science starves the innovation pipeline at its source, limiting the discovery of novel biological targets and mechanisms of action.11 Without a strong foundation in basic research, the ecosystem naturally gravitates towards developing “me-too” or “me-better” drugs that target well-understood pathways, rather than pioneering new ones.
This leads to a phenomenon of intense homogenization and redundant competition. The focus on a limited number of validated targets, such as PD-1 and EGFR in oncology, is significantly more concentrated in China than it is globally.24 While this strategy can be commercially viable in the short term, it reflects a weakness in original discovery and leads to a hyper-competitive landscape where numerous companies are chasing the same patient populations with similar products.12 China’s main strengths currently lie in cost-effective manufacturing, contract research, and generating these “me-too” and “me-better” versions of existing treatments, rather than in originating breakthrough therapies.14
The problem is compounded by systemic issues within academia and talent development. The “SCI-oriented” research assessment system in Chinese universities often incentivizes academics to produce a high quantity of publications rather than focusing on the quality, impact, or industrial relevance of their work.12 This creates a disconnect between academic research and the needs of the pharmaceutical industry. Furthermore, there is a recognized shortage of high-level talent with experience in translational science and clinical research leadership—the very skills needed to bridge the gap between a laboratory discovery and a viable drug candidate.23 The innovation conversion rate in China is estimated to be less than 8% annually, significantly lower than the 50-70% seen in developed countries, highlighting a structural separation between research resources and commercialization capabilities.24
The success of the NMPA’s regulatory reforms has, paradoxically, thrown these deeper weaknesses into sharp relief. In the past, the slow approval system and “drug lag” were often blamed for China’s weak innovation output.12 Now that these administrative bottlenecks have been largely removed, with approval times approaching or even beating Western standards, it has become clear that the true obstacles lie deeper within the ecosystem.8 China has successfully built the “superhighway” of regulatory pathways, but it is now grappling with the more fundamental challenge of producing a sufficient number of truly innovative “vehicles”—first-in-class drug candidates—to run on it. The next phase of China’s innovation journey will require addressing these core issues in basic science, talent development, and academic-industrial linkage, a far more complex and long-term undertaking than reforming administrative processes.
| Table 4: Global Innovation Scorecard: A Comparative Analysis (China vs. US vs. EU) |
| Metric |
| R&D Spend Growth (2010-2022) |
| Public R&D Funding Focus |
| Share of Global Drug Pipeline |
| Share of New Clinical Trials (2022) |
| Venture Capital (VC) Access |
| Innovation Conversion Rate |
3.3 The Intellectual Property Gauntlet: A Double-Edged Sword
China’s intellectual property (IP) environment for pharmaceuticals is a landscape of profound contradictions. On one hand, the country has made substantial and undeniable progress in strengthening its legal framework for patent protection. On the other, persistent challenges in practical enforcement and a perception of systemic bias create a high-risk environment for both foreign and domestic innovators. This duality makes navigating IP in China akin to wielding a double-edged sword: it offers powerful tools for protection but carries significant risks.34
The legislative progress has been significant. The Fourth Amendment to China’s Patent Law, effective in 2021, introduced several measures designed to align the system with international standards and provide stronger recourse for patent holders. These include the introduction of punitive damages of up to five times the compensatory amount for willful infringement, a substantial increase in the cap for statutory damages, and a shift in the burden of proof for damages calculation.34 Furthermore, China has established a patent linkage system, similar to the Hatch-Waxman Act in the US, which allows innovators to challenge generic drug applications before they are approved, triggering a 9-month stay on the generic approval.34 Specialized IP courts have been established in major cities, and data shows that foreign plaintiffs enjoy a high success rate (77%) in infringement cases against Chinese defendants, suggesting that on paper, the system is robust.34
However, these formal improvements are tempered by persistent practical challenges. One of the most significant hurdles is the lack of a US-style discovery process. In Chinese litigation, parties are responsible for collecting their own evidence, which can be exceptionally difficult in complex pharmaceutical cases involving trade secrets and proprietary manufacturing processes.34 This places a heavy and often insurmountable burden of proof on the plaintiff.
Moreover, despite high win rates in major urban courts, concerns about local protectionism and systemic bias remain, particularly in cases involving strategic industries or state-owned enterprises.35 A 2021 EU survey highlighted patent invalidation as a serious problem, with respondents expressing concern that court rulings tend to favor Chinese stakeholders in strategic sectors.35 There is evidence of Chinese courts being used to countersue foreign firms and issue anti-suit injunctions to halt litigation in other jurisdictions, adding another layer of strategic risk.35 For foreign companies, these legal complexities are compounded by language and cultural barriers, making expert local counsel an absolute necessity.34 This creates the “double-edged sword” scenario: the laws provide a strong basis for protection, but the reality of enforcing those rights can be a challenging, costly, and unpredictable gauntlet.34
3.4 The Hyper-Competitive Arena: Fragmentation and Redundancy
The Chinese pharmaceutical market is not only vast but also intensely competitive and highly fragmented. This competition comes not just from the traditional rivalry between MNCs and domestic firms, but also from a crowded field of thousands of local players, many of whom are pursuing similar strategies and targeting the same therapeutic areas.
The market structure is fundamentally different from that of most developed countries. China’s thousands of domestic companies account for approximately 70% of the market, yet the top 10 players control only about 20% of the market share.18 In contrast, in most developed nations, the top 10 companies command roughly half the market.18 This high degree of fragmentation means that no single company has a dominant position, leading to fierce competition for market share.
This landscape is still heavily influenced by the legacy of the generics industry. Generic drugs continue to command a large portion of sales, creating a “race-to-the-bottom” pricing environment that puts constant pressure on margins.18 While VBP is pushing companies towards innovation, the competitive dynamics of the generic market still cast a long shadow over the industry.
The innovation space itself is becoming dangerously crowded. As discussed, the focus on “me-too” and “me-better” drugs has led to severe product homogeneity, particularly in hot therapeutic areas like oncology.14 This results in multiple companies launching similar products targeting the same patient populations and competing for the same limited reimbursement quotas under the NRDL, further intensifying price pressure and making it difficult to achieve a return on investment.
In this environment, MNCs are facing unprecedented pressure. They are no longer just competing with each other but are also being challenged by increasingly sophisticated domestic firms that can offer high-quality alternatives at a fraction of the price. This is particularly evident in the vaccine market, where local manufacturers have developed their own HPV and shingles vaccines, disrupting the premium pricing strategies of global players like MSD and GSK and forcing them to rethink their commercial models in China.37 The government’s clear priority to boost its domestic industry means that the competitive headwinds for foreign firms are likely to intensify.37
3.5 Geopolitical and Cross-Border Complexities
Superimposed on these domestic challenges is a growing layer of geopolitical and cross-border risk. Rising strategic competition between the United States and China, coupled with Beijing’s own increasingly stringent data security regulations, is creating significant new complexities for investment, collaboration, and the conduct of global clinical trials.
From the US side, there is a growing tendency to view biotechnology as a sector of strategic national importance, on par with semiconductors and AI. This has led to increased scrutiny of cross-border deals and investments by bodies like the Committee on Foreign Investment in the United States (CFIUS).16 There is a palpable “decoupling” impulse in some policy circles, which could lead to US companies reducing or severing ties with their Chinese partners and a more protectionist stance towards the industry.16
Simultaneously, China has implemented its own set of strict data security and biosecurity laws. While these regulations provide a competitive advantage to domestic firms, which have unparalleled access to vast pools of Chinese patient data for R&D and AI-driven drug discovery, they create significant hurdles for MNCs.38 The laws place tight controls on the cross-border transfer of human genetic resources and other health-related data, complicating the execution of global clinical trials and the integration of Chinese data into global development programs.29
This dynamic creates a strategic paradox, particularly for the United States. On one hand, national security concerns are driving a policy push to de-risk supply chains and compete with China’s biotech ascent.16 On the other hand, America’s own flagship pharmaceutical companies, facing internal R&D productivity challenges and patent cliffs, are becoming increasingly reliant on the Chinese biotech ecosystem as a vital source of rapid, cost-effective innovation to fill their pipelines.16 This creates a fundamental tension where US policy may seek to contain the very ecosystem that its own industry is increasingly partnering with for its growth and survival.
In response to these dual pressures, some Chinese firms are beginning to hedge their bets by pursuing regional strategies, such as strengthening ties with Southeast Asian nations through frameworks like the Regional Comprehensive Economic Partnership (RCEP). This allows them to build more resilient supply chains for active pharmaceutical ingredients (APIs) and access new growth markets outside the direct glare of US-China competition.38 For all players, the intersection of science, strategy, and security means that a geopolitically realistic and agile approach is no longer optional, but essential for navigating the future.16
Section 4: Strategic Imperatives and Future Outlook
The transformation of China’s pharmaceutical sector into a global innovation player presents a complex matrix of high-stakes opportunities and deeply entrenched systemic risks. Success in this new era requires moving beyond tactical market-entry approaches to crafting sophisticated, resilient, and differentiated strategies that account for the unique dynamics of the ecosystem. The future trajectory of Chinese pharma will be defined by how stakeholders—multinational corporations, domestic biotechs, and global investors—navigate the central tension between state-driven ambition for innovation and state-enforced pressure for cost containment.
4.1 Crafting a Resilient China Strategy: Differentiated Approaches
The one-size-fits-all strategies of the past are no longer viable. Different players in the ecosystem must adopt tailored approaches to capitalize on the opportunities while mitigating the significant risks.
For Multinational Corporations (MNCs): The strategic imperative for MNCs is to evolve from a model of “In China, For China” (selling products to the domestic market) to one of “In China, For Global” (sourcing innovation from the domestic ecosystem for the worldwide market). This requires a fundamental shift in mindset and operations. It involves not just maintaining a commercial presence, but actively establishing on-the-ground incubators, expanding deal-sourcing capabilities, and forging multifaceted partnerships with local biotechs and CROs to tap into the stream of novel assets.9 Commercially, MNCs must adapt their go-to-market models to survive the VBP/NRDL pricing gauntlet. This may involve pivoting away from an over-reliance on top-tier hospitals towards retail and online pharmacy channels, as well as dedicating more resources to penetrating lower-tier cities and rural markets where growth potential remains.29 Operationally, building robust compliance frameworks to navigate China’s complex data security and cybersecurity laws is non-negotiable to manage the risks of cross-border data transfer.29
For Domestic Biotechs: For the majority of Chinese biotechs, the most direct path to significant value creation lies in global out-licensing deals. Success in this arena depends on a relentless focus on generating high-quality clinical data that meets the rigorous standards of global regulators like the FDA and EMA. This requires planning for internationalization early in the development process, aligning with global quality standards (such as USP), and designing clinical trials with global submission in mind.39 For the few companies with the ambition and resources to commercialize independently in China, success will hinge on pipeline differentiation. They must avoid the hyper-competitive “me-too” spaces and focus on developing assets with a clear clinical advantage that can justify their value in the tough NRDL pricing negotiations.
For Investors: The investment landscape demands a more nuanced and sophisticated approach than ever before. The “capital winter” and depressed valuations in recent years may have created a buyer’s market, but the highest-quality assets with truly differentiated science are still being acquired quickly, requiring speed and decisiveness.16 Due diligence must now extend beyond the traditional assessment of scientific, clinical, and commercial risk. A thorough evaluation must also incorporate the unique pressures of the VBP/NRDL system, the strength of a company’s IP portfolio and its defensibility in Chinese courts, and the rising geopolitical risks that could impact cross-border partnerships and future exit opportunities. Creative deal structures, such as the formation of new companies (NewCos) to house specific assets, are emerging as a way to manage risk and provide flexible financing and exit channels.39
4.2 The Future Trajectory: From “Fast Follower” to “Global Leader”?
The evolution of China’s pharmaceutical innovation ecosystem is likely to occur in distinct phases over the next decade, with the central challenge shifting from building capacity to enhancing the quality and originality of its output.
Short-Term (1-3 years): The current trends are expected to continue and intensify. The ecosystem will remain dominated by “best-in-class” and “me-better” innovation, as this model is well-suited to the current incentive structure. The out-licensing boom will likely persist as Chinese biotechs continue to monetize their key advantages in development speed and cost-efficiency for incremental innovation. On the commercial side, pricing pressures from VBP and NRDL will remain a defining feature of the market, forcing continued consolidation and a focus on cost control. Geopolitical tensions will continue to create uncertainty, prompting companies to build more resilient, localized strategies.
Mid-Term (3-5 years): The focus of both government policy and industry strategy will likely begin to shift more concertedly towards addressing the R&D-to-commercialization chasm. We can anticipate a significant increase in state-led investment into basic research and the creation of targeted talent development programs aimed at cultivating translational science expertise.23 During this period, the first wave of assets that were out-licensed in the early 2020s will generate pivotal late-stage clinical data or reach the market. The success or failure of these assets on the global stage will be a critical validation point, heavily influencing future investment trends and strategic partnerships.
Long-Term (5-10 years): China’s ultimate ability to ascend to the status of a true global leader in pharmaceutical innovation, on par with the United States and Europe, will hinge on its success in resolving the fundamental bottlenecks in its scientific ecosystem. The key metric of success will no longer be the quantity of new drug approvals or the value of licensing deals, but the number of truly novel, first-in-class medicines originating from Chinese labs that become global standards of care. This will require a cultural and strategic shift within the industry, moving from an emphasis on rapid implementation to fostering independent, curiosity-driven innovation and long-term strategic thinking.23
4.3 Concluding Analysis: Balancing Ambition with Reality
China has, in less than a decade, successfully engineered a world-class pharmaceutical innovation ecosystem. Through a combination of unwavering political will, sweeping regulatory reform, and immense capital investment, it has transformed itself from the world’s factory for generics into a global force in drug discovery and development that can no longer be ignored.15 Its share of the global R&D pipeline now rivals that of the United States, and its biotech hubs are vibrant centers of scientific activity.17
However, this remarkable achievement is defined by a central, unresolved tension: the state’s powerful ambition to foster world-leading innovation is in direct collision with its equally powerful mandate to control healthcare costs. The result is the “double-edged sword” that defines the market today. The system’s regulatory speed and vast patient pool create unparalleled opportunities to accelerate development. Yet, its aggressive pricing mechanisms can severely diminish the rewards of that innovation. Its strengthened IP laws offer protection, but enforcement remains a complex gauntlet. Its massive scale offers immense market potential, but also breeds hyper-competition and redundancy.
The future of Chinese pharmaceutical innovation will be shaped by the navigation of these paradoxes. The winners—be they multinational corporations, domestic biotechs, or global investors—will be those who can master this complex environment. They must develop strategies that leverage China’s undeniable strengths in speed, scale, and cost-efficiency while insulating themselves from its intense pricing pressures, its systemic weakness in basic research, and its growing geopolitical complexities. China is no longer just a market to sell to or a factory to produce in; it is a critical, complex, and indispensable node in the global biopharmaceutical network, and engaging with it requires a strategy as sophisticated and multifaceted as the ecosystem itself.
Works cited
- Healthy China 2030: China’s healthcare journey | Bayer Global, accessed July 30, 2025, https://www.bayer.com/en/pharma/healthy-china-2030-chinas-healthcare-journey
- China Pharmaceutical Market Estimated to Reach a CAGR of 7.50% during 2024-2032, Impelled by the Rising Geriatric Population – BioSpace, accessed July 30, 2025, https://www.biospace.com/china-pharmaceutical-market-estimated-to-reach-a-cagr-of-7-50-during-2024-2032-impelled-by-the-rising-geriatric-population
- China’s pharmaceutical sector: innovation meets demographic …, accessed July 30, 2025, https://www.imd.org/ibyimd/asian-hub/chinas-pharmaceutical-sector-innovation-meets-demographic-driven-demand/
- The dawn of China biopharma innovation | McKinsey, accessed July 30, 2025, https://www.mckinsey.com/industries/life-sciences/our-insights/the-dawn-of-china-biopharma-innovation
- Five trends of China’s pharmaceutical industry in 2022 – PMC, accessed July 30, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC10326289/
- Bridging the new drug access gap between China and the United States and its related policies, accessed July 30, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC10800674/
- China Proposes Reforms to Optimize Drug Trials – Cisema, accessed July 30, 2025, https://cisema.com/en/innovative-drug-trial-optimization-proposals/
- China proposes shorter clinical trial reviews in efforts to accelerate drug development, accessed July 30, 2025, https://www.fiercebiotech.com/biotech/accelerate-drug-development-china-proposes-shorten-clinical-trial-review-time
- China Market Opportunities for International … – L.E.K. Consulting, accessed July 30, 2025, https://www.lek.com/sites/default/files/PDFs/china-market-opportunities-pharma.pdf
- Bridging the new drug access gap between China and … – Frontiers, accessed July 30, 2025, https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2023.1296737/pdf
- Obstacles and opportunities in Chinese pharmaceutical innovation – ResearchGate, accessed July 30, 2025, https://www.researchgate.net/publication/315631213_Obstacles_and_opportunities_in_Chinese_pharmaceutical_innovation
- Obstacles and opportunities in Chinese pharmaceutical innovation …, accessed July 30, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC5366105/
- 5 private biotech companies in China you should know about – Labiotech.eu, accessed July 30, 2025, https://www.labiotech.eu/best-biotech/china-healthcare-private-biotech-company/
- Lab leader, market ascender: China’s rise in biotechnology | Merics, accessed July 30, 2025, https://merics.org/en/report/lab-leader-market-ascender-chinas-rise-biotechnology
- China, the new epicenter of global pharmaceutical innovation – Servier, accessed July 30, 2025, https://servier.com/en/newsroom/china-global-pharmaceutical-innovation/
- China is Making Large Inroads into Biotech: Is Investment Money …, accessed July 30, 2025, https://pharmaceuticalintelligence.com/2025/07/28/china-is-making-large-inroads-into-biotech-is-investment-money-following/
- The Dragon Awakes: Charting the Unstoppable Growth of Chinese …, accessed July 30, 2025, https://www.drugpatentwatch.com/blog/the-dragon-awakes-charting-the-unstoppable-growth-of-chinese-pharmaceuticals-in-the-global-market/
- Pharmaceutical industry in China – Wikipedia, accessed July 30, 2025, https://en.wikipedia.org/wiki/Pharmaceutical_industry_in_China
- China’s Population Decline: Impact on Business and the Economy – China Briefing, accessed July 30, 2025, https://www.china-briefing.com/news/chinas-demographic-shift-how-population-decline-will-impact-doing-business-in-the-country/
- Integrating artificial intelligence into the modernization of … – Frontiers, accessed July 30, 2025, https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2024.1181183/full
- Integration of TCM, modern medicine in the pipeline – Chinadaily …, accessed July 30, 2025, https://www.chinadaily.com.cn/a/202503/22/WS67ddfec9a310c240449dc3e7.html
- China’s Medicines: A New Frontier in Global Innovation – Linkdood Technologies, accessed July 30, 2025, https://linkdood.com/chinas-medicines-a-new-frontier-in-global-innovation/
- Shaping Future-Oriented R&D Talent Innovation in China: Part 2, accessed July 30, 2025, https://globalforum.diaglobal.org/issue/april-2022/shaping-future-oriented-rd-talent-innovation-in-china-part-2/
- New Drug Approvals in China: An International Comparative …, accessed July 30, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11974572/
- Does China’s national volume-based drug procurement policy promote or hinder pharmaceutical innovation? – PubMed Central, accessed July 30, 2025, https://pmc.ncbi.nlm.nih.gov/articles/PMC11236746/
- Healthy China 2030: Pressure on Prices Under Pooled Procurement – Bayer Global, accessed July 30, 2025, https://www.bayer.com/en/pharma/healthy-china-2030-pressure-prices-under-pooled-procurement
- Pricentric Insights: China Issues New Volume-Based Procurement …, accessed July 30, 2025, https://www.eversana.com/2021/02/02/pricentric-insights-china-issues-new-volume-based-procurement-policy-document/
- 2024 China Drug Pricing Update – Pacific Bridge Medical, accessed July 30, 2025, https://www.pacificbridgemedical.com/publication/2024-china-drug-pricing-update/
- Navigating China’s pharmaceutical industry landscape – PwC, accessed July 30, 2025, https://www.pwc.com/us/en/industries/pharma-life-sciences/china-pharmaceutical-strategy.html
- Obstacles and opportunities in Chinese pharmaceutical innovation – PubMed, accessed July 30, 2025, https://pubmed.ncbi.nlm.nih.gov/28340579/
- A comparative analysis of public R&I funding in the EU, US, and China, accessed July 30, 2025, https://research-and-innovation.ec.europa.eu/document/download/6eaa3b4e-2af3-46fd-87b5-aa92e4ad10ee_en?filename=ec_rtd_comparative-analysis-public-funding.pdf&utm_source=substack&utm_medium=email
- Despite a decade of gradual growth, R&D spending in Europe …, accessed July 30, 2025, https://www.efpia.eu/news-events/the-efpia-view/statements-press-releases/despite-a-decade-of-gradual-growth-rd-spending-in-europe-outpaced-by-the-us-with-increasing-competition-from-china-new-data-shows/
- Biotechnologies: Europe struggles with China-US rivalry – Coface, accessed July 30, 2025, https://www.coface.com/news-economy-and-insights/biotechnologies-europe-struggles-with-china-us-rivalry
- The Double-Edged Sword: Opportunities and Challenges in China’s …, accessed July 30, 2025, https://www.drugpatentwatch.com/blog/the-double-edged-sword-opportunities-and-challenges-in-chinas-patent-litigation-system/
- Tracking China’s Push To Invalidate Foreign Patents | Selendy Gay PLLC, accessed July 30, 2025, https://www.selendygay.com/news/publications/2024-06-20-tracking-chinas-push-to-invalidate-foreign-patents
- Domestic Competition in the Chinese Pharmaceutical Industry – Western University, accessed July 30, 2025, https://www.democracylab.uwo.ca/Archives/2016__2017_research_/pharma_in_china/domestic_competition_in_the_chinese_pharmaceutical_industry.html
- Big pharma faces headwinds in China as vaccine sales decline …, accessed July 30, 2025, https://www.pharmaceutical-technology.com/features/big-pharma-faces-headwinds-in-china-as-vaccine-sales-decline/
- Biopharmaceuticals Rising: China’s Strategic Pivot to Southeast Asia Amid Great Power Tech Competition, accessed July 30, 2025, https://carnegieendowment.org/research/2025/01/biopharmaceuticals-rising-chinas-strategic-pivot-to-southeast-asia-amid-great-power-tech-competition?lang=en
- Accelerating Chinese Innovative Drugs Going Global: International …, accessed July 30, 2025, https://cbiic.phirda.com/portal/article/index/id/896.html