Part 1: What Branded Generics Are (and Why the Distinction Matters)
Defining the Category

A branded generic is a pharmaceutical compound whose active molecule has lost patent protection and whose manufacturer markets it under a proprietary trade name rather than its International Nonproprietary Name (INN). The active ingredient, dosage form, route of administration, and bioequivalence profile are identical to the reference listed drug (RLD). The brand is the differentiator, not the molecule.
This distinction carries real commercial weight. An unbranded atorvastatin competes on price alone. A branded atorvastatin, sold under a proprietary name by a manufacturer with an established quality reputation, can sustain a meaningful price premium over commodity generics in markets where brand equity moves prescribing behavior. That premium is the entire thesis.
The category sits between two poles. Originator brands like Lipitor or Humira carry the full burden of phase I through phase III clinical trials, NDA or BLA submission, and post-marketing pharmacovigilance. Their gross margins can reach 80% to 90%, but the R&D cost to get there averages $1.3 billion per approved drug by most industry estimates. Traditional unbranded generics strip all of that away. They compete on COGS, manufacturing scale, and distributor relationships. Gross margins run closer to 30% to 40%.
Branded generics occupy a calculated middle position. Gross margins average approximately 40%, achieved without the originator’s clinical development expenditure but with marketing spend sufficient to build prescriber and patient trust. The operating leverage is real: a company that can convert an off-patent molecule into a recognized brand name captures the affordability advantage of generics while shedding the commodity pricing dynamic that destroys margins in the unbranded segment.
Key Distinctions Across the Pharmaceutical Spectrum
Understanding where branded generics sit relative to adjacent categories is foundational for IP valuation and portfolio construction.
Traditional generics require ANDA submission in the U.S. and Article 10(1) applications in the EU. Both pathways require bioequivalence demonstration rather than full clinical trial packages. The regulatory rationale: the originator’s clinical data already establishes safety and efficacy for the active molecule. The generic manufacturer’s obligation is to prove their formulation delivers the drug to systemic circulation at the same rate and extent. An ANDA demonstrating AUC and Cmax within 80% to 125% of the RLD satisfies that requirement.
Biosimilars operate under a fundamentally different framework. The complexity of large-molecule biologics, typically monoclonal antibodies, fusion proteins, or glycoproteins produced in living cell systems, means exact replication is chemically impossible. The FDA’s 351(k) pathway and the EMA’s biosimilar guidelines require ‘high similarity’ and ‘no clinically meaningful differences’ in safety, purity, and potency, not bioequivalence. Interchangeability designation, which allows automatic substitution at the pharmacy level without prescriber intervention, requires additional switching study data. The distinction between a biosimilar and an interchangeable biosimilar matters enormously for market uptake, payer contracting, and formulary positioning.
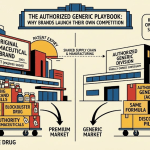
Authorized generics represent a fourth category with direct implications for Paragraph IV litigation strategy. An authorized generic is a version of a brand-name drug that the NDA holder licenses or manufactures itself, often to compete during the 180-day generic exclusivity period granted to the first Paragraph IV filer. AbbVie, Pfizer, and AstraZeneca have all used authorized generic launches to blunt the revenue advantage that first-filers seek to capture. Between 2010 and 2019, authorized generics appeared in roughly 45% of all 180-day exclusivity periods, according to Health Affairs research, materially eroding the first-filer’s period of profit.
Key Takeaways: Category Definition
Branded generics generate margin through brand equity applied to off-patent molecules, not through novel chemistry. The regulatory pathway is the same as unbranded generics. The commercial strategy is categorically different. For IP teams, the implication is clear: the molecule’s patent clock is secondary to the brand architecture built on top of it. For portfolio managers, the distinction between a commodity generic position and a branded generic position in the same therapeutic area can represent a two- to three-turn difference in EBITDA multiple.
Part 2: Market Size, Growth Trajectories, and What the Data Actually Says
Current Market Valuation
The global branded generics market was valued at approximately $261 billion in 2024. That figure comes with a wide range of forward projections depending on methodology and assumed market boundaries. Research and Markets projects $601 billion by 2029 at a 10.9% CAGR. Straits Research puts the 2033 figure at $431 billion on a 5.65% CAGR. GMI Insights estimates $448 billion by 2032 at 6.1%. The spread between these forecasts is wide enough to matter for investment decisions.
The divergence reflects genuine uncertainty about several variables: the pace of patent expirations for high-revenue biologics, the rate of biosimilar penetration, the trajectory of generic drug pricing in the U.S. under the Inflation Reduction Act, and the speed of healthcare infrastructure buildout in South and Southeast Asia. Anyone citing a single market projection as authoritative is being imprecise.
What the data points agree on: North America held approximately 38% of global market share in 2024, driven by insurance coverage dynamics, high per-capita drug spending, and FDA generic drug approval throughput. Asia-Pacific held roughly 30% and is growing faster, driven by India, China, Japan, and rising middle-class healthcare expenditure in Indonesia, Vietnam, and the Philippines.
Primary Growth Drivers
Patent expirations for high-revenue branded drugs are the primary mechanical driver. When a drug loses exclusivity, the entry of a branded generic gives prescribers an alternative that carries quality signals without the originator’s price tag. The scale of upcoming LOE (loss of exclusivity) events through 2030 is substantial. Humira’s principal small-molecule-equivalent exclusivity window has been systematically prolonged through a 100-patent thicket, but key formulations have opened to biosimilar competition since 2023. Merck’s Januvia and Janumet, AstraZeneca’s Symbicort, Bristol Myers Squibb’s Eliquis, and Pfizer’s Ibrance all have exclusivity events in the 2025-2028 window that will generate genericization opportunities.
The chronic disease burden compounds this effect. The WHO attributes 74% of global deaths to non-communicable diseases. The treatment courses for type 2 diabetes, hypertension, heart failure, COPD, and rheumatoid arthritis run for years or decades. A patient who cannot afford consistent access to a branded originator faces direct health consequences. Branded generics at intermediate price points improve the adherence economics for these patients, which is both a public health outcome and a commercial opportunity.
Government-driven generic substitution policies accelerate the market in both high-income and lower-middle-income countries. India’s Jan Aushadhi scheme, Brazil’s Ministry of Health generic preference policies, and EU member state reference pricing systems all create structural demand for generic alternatives, with branded generics frequently capturing the upper tier of that demand.
Key Takeaways: Market Sizing
Do not anchor to a single market size figure. Use the range as a sensitivity band. The variables most likely to compress growth are IRA-driven price negotiation dampening generic entry incentives for certain molecules, and continued REMS abuse by brand manufacturers delaying the generic sample access necessary for bioequivalence studies. The variables most likely to accelerate growth are continued biologic LOE events generating biosimilar opportunities and generic-adjacent product categories, and accelerating healthcare penetration in emerging markets where branded generics often constitute the majority of the pharmaceutical market by prescription volume.
Part 3: The IP Valuation Core: How Patent Architecture Drives Asset Worth
Why Patent Landscape Determines Branded Generic Asset Value
For any branded generic program, IP valuation starts with a systematic mapping of the originator’s patent estate. A molecule with a single composition-of-matter patent expiring in 2026 and no secondary patents is a clean genericization target. A molecule with a composition-of-matter patent expiring in 2024 but a cascade of secondary formulation, polymorph, metabolite, and method-of-use patents running to 2034 is a different problem. The difference between these two scenarios can represent five to eight years of forecasted revenue, which at a standard DCF with a 10% discount rate translates to a 30% to 50% swing in NPV for the asset.
Patent thickets, defined as overlapping clusters of patents on a single drug that collectively extend effective market exclusivity well beyond the original compound patent, are systematically deployed by major originators. AbbVie’s Humira is the most cited example. The compound patent expired in 2016 in the U.S. AbbVie filed more than 165 subsequent patents on formulation, concentration, and manufacturing process, pushing effective exclusivity toward 2034 on multiple dimensions. The result: U.S. biosimilar entry did not occur until January 2023, seven years after the primary patent lapsed, and only after Amgen negotiated a settlement with AbbVie that included a royalty payment in exchange for the settlement license.
Patent Categories and Their Commercial Implications
Branded generic and biosimilar developers must categorize each patent in a target drug’s estate by type, since the commercial and litigation implications differ substantially across categories.
Composition-of-matter patents cover the chemical structure of the active ingredient. They are the foundational IP and the hardest to design around. When they expire, the molecule is genericizable. These patents typically last 20 years from filing date, though effective patent life after regulatory approval averages seven to ten years. Manufacturers frequently apply for Patent Term Extension (PTE) under the Hatch-Waxman Act to restore up to five years of patent life lost during FDA review.
Formulation patents cover the physical form of the drug product: tablet, capsule, extended-release pellet, nasal spray, injectable suspension. These patents can be filed after the composition-of-matter patent and are frequently the mechanism for evergreening. A manufacturer might obtain a formulation patent on an extended-release version of a drug whose immediate-release composition-of-matter patent is expiring, then aggressively market the extended-release version and attempt to migrate patients to it before generic entry on the immediate-release formulation. This is the Nexium strategy from AstraZeneca, which converted Prilosec patients to the S-enantiomer Nexium as the Prilosec patent window closed.
Polymorph patents cover specific crystalline forms of a compound. Since pharmaceutical manufacturing typically requires a specific polymorph for consistent bioavailability and stability, a polymorph patent can block generic entry even after the underlying composition-of-matter patent expires. Generic manufacturers must either demonstrate their formulation uses a different polymorph with equivalent bioavailability or challenge the polymorph patent’s validity.
Method-of-use patents cover the therapeutic application: treating a specific disease indication, using a specific dosing schedule, administering to a specific patient population. These are directly challengeable via Paragraph IV certification if the generic manufacturer’s labeling carves out the patented indication (a ‘skinny label’ strategy). The Caraco v. Novo Nordisk ruling clarified that FDA may require a generic to delete infringing use codes from its labeling at the brand’s request, complicating skinny label strategies.
Process patents cover manufacturing methods. They rarely block generic entry directly because a generic manufacturer typically uses a different synthetic route, but they add litigation complexity and cost to the clearance process.
IP Valuation Framework for Branded Generic Assets
A rigorous IP valuation for a branded generic program requires four analytical layers.
First, freedom-to-operate (FTO) clearance. For each active patent in the Orange Book listing for the target drug, the development team must assess whether the generic formulation infringes and, if so, whether the patent is validity-challengeable. FTO clearance determines the earliest legally viable launch date, which is the foundational variable in any NPV model.
Second, Paragraph IV litigation risk assessment. If the generic manufacturer files an ANDA with a Paragraph IV certification asserting a listed patent is invalid or not infringed, the brand manufacturer has 45 days to file a patent infringement suit. Filing triggers an automatic 30-month stay on FDA approval. The generic team must model the probability of prevailing in litigation, the likely litigation duration (typically 18 to 30 months), and the settlement outcome distribution. First-to-file status confers 180 days of generic market exclusivity, a period that can generate revenues sufficient to fund an entire generic drug development program if the underlying drug is high-volume.
Third, lifecycle positioning of the branded generic relative to the originator’s remaining commercial presence. A branded generic launched at 60% of originator price into a market where the originator still has 40% formulary share captures different economics than one entering a fully genericized market. The brand premium the generic manufacturer can sustain depends on the originator’s continued presence, prescriber inertia, and payer tier assignment.
Fourth, secondary patent filing opportunities. For branded generics that incorporate value-added formulation elements (extended release, fixed-dose combination, novel delivery mechanism), the manufacturer can file its own secondary patents on those improvements. These patents do not extend the underlying molecule’s exclusivity for other generic manufacturers but do protect the specific value-added formulation, creating a proprietary position within the branded generic segment.
Case Study: Teva and the Multi-Jurisdictional Patent Estate for Copaxone
Teva’s Copaxone (glatiramer acetate) for multiple sclerosis is an instructive example of IP lifecycle management in the branded generic context. Glatiramer acetate is a complex mixture of polypeptides, which complicates genericization because it does not fit cleanly into the small-molecule ANDA framework or the large-molecule 351(k) framework. Teva filed a series of patents on the 40 mg/mL formulation and the three-times-weekly dosing regimen as the original 20 mg/mL daily formulation approached patent expiry. Generic manufacturers challenged these secondary patents via Paragraph IV, and Teva pursued litigation in multiple jurisdictions.
The commercial result: by the time Momenta and Sandoz achieved generic entry on the 20 mg/mL formulation, Teva had migrated a substantial portion of the Copaxone patient base to the 40 mg/mL three-times-weekly product, which remained under patent protection. The 40 mg/mL product’s formulation patents and the proprietary autoinjector device created a defensible IP position that temporarily insulated Teva’s market share. The asset’s IP valuation at any point in this cycle depended critically on which formulation’s patent estate the analyst was modeling.
Key Takeaways: IP Valuation
For IP teams: conduct a layered patent landscape analysis before committing to any branded generic development program. FTO clearance on the composition-of-matter patent is necessary but not sufficient. Map all secondary patents, assess Paragraph IV challenge viability, and model the litigation duration distribution. For portfolio managers: the IP estate quality of a branded generic program is more predictive of revenue timing than the molecule’s underlying therapeutic utility. A drug with clear FTO and no secondary patent exposure is worth more at launch than an equivalent drug surrounded by a thicket, even if the therapeutic profile is identical.
Part 4: The Business Case: Margin Structure, Operating Models, and Revenue Mechanics
The Cost Structure Advantage
The economic rationale for branded generics starts with what the manufacturer does not spend. An originator brand program absorbs the full cost of preclinical development, IND filing, phase I through phase III clinical trials, regulatory submission, and post-marketing surveillance. Total costs across a successful program routinely exceed $1 billion. The probability of reaching approval from IND filing is roughly 10% to 15%, meaning the expected cost per approved drug, when losses on failed programs are amortized, is substantially higher.
A branded generic manufacturer avoids all of this. The ANDA pathway requires bioequivalence studies, which typically run $1 million to $5 million depending on the drug’s complexity. The ANDA submission itself costs roughly $300,000 in PDUFA user fees for FY2025. Post-approval chemistry, manufacturing, and controls (CMC) obligations apply, but the clinical development burden simply does not exist. This is why branded generic manufacturers can generate healthy returns at price points 50% to 80% below the originator.
The marketing cost structure also differs. Originator brands deploy medical science liaisons, DTC advertising, speaker bureaus, and patient support programs. A branded generic program relies on a tighter promotional model: targeted pharmacist and physician outreach, formulary tier negotiation with PBMs, and often a brand identity built around quality signaling rather than clinical differentiation messaging. Total promotional expenditure as a percentage of revenue is lower, contributing to margin sustainability.
Pricing Dynamics and Competitive Entry Erosion
The pricing trajectory of a branded generic market follows a predictable pattern that is now well-documented in FDA and HHS data. When the first generic enters an originator market, it typically prices at 70% to 80% of the originator’s WAC. With two to three competitors, prices fall to 40% to 60% of originator pricing. With ten or more competitors, commodity generics frequently price at 10% to 20% of original WAC.
Branded generics resist this erosion more effectively than unbranded generics for two reasons. First, prescribers and patients who have established familiarity with a brand name do not automatically switch to a lower-priced alternative when a new generic enters. Therapeutic inertia is real. Second, formulary positioning matters: payers who place a branded generic on Tier 2 rather than Tier 1 are implicitly creating a price floor by ensuring some patient cost-sharing even at the branded generic’s price. This contrasts with commodity generic positioning, where intense Tier 1 competition drives near-zero patient out-of-pocket cost and maximum price erosion.
The practical implication: branded generic manufacturers with established prescriber relationships can sustain 15% to 25% price premiums over commodity generics for months or years after additional unbranded competitors enter. This premium duration depends on the strength of brand recognition and the therapeutic area’s prescribing complexity. In specialty areas like neurology, psychiatry, or endocrinology, where prescribers have strong product preferences and patient monitoring requirements, brand equity endures longer.
Revenue Contribution by Therapeutic Area
Branded generics generate disproportionate revenue in therapeutic areas where the following conditions apply: long treatment duration, chronic disease burden requiring consistent adherence, high prescriber sensitivity to quality concerns, or complex dosing regimens where brand trust reduces medication errors. Cardiovascular, diabetes, central nervous system, and respiratory therapy areas consistently rank as the highest-value branded generic segments globally.
In emerging markets, the branded generic share of total prescription volume is substantially higher than in mature markets. In India, branded generics represent 70% to 80% of prescription volumes by some estimates, because the regulatory system for decades required brand-level manufacturing and quality oversight. In China, the Volume-Based Procurement (VBP) program has disrupted traditional branded generic economics for centrally tendered drugs, but branded generics outside VBP tender scope retain pricing power.
Key Takeaways: Business Case
The profitability of a branded generic program is not driven by high per-unit margins but by the combination of low development cost, moderate promotional efficiency, and the ability to resist commodity pricing through brand equity. The financial model to construct: ANDA development cost as the primary capital outlay, first-to-market exclusivity period revenue as the peak cash flow window, and the branded premium duration post-exclusivity as the long-tail revenue variable. Programs with strong brand recognition and prescriber loyalty can sustain meaningful cash generation for five to ten years post-exclusivity. Programs without that brand investment typically price-compete into commodity territory within 24 months of losing exclusivity.
Part 5: Evergreening, Pay-for-Delay, and REMS Abuse: The Anti-Competitive Playbook
Evergreening: The Patent Extension Toolkit
Evergreening refers to the systematic use of secondary patent filings to extend effective market exclusivity beyond the expiration of the original composition-of-matter patent. It is not a single tactic but a suite of strategies applied in combination. IP teams at branded generic companies must recognize each mechanism and develop distinct counter-strategies.
Minor molecular modifications involve filing patents on enantiomers, metabolites, or prodrugs of an existing compound. AstraZeneca’s esomeprazole (Nexium) is the S-enantiomer of omeprazole (Prilosec). When omeprazole’s patents expired, AstraZeneca had already obtained independent composition patents on esomeprazole and aggressively marketed it as a newer, improved product. The clinical differentiation was marginal, but the IP differentiation was real: a generic manufacturer had to clear esomeprazole-specific patents, not just demonstrate bioequivalence to the old omeprazole.
Formulation and delivery system patents protect modified-release systems, novel salt forms, specific particle size distributions, or novel dosage forms. A once-daily extended-release formulation of a drug previously given three times daily is a genuine patient convenience improvement, but it also generates a new patent clock independent of the original compound. Pharma companies routinely attempt to migrate patients to these new formulations before the original patent expires, hoping to obsolete the soon-to-be-genericized immediate-release product commercially.
Pediatric exclusivity, granted by FDA under the Best Pharmaceuticals for Children Act (BPCA), adds six months of exclusivity to all existing patent and exclusivity protections when a manufacturer completes requested pediatric studies. It is explicitly a policy incentive to generate pediatric data, but the commercial effect is to delay all generic entry by six months. For a drug generating $1 billion annually, six months of exclusivity is worth $500 million at originator pricing.
New indication patents cover expanded therapeutic uses discovered post-approval. If a drug approved for hypertension is found to reduce cardiovascular mortality in heart failure patients, a new use patent covers that indication. A generic manufacturer targeting the original indication can theoretically use a skinny label carving out the patented heart failure indication, but prescribers frequently prescribe the generic for all indications regardless of label, creating infringement risk.
Pay-for-Delay: Reverse Payment Settlements
Reverse payment settlements, often called pay-for-delay agreements, occur when a brand manufacturer pays a generic manufacturer to abandon a Paragraph IV challenge and delay market entry. The logic: the brand pays the generic manufacturer less than the value of the brand’s retained monopoly profits, while the generic manufacturer receives more than it would have expected to earn from launching early. Both parties are better off than the litigation outcome. Consumers pay higher drug prices for the duration of the delay.
The FTC has consistently opposed these settlements. In FTC v. Actavis (2013), the Supreme Court held that reverse payment settlements are not automatically legal under antitrust law and must be evaluated under the rule of reason. This decision created exposure for existing and future pay-for-delay deals, but it did not categorically prohibit them. Settlements continue, with structuring that makes the value transfer less obviously monetary, including agreements on supply, co-promotion rights, licensing of other products, or litigation cost reimbursement.
For branded generic developers, the Actavis decision is a double-edged constraint. A Paragraph IV filer challenging a major brand’s secondary patents now faces a brand manufacturer less willing to pay a large cash settlement for delay, since that cash payment itself creates antitrust exposure. Settlements are more likely to be structured as licensed early entry agreements at a specific future date, which preserves some certainty of revenue while eliminating the antitrust vulnerability of a straight cash payment.
REMS Abuse: Weaponizing Safety Programs Against Generics
Risk Evaluation and Mitigation Strategy (REMS) programs are FDA-mandated when the agency determines that a drug’s benefits outweigh its risks only if specific safety measures are in place. The most restrictive category, Elements to Assure Safe Use (ETASU), limits dispensing to certified pharmacies or prescribers and may require patient enrollment in monitoring programs.
The abuse mechanism: brand manufacturers claim that shared REMS programs (required by statute for generic entry into REMS-restricted markets) cannot be implemented because of concerns about patient safety, liability, or logistical complexity. Meanwhile, they refuse to supply the drug samples that generic manufacturers need to conduct bioequivalence studies. Without samples, there is no ANDA. Without an ANDA, there is no generic entry. The REMS program becomes a permanent exclusivity mechanism with no patent required.
Celgene’s behavior with thalidomide (Thalomid) and lenalidomide (Revlimid) under the RevASSIST and REMS programs has been extensively scrutinized. Celgene denied sample access to generic manufacturers for years, arguing that the teratogenic risk required tight supply chain control. The FTC and several state attorneys general pursued litigation. Celgene ultimately paid $843 million to settle the FTC case in 2022.
The Creating and Restoring Equal Access to Equivalent Samples (CREATES) Act, enacted in 2019, created a private right of action allowing generic and biosimilar developers to sue brand manufacturers who refuse to provide drug samples necessary for bioequivalence or comparative clinical studies. It is a direct legislative response to REMS abuse. Enforcement activity under CREATES has been modest but building, and it represents the primary legal tool available to branded generic developers facing sample access refusal.
Key Takeaways: Anti-Competitive Practices
For IP teams and legal counsel at branded generic companies: map the originator’s full REMS footprint alongside its patent estate in any target selection analysis. A drug with clean patent FTO but REMS-protected distribution is not a clean target. For portfolio managers: pay-for-delay exposure is now an antitrust risk that brands carry, which paradoxically makes some settlement structures less favorable for generic developers. The CREATES Act creates litigation leverage that did not exist before 2019. Companies with pending ANDA applications facing sample access refusal should evaluate CREATES actions as a tool.
Part 6: Regulatory Pathways: ANDA, EMA, and the 505(b)(2) Bridge
The U.S. ANDA Framework
The Abbreviated New Drug Application process, established by the Hatch-Waxman Act of 1984, is the regulatory foundation for generic drug entry in the United States. It is abbreviated because it relies on FDA’s prior findings of safety and efficacy for the RLD, requiring the generic applicant only to demonstrate that its product is pharmaceutically equivalent and bioequivalent.
Pharmaceutical equivalence requires identical active ingredient, identical route of administration, identical dosage form, and identical strength. Bioequivalence requires that the 90% confidence interval for the ratio of AUC and Cmax between the generic and RLD falls within 80% to 125%. This two-sided confidence interval approach, derived from the 1992 FDA bioequivalence guidance, is now standard internationally.
ANDA review timelines have historically averaged 30 to 40 months from submission to approval, though the 2017 Generic Drug User Fee Amendments (GDUFA II) set targets of 10 months for complete responses to standard original ANDAs and faster timelines for priority review applications. FDA’s Generic Drug Program has made measurable progress toward these targets, approving 1,003 generic drugs in FY2024 alone.
The Orange Book (FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations) is the operative document for ANDA strategy. It lists all patents that the NDA holder has certified cover the approved drug, by active ingredient, formulation, and use. Each listed patent requires a certification from the ANDA applicant: Paragraph I (patent not listed), Paragraph II (patent expired), Paragraph III (applicant will wait for patent expiry), or Paragraph IV (patent invalid, not infringed, or both). Paragraph IV certifications are the mechanism for accelerated entry and the trigger for brand litigation and the 30-month stay.
The EU Framework: EMA Article 10(1) and the Hybrid Application
The European Medicines Agency authorizes generic medicines under Article 10(1) of Directive 2001/83/EC, the same conceptual framework as the ANDA: reliance on the reference medicinal product’s (RMP) established safety and efficacy data. The data exclusivity period is eight years from initial RMP authorization, during which generic applicants cannot rely on the RMP’s preclinical and clinical data. The market exclusivity period extends to ten years (eleven years for new indications granted within eight years of initial authorization), during which approved generic applications cannot be marketed even if the data exclusivity period has passed.
The centralized procedure, managed directly by EMA, covers products authorized across all EU member states with a single marketing authorization. Not all products are eligible for centralized procedure; automatic access applies if the reference product was centrally authorized. For products authorized through national procedures, EMA centralized access for generics requires a demonstration of ‘significant therapeutic, scientific, or technical innovation or an interest to patients at Union level.’ This is a non-trivial bar, and most EU generic applications proceed through mutual recognition or decentralized procedures.
Bioequivalence criteria in the EU align closely with FDA standards: 90% CI within 80% to 125% for AUC and Cmax. For highly variable drugs, EMA guidance allows widened acceptance criteria up to 69.84% to 143.19% under specific conditions including reference-scaling approaches. FDA has adopted similar reference-scaled average bioequivalence approaches for highly variable drugs, reflecting ongoing regulatory convergence between the two major markets.
The 505(b)(2) Strategic Bridge
Section 505(b)(2) of the Federal Food, Drug, and Cosmetic Act is an underutilized pathway that sits between a full NDA and an ANDA. It allows an applicant to rely on FDA’s prior findings of safety and efficacy for a previously approved drug, combined with new applicant-generated data, to support approval of a modified version of that drug.
For branded generics with value-added formulation elements, 505(b)(2) is frequently the correct pathway rather than ANDA. An ANDA requires the generic to be pharmaceutically equivalent to the RLD, meaning identical active ingredient, dosage form, route, and strength. A 505(b)(2) application can support approval of a different dosage form (e.g., an oral solution version of a tablet), a different strength, a modified-release formulation, or a new route of administration, provided the applicant generates the additional clinical or PK data needed to bridge from the RLD’s established safety and efficacy.
The commercial advantage of the 505(b)(2) approval: the modified product is a distinct approved drug with its own NDA number, its own Orange Book listing, and potentially its own patent protection on the formulation innovations. This creates a defensible IP position that an ANDA approval on the base molecule cannot. Noven Pharmaceuticals, Jazz Pharmaceuticals, and Supernus Pharmaceuticals have built significant portfolio value on 505(b)(2) programs, applying novel formulation technology to established molecules and generating proprietary branded products at a fraction of the cost of a de novo NDA.
Key Takeaways: Regulatory Strategy
For R&D leads: pathway selection (ANDA vs. 505(b)(2)) should be a commercial and IP strategy decision, not a regulatory affairs default. If the formulation innovation is substantial enough to generate a defensible patent position and sustain a meaningful price premium, 505(b)(2) is generally the correct pathway even if it requires additional clinical data generation. For IP teams: 505(b)(2) approvals create Orange Book-listable patents that are separately challengeable, adding a new layer of IP complexity to the target landscape for any company subsequently pursuing ANDA entry against the 505(b)(2) product.
Part 7: Leading Players: Portfolio Strategy and Competitive Positioning
Teva Pharmaceutical Industries
Teva remains the world’s largest generic drug manufacturer by revenue, posting $16.5 billion in 2024. Its branded generic portfolio operates across more than 60 countries, with particularly strong positions in the U.S., European, and Israeli markets. Teva’s ‘Pivot to Growth’ strategy, outlined in 2023, shifts R&D investment toward innovative assets in neuroscience, immunology, and immuno-oncology while maintaining its generics base for cash generation.
IP valuation note: Teva’s branded generic assets derive value from two distinct mechanisms. In the U.S., high-value first-to-file ANDA exclusivity periods on major molecules contribute disproportionate cash flow. In international markets, established brand recognition for Teva-labeled products in specific therapeutic categories (particularly CNS and cardiovascular) sustains premiums above commodity generic alternatives. The Copaxone litigation history demonstrates both the opportunity and the risk: the company generated enormous cash flow from Copaxone exclusivity but took multiple impairment charges as competitors successfully challenged the secondary patent estate.
Viatris
Viatris, formed from the 2020 merger of Pfizer’s Upjohn division and Mylan, generated $14.7 billion in 2024. The company’s portfolio spans branded generics, complex generics, and biosimilars across global markets. Viatris has been systematically divesting non-core assets, including its biosimilars portfolio to Biocon Biologics and its OTC business, to focus on complex generics and differentiated branded products.
The Mylan legacy includes an extensive Paragraph IV litigation history, with Mylan having been among the most aggressive first-to-file ANDA filers in U.S. generics history. That institutional capability is an IP asset in its own right. The company’s current pipeline focuses on complex formulations, ophthalmic products, and transdermal delivery systems, all of which are candidate 505(b)(2) programs with proprietary IP positions.
Sandoz
Sandoz, spun off from Novartis in October 2023, is now a pure-play off-patent pharmaceutical company with $10.4 billion in 2024 net sales. Its explicit strategic focus is biosimilars, where it has the largest approved portfolio globally. The 2024 launch of its adalimumab biosimilar in the U.S. (Hyrimoz) was among the most commercially significant biosimilar launches in the market’s history, given the scale of the Humira opportunity.
Sandoz’s biosimilar IP position is complex. The company has invested in biologic manufacturing capabilities including mammalian cell culture, downstream purification, and device partnerships, all of which require proprietary know-how that is not easily replicable. While biologic molecules themselves cannot be patented after the reference product’s exclusivity expires, the manufacturing processes, formulation characteristics, and device integration that Sandoz has developed represent protectable IP distinct from the biologic molecule.
Sun Pharmaceutical Industries
Sun Pharma generated $5.77 billion in 2024 revenues with a model that spans Indian branded generics, U.S. specialty and complex generics, and an emerging specialty pharmaceutical segment targeting dermatology and oncology. The company’s Indian branded generic business represents the archetype of the emerging market branded generic model: strong brand recognition among prescribers, established distribution networks, and pricing power that commodity generics cannot match.
Sun Pharma’s acquisition of Ranbaxy Laboratories in 2015 consolidated a major branded generic portfolio, particularly in emerging markets, though the Ranbaxy legacy also included FDA import alerts and manufacturing consent decrees that have been progressively resolved. The Taro Pharmaceutical subsidiary provides a high-margin dermatology branded generic position in the U.S. and Canada.
Dr. Reddy’s Laboratories
Dr. Reddy’s ($3.4 billion FY2025 revenues) has built value through two parallel strategies: aggressive Paragraph IV filings in the U.S. to capture first-to-file exclusivity revenue, and a differentiated branded generic business in India and other emerging markets. The company’s API manufacturing integration is a cost advantage that both supports its generic drug economics and provides revenue from API supply to third parties.
Key Takeaways: Competitive Positioning
For portfolio managers: the most defensible branded generic positions are those where the manufacturer has both regulatory track record (ANDA approval history, GDUFA compliance, EMA manufacturing inspection outcomes) and brand recognition in specific therapeutic areas or geographies. Companies with strong Indian branded generic franchises (Sun, Dr. Reddy’s, Cipla) have durable competitive advantages in markets where brand trust drives prescribing. In mature markets (U.S., EU), differentiation requires formulation innovation, 505(b)(2) pathways, or biosimilar interchangeability designation rather than brand name alone.
Part 8: The Patient Access Equation: Adherence, Affordability, and Trust
The Affordability Gap and Its Downstream Consequences
Generic drugs, including branded generics, price at 70% to 90% below originator WAC on average after full market entry. This price differential does not exist in a vacuum. For patients managing type 2 diabetes, chronic heart failure, HIV, or schizophrenia, the ability to consistently access medication over years or decades is the determining factor in health outcomes. Adherence studies across chronic disease categories consistently show that cost-related non-adherence reduces medication possession ratios below the 80% threshold associated with adequate chronic disease control.
The cost savings from generic substitution across the U.S. healthcare system from 2009 to 2019 amounted to an estimated $2.2 trillion. Generics account for approximately 90% of U.S. prescription volume but only 13% to 18% of total retail pharmaceutical expenditure. This ratio is the clearest quantitative expression of the value transfer from branded generics to the healthcare system.
For individual patients, the impact is direct. A branded generic cardiovascular drug at $30 per month rather than a brand-name equivalent at $200 per month reduces the probability that a patient skips refills when cash is constrained. Every skipped refill on an antihypertensive or antidiabetic medication represents a measurable increase in downstream risk for myocardial infarction, stroke, or hospitalization. The adherence benefit of affordability is a genuine clinical outcome, not a marketing claim.
The Quality Perception Problem and How Branded Generics Address It
Generic drugs face a persistent perception problem. Studies published in BMJ Open found that 35.6% of general population respondents perceived generics as less effective than branded medications. Among physicians the figure was 28.7%, and among pharmacists 23.6%. These perceptions persist despite bioequivalence regulatory requirements and despite decades of post-market evidence that FDA-approved generics perform equivalently to their reference products.
Branded generics directly address this perception gap by attaching a proprietary name and associated quality signals to an off-patent compound. The prescriber who has years of experience prescribing an originator-branded product for a specific indication is more likely to prescribe a recognizable branded generic from a manufacturer they know than to write for an unbranded generic they cannot visually distinguish from competitors. The pharmacist who stocks a branded generic from a manufacturer with a known quality history faces less patient resistance to substitution than when dispensing an unbranded generic.
This is not irrational behavior. Regulatory bioequivalence establishes that the active ingredient reaches systemic circulation at equivalent rates and extents. It does not govern inactive ingredient quality, tablet coating consistency, or supply chain documentation standards. Patients with drug hypersensitivities may respond differently to different inactive ingredients. In practice, FDA-approved generics are uniformly safe and effective, but the brand name provides a quality signal shortcut that operates independently of regulatory fact. Branded generic manufacturers who understand this perception dynamic build manufacturing quality programs that go beyond regulatory compliance to justify the quality signal their brand name implies.
Key Takeaways: Patient Access
For R&D leads evaluating branded generic program selection: prioritize therapeutic areas where adherence economics are demonstrably poor and where the prescriber-quality trust dynamic is active. CNS, cardiovascular, endocrine, and inflammatory disease categories are the strongest candidates. For corporate communications and access teams: the case for branded generics as a patient access tool is empirically strong. The price premium over unbranded generics must be justified by genuine quality differentiation or brand trust, not merely by marketing spend. Programs that cannot sustain that justification will lose formulary positioning to commodity generics over time.
Part 9: Value-Added Formulations: The ‘Super Generic’ Technology Roadmap
Beyond Bioequivalence: The Value-Added Medicine Category
Value-added medicines (VAMs), sometimes called ‘super generics,’ are pharmaceutical products based on known, off-patent molecules that incorporate formulation or delivery innovations delivering measurable improvements over the existing product. They are distinct from standard branded generics in that the value addition is technical and clinically demonstrable, not merely commercial. The European Federation of Pharmaceutical Industries and Associations (EFPIA) has invested in formal VAM categorization frameworks, recognizing the category’s distinct regulatory and commercial positioning.
The VAM landscape covers several distinct technology platforms. Each platform represents both a differentiation strategy for branded generic developers and a distinct IP architecture with different patent durability characteristics.
Extended and Modified Release Technology
Modified-release formulations extend drug action, reduce dosing frequency, and in some cases improve the tolerability profile by reducing peak plasma concentration-related adverse effects. Osmotic delivery systems (OROS technology), matrix tablet systems, and multi-particulate pellet technologies each produce modified-release profiles through distinct mechanisms, each generating patentable formulation innovations.
The commercial rationale is well-established: once-daily versions of twice-or-three-times-daily drugs improve patient adherence, reduce caregiver burden in institutionalized populations, and generate prescriber preference independent of price. For branded generic developers, a 505(b)(2)-approved once-daily formulation of a drug whose immediate-release formulation is fully genericized occupies a defensible niche with meaningful pricing power.
Examples with IP implications: Supernus Pharmaceuticals built significant value with Oxtellar XR (oxcarbazepine extended release) and Trokendi XR (topiramate extended release), both 505(b)(2) approvals based on proprietary extended-release formulations of established CNS drugs. The formulation patents supporting these products have been subject to Paragraph IV challenges, but the company has successfully litigated or settled most challenges, preserving exclusivity windows that generate revenue disproportionate to the program’s development cost.
Nanoparticle and Nanotechnology Delivery Platforms
Nanoparticle drug delivery transforms the pharmacokinetic and pharmacodynamic profile of established molecules in ways that generate genuine clinical differentiation. Abraxane (nab-paclitaxel, Celgene/BMS) is albumin-nanoparticle-bound paclitaxel that eliminates the Cremophor EL-based vehicle required for conventional paclitaxel, removing the premedication regimen for hypersensitivity prophylaxis and improving tumor uptake. Abraxane achieved FDA approval under 505(b)(2) and generated peak revenues exceeding $1.5 billion annually before biosimilar-adjacent competition developed.
Nanoparticle-formulated candesartan cilexetil, to cite a more recent example from the academic literature, improves bioavailability by approximately 40% over conventional tablet formulations in preclinical and early clinical data, allowing dose reduction and potentially improving tolerability. The nanotechnology platform itself, rather than candesartan as a molecule, is the protectable IP.
For IP teams: nanoparticle delivery patents cover particle size distribution, surface coating characteristics, stabilizer systems, and manufacturing processes. They are not composition-of-matter patents on the active drug but are frequently as durable in litigation because the manufacturing complexity creates a non-obvious inventive step that survives obviousness challenges. A branded generic program built on a proprietary nanoparticle platform can sustain exclusivity well beyond the underlying molecule’s patent cliff.
Fixed-Dose Combinations
Fixed-dose combinations (FDCs) integrate two or more active ingredients into a single dosage form. They improve adherence by reducing pill burden, ensure consistent co-administration ratios where combination therapy is standard of care, and can generate regulatory exclusivity and patent protection on the combination itself even when each component is individually off-patent.
The cardiovascular sector has been particularly active in FDC development. Polypill approaches combining aspirin, a statin, and an antihypertensive target the majority of cardiovascular risk reduction achievable with pharmacotherapy in a single tablet. Several academic and commercial programs have advanced FDC polypills through phase III trials demonstrating superior adherence outcomes compared to equivalent separate medications.
The regulatory pathway for FDC products where both components are individually approved typically proceeds through 505(b)(2), relying on existing individual component NDA data while generating new PK bridging data for the combination. If the combination is a genuinely new mixture, a full 505(b)(1) NDA may be required. For branded generic developers, the patent opportunity is on the specific combination ratio, formulation, and release characteristics, creating IP that neither component’s originator owns.
3D Printing and Precision Dosing
Additive manufacturing (3D printing) in pharmaceutical production enables dose forms that are geometrically impossible by conventional tablet compression. Spritam (levetiracetam), approved by FDA in 2015 and manufactured by Aprecia Pharmaceuticals using ZipDose technology, was the first 3D-printed drug approved in the United States. Its rapidly disintegrating, porous structure enables doses up to 1,000 mg in a single tablet that disperses in seconds with minimal liquid, addressing the specific compliance challenge of high-dose epilepsy treatment in patients with swallowing difficulties.
Amneal Pharmaceuticals’ LYVISPAH (baclofen oral granules) represents a later application of precision manufacturing to pediatric dosing, using granule technology to enable weight-based dose titration in a population that cannot swallow conventional tablets. The formulation patents and manufacturing process IP supporting LYVISPAH are the differentiating assets that justify a branded positioning and premium pricing over compounded baclofen alternatives.
The long-term implication of 3D printing for branded generics: as printer technology matures and regulatory frameworks for pharmaceutical additive manufacturing firm up (FDA issued a guidance document on technical considerations for additive-manufactured devices in 2017, with pharmaceutical-specific guidance expected to follow), the manufacturing process itself becomes a barrier to entry and a source of IP protection distinct from the underlying molecule.
Key Takeaways: Value-Added Formulations
For R&D leads: the most defensible VAM programs are those where the formulation technology creates measurable and clinically meaningful patient benefit, not merely commercial differentiation. FDA and EMA are increasingly attentive to whether 505(b)(2) and hybrid applications add genuine therapeutic value or simply manufacture incremental IP protection. Programs with robust PK data demonstrating bioavailability improvements, tolerability benefits, or adherence advantages are substantially more defensible. For IP teams: the patent durability of VAM IP depends on the obviousness assessment at the time of filing. Nanoparticle and 3D printing platform patents filed early in the technology’s development tend to be broader and harder to invalidate. Programs entering established technology platforms face narrower patent scopes and higher validity challenge risk.
Part 10: Digital Health Integration and Combination Product Strategy
The Combination Product Framework
FDA defines a combination product as one that combines a drug with a device, a biologic with a device, or a drug with a biologic. The regulatory pathway depends on the primary mode of action. If the device component (e.g., a smart autoinjector) primarily drives the therapeutic effect, the product is device-led and regulated primarily by CDRH. If the drug component is primary, the combination is drug-led and regulated primarily by CDER. In practice, most combination products involving branded generics, such as drug-device combinations integrating sensors or connectivity, are drug-led.
The commercial rationale for combination product strategy in branded generics: a drug delivered via a proprietary connected device is not directly substitutable by a generic formulation of the same molecule in a conventional syringe. The device component creates switching costs, generates separate patent protection (device patents, software patents), and enables data collection that supports real-world evidence generation and differentiated patient support programs.
Connected Drug Delivery Devices
Bayer’s Betaconnect autoinjector for Betaseron (interferon beta-1b) in multiple sclerosis is an early-stage prototype of what combination product strategy looks like in practice. The Betaconnect records injection time, date, speed, and patient adherence data, transmitting it to an app and potentially to the treating neurologist. The device improves injection technique, reduces injection site reactions, and generates data that documents patient adherence for payer purposes.
For branded generics specifically, connected delivery devices create durable commercial advantages that outlast the drug’s underlying patent position. A pharmaceutical company that has invested in a proprietary connected autoinjector and built a patient data ecosystem around it has created an asset with commercial value independent of the specific drug molecule. When the molecule’s patents expire, the device and data ecosystem remain proprietary, sustaining brand loyalty and creating barriers to unbranded generic substitution that no generic manufacturer can easily replicate.
AI-Driven Precision Dosing and Real-World Evidence
Smart inhaler platforms for respiratory diseases like COPD and asthma integrate spirometric monitoring with drug delivery, enabling machine learning algorithms to adjust dosing recommendations based on real-time lung function data. A salmeterol-containing inhaler that adjusts its delivery recommendations based on the patient’s daily FEV1 readings is not the same product as a commodity salmeterol inhaler, even if the active molecules are identical. The clinical utility of precision dosing is demonstrable, and it generates real-world evidence data that can support label expansions, formulary negotiations, and comparative effectiveness arguments with payers.
The IP strategy around AI-driven dosing is evolving. Algorithm patents, training data ownership, and data exclusivity are all active areas of pharmaceutical and medtech IP litigation. For branded generic developers entering this space, the core intellectual property is not the drug molecule but the software architecture, the training data set, the device hardware, and the clinical protocols that generate and validate the dosing algorithm. This is a fundamentally different IP strategy from traditional branded generic development, and it requires legal counsel with software patent expertise in addition to pharmaceutical IP specialists.
Digital Pills and the Second-Generation Adherence Model
Digital pills integrate an ingestible sensor (typically a small RFID or electrochemical chip embedded in the tablet) that activates when the drug contacts gastric fluid, transmitting a signal to a wearable patch that records the ingestion event and time. Abilify MyCite (aripiprazole with sensor), approved by FDA in 2017, was the first digital pill approved in the United States.
The adherence data generated by digital pills has direct clinical and regulatory implications. In conditions like schizophrenia, bipolar disorder, or HIV, where medication adherence is both critical and frequently problematic, documented adherence data can support patient monitoring, adjust treatment decisions, and provide payers with evidence of therapy optimization. For institutional investors, the value creation from digital pill integration is not in the drug molecule but in the adherence data infrastructure and the clinical decision support ecosystem built around it.
Key Takeaways: Digital Integration
For corporate strategy and business development: digital health integration transforms a branded generic from a commodity at risk of price erosion into a combination product ecosystem with distinct IP, distinct switching costs, and distinct value propositions for payers and prescribers. The investment required is meaningful, but the commercial durability substantially exceeds what brand marketing alone can achieve. For M&A teams: digital health company acquisitions that bring connected device platforms, adherence monitoring software, or AI-driven dosing algorithms represent ways to create differentiated branded generic assets without molecule-level IP.
Part 11: Biosimilar Interchangeability: Separate Rules, Shared Stakes
The Interchangeability Designation and Its Commercial Implications
A biosimilar is not automatically substitutable at the pharmacy counter without prescriber intervention. Standard biosimilar approval under 351(k) establishes that the product is highly similar to the reference biologic and has no clinically meaningful differences in safety, purity, and potency. This does not permit automatic pharmacy-level substitution in U.S. states with substitution laws. For that, the manufacturer must pursue the additional interchangeability designation.
Interchangeability requires the manufacturer to demonstrate that the biosimilar can be expected to produce the same clinical result as the reference product in any given patient, and, for products administered more than once, that the risk in terms of safety or diminished efficacy from alternating between the biosimilar and the reference product is not greater than the risk of using the reference product alone. In practice, this requires pharmacokinetic switching studies in patients to demonstrate that transitions between the biosimilar and the reference biologic do not introduce clinically meaningful variability.
The commercial value of interchangeability is substantial. A pharmacist in a state with automatic substitution laws can substitute an interchangeable biosimilar for the reference biologic without prescriber intervention, identical to how a pharmacist substitutes a generic for a brand-name small-molecule drug. This bypasses the prescriber inertia and system-level change management that non-interchangeable biosimilar uptake requires, accelerating market penetration and reducing the sales force investment necessary to drive adoption.
As of early 2026, FDA has granted interchangeability designation to a limited but growing set of biosimilars, including Semglee (insulin glargine), Rezvoglar (insulin glargine), Cyltezo (adalimumab), and a small number of others. The pipeline of interchangeability applications is expanding as manufacturers recognize the commercial premium that designation confers.
Biosimilar Market Dynamics and the Adalimumab Precedent
AbbVie’s Humira (adalimumab) achieved peak global revenues of approximately $21 billion. The U.S. biosimilar market for adalimumab opened in January 2023 following AbbVie’s settlement with multiple biosimilar manufacturers that licensed U.S. market entry as early as January 2023 under royalty-bearing agreements. The European market had biosimilar competition from 2018. The pricing dynamics have differed substantially between markets.
In Europe, where single-payer or reference pricing systems enable rapid biosimilar substitution, adalimumab biosimilar penetration has exceeded 80% in some markets. In the U.S., despite approved biosimilars from Sandoz, Amgen, Boehringer Ingelheim, Pfizer, and others, the penetration trajectory has been slower. Payer and PBM contracting dynamics, rebate structures favoring branded Humira, and formulary tier management have all contributed to a slower penetration rate than the European experience suggested.
The Humira biosimilar experience contains critical lessons for branded generic developers operating in the adjacent small-molecule branded generic space. Regulatory approval does not automatically translate to commercial penetration. Formulary positioning, payer contracting, and prescriber switching inertia must be addressed with dedicated commercial investment. Programs without that commercial infrastructure cannot capture the theoretical value of regulatory approval.
Key Takeaways: Biosimilar Strategy
For R&D and regulatory teams: biosimilar interchangeability designation is a commercially valuable regulatory milestone that should be pursued systematically for biologics with significant self-injection patient populations. The switching study investment is modest relative to the commercial return from pharmacy-level automatic substitution. For portfolio managers: biosimilar assets with interchangeability designation command premiums in licensing and acquisition negotiations that non-interchangeable biosimilars do not. Model the interchangeability probability and timeline explicitly in any biosimilar program NPV analysis.
Part 12: Supply Chain Exposure and Quality Risk
API Geographic Concentration and the Tariff Disruption Vector
By 2020, 74% of API manufacturing establishments supplying U.S. generic drug manufacturers were located outside the United States. China and India together account for the majority of this capacity, with China dominant in chemical synthesis of API precursors and India dominant in API finishing and generic finished-dose manufacturing.
This concentration creates a fragile supply chain for branded generics in several scenarios. A quality enforcement action by FDA against a major Indian API manufacturer, such as the Ranbaxy and later Sun Pharma API facility consent decrees, can interrupt supply for multiple branded generic products simultaneously. A trade policy disruption affecting pharmaceutical raw material imports from China, whether tariff-based or export-control-based, propagates upstream through the API supply chain and downstream into generic drug shortages within months.
The COVID-19 pandemic demonstrated this vulnerability in concrete terms. For sterile injectable generics used in hospital ICUs (paralytic agents, sedatives, vasopressors), 90% to 95% of raw materials or API originated from China or India. When COVID-19 disrupted Chinese manufacturing in Q1 2020, U.S. hospitals faced acute shortages of critical drugs within weeks. The FDA’s foreign drug facility inspection program, which requires advance notice and visa applications for foreign inspections, means that compliance monitoring of overseas facilities is fundamentally less frequent and less rigorous than for domestic facilities.
For branded generic manufacturers with premium quality positioning, this supply chain vulnerability is both an operational risk and a competitive opportunity. Companies that invest in domestic or near-shored API supply, or that maintain dual-source API relationships, can sustain supply continuity during disruptions that shut down competitors. That continuity, marketed as a quality and reliability differentiator, supports the branded premium.
The Quality-Price Paradox in Generic Drug Markets
The generic drug market’s intense price competition creates a structural disincentive to invest in quality beyond regulatory compliance. Once FDA approves multiple generics for the same product, they are therapeutically equivalent on label. Payers and pharmacy benefit managers select primarily on price. A generic manufacturer that invests in superior manufacturing quality, tighter impurity controls, and better supply chain documentation cannot recover that investment through pricing, because the commodity market structure prevents price differentiation.
This dynamic explains several of the industry’s quality failures. The 2018 discovery of nitrosamine impurities in valsartan and other sartan ARBs, initially from a Chinese API manufacturer that had changed its synthetic route without proper risk assessment, resulted in widespread recalls across multiple generic manufacturers. The impurity, N-nitrosodimethylamine (NDMA), is a probable human carcinogen. The root cause was a process chemistry change that the manufacturer made to increase yield and reduce cost, which is exactly the behavior a commodity price-competitive market incentivizes.
Branded generic manufacturers can use quality investment as genuine differentiation, but only if they communicate that quality investment to prescribers and payers in ways that justify the price premium. Manufacturers who build reputations for supply continuity, impurity control beyond regulatory minimums, and transparent CMC documentation command prescriber preference that generic commodity manufacturers cannot match. That preference is the commercial justification for the branded premium, not simply the brand name itself.
Key Takeaways: Supply Chain and Quality
For operations and procurement teams: dual-source API qualification and domestic supply chain investment are risk management investments with quantifiable value when modeled against supply interruption scenarios. The probability of a disruption event in any given year is material given the current geopolitical and regulatory environment. For commercial and medical affairs teams: supply continuity and quality documentation are differentiating marketing assets in the branded generic space. They should be part of the prescriber and payer value proposition explicitly, not assumed to be obvious.
Part 13: The IRA Effect: What Medicare Negotiation Does to Generic Incentives
The Medicare Drug Price Negotiation Program
The Inflation Reduction Act of 2022 created the first-ever mechanism for CMS to negotiate prices directly with pharmaceutical manufacturers for high-expenditure single-source Medicare Part D and Part B drugs. The program began with ten drugs for 2026 pricing, expanding to a larger number in subsequent years.
For branded generics and unbranded generics, the IRA’s negotiation program does not directly apply: the statute exempts drugs with approved generic or biosimilar competition from the small-molecule negotiation universe (and uses a biologic-specific framework for large-molecule drugs). But the downstream effects on the generic incentive structure are real and require explicit modeling.
When CMS negotiates a substantially lower price for a branded drug, the effective price differential between the negotiated brand price and the generic price compresses. Generic entry economics depend on that differential: the generic manufacturer captures market share by offering a meaningful price discount to payers, PBMs, and patients relative to the brand. If the brand’s negotiated price narrows the differential, the revenue opportunity for generic entry on that molecule shrinks. In extreme cases, if the negotiated brand price approaches the generic’s manufacturing cost, the economic rationale for generic entry disappears entirely.
This effect will be most pronounced for drugs near the end of their patent exclusivity whose branded prices CMS has aggressively negotiated. A drug negotiated down to 65% of list price in 2026 leaves less room for a generic manufacturer to generate significant revenue at 50% of list price. The practical result: generic manufacturers may defer or deprioritize ANDA development for certain molecules on CMS’s negotiation list, reducing the pipeline of future generic competition.
The Small-Molecule Exclusivity Gap
The IRA’s negotiation timeline differs between small molecules and biologics. For small-molecule drugs, CMS can initiate negotiation after nine years of approval. For biologics, the timeline is thirteen years. This disparity has generated substantial industry pushback, as it reduces the effective commercial exclusivity period for small-molecule drugs relative to biologics and may reduce R&D investment in small-molecule innovation.
For branded generic developers, the IRA’s small-molecule timeline creates a counterintuitive opportunity in specific cases: drugs where CMS negotiation has reduced brand pricing may become less attractive targets for commodity generic entry, but may remain viable targets for branded generic entry at a price point that patients and prescribers prefer over the commodity market. The branded quality premium may sustain revenue in a market where commodity generics struggle to justify ANDA investment.
Key Takeaways: IRA Impact
For corporate strategy and portfolio planning teams: model the IRA negotiation pipeline explicitly when evaluating ANDA development investment decisions. Drugs on CMS’s current or projected negotiation list represent reduced-opportunity generic targets. Divert development resources toward molecules outside the negotiation universe. For government affairs teams: the small-molecule versus biologic exclusivity asymmetry in the IRA is an active policy debate. Engaging CMS and congressional stakeholders on the downstream generic incentive implications of aggressive price negotiation is both legitimate and commercially relevant.
Part 14: Emerging Markets: India, China, and the Branded Generic Default
India: The Branded Generic System as the Default Model
In India, branded generics are not a niche category; they are the pharmaceutical market’s operating default. The Drug Price Control Order (DPCO) system governs pricing for essential medicines, but outside that regulated universe, branded generic manufacturers set their own prices, and brand recognition among prescribers directly determines market share.
Indian pharmaceutical companies have built multi-decade competitive advantages through physician relationship programs, field force density, and therapeutic area brand franchises that operate independently of the molecule’s patent status. Sun Pharma’s Pantocid (pantoprazole), Cipla’s Ciprobid (ciprofloxacin), and Dr. Reddy’s Omez (omeprazole) are examples of branded generics with market share that reflects brand trust rather than patent protection. These brands command premiums over unbranded generics because prescribers recognize them and patients request them.
The Jan Aushadhi program, India’s government-run generic dispensing network, has expanded to over 10,000 outlets and represents a policy push toward unbranded generic use. Its penetration into the broader prescription market remains limited, however, because physician prescribing behavior in India’s predominantly private healthcare system favors brand-name prescriptions. The program primarily serves economically marginalized populations who cannot afford branded alternatives. This structural reality sustains the branded generic premium across the broader Indian market.
China: Volume-Based Procurement and the Branded Generic Disruption
China’s centralized Volume-Based Procurement program, launched nationally in 2019, has fundamentally disrupted the branded generic economics for drugs included in VBP tender rounds. Under VBP, manufacturers bid for hospital-based contracts with winner-takes-most volume guarantees. Winning bids typically price 60% to 95% below pre-VBP branded generic pricing, immediately commoditizing the included drug category within the hospital channel.
For branded generic manufacturers operating in China, VBP inclusion is an existential commercial event for a product. Chinese pharmaceutical companies that built significant revenue on branded generics of now-VBP-tendered molecules have been forced to pivot to retail channels, new markets outside VBP scope, or innovative drug pipelines. The strategic response from international branded generic players has generally been to de-emphasize VBP-affected products and concentrate resources on therapeutic categories not yet subject to VBP expansion.
Outside hospital channels, China’s retail pharmacy and private clinic markets retain more traditional branded generic dynamics, with brand recognition continuing to drive prescribing in categories like dermatology, psychiatric medication, and primary care. The tension between government-directed commodity pricing in the institutional channel and brand-driven dynamics in the retail channel creates a bifurcated China market that requires distinct commercial strategies by channel.
Key Takeaways: Emerging Market Strategy
For international market teams: India’s branded generic market requires field-force capability and physician relationship investment commensurate with the market’s relationship-driven structure. Financial models built on branded generic pricing that do not account for field force cost structures will misstate actual market economics. For China strategy teams: VBP expansion scope is the primary risk variable for any branded generic China asset. Build scenario models that account for VBP inclusion at 12 to 24 months from any new product launch, and size the retail channel opportunity as the primary long-term revenue base for non-hospital branded generic positioning.
Part 15: Sustainable Manufacturing and ESG Positioning
The Environmental Footprint of Generic Drug Manufacturing
Pharmaceutical manufacturing generates significant environmental load, particularly in API production. Chemical synthesis routes for small-molecule APIs frequently use large volumes of organic solvents, strong acids and bases, and heavy metal catalysts. Wastewater from API manufacturing facilities has been documented to contain active pharmaceutical ingredients at concentrations with measurable ecotoxicological effects on aquatic organisms. The problem is most acute in clusters of generic API manufacturing around Hyderabad, India and Taizhou, China, where regulatory oversight of environmental discharge has historically been less stringent than in North America or Europe.
For branded generic manufacturers with quality and reliability positioning, environmental compliance is not merely a regulatory obligation but a brand risk management issue. A major environmental enforcement action against a branded generic manufacturer’s key API supplier is a supply continuity event and a reputational event simultaneously. Companies that source from environmentally audited API suppliers, require ISO 14001 certification as a procurement condition, or invest in vertically integrated manufacturing with controlled effluent treatment are systematically reducing both supply chain risk and ESG-related investor concern.
Green Chemistry in Formulation Development
Green chemistry principles applied to generic drug formulation development reduce solvent use, energy consumption, and waste generation while frequently improving manufacturing economics. Continuous manufacturing, which FDA has actively encouraged through process analytical technology (PAT) guidance and the 21st Century Cures Act’s provisions, reduces batch-to-batch variability, cuts manufacturing cycle time, and lowers energy consumption per unit versus traditional batch processing.
Pfizer’s Groton facility and Vertex Pharmaceuticals’ CF drug manufacturing programs have demonstrated that continuous manufacturing can produce pharmaceutical-grade product with lower environmental footprint and better quality consistency than equivalent batch processes. For generic manufacturers, the capital investment in continuous manufacturing infrastructure is substantial, but the long-term operating cost advantage is material.
Sustainable packaging, covering recyclable secondary packaging, biodegradable blister materials, and right-sized dosing unit designs that reduce overall packaging material per dose, is increasingly a procurement criterion for hospital group purchasing organizations and retail pharmacy chains with ESG commitments. Branded generic manufacturers who lead on sustainable packaging can use it as a genuine commercial differentiator with institutional customers.
Key Takeaways: Sustainability
For operations and procurement teams: ESG-linked supply chain requirements from institutional customers (hospital GPOs, pharmacy chains, government tender authorities) are hardening from preference to requirement. Companies without visible ESG programs in manufacturing and supply chain face increasing commercial risk with these customers over a five-year horizon. For investor relations teams: branded generic companies with credible carbon reduction roadmaps, sustainable packaging programs, and API supplier environmental audit standards attract institutional ESG investors and reduce the cost of capital relative to peers without these programs.
Part 16: Investment Strategy for Portfolio Managers and Analysts
Valuation Framework for Branded Generic Assets
Branded generic companies and assets are best valued through a sum-of-the-parts DCF approach that explicitly models each key commercial product’s patent-adjusted revenue trajectory and the corporate overhead required to maintain it. The components:
Revenue timing model: Map each key asset’s expected generization date, incorporating Paragraph IV challenge probability, litigation duration distribution, and authorized generic entry risk. This produces a probabilistic launch window rather than a single date, which should be used to generate expected value revenue projections rather than point estimates.
Margin trajectory model: Estimate gross margin at launch (typically 50% to 60% for a first-to-market generic), post-exclusivity margin erosion rate (generic entry typically compresses margins 15 to 25 percentage points within 12 months), and the branded premium that sustains above commodity pricing over the long tail. The branded premium duration is the critical variable: well-established brands in high-prescriber-loyalty therapeutic areas can sustain 15% to 25% premiums for 3 to 7 years post-exclusivity. Poorly differentiated branded generics revert to commodity pricing within 12 to 24 months.
IP portfolio quality score: Assess the percentage of current revenue covered by internally held patents, ANDA exclusivity periods, or Paragraph IV-related first-filer advantages. Companies with a high percentage of near-term revenue from assets with clear IP protection trade at higher multiples than equivalent revenue companies with exposed assets. For biosimilar assets, interchangeability designation converts a non-automatic-substitution product into one that can penetrate pharmacy channels without prescriber action, representing a step-change in market access economics.
Regulatory track record premium: Companies with clean FDA inspection histories, no import alerts, no consent decrees, and low Complete Response Letter (CRL) rates on ANDA submissions deserve a discount rate reduction relative to peers with compliance burdens. Regulatory risk is a real capital cost that is frequently underweighted in analyst models.
Sector-Specific Risks and Investment Signals
For long positions in branded generics companies: key positive signals include a concentrated pipeline of first-to-file Paragraph IV ANDAs approaching approval, value-added formulation 505(b)(2) programs with proprietary IP, biosimilar interchangeability designations in the pipeline, and emerging market branded generic franchises with demonstrated prescriber loyalty. EBITDA margin expansion from manufacturing efficiency programs, combined with a clean regulatory track record, supports multiple expansion arguments.
For short positions or underweight recommendations: signals include heavy dependence on single molecules approaching commoditization without a next-generation portfolio, REMS litigation exposure that could delay generic entry and reduce first-mover revenue, supply chain concentration in single-source API suppliers with active FDA warning letters, and Inflation Reduction Act negotiation exposure on key revenue molecules.
M&A signal watch: branded generic companies with undervalued emerging market branded franchises, particularly in India or Southeast Asia, are perennial acquisition targets for companies seeking geographic diversification. Sun Pharma’s acquisition of multiple Indian branded generic companies at 3 to 5x revenue multiples reflects the durable value embedded in physician relationship networks and brand equity that cannot be easily replicated organically.
The Biosimilar Interchangeability Premium in Deal Valuation
In acquisition and licensing transactions involving biosimilar assets, interchangeability designation should command a specific premium. The most defensible basis for quantifying this premium: model the additional penetration rate achievable through pharmacy-level automatic substitution versus the baseline penetration achievable without interchangeability, multiply by expected price per unit, discount at the appropriate risk-adjusted rate, and compare the NPV differential. In high-volume self-injection markets (insulin, adalimumab, etanercept), the interchangeability premium on NPV is material, frequently in the range of 15% to 30% of total asset NPV.
Key Takeaways: Investment Strategy
Branded generics are not generic stocks. The spread between a well-managed branded generic company with diversified IP and a commodity generic manufacturer with no brand investment can be 3 to 5 EBITDA multiple turns. The variables that drive that spread, patent estate quality, first-to-file pipeline, brand durability, regulatory compliance, and emerging market franchise strength, are all analytically accessible with the right data. The companies that systematically compound these variables over time generate durable shareholder value independent of drug pricing political cycles. Those that fail to invest in brand differentiation and quality infrastructure get commoditized along with their molecules.
Conclusion
The branded generic sector’s commercial logic is straightforward: apply brand architecture to off-patent molecules, generate margins above the commodity generic floor, and build patient and prescriber trust that sustains those margins longer than pure price competition would allow. The execution is not simple. It requires rigorous patent landscape analysis, disciplined regulatory strategy, consistent quality investment, and commercial infrastructure capable of sustaining brand relationships against commodity competition.
The sector’s trajectory over the next decade runs through three structural forces. The patent cliff for major biologics and small-molecule drugs generates the raw material. Value-added formulation technology and digital health integration create the differentiation. Emerging market healthcare infrastructure growth provides the volume. Companies positioned at the intersection of all three, with defensible IP, operational quality, and geographic diversification, will generate durable returns. Those positioned in only one or two dimensions face compression as patent cliffs commoditize their core and competition erodes their margins.
For IP teams, the branded generic opportunity is a patent strategy exercise as much as a development exercise. For R&D leads, it is a formulation science and regulatory pathway choice with 505(b)(2) increasingly preferred over ANDA for programs with genuine technical differentiation. For portfolio managers, it is a sector where the spread between well-positioned and poorly-positioned companies is wide and persistent, driven by factors that are analytically distinguishable with the right data and frameworks.
Data sourced from FDA Orange Book, EMA public assessments, Research and Markets, GMI Insights, Straits Research, Health Affairs, HHS ASPE generic competition analysis, Association for Accessible Medicines, FTC published enforcement actions, and company public financial disclosures. Patent landscape data references DrugPatentWatch pharmaceutical patent database.