A comparative deep dive for pharma and biotech IP teams, R&D leads, and institutional investors navigating the two most consequential patent systems in the global drug economy.
Section 1: Executive Summary and Strategic Context

The U.S. and Chinese patent systems are no longer parallel frameworks that global pharmaceutical companies can treat as independent filing targets. They are deeply entangled competitive arenas where strategic moves in one jurisdiction increasingly dictate outcomes in the other. A Paragraph IV challenge filed in Newark can now trigger a parallel invalidation petition at CNIPA’s Re-examination and Invalidation Department in Beijing. A Chinese court’s anti-suit injunction can block a German licensor from enforcing a Standard-Essential Patent in any other jurisdiction. A drug that launches commercially in the U.S. two years before China now forfeits its full five-year Patent Term Extension eligibility in China under the 2021 Patent Law amendments.
This is no longer a story about differing procedural rules. It is a story about two competing models of state-directed technological accumulation, and about how the global pharmaceutical supply chain is being reshaped by the divergent incentive structures each model creates.
The U.S. system is, at its constitutional core, a private property framework. Patents are property rights granted in exchange for public disclosure — the classical Lockean bargain transplanted into Article I, Section 8, Clause 8. The system is adversarial, expensive, and slow, but when it works, it generates extraordinarily durable and commercially certain exclusivity windows. For a blockbuster oncology drug generating $5 billion annually, a single valid patent extending exclusivity by 18 months is worth more than most mid-cap pharmaceutical companies.
China’s system is something different. It began as a diplomatic concession to WTO accession in 2001, accelerated through a decade of subsidy-driven volume maximization, and has now matured into a precise instrument of industrial policy. The 14th Five-Year Plan is explicit: China intends to lead in biopharmaceuticals, AI drug discovery, and next-generation vaccines. Its patent system, enforcement courts, data exclusivity rules, and PTE framework are all calibrated to make that outcome more likely. The drop of nearly 29% in Chinese invention patent grants in the first half of 2025 is not a sign of weakness — it is the controlled demolition of a low-quality portfolio to make room for a smaller, more powerful one.
Understanding both systems at the technical level — how examination standards differ, what dual filing buys you, how Paragraph IV maps onto China’s Type IV declaration, and what Telitacicept’s June 2025 PTE precedent means for global launch sequencing — is no longer the exclusive domain of in-house patent counsel. For investors modeling terminal values on drug assets, for R&D leads making molecule prioritization decisions, and for business development teams structuring licensing deals, this knowledge is material.
Key Takeaways — Section 1
- China’s 2021 Patent Law amendments created a “launch sequencing trap”: drugs first approved overseas before Chinese approval lose access to full PTE and data exclusivity protections.
- The ~29% decline in Chinese invention patent grants in H1 2025 reflects deliberate policy, not systemic failure. CNIPA is pruning subsidized low-quality applications.
- A valid, well-structured patent position in both jurisdictions is not just a legal defensive asset — it is a primary determinant of a drug asset’s DCF-based terminal value.
- IP strategy and geopolitical strategy are no longer separable decisions for pharmaceutical companies with China exposure.
Section 2: Foundational Philosophies and Legal Architecture
2.1 The U.S. System: Constitutional Origins and the Quid Pro Quo
U.S. patent law derives its authority from Article I, Section 8, Clause 8 of the Constitution, which grants Congress power to “promote the Progress of Science and useful Arts, by securing for limited Times to Authors and Inventors the exclusive Right to their respective Writings and Discoveries.” Two structural principles flow from this mandate.
First, the system is utilitarian in purpose but property-based in mechanism. The patent is a temporary monopoly granted as an incentive for inventors to disclose, not hoard, their inventions. This quid pro quo logic — full public disclosure in exchange for a bounded exclusive right — is the basis for the entire prosecution and claims-drafting regime. A claim that discloses less than it covers is invalid for lack of written description. A claim that covers more than one distinct invention can be restricted. The system polices the boundaries of the bargain on both sides.
Second, because patents are property, disputes about their scope and validity are adjudicated by the judicial branch with all the procedural rigor that implies: Markman hearings for claim construction, jury trials for infringement and willfulness, expert witnesses on both sides, and the full machinery of U.S. federal discovery. This process costs between $3 million and $17 million per side depending on the amount at risk, but generates decisions with significant precedential weight and finality.
The four substantive requirements for U.S. patent protection have generated decades of litigation that define the practical scope of the system:
35 U.S.C. §102 (Novelty) requires that the claimed invention be new as of the effective filing date. The America Invents Act (AIA), enacted in 2011, converted the U.S. from a first-to-invent to a first-inventor-to-file system, eliminating the “swearing behind” practice by which inventors could antedate prior art by proving an earlier conception date. For pharmaceutical companies, this shift increased the strategic importance of provisional application filing dates, particularly for molecules in early-stage development where the prior art landscape is moving fast.
35 U.S.C. §103 (Non-Obviousness) is the workhorse of pharmaceutical patent prosecution and the battleground for most ANDA Paragraph IV challenges. The Graham v. John Deere four-factor framework — scope of prior art, differences from the claim, level of ordinary skill, and secondary considerations — sounds manageable in the abstract. In practice, the Federal Circuit has produced a body of obviousness jurisprudence so granular that pharmaceutical patent counsel must monitor weekly decisions to understand where the line sits on any given structural class of molecule. The “obvious to try” doctrine, which the Federal Circuit applied aggressively in Pfizer v. Apotex (2007) to invalidate a besylate salt patent, is particularly threatening for formulation and polymorph claims.
35 U.S.C. §101 (Utility and Patent-Eligible Subject Matter) had been relatively uncontroversial in pharmaceuticals until Mayo Collaborative Services v. Prometheus Laboratories (2012) and its application to diagnostic method claims. A claim covering a method of measuring a metabolite level and adjusting drug dosage accordingly was held ineligible as a law of nature. The consequences for personalized medicine patent strategies were profound: companion diagnostic claims, biomarker-based dosing methods, and genomic testing methods all faced renewed eligibility scrutiny.
The enablement requirement under 35 U.S.C. §112 deserves separate treatment for biologics. In Amgen v. Sanofi (2023), the Supreme Court unanimously invalidated Amgen’s broad PCSK9 antibody claims covering all antibodies that bind a defined epitope and block PCSK9 activity, holding that requiring a skilled artisan to generate and screen potentially millions of antibodies to find those within the claim’s scope did not constitute adequate enablement. For antibody patent strategy, this ruling fundamentally reshaped the appropriate claim breadth, shifting emphasis toward claims defined by specific sequences or well-characterized structural features rather than functional properties alone.
2.2 The Chinese System: Statutory Architecture and the Three Natures
China’s Patent Law entered force on April 1, 1985 — a deliberate product of the reform and opening-up policy, not an organic outgrowth of domestic innovation culture. It was designed, from the outset, to signal alignment with international norms and attract technology transfer from foreign companies that required IP protection before committing to Chinese joint ventures. The system has been revised four times (1992, 2000, 2008, 2021), each revision driven by a combination of external treaty obligations and internal industrial policy objectives.
Article 1 of the Chinese Patent Law states the system’s purpose explicitly: to protect patentees’ rights, encourage invention-creation, “promote the exploitation of invention-creation,” enhance innovation capability, and promote economic and social development. The word “exploitation” is deliberate. Chinese patent law does not merely reward disclosure; it seeks to ensure that protected inventions are actively commercialized and contribute to national economic development. This distinction has direct legal consequences: compulsory licenses in China can be granted when a patent is “not exploited” without justification, a mechanism unavailable in the U.S. outside narrow national emergency provisions.
The three patentability criteria for a Chinese invention patent — novelty (新颖性), inventiveness/creativity (创造性), and practical applicability (实用性) — broadly mirror the U.S. standard, but their application differs in ways that matter:
Novelty in China is assessed against worldwide prior art as of the filing date. China’s grace period is narrowly confined to disclosures at government-recognized international exhibitions or academic conferences. Its application is unreliable in practice. For pharmaceutical companies used to publishing clinical trial results before filing continuation patents in the U.S., this creates material risk: a journal publication describing a new indication or formulation can constitute prior art in China that destroys novelty of a subsequently filed Chinese application. Pre-filing publication strategies must be re-engineered for companies with serious China market ambitions.
Practical applicability in China is slightly broader than U.S. utility in one respect: it does not require a “specific, substantial, and credible” utility as the USPTO does post-In re Fisher. Claims covering new chemical entities with demonstrated in vitro activity generally satisfy Chinese practical applicability requirements even where a U.S. examiner might push back on utility adequacy.
2.3 The Structural Divergence: What It Means for Drug Companies
The different philosophical foundations create predictable, systematic differences in behavior that drug companies should factor into IP strategy at the portfolio level.
A U.S. patent portfolio is primarily a litigation asset. Its value is realized in the courtroom through Hatch-Waxman 30-month stays, Federal Circuit appeals, and district court enforcement actions. Building a U.S. portfolio means building for litigation defensibility: broad primary claims supported by extensive written description, narrow fallback claims to survive obviousness attacks, and continuation strategies that allow claims to be tuned to the actual products entering the market years after filing.
A Chinese patent portfolio is primarily a market access and enforcement tool. Its value is realized through the speed of the utility model system, the favorable injunction rates of the specialized IP courts, and the strategic use of administrative enforcement through local IP offices. Building a Chinese portfolio means thinking about grant speed first, then litigation stability second. Dual filing an invention patent and a utility model on the same day secures an enforceable right in under a year while the stronger 20-year invention patent matures through examination.
Key Takeaways — Section 2
- The U.S. grace period allows inventors to publish before filing; China’s does not. Publication before filing destroys Chinese novelty for most pharmaceutical claims.
- Amgen v. Sanofi (2023) fundamentally limits functional antibody claims in the U.S. Sequence-defined or structurally specific claims are the appropriate standard for biologic prosecution.
- China’s practical applicability standard is slightly more permissive than U.S. utility for early-stage NCE claims.
- U.S. portfolio strategy optimizes for litigation defensibility. Chinese portfolio strategy optimizes for enforcement speed and technical problem/effect framing.
Section 3: The Administrative Gatekeepers: USPTO and CNIPA Compared
3.1 The USPTO: Fee-Funded, Policy-Constrained, and Under Legislative Pressure
The USPTO operates under the Department of Commerce and is funded entirely by fees collected from patent and trademark applicants — approximately $4.4 billion annually. This self-funding model creates a structural tension that is particularly acute for pharmaceutical companies. The agency has a genuine incentive to issue high-quality patents, because invalid patents that later get canceled undermine the system’s credibility. At the same time, it faces constant pressure to manage its budget, control pendency times, and process a large volume of applications efficiently.
The USPTO’s examination corps includes approximately 12,000 patent examiners organized into Technology Centers. Technology Center 1600 handles biotechnology and organic chemistry. TC1600 examiners have PhDs in biology, biochemistry, and medicinal chemistry, and the quality of examination in this technology area is generally high. However, the backlog remains significant: average total pendency from filing to final disposition sits at approximately 24.6 months for a standard examination track. Track One prioritized examination reduces this to 8-12 months for an additional fee of $4,000 (large entity), and pharmaceutical companies launching products with uncertain IP positions routinely use this mechanism.
The USPTO’s Patent Trial and Appeal Board (PTAB) is the venue for Inter Partes Review (IPR) and Post-Grant Review (PGR) proceedings, which have become standard defensive tools for generic pharmaceutical companies challenging branded drug patents. IPR can be filed by any party who has not already challenged the patent in district court, within one year of being served with an infringement complaint. The challenger’s burden is “preponderance of the evidence” — easier than the “clear and convincing evidence” required in district court. The PTAB institution rate on pharmaceutical patent IPR petitions has historically run around 60-70%, and it has been called a “patent death squad” by critics in the pharmaceutical industry.
3.2 CNIPA: An Executor of Industrial Policy, Not a Neutral Examiner
CNIPA’s reorganization in 2018 — from SIPO to the China National Intellectual Property Administration under the State Administration for Market Regulation — was not cosmetic. It embedded China’s primary IP authority within the executive apparatus responsible for market regulation, competition policy, and anti-monopoly enforcement. This placement gives CNIPA direct lines of coordination with the agencies that regulate pharmaceutical pricing, market access, and domestic company support.
CNIPA’s mandate goes well beyond examining patents. It formulates national IP strategy, oversees administrative enforcement at the local IP office level, manages compulsory licensing procedures, sets data exclusivity rules through regulatory coordination with the National Medical Products Administration (NMPA), and operates the China Patent Information Registration Platform for Marketed Drugs — the domestic Orange Book equivalent.
The most revealing indicator of CNIPA’s policy-driven character is its subsidy elimination campaign. For years, provincial and municipal governments paid per-patent subsidies to domestic filers. Shenzhen, for example, offered RMB 3,000 per granted invention patent and RMB 1,000 per utility model. These subsidies created a predictable response: a flood of low-quality applications filed to collect fees, not to protect real innovations. CNIPA now defines and actively pursues “abnormal patent applications” — fabricated inventions, plagiarized applications, mass applications from patent farms. The direct consequence was a 28.8% decline in Chinese invention patent grants in the first half of 2025 relative to the same period in 2024. This was a controlled policy outcome: CNIPA trading volume for quality, consistent with the 14th Five-Year Plan’s explicit shift from patent quantity to “high-quality development.”
For foreign pharmaceutical companies evaluating China patent exposure, this shift matters in two ways. First, the patent thicket created by years of subsidized low-quality utility models will persist for a decade as those patents reach their 10-year terms. FTO analyses in China remain extraordinarily complex. Second, the new high-quality patents being issued to Chinese domestic companies in biopharmaceuticals — from BeiGene, Zymeworks China, and Hengrui Medicine — are substantively more robust than the subsidy-era filings, making them harder to invalidate in CNIPA proceedings.
3.3 The Administrative Enforcement Track: China’s Structural Advantage
One feature of CNIPA’s architecture with no U.S. equivalent is the administrative IP enforcement route. A patent holder in China can file an infringement complaint with a local IP office rather than a court. The local IP office can investigate, issue an order to stop infringing activity, and impose fines — typically within three to six months, compared to the one to two years a first-instance court judgment takes. The trade-off is that administrative enforcement does not award damages. But for a foreign pharmaceutical company that has discovered a Chinese company manufacturing and exporting infringing product, an administrative injunction that stops the production line within six months may be worth more than a damages award two years later.
Companies with active China patent portfolios should map their enforcement strategy across judicial, administrative, and customs tracks simultaneously. All three mechanisms — local IP office complaint, court action, and GACC border recording — can run in parallel and are most effective when deployed together.
Key Takeaways — Section 3
- USPTO TC1600 examination quality for pharmaceutical biotech claims is high, but IPR remains a potent invalidation weapon for generic challengers. PREVAIL Act passage would shift the IPR calculus significantly.
- CNIPA’s mandate includes market regulation coordination, compulsory licensing, and data exclusivity rule-setting — functions the USPTO does not perform. This integration matters for pharmaceutical deal-making in China.
- The 29% drop in Chinese patent grants in H1 2025 is policy-driven quality control. Domestic champions like BeiGene and Hengrui are filing more — and better — patents.
- China’s administrative enforcement track can secure an effective production stop in 3-6 months. This mechanism is underused by foreign pharmaceutical companies.
Section 4: Patent Typology — The Utility Model Arbitrage Opportunity
4.1 U.S. Patent Types: The Pharmaceutical Practitioner’s Toolkit
U.S. utility patents cover processes, machines, manufactures, and compositions of matter. For the pharmaceutical industry, the relevant claim categories are:
Composition claims cover the NCE itself, defined by structure (Markush groups for related analogs, or specific stereochemical configurations), crystal form (polymorphs), or salt forms. These are the primary patent for any small molecule drug and typically command the longest market exclusivity window because they are filed earliest in development.
Method-of-use claims cover the specific therapeutic indication, dosing regimen, or patient population. These can be filed after the composition patent and, under Hatch-Waxman, can be listed in the Orange Book if they cover an approved method of use. They are particularly valuable for drugs that gain major new indications post-launch.
Formulation claims cover the specific drug delivery system, dosage form, or excipient combination. These are frequently the target of obviousness attacks in Paragraph IV proceedings because pharmaceutical formulation is a mature science.
Process claims cover the synthetic route to the API or a critical intermediate. They are valuable when a novel and non-obvious route produces meaningfully higher purity or yield, but are easier for generic manufacturers to design around than composition claims.
4.2 China’s Three Patent Categories and Pharmaceutical Relevance
China’s invention patent is the full analog of the U.S. utility patent: 20 years from filing, substantive examination required, novelty plus inventiveness plus practical applicability. The 2021 Patent Law added Patent Term Adjustment for unreasonable examination delays and PTE for qualifying new drugs, making the effective duration of Chinese invention patents potentially competitive with U.S. exclusivity windows for the first time.
China’s design patent was extended to 15 years from the grant date under the 2021 amendments, and the scope now covers partial product designs — meaning a pharmaceutical company can protect not just the entire form of an inhaler but specific ergonomic features.
The utility model is where the real strategic action lies for pharmaceutical companies, and it is also where the most confusion occurs. A Chinese utility model can only protect a product with a definite physical shape or structure. This means:
It can protect a novel drug delivery device, a prefilled syringe mechanism, a novel capsule design, or a specific physical configuration of a pharmaceutical dosage form, provided the claim is directed to the physical structure itself.
It cannot protect a chemical compound, a process, a method of treatment, a formulation defined by its chemical composition, or a software algorithm for drug discovery.
For a pharmaceutical company launching a combination product — an autoinjector prefilled with a biologic, for example — the utility model can protect the device component with near-immediate enforceability, while an invention patent application on the biologic itself works through the multi-year substantive examination process. For GLP-1 receptor agonist pen injectors, anti-VEGF biologic prefilled devices, and subcutaneous biologic formulation delivery systems, this is not a trivial commercial distinction.
4.3 The Dual Filing Mechanism: Mechanics and Strategic Parameters
The Chinese dual filing system — filing an invention patent and a utility model on the same invention on the same day — is the single most underutilized strategic tool available to foreign pharmaceutical companies with Chinese device or product claims. The mechanism is available for Paris Convention national phase entries but not for PCT national phase entries, where the applicant must choose one type.
The prosecution tracks run in parallel. The utility model application undergoes only a formal examination (checking completeness and form) and typically grants within six to twelve months. The granted utility model is immediately enforceable and can be asserted in both judicial and administrative enforcement proceedings. When the invention patent application subsequently reaches an allowability determination, CNIPA notifies the applicant, who must then abandon the utility model to allow the invention patent to issue, preventing double protection on the same invention. If the invention patent is rejected on inventiveness grounds, the utility model remains in force as a fallback.
The financial arithmetic is straightforward. Official fees for a Chinese utility model application are roughly RMB 500 (approximately $70). Attorney drafting costs for a utility model that parallels an already-drafted invention patent application are modest — claims must be modified to focus on structural features, but the specification is shared. Total incremental cost of the dual filing is typically under $500. Against the commercial value of having an enforceable right within twelve months in a market of 1.4 billion people, this is a trivially small investment.
Key Takeaways — Section 4
- Composition, method-of-use, formulation, and process claim types each have distinct vulnerability profiles in Paragraph IV proceedings. Portfolio strategy should include explicit modeling of which claim type is most likely to survive a generic challenge.
- Chinese utility models cannot protect chemical compounds, processes, or methods. They can protect drug delivery devices, novel dosage form structures, and combination product device components.
- Dual filing provides enforceable Chinese rights within 12 months for under $500 in incremental cost. It is the most underutilized tool for foreign pharma companies with device-related IP.
- The 2021 Chinese design patent extension to 15 years now covers partial product designs, enabling protection of specific ergonomic or functional features of drug delivery systems.
Section 5: Prosecution Strategy — Timelines, Costs, and Examination Standards
5.1 Application Pathways and Timing Strategy
In the United States, the standard pharmaceutical prosecution path begins with a provisional application establishing priority. The provisional buys 12 months to refine claims, gather additional data, and decide whether to pursue full prosecution. In fast-moving areas — CRISPR applications, cell therapy manufacturing processes, mRNA synthesis methods — the ability to lock in a priority date while the science continues is commercially critical.
Patent term adjustment (PTA) compensates for USPTO delays in meeting statutory examination windows and can extend the 20-year patent term by months or years. For blockbuster drugs, PTA tracking and optimization is a material commercial activity. Pfizer’s Lyrica (pregabalin) benefited from significant PTA accumulation, as have multiple oncology drugs where examination was prolonged by biomarker patentability disputes.
In China, the prosecution path for an invention patent begins with filing at CNIPA. Within 18 months, the application is published. The applicant must then separately request substantive examination — if this request is not made within three years of the filing date, the application is deemed withdrawn. The substantive examination process then takes two to four additional years from the examination request, meaning total pendency from filing to grant is typically three to five years. PTA was introduced in China under the 2024 Implementing Regulations to compensate for examination delays exceeding statutory periods.
The Patent Prosecution Highway (PPH) between the USPTO and CNIPA allows an applicant who has received a U.S. allowance to request accelerated examination in China, leveraging the USPTO’s prior art search and examination findings. Eligibility requires that the Chinese application have already been published and that a substantive examination request has been filed. The claims in the Chinese application must “sufficiently correspond” to the allowed U.S. claims. For pharmaceutical companies prosecuting parallel portfolios in both countries, PPH is a standard cost-efficiency tool that can reduce Chinese prosecution costs by eliminating duplicative prior art searches.
5.2 The U.S. Obviousness Standard in Pharmaceutical Practice
The Graham v. John Deere four-factor framework is the formal test, but pharmaceutical patent litigation has developed its own body of obviousness doctrine so rich and specific that it operates almost as a separate analytical system. The critical battlegrounds are:
Structural obviousness for NCEs: The Federal Circuit has long held that structural similarity to known compounds, combined with overlapping biological activity, creates a prima facie case of obviousness. The pharmaceutical applicant must then rebut with evidence of unexpected results — superior potency, better selectivity, longer half-life, reduced toxicity — that the prior art would not have predicted.
Lead compound selection: In many pharmaceutical obviousness cases, the central question is whether a skilled artisan would have selected the asserted compound as a “lead compound.” If the prior art provides a reason to select a specific structural class, and if the claimed compound is a routine modification of that class, obviousness is often found. AstraZeneca’s esomeprazole patent litigation illustrated this: the pure (S)-enantiomer of omeprazole was held obvious where the prior art disclosed both enantiomers and where the benefits of enantiopure drug development were well-known.
Secondary considerations: Commercial success, long-felt but unsolved need, failure of others, and unexpected results are the standard secondary considerations. The Federal Circuit requires a “nexus” between the patent claims and the commercial success evidence — the success must be attributable to the claimed invention, not to other product features.
5.3 China’s Three-Step Inventive Step Method: A Pharmaceutical Practitioner’s Guide
CNIPA’s three-step inventive step analysis differs structurally from the U.S. Graham framework in ways that create systematically different outcomes for the same pharmaceutical inventions:
Step 1 identifies the closest single piece of prior art. The CNIPA examiner selects the reference most similar to the claimed invention. This is more rigid than the U.S. approach, which can consider combinations of references from the outset.
Step 2 determines the distinguishing features and the “technical problem actually solved.” The examiner compares the claim to the closest prior art, identifies the structural or functional differences, and asks: given those distinguishing features, what is the technical problem actually solved by the invention? This framing is often dispositive. If the technical problem is framed narrowly — providing a compound with a specific enzyme binding profile — it may not be obvious. If framed broadly — providing an alternative kinase inhibitor — the invention may appear obvious.
Step 3 assesses obviousness: would a skilled artisan, starting from the closest prior art and seeking to solve the technical problem identified in Step 2, have arrived at the claimed invention with a reasonable expectation of success?
The key pharmaceutical prosecution implication is that the specification must explicitly articulate the technical problem the invention solves and provide data demonstrating the technical effect achieved. A U.S.-style specification that emphasizes structural novelty and mechanism of action, without explicitly framing a technical problem and demonstrating a superior technical effect, is underoptimized for Chinese examination. A Chinese specification that clearly demonstrates an unexpected technical effect — a 10-fold improvement in selectivity over the structurally closest prior art compound, with supporting data in the examples section — is difficult to reject on inventive step grounds even if the structural modification appears modest.
5.4 The Conflicting Application Rule: A Chinese Prosecution Trap
China’s “conflicting application” rule has no direct U.S. equivalent and routinely catches foreign pharmaceutical filers by surprise. An application filed earlier by a different applicant but published after the applicant’s own filing date can destroy novelty — but it cannot be used against inventive step. A competitor who filed a similar compound claim one week before your filing, but whose application was not yet published at your priority date, can retroactively destroy the novelty of your claim.
The U.S. system provides the 35 U.S.C. §102(b)(2) exception for common assignment or joint research agreement — meaning prior-filed applications from your own organization do not count as prior art. China has no equivalent exception, making internal organizational patent filing coordination within multinational pharmaceutical companies an important risk-management function.
Key Takeaways — Section 5
- Track One prioritized examination ($4,000 large entity) is standard practice for high-priority pharmaceutical assets with imminent generic exposure. It is underutilized as a tactical tool in portfolio management.
- Chinese prosecution specifications must explicitly state the technical problem solved and demonstrate the technical effect with comparative data. U.S.-drafted specifications typically do not do this and require purposeful adaptation for Chinese filing.
- The conflicting application rule in China can destroy novelty retroactively. Portfolio management must include monitoring competitor filing activity, not just publication dates.
- PPH between USPTO and CNIPA is a standard cost-efficiency tool for parallel prosecution; it requires that the Chinese application be published and substantive examination be requested before the PPH acceleration request can be filed.
Section 6: Enforcement Architecture — Where to Fight and How to Win
6.1 China’s Bifurcated System: Strategic Separation of Validity and Infringement
China handles patent validity and patent infringement in separate proceedings — a bifurcated system similar to Germany’s. In an infringement suit before one of China’s specialized IP courts (Beijing, Shanghai, Guangzhou, or regional IP tribunals in Chengdu, Wuhan, Suzhou, and others), the defendant cannot raise invalidity as a defense in the infringement case. The court proceeds on the assumption that the patent is valid. The defendant’s rational response is to file an invalidation petition with CNIPA simultaneously with the answer in the infringement proceeding, and to request a stay of the infringement case pending the CNIPA decision.
Courts exercise discretion on whether to grant such stays. Beijing IP Court tends to continue infringement proceedings even when a CNIPA invalidity proceeding is pending, if the infringement evidence is strong and the invalidity arguments are unlikely to succeed. This approach is a conscious policy choice designed to make Chinese IP enforcement fast and credible.
Appeals from the specialized IP courts in infringement matters go directly to the IP Tribunal of the Supreme People’s Court (SPC-IP Tribunal). Appeals from CNIPA invalidity decisions go first to the Beijing IP Court and then, if further appealed, also to the SPC-IP Tribunal. For high-stakes pharmaceutical patent cases, final resolution of both infringement and validity questions ultimately lands before the same specialized judicial body — a structure that promotes doctrinal consistency and predictability.
6.2 U.S. Enforcement: Hatch-Waxman Litigation, PTAB, and the ITC
The U.S. pharmaceutical patent enforcement system is defined by the Hatch-Waxman Act framework, which built a structured patent dispute resolution process directly into the drug approval pathway. When a generic pharmaceutical company files an ANDA challenging a patent listed in the FDA Orange Book under a Paragraph IV certification — asserting that the patent is invalid, unenforceable, or not infringed — it triggers a statutory mechanism with the following sequence:
The generic ANDA filer must notify the patent holder and NDA holder of its Paragraph IV certification. The patent holder has 45 days to file an infringement lawsuit. Filing within 45 days automatically triggers a 30-month stay of ANDA approval, during which the FDA cannot approve the generic application regardless of scientific review status. This stay effectively gives the branded company 2.5 years to litigate the patent before generic entry, even if the generic ultimately prevails.
The first ANDA filer with an unresolved Paragraph IV certification has a 180-day exclusivity period once it receives a final court judgment of invalidity or non-infringement. This exclusivity — which blocks FDA from approving subsequent ANDA filers — is the primary economic incentive driving the generics industry to file Paragraph IV certifications aggressively. For a drug with $2 billion in annual U.S. sales, the 180-day exclusivity period is worth $400-600 million in generic profit.
For pharmaceutical patentees where infringing product is manufactured outside the United States and imported, the ITC’s Section 337 proceeding offers an exclusion order blocking U.S. Customs from allowing the infringing product to enter. The ITC timeline is 15 to 18 months from institution to initial determination — faster than district court. The primary remedy is an exclusion order rather than monetary damages. To bring a case, the complainant must prove the existence of a “domestic industry” related to the patented technology.
6.3 The Discovery Divide and Its Strategic Consequences
No procedural difference between U.S. and Chinese patent litigation is more commercially significant than discovery. U.S. pharmaceutical patent litigation involves extensive document production, depositions of named inventors and corporate witnesses, interrogatories, requests for admission, and expert depositions. In a complex ANDA case covering multiple patents, total document production can run to millions of pages. The cost of U.S. pharmaceutical patent litigation through trial routinely exceeds $10 million per side for branded companies.
Chinese patent litigation has no pre-trial discovery. The plaintiff must arrive at the courthouse with all evidence of infringement already assembled, authenticated, and notarized. For pharmaceutical cases, this typically means purchasing the accused infringing product from Chinese market channels, notarizing the purchase transaction, conducting comparative compositional analysis, and commissioning an expert technical report — all before filing. The burden on the plaintiff is front-loaded, but the case moves faster and for less money once it is filed.
The no-discovery rule creates a structural challenge for pharmaceutical patentees suing Chinese generic manufacturers: proving damages. Chinese courts increasingly award damages based on a multiple of the patentee’s reasonable royalty rate, calculated by reference to industry licensing benchmarks. The 2021 Patent Law raised the statutory damages ceiling to RMB 5 million (~$715,000) and introduced punitive damages of up to five times the base award for willful infringement.
6.4 Injunction Availability: The Post-eBay Asymmetry
In the U.S., the Supreme Court’s 2006 eBay decision eliminated the historical presumption that a patent victory entitles the patent holder to a permanent injunction. Courts must now apply a four-factor equitable test: irreparable injury to the patentee, inadequacy of monetary damages, balance of hardships, and whether a permanent injunction serves the public interest. For pharmaceutical cases, this test generally favors injunctive relief for practicing entities, but the post-eBay test has proven much harder to satisfy for non-practicing entities and for cases where the patent covers a minor component of a complex product.
In China, injunction follows infringement essentially automatically. Foreign plaintiffs who win Chinese patent infringement cases receive permanent injunctions in more than 90% of cases. Preliminary injunctions must be ruled on by courts within 48 hours of application. For a foreign pharmaceutical company that has discovered a Chinese competitor mass-producing an infringing API or drug product, a preliminary injunction obtained within 48 hours can stop the entire production line before the product reaches commercial channels.
The combination of high injunction rates, short timelines, no discovery, and high foreign plaintiff win rates (77% to 87% in empirical studies of Chinese IP court outcomes) has transformed China from a jurisdiction to avoid into, for certain types of disputes, the most favorable enforcement venue in the world. A foreign pharmaceutical company whose technology is being infringed by a Chinese manufacturer that also exports to third-country markets should consider Chinese litigation as the primary enforcement vehicle, not a secondary or defensive option.
Key Takeaways — Section 6
- China’s bifurcated validity/infringement system means patentees can pursue an injunction in infringement proceedings while the defendant contests validity separately at CNIPA. Courts often proceed even when CNIPA invalidity is pending.
- Hatch-Waxman’s 30-month stay is the most economically valuable feature of U.S. pharmaceutical patent law. Filing an infringement lawsuit within 45 days of receiving a Paragraph IV certification notification is non-negotiable for any branded company with Orange Book-listed patents.
- Post-eBay, U.S. injunctions remain obtainable for practicing pharmaceutical entities but require equitable analysis. The RESTORE Act would restore a presumption of injunction.
- Chinese IP courts grant permanent injunctions in over 90% of cases won by the patentee, and preliminary injunctions must be ruled on within 48 hours. For infringers with Chinese manufacturing operations, this is the most powerful enforcement tool available.
Section 7: Pharmaceutical IP Deep Dive — Evergreening, PTE, Linkage, and Data Exclusivity
7.1 Evergreening: The Technology Roadmap
Evergreening is not a single strategy but a layered portfolio construction methodology that uses the continuation and divisional prosecution system, combined with targeted NDA/sNDA submissions, to extend effective market exclusivity beyond the expiration of the primary composition patent. The term is used pejoratively by critics but describes a legally protected practice built into the Hatch-Waxman framework.
A pharmaceutical company’s evergreening technology roadmap for a small molecule drug typically proceeds along the following sequential layers, with each layer representing both a patent filing opportunity and a potential Orange Book listing:
Layer 1 — Primary Composition Patent: Filed at discovery or lead optimization. Covers the NCE by structure. Typically expires 20 years from filing, adjusted for PTA and PTE. This patent is the primary Paragraph IV target. It is also the longest-lived piece of protection and, when valid, the most effective.
Layer 2 — Salt and Polymorph Patents: Filed 2-4 years after lead optimization when the formulation team has identified the commercial salt form and characterized the crystalline polymorphs. Claims covering a specific salt form or crystal polymorph are valid when the chosen form has unexpected properties — solubility, bioavailability, stability, or processability — not predictable from the prior art. The Federal Circuit’s Pfizer v. Apotex (2007) decision striking the besylate salt patent for Norvasc established that unexpected properties must be substantially superior, not incrementally better.
Layer 3 — Formulation Patents: Filed during clinical development or Phase III when the final commercial formulation is characterized. Covers the specific excipient combination, release mechanism, particle size range, or coating technology. These are the most vulnerable to obviousness attack because pharmaceutical formulation science is mature and ANDA filers can argue that combining known excipients for known purposes is obvious.
Layer 4 — Method-of-Use Patents: Filed for each new indication or patient population. Second-indication method-of-use patents can be listed in the Orange Book for the approved method of use, triggering the 30-month stay against any ANDA that includes a Paragraph IV certification against that patent. These patents are attractive because they can be filed years after the primary composition patent.
Layer 5 — Dosing Regimen and Companion Diagnostic Patents: Filed during the clinical optimization phase. Dosing regimen patents cover the specific administration schedule that achieves clinical endpoints. Companion diagnostic patents cover the biomarker selection or testing method that identifies the patient population for the drug. Post-Mayo, companion diagnostic claims face §101 eligibility scrutiny that composition claims do not.
Layer 6 — Combination Product and Device Patents: Filed when the drug is reformulated into a combination product (pen injector, autoinjector, inhaler). These can be particularly valuable because they are filed late in the product lifecycle, provide protection extending well past the primary composition patent expiration, and are difficult for generic challengers to design around while maintaining the same user experience.
7.2 AbbVie and Humira: The IP Valuation Case Study
Humira’s patent portfolio provides the most extensively documented example of evergreening economics at scale. At its commercial peak, Humira generated approximately $21 billion in annual global revenues. AbbVie has asserted more than 130 patents covering Humira in various jurisdictions. The core composition patents expired in 2016, but a forest of formulation, manufacturing process, and method-of-use patents extended effective U.S. exclusivity until the January 2023 settlement-date launches of biosimilars.
The IP valuation of the Humira portfolio is not captured in any single patent’s filing date. It is the aggregate financial value of the extended exclusivity period — roughly 2016 to 2023 — attributable to the secondary patent layer. At $21 billion annual revenue and an operating margin of roughly 65%, each additional year of U.S. exclusivity was worth approximately $13.5 billion in operating income. The secondary patent portfolio, in aggregate, was a $40-50 billion asset measured by the value of the exclusivity extension it provided.
For institutional investors evaluating this position, the complexity and legal cost of challenging 130 patents across multiple jurisdictions was itself a deterrent to earlier biosimilar entry, independent of the technical merits of individual patent claims. This “complexity as a deterrent” dynamic is a quantifiable strategic benefit of large, well-constructed secondary patent portfolios that DCF models typically do not capture explicitly.
7.3 The Paragraph IV Litigation Ecosystem
A Paragraph IV certification is a legal assertion by an ANDA filer that a listed Orange Book patent is invalid, unenforceable, or will not be infringed by the proposed generic product. It is the formal starting point of Hatch-Waxman pharmaceutical patent litigation and one of the most commercially consequential legal filings in the U.S. economy.
The branded company’s response to a Paragraph IV filing has a well-established structure. Within 45 days of receiving the notification letter, it files in district court. The Southern District of New York, District of Delaware, and District of New Jersey are the most common venues, largely because of their experienced pharmaceutical patent judges. The case then proceeds through Markman (claim construction), expert discovery, potentially summary judgment, and trial over a 2-3 year period, all while the 30-month stay blocks ANDA approval.
Strategic outcomes of Paragraph IV litigation can involve: (1) a court judgment upholding the patent and blocking generic entry; (2) a court judgment invalidating the patent allowing generic entry; (3) a settlement with an authorized generic arrangement; or (4) a settlement with a delayed-entry date and value transfer from brand to generic. The FTC v. Actavis (2013) decision created significant antitrust exposure for pharmaceutical companies that want to settle Paragraph IV cases with large cash payments to generic challengers, making settlement structures considerably more complex.
7.4 China’s Patent Linkage System: Paragraph IV by Another Name
China’s patent linkage system, established by the 2021 Patent Law amendments, mirrors the Hatch-Waxman architecture with important structural modifications.
When a generic drug applicant files a marketing approval application in China and the drug is listed on the CPIRPMD, the generic applicant must file one of four patent status declarations:
Declaration Type I: The relevant patent has expired or been declared invalid. Declaration Type II: No relevant patent applies to the generic product. Declaration Type III: The generic company will not enter the market until the patent expires (equivalent to Paragraph III). Declaration Type IV: The generic product does not infringe the listed patent(s), or the listed patent is invalid (equivalent to Paragraph IV).
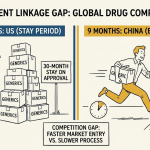
A Type IV declaration triggers a notification to the patent holder. If the patent holder files a lawsuit or requests an administrative proceeding within 45 days, NMPA imposes a nine-month stay on the generic marketing approval decision. This nine-month stay is significantly shorter than the U.S. 30-month stay — a deliberate policy choice to provide branded companies with notice and a litigation window while not indefinitely delaying generic access.
The first generic applicant to successfully challenge a patent through a Type IV declaration and receive marketing approval is entitled to a 12-month market exclusivity period. During that 12 months, NMPA will not approve subsequent generic applications for the same drug, mirroring the 180-day exclusivity in the U.S. system. Companies with Chinese CPIRPMD registrations must have a litigation readiness protocol in place before any generic applications are filed, because the 45-day window for patent holders to file suit is tight.
7.5 Patent Term Extension: The Global Launch Sequencing Imperative
Patent Term Extension is designed to compensate pharmaceutical patent holders for the patent term consumed during regulatory review — the years between the patent filing date and the drug’s commercial launch date. Both the U.S. and China provide PTE, but their eligibility criteria differ in a way that has become the central driver of global drug launch strategy for multinational pharmaceutical companies.
United States PTE (Hatch-Waxman Act, 35 U.S.C. §156): The extension compensates for time spent in Phase II and Phase III clinical trials (at a 50% weighting) and time spent in FDA review from initial submission to approval (at 100% weighting). The maximum extension is five years. After PTE, the total post-approval term cannot exceed 14 years. Only one patent per drug can receive PTE, and it must cover the approved drug.
China PTE (2021 Patent Law, Article 42.3): China’s PTE mechanism closely mirrors the U.S. framework — five-year maximum extension, 14-year post-approval cap, one patent per drug — with one critical difference in eligibility. The Chinese PTE is available only for a “new drug” that has not been previously approved for marketing overseas or domestically before the Chinese approval. A drug that received FDA approval before its Chinese marketing approval is ineligible for the full Chinese PTE. The extension available is reduced by the length of the time gap between the first overseas approval date and the date the Chinese marketing approval application was accepted by NMPA.
The June 2025 grant of China’s first full five-year PTE for Telitacicept — an anti-BLyS/APRIL antibody fusion protein developed by RemeGen — illustrates the system in practice. Telitacicept received Chinese marketing approval before any overseas approval, making it eligible for the full five-year extension. This was a Chinese-first launch, precisely the outcome the PTE incentive was designed to encourage.
For a multinational pharmaceutical company with a drug in late-stage development, the PTE calculus is now explicitly part of global launch sequencing decisions. What is the value of full Chinese PTE (additional billions in discounted future revenues protected by 3-5 extra years of exclusivity) versus the cost of prioritizing the Chinese regulatory approval process to achieve China-first or simultaneous global launch? For high-revenue drugs in large Chinese indication populations — oncology, cardiovascular, metabolic diseases — this analysis frequently favors accelerating Chinese development timelines.
7.6 Regulatory Data Exclusivity: The Six-Year Shield and Its Conditions
Data exclusivity protects the NDA holder’s clinical trial data from being relied upon by generic drug applicants for a defined period after initial approval, regardless of patent status. It is a separate protection from patent exclusivity and can be commercially critical for drugs that lack strong patent protection or for which patents have been invalidated.
In the U.S., the FDA grants five years of data exclusivity for New Chemical Entities upon initial approval. Three years of data exclusivity apply to new clinical investigations submitted in support of a supplemental NDA for a new indication, new formulation, or new combination. Twelve years of data exclusivity apply to biological reference products (BRPs) under the BPCIA.
China’s data exclusivity framework has been under development, with NMPA publishing draft rules in 2022 for a six-year data exclusivity period for innovative new drugs. The most strategically significant feature of the proposed Chinese framework is that the exclusivity term is reduced by the period between the drug’s first overseas approval and the date the Chinese marketing approval application was accepted. A drug approved by FDA in year zero and applied for in China in year two would receive only four years of Chinese data exclusivity rather than the full six. This condition reinforces the same incentive as the PTE eligibility rule: global launch strategies that treat China as a secondary market risk losing both PTE and full data exclusivity protection.
Investment Strategy — Section 7
For institutional investors modeling pharmaceutical company valuations, the following IP-specific analytics are material:
Patent cliff analysis: A company’s revenue-weighted patent expiration schedule determines the timing and magnitude of generic entry risk. The standard methodology is to identify all Orange Book-listed patents for each product, model the probability distribution of Paragraph IV success by patent layer, and discount future revenues by the probability-weighted generic entry date. The discrepancy between a drug’s nominal patent expiration and its expected effective exclusivity date — accounting for secondary patents, authorized generics, and PTE — is frequently a source of valuation error in sell-side models.
China PTE and data exclusivity optionality: For drugs currently in Phase III development with significant China market potential, the incremental value of a China-first or simultaneous global launch — capturing full PTE and six-year data exclusivity rather than the reduced versions available after a delayed China launch — can be modeled as a real option. For high-revenue drugs in large Chinese indication populations, this analysis frequently supports accelerating Chinese development timelines.
Evergreening sustainability: Regulatory and litigation pressure on evergreening practices has increased. The FTC’s pharmaceutical competition investigations, the Actavis antitrust litigation legacy, and PTAB IPR proceedings against secondary patent claims have all increased the cost and risk of aggressive evergreening strategies. Investors should model the probability that secondary patent layers survive challenge, using the PTAB grant rate for IPR petitions as a base rate (historically approximately 60-70% institution, with claim cancellation in a majority of instituted cases) and adjusting for claim quality indicators.
Key Takeaways — Section 7
- Evergreening is a legally protected, systematically constructed portfolio strategy built on continuation prosecution, sNDA submissions, and targeted Orange Book listings. The Humira portfolio demonstrates its multi-decade commercial value at scale — the secondary patent portfolio was a $40-50 billion asset measured by exclusivity extension value.
- Paragraph IV certification economics are driven by 180-day exclusivity. Post-Actavis, settlement structures involving cash payments from brand to generic carry antitrust exposure. Authorized generic strategies by branded companies partially neutralize 180-day exclusivity value.
- China’s Type IV declaration triggers only a nine-month stay on generic approval, compared to the U.S. 30-month stay. Patent holders must have litigation readiness protocols in place before any Chinese generic applications are filed.
- Chinese PTE eligibility requires that the drug not have been marketed overseas before the Chinese approval. Telitacicept’s June 2025 five-year PTE grant is the first practical demonstration of this system at work.
- For a $5 billion annual peak revenue drug, the incremental value of full Chinese PTE versus a reduced term is potentially a $2-5 billion present value difference. This is a material input to any R&D capital allocation model.
Section 8: Biologics and Biosimilars — Patent Thicket Construction and Defense
8.1 The Biologic Patent Estate: Construction Principles
Biologic drugs — therapeutic proteins, monoclonal antibodies, antibody-drug conjugates, gene therapies, cell therapies, and mRNA products — present a distinct patent strategy challenge compared to small molecules. The primary composition patent, covering the molecule by its amino acid sequence or nucleic acid sequence, is typically less vulnerable to generic challenge than an NCE composition patent because biological complexity makes it impossible to produce a “chemically identical” copy. But the patent landscape for a successful biologic is typically broader and more layered than for a small molecule.
A comprehensive biologic patent estate, constructed for maximum commercial durability, covers at minimum the following layers:
Sequence claims cover the specific amino acid sequence of the therapeutic protein or the nucleotide sequence of the encoding gene. These are the primary claims and the hardest to design around, because the biological activity of the molecule depends on its precise sequence.
Cell line and production process claims cover the specific host cell line, culture conditions, bioreactor parameters, and purification processes used to manufacture the biologic. These protect the specific commercial manufacturing process and can capture efficiency improvements that competitors would want to replicate.
Formulation claims cover the specific buffer, excipient, stabilizer, and preservative combination in the commercial drug product. Biologic formulation is technically complex — maintaining protein stability across storage conditions requires specific combinations of stabilizers — and unexpected stability data can support non-obvious formulation claims.
Glycoform and post-translational modification claims cover the specific glycosylation pattern or other post-translational modifications present in the commercial product that contribute to its pharmacokinetic or pharmacodynamic profile. These are particularly valuable because biosimilar manufacturers using different cell lines will produce proteins with different glycoform profiles, potentially allowing the originator to argue that only the specifically claimed glycoform produces the clinical benefits.
Dosing regimen and method-of-use claims cover specific administration schedules and patient population criteria. These are particularly valuable for biologics that achieve best-in-class differentiation through their dosing schedule.
8.2 The BPCIA Patent Dance and Its Strategic Application
The Biologics Price Competition and Innovation Act (BPCIA) of 2009 created a complex pre-litigation information exchange process — the “patent dance” — between biological reference product (BRP) sponsors and biosimilar applicants. The process determines which patents will be litigated before the biosimilar’s commercial launch.
Under the BPCIA, a biosimilar applicant who files an application with FDA must provide the BRP sponsor with a copy of the application and information about the manufacturing process. The BRP sponsor then identifies patents it believes are infringed. The parties negotiate which patents will be litigated in an immediate patent suit. If they cannot agree, the default rule allows the BRP sponsor to choose up to 35 patents for immediate litigation, with the remaining identified patents to be litigated in a later suit.
The 12-year data exclusivity period for biological reference products provides a parallel protection floor that ensures no biosimilar approval can be based on reliance on the BRP sponsor’s clinical data for 12 years after the BRP’s approval, regardless of patent status.
8.3 Chinese Biosimilar Entry and Patent Implications
China is the world’s largest injectable drug market by volume, and the entry of domestic biosimilars for blockbuster biologics including adalimumab (Humira equivalent), bevacizumab (Avastin equivalent), and trastuzumab (Herceptin equivalent) has reshaped pricing and market share dynamics.
For originator biologic companies, the Chinese patent linkage system applies to biologics as it does to small molecules — any invention patent covering the approved biologic can be registered on the CPIRPMD. Process patents covering cell culture and purification are the most practically valuable for Chinese enforcement because they are more likely to be infringed by a biosimilar manufacturer replicating the commercial manufacturing process than by a totally independent developer.
The post-Amgen v. Sanofi implication for biologic patent prosecution in China is strategically significant: CNIPA has not adopted an enablement doctrine as restrictive as the U.S. Supreme Court’s Amgen ruling. Chinese invention patent claims covering antibodies defined by their functional properties are subject to inventive step analysis but not to the same functional claim breadth limitation imposed by the U.S. This creates a jurisdiction-specific opportunity: antibody claims too broad for U.S. prosecution post-Amgen may be appropriately filed in China with reasonable prospects for allowance.
Key Takeaways — Section 8
- Biologic patent estates should be constructed across at least five layers: sequence, cell line/process, formulation, glycoform, and dosing regimen/method-of-use. Each layer adds commercial durability and raises the threshold for biosimilar entry.
- Amgen v. Sanofi (2023) prohibits broad functional antibody claims in the U.S. Sequence-defined or structurally characterized claims are the appropriate standard for U.S. prosecution.
- China has not adopted an Amgen-equivalent enablement limitation. Functional antibody claims may be prosecutable in China at broader scope than in the U.S. — a deliberate jurisdiction-differentiation opportunity.
- The BPCIA patent dance selection of up to 35 patents for immediate litigation must be strategic: prioritize patents most likely to survive invalidity challenge and most likely to result in a preliminary injunction preventing biosimilar launch.
Section 9: AI and Drug Discovery — Patenting Computational Inventions
9.1 The U.S. Eligibility Minefield for AI Drug Discovery Claims
AI is reshaping pharmaceutical R&D across target identification, lead optimization, ADMET prediction, clinical trial design, and manufacturing process control. Insilico Medicine, Recursion Pharmaceuticals, and Exscientia have each advanced AI-designed molecules into clinical trials. But the U.S. patent system has not kept pace with the technical reality.
Under the Alice/Mayo two-step test, an AI algorithm is frequently characterized as an “abstract idea.” Most AI drug discovery claims — algorithms that predict protein structure, generate molecular candidates, or identify biomarkers from genomic data — are directed to mathematical concepts or mental processes, falling within the abstract idea exception. The second step of Alice asks whether the claim adds “significantly more” than the abstract idea itself. Simply implementing the algorithm on a computer, or applying it in a pharmaceutical context, is generally insufficient.
The practical drafting strategy for AI drug discovery claims in the U.S. focuses on “technical improvement” framing: claims that present the AI method as improving the computer system itself (e.g., a more efficient training algorithm for a specific neural network architecture) or that are specifically tied to the concrete production of a specific chemical compound or experimental result tend to fare better under Alice step two. The specific architecture of the neural network, the specific training data set, the specific input/output parameters, and the specific application to a concrete drug target should all be explicitly recited to anchor the claim in practical application.
9.2 China’s Technical Solution Framework: More Permissive for AI Claims
CNIPA’s 2024 “Guidelines for Patent Applications for Artificial Intelligence” clarify that AI algorithms are patentable when they are directed to a “technical solution” — one that uses “technical means” to achieve a “technical effect.” The focus is on whether the AI method solves a technical problem and produces a technical result, rather than on the abstract/concrete distinction that drives U.S. §101 analysis.
In practice, an AI method for predicting binding affinity of candidate molecules to a specific protein target satisfies Chinese patentability requirements when the specification demonstrates that the method takes specific technical inputs (molecular descriptor data, protein structural data), uses a specific technical architecture (a defined neural network trained on specific data), and produces a specific technical output (a numerical affinity score with demonstrated predictive validity). The method solves the technical problem of efficient lead compound identification and produces a technical effect measured by the accuracy of the affinity prediction. This framing is not subject to a §101-equivalent eligibility rejection.
China accounts for over 70% of all global AI patent filings and an even higher share in generative AI applications. Chinese companies including XtalPi, Deep Pharmaceutical, and TenboMed are filing AI drug discovery patents in China at scale. The Chinese domestic patent grants for AI drug discovery methods will create FTO complications for U.S. and European companies operating AI-assisted discovery programs in China.
9.3 Inventorship in the Age of AI-Assisted Drug Discovery
Both the USPTO and CNIPA currently require that an inventor be a natural person who made a significant intellectual contribution to the conception of the invention. An AI system cannot be named as an inventor. This creates a compliance challenge for AI-assisted drug discovery programs where the boundary between human intellectual contribution and AI-generated output is increasingly blurry.
The USPTO’s guidance on AI-assisted inventions requires that a human inventor have made a “significant contribution” to the conception of each claim. For an AI drug discovery program where the human researcher defines the target, selects training data, validates the model architecture, and exercises judgment in selecting AI-generated candidates for synthesis and testing, the human inventorship argument is generally supportable.
This uncertainty is commercially material. The patent strategy implication is to document human decision-making at each stage of the discovery process — target selection, model architecture decisions, data set curation, lead selection criteria — to support inventorship claims. Companies that fail to maintain this documentation risk having their AI-derived patents challenged on inventorship grounds.
Key Takeaways — Section 9
- U.S. Alice/Mayo eligibility is the primary prosecution risk for AI drug discovery patents. Claims must emphasize specific technical architecture, specific training parameters, and specific practical outputs to survive §101 scrutiny.
- CNIPA’s 2024 AI guidelines take a technical solution approach that is more permissive for AI drug discovery claims than the U.S. §101 framework. AI drug discovery methods that solve a defined technical problem and produce a measurable technical effect are generally allowable in China.
- China’s 70%+ share of global AI patent filings creates FTO risk for foreign companies operating AI discovery programs in China. Systematic monitoring of CNIPA AI drug discovery grants is now routine IP due diligence.
- Human inventorship documentation at every stage of AI-assisted discovery programs is essential to support patent validity against inventorship challenges.
Section 10: Portfolio Valuation — How Patent Position Affects Drug Asset Pricing
10.1 The Patent Cliff as a Financial Event
In pharmaceutical M&A and equity markets, the patent cliff is the single most scrutinized event in a drug company’s financial forecast. When the primary composition patent for a blockbuster drug expires or is invalidated, generic entry typically drives price declines of 70-90% within 12-18 months, and revenue can fall by 60-80% within two to three years. The patent cliff for Pfizer’s Lipitor in 2011 — with $10 billion in U.S. annual revenues at peak — reduced U.S. sales to under $1 billion within two years of generic entry.
For institutional investors, the patent cliff analysis requires modeling each of the following variables for each material asset:
Effective patent expiry date: The later of (a) the primary composition patent expiration date, adjusted for PTA and PTE; (b) the expiration date of the last Orange Book-listed secondary patent that must be successfully challenged before generic entry; and (c) any non-patent exclusivity periods including NCE data exclusivity, orphan drug exclusivity, or pediatric exclusivity. The gap between nominal and effective patent expiry is the practical measure of evergreening value.
Paragraph IV litigation probability and timeline: The probability that a generic company will file a Paragraph IV certification, the expected court timeline assuming a lawsuit is filed within 45 days, and the historical success rate of Paragraph IV challenges to each patent layer. For formulation patents, the historical invalidity rate is high — above 60% — reflecting the vulnerability of obvious formulation claims. For well-constructed NCE composition patents, the invalidity rate is lower, typically 30-50%.
Authorized generic strategy: Many branded companies launch their own authorized generic at the time of first generic entry to capture a share of the 180-day exclusivity period and mitigate revenue cliff depth. Modeling the authorized generic competition reduces the revenue impact of the first generic entry but also reduces the value of primary composition patent protection.
10.2 IP Valuation Methodologies for Drug Assets
Drug asset valuation for M&A and licensing transactions is generally performed through risk-adjusted net present value (rNPV) analysis, where the future revenue streams from the drug product are discounted by both a probability of technical and regulatory success and a capital charge. Patent position enters this model at several points:
The primary patent expiration date defines the terminal value window during which the drug generates monopoly-equivalent revenues at full pricing power. Extending this window by one year for a drug generating $2 billion in net profits increases the asset’s rNPV by approximately $1.5-1.8 billion, assuming a discount rate of 10-12%.
Secondary patent layers add optionality value: the probability-weighted expected duration of each layer of protection beyond the primary patent contributes incremental present value. The market consistently prices this optionality, as demonstrated by AbbVie’s ability to sustain its premium valuation through the Humira biosimilar wave because investors correctly modeled that the secondary patent thicket would delay biosimilar adoption.
China-specific patent rights contribute to asset value through two channels: Chinese market revenue exclusivity and global supply chain enforcement leverage. For oncology drugs with large Chinese patient populations, Chinese patent protection at full PTE value can contribute 10-20% of total global asset NPV.
10.3 Due Diligence Standards for Pharmaceutical IP
In pharmaceutical M&A, patent due diligence has a defined scope that investors and acquirers should understand as a quality signal. The minimum standard for a primary drug asset acquisition includes:
Freedom-to-operate analysis: Confirmation that the drug can be manufactured, sold, and used without infringing valid third-party patents in all material jurisdictions.
Patent coverage analysis: Confirmation that the claims as granted actually cover the commercial drug product in each relevant jurisdiction, with particular attention to claim scope after any prosecution history estoppel events.
Validity assessment: Review of the closest prior art and an assessment of the probable outcome of a Paragraph IV challenge or IPR proceeding against the key Orange Book-listed patents.
Orange Book listing audit: Confirmation that all listable patents are actually listed, and that the listing is accurate with respect to which aspects of the product each patent covers.
China-specific assessment: Review of Chinese patent filings, CNIPA prosecution history, and the CPIRPMD registration status for any drug with material China market revenues.
Key Takeaways — Section 10
- Effective patent expiry — accounting for PTA, PTE, secondary patents, and non-patent exclusivity — routinely differs from nominal expiry by 3-7 years. This gap is the measure of evergreening value and a primary source of valuation error in financial models.
- Each additional year of exclusivity for a $2 billion operating profit drug is worth approximately $1.5-1.8 billion in asset NPV at a 10-12% discount rate. Patent term optimization is a multi-billion dollar capital allocation decision.
- China-specific patent rights contribute 10-20% of total global asset NPV for drugs with large Chinese indication populations, when modeled with full PTE and data exclusivity.
- Standard pharmaceutical IP due diligence for an M&A target must include a China-specific assessment: CNIPA prosecution history, CPIRPMD registration status, and Type IV declaration exposure.
Section 11: Recent Reforms and the Legislative Pipeline
11.1 U.S. Legislative Proposals: PERA, PREVAIL, and RESTORE
Three bipartisan bills are advancing through Congress as of 2025 that would collectively represent the most significant legislative revision to U.S. patent law since the America Invents Act of 2011. Each targets a specific perceived weakness in the current system.
The Patent Eligibility Restoration Act (PERA) would eliminate the judicially created exceptions to §101 patent eligibility by statute. The effect would be to overrule Alice Corp. v. CLS Bank and Mayo Collaborative Services v. Prometheus Laboratories — the two Supreme Court decisions that created §101 uncertainty particularly damaging to pharmaceutical diagnostic method patents, AI invention patents, and software-based medical device patents.
For pharmaceutical companies, PERA’s most immediate impact would be on companion diagnostic patents. Claims covering methods of measuring metabolite levels to guide drug dosing, genetic testing methods identifying patients likely to respond to a drug, and biomarker-based disease staging methods would no longer face Mayo-based §101 rejections. This would restore robust patent protection for the precision medicine IP portfolio that has been largely stranded in §101 uncertainty since 2012.
The PREVAIL Act would reform PTAB proceedings. The most commercially significant provisions: raising the standard for invalidating a patent from “preponderance of the evidence” to “clear and convincing evidence” (the same standard used in district court); requiring that PTAB challengers have standing to file; and prohibiting serial petitions challenging the same patent multiple times. These changes would meaningfully reduce PTAB’s utility as a generic company tool for challenging branded pharmaceutical patents during Hatch-Waxman litigation. Generic companies have routinely filed IPR petitions against branded drug patents simultaneously with district court Paragraph IV defenses, creating dual-track invalidity pressure at lower combined cost than district court litigation alone. PREVAIL would constrain this strategy.
The RESTORE Act would address the post-eBay injunction problem by creating a rebuttable presumption that a permanent injunction should be granted upon a finding of infringement. This provision primarily benefits practicing entities and would have modest direct impact on Hatch-Waxman pharmaceutical litigation, where injunctions after a successful trial are already relatively available to practicing branded companies.
If all three bills pass in their current form — which remains uncertain — the combined effect on pharmaceutical IP would be an increase in the value and enforceability of branded drug patents across all claim categories. This would mechanically improve the NPV of pharmaceutical R&D pipelines and increase barriers to early generic entry.
11.2 China’s Quality Revolution: 2024 Implementing Regulations and the 2025 Work Plan
China’s 2024 Implementing Regulations of the Patent Law, which entered force in January 2024, are the most comprehensive revision to Chinese patent prosecution rules in over a decade. Key pharmaceutical-relevant provisions:
The PTA system for Chinese invention patents now compensates patent holders when CNIPA delays extend prosecution beyond statutory examination windows. For pharmaceutical companies with Chinese invention patent applications filed more than 3-5 years ago, PTA analysis should be a routine part of portfolio management to calculate the true effective patent term.
The “good faith” principle codified in the 2024 regulations adds a conduct dimension to Chinese patent prosecution. Applicants who engage in “abnormal” prosecution behaviors — filing multiple nearly identical applications, using patent agencies to file large batches of applications without genuine inventive content — face administrative sanctions and patent cancellation.
CNIPA’s 2025 Work Plan explicitly targets the cleanup of “abnormal” patents while accelerating substantive examination for high-quality applications in priority technology areas including biopharmaceuticals, AI, and new energy. For foreign pharmaceutical companies, well-prepared applications with clear technical problem/effect framing and strong comparative data examples should expect more efficient examination, not more resistance.
Key Takeaways — Section 11
- PERA, if enacted, would restore §101 patent eligibility for companion diagnostic methods and biomarker-based precision medicine IP — a category currently unprotectable in the U.S. under Mayo/Alice. The financial value unlocked for the precision medicine sector would be substantial.
- PREVAIL Act would raise PTAB invalidity burden to “clear and convincing evidence” and restrict serial IPR petitions. This would reduce generic challengers’ ability to run parallel PTAB/district court campaigns against Orange Book patents.
- China’s 2024 implementing regulations introduced PTA, a good-faith prosecution principle, and accelerated examination for biopharmaceutical priority technology areas. Foreign pharma applications in priority technology areas should see more efficient examination.
- CNIPA’s deliberate 29% reduction in invention patent grants in H1 2025 signals that the quality-over-quantity pivot is real, sustained, and supported by enforcement action against subsidy-driven low-quality filers.
Section 12: Geopolitical Risk — IP as an Instrument of Industrial Policy
12.1 Anti-Suit Injunctions and the Weaponization of IP Jurisdiction
The most aggressive recent development in U.S.-China IP conflict is China’s deployment of anti-suit injunctions (ASIs) by Chinese courts in global patent disputes. An anti-suit injunction is a court order that prohibits a party from pursuing litigation in another jurisdiction. Chinese courts have issued ASIs in SEP licensing disputes that prohibit foreign patent holders from litigating their patents in European or U.S. courts — on penalty of enormous daily fines for non-compliance — while the Chinese rate-setting proceeding proceeds to completion.
The practical effect is a jurisdictional power grab: by setting a “global FRAND rate” for Standard-Essential Patents in a Chinese proceeding, and then prohibiting the SEP holder from pursuing enforcement at that rate in other jurisdictions, Chinese courts have arrogated to themselves the authority to determine the licensing economics for technology incorporated into every smartphone and broadband device sold worldwide.
For pharmaceutical companies, ASIs in their pure patent licensing form are less immediately applicable because drug patents are generally not SEPs. But the underlying mechanism — using domestic courts to project jurisdictional authority over global IP disputes — is a demonstrated tool. As Chinese pharmaceutical companies including Hengrui, BeiGene, and Zymeworks China build significant global patent portfolios, the risk that this mechanism is applied in pharmaceutical licensing disputes increases.
12.2 Compulsory Licensing: Latent Risk for High-Revenue Drugs
China’s compulsory licensing provisions remain a latent risk for pharmaceutical companies with major Chinese market exposure. Article 48 of the Chinese Patent Law permits compulsory licensing when a patentee has not exploited the patent without justification for three years from grant (or four years from filing), or where exploitation is for national emergency purposes. The COVID-19 pandemic demonstrated that multiple governments, including China, were prepared to invoke or threaten emergency compulsory licensing to ensure domestic access to antiviral and vaccine technologies.
For pharmaceutical companies with Chinese manufacturing partners, joint ventures, or technology transfer agreements, the compulsory licensing risk is managed through contract design: the licensing agreement should specify minimum exploitation standards, include audit rights, and be structured to avoid triggering the “not exploited” standard.
12.3 Export Controls and Their Intersection with IP
U.S. export control regulations — particularly Entity List additions and restrictions under the Export Administration Regulations (EAR) — increasingly affect the ability of U.S. pharmaceutical companies to engage in technology transfer activities with Chinese partners. An IP licensing deal that involves the transfer of controlled technology (including certain biotechnology know-how and dual-use materials) to a Chinese entity requires export license authorization. The intersection of export controls and IP licensing is a rapidly evolving compliance area that must be integrated into any China-related IP transaction.
Key Takeaways — Section 12
- Chinese ASIs in SEP disputes have demonstrated China’s willingness to use its courts to project jurisdictional authority over global IP licensing. The pharmaceutical industry should monitor this mechanism’s potential extension to biopharmaceutical licensing disputes as Chinese domestic companies become larger global IP holders.
- Compulsory licensing provisions in China are a real risk for pharmaceutical companies with drugs addressing public health emergencies. Contract structures for China market IP licensing should include minimum exploitation commitments and audit rights.
- Export control compliance is now an integral part of China IP licensing deal structuring. Technology transfer involving controlled biotechnology know-how requires EAR license analysis before execution.
Section 13: Investment Strategy — Actionable Frameworks for Analysts
13.1 Evaluating Patent Position in Pharmaceutical Equity Models
For pharmaceutical equity analysts, patent position analysis is a required input to any drug-by-drug revenue model. The following framework systematizes this analysis:
Step 1 — Map the Orange Book. For each U.S.-marketed product, identify all patents listed in the FDA Orange Book, their expiration dates (nominal and adjusted for PTA/PTE), and whether they have been challenged by a Paragraph IV certification. The existence of an unchallenged Paragraph IV certification from a major generic company is a forward indicator of imminent litigation.
Step 2 — Score patent layer vulnerability. Assign probability-of-invalidity estimates to each Orange Book-listed patent by layer. Use published Paragraph IV litigation success rates as base rates (approximately 60-70% invalidity rate for formulation patents, 30-50% for NCE composition patents), and adjust for specific patent quality indicators: examination history, number of prior art references cited, claims specificity, and secondary consideration evidence available.
Step 3 — Model the Chinese IP optionality. For any drug with estimated China market revenues exceeding $200 million at peak, model the incremental NPV contribution of full Chinese PTE and data exclusivity versus a reduced-term scenario. This calculation requires: (a) the China marketing approval timeline estimate; (b) the applicable PTE and data exclusivity reduction for any expected delay relative to first global approval; and (c) the revenue contribution of the China market during the extended exclusivity period.
Step 4 — Assess PTAB exposure. Identify whether any material patents have been petitioned at PTAB. Use the PTAB institution rate (historically approximately 60-70% for pharmaceutical patents) and claim cancellation rate (approximately 70% of challenged claims in instituted proceedings) as probability inputs. Model the revenue impact of the claim cancellation scenario.
Step 5 — Evaluate the biosimilar pathway for biologic assets. For biologic drugs within 3-5 years of BLA approval, assess the 12-year data exclusivity cliff separately from the patent cliff. Model the probability and timeline of biosimilar entry based on the number of filed biosimilar applications (visible through FDA’s Purple Book) and the likely price erosion trajectory based on comparable biosimilar launch histories. Adalimumab biosimilars achieved approximately 85% price discount from reference product within 18 months of U.S. launch — a useful benchmark for modeling competitive biologic scenarios.
13.2 Patent Position as an M&A Signal
In pharmaceutical M&A, the acquirer’s IP due diligence process generates information not available to public market investors, but whose conclusions drive acquisition premium justifications. For institutional investors evaluating acquisition rumors or announced deals, the patent position indicators publicly available include:
Orange Book patent count and expiry spread: A drug with numerous Orange Book-listed patents with staggered expiration dates through the 2030s has a more defensible IP position than one with two patents expiring in 2027. Count and expiry spread are observable without non-public information.
Paragraph IV litigation history: Visible through PACER court filings and FDA’s Orange Book challenge notifications. A drug that has successfully defended three Paragraph IV challenges over five years has a demonstrated patent estate quality signal.
CNIPA patent registration count and claim structure: Visible through CNIPA’s public patent database. A drug with 15 Chinese patent applications covering multiple claim layers — NCE composition, formulation, process, method-of-use — across two prosecution firms is better positioned for China exclusivity than one with a single Chinese filing still pending.
13.3 Risk-Adjusted Portfolio Allocation Considering China IP Dynamics
For institutional investors with pharmaceutical sector allocations, the China IP dynamics described throughout this guide create the following portfolio construction considerations:
Companies with drugs in pre-commercial development that have not yet received global marketing approval have the ability to sequence their China launches to maximize PTE and data exclusivity — a real option with positive expected value. Portfolio screens that identify companies with late-stage development pipelines and major China indication populations (oncology, metabolic disease, cardiovascular) that have announced China-first or simultaneous global regulatory strategies should flag these companies as potential beneficiaries of the 2021 Patent Law incentive structure.
Companies with drugs currently marketed in the U.S. but not yet in China face a more complex calculus: full Chinese PTE is likely unavailable because the drug has already been approved overseas, and the six-year data exclusivity term will be reduced for any application delay. The NPV impact of the data exclusivity reduction should be modeled for any China-market-material drug.
Companies with large Chinese manufacturing footprints or contract manufacturing relationships in China face an additional dimension of IP risk: FTO complexity created by China’s utility model patent thicket for any product with physical device components manufactured in China. Due diligence on Chinese partner company patent portfolios is now a routine part of manufacturing relationship risk assessment.
Key Takeaways — Section 13
- The five-step patent position analysis framework (Orange Book mapping, layer vulnerability scoring, China IP optionality modeling, PTAB exposure assessment, biosimilar pathway evaluation) is the standard analytical toolkit for pharmaceutical equity modeling.
- Orange Book patent count and expiry spread, Paragraph IV litigation history, and CNIPA patent registration scope are all publicly observable proxies for patent estate quality.
- Companies with pre-commercial assets and major China indication populations have a real and positive-NPV option to sequence global launches to maximize Chinese PTE and data exclusivity. This option is a differentiating value driver that public market models frequently underweight.
- Chinese manufacturing relationships create FTO risk from the domestic utility model patent thicket. This risk is structural, not transient, and requires ongoing active monitoring and contract-level IP risk management.
Conclusion: The Two Systems, One Strategy
The U.S. and Chinese patent systems will continue to diverge in their foundational logic while converging in specific procedural features. The U.S. system is moving toward stronger, more certain patent rights if the PERA/PREVAIL/RESTORE legislative package advances — but it remains a rights-based, adversarial, private property framework where value is realized through litigation. The Chinese system is moving toward fewer but higher-quality patents, with China-specific incentive structures — PTE, data exclusivity, patent linkage — explicitly engineered to alter global pharmaceutical commercial strategy in ways that favor China-first development.
For pharmaceutical IP teams, R&D leads, and institutional investors, the practical implication is not a choice between these systems but a requirement to operate in both simultaneously, with each jurisdiction informing the strategy in the other. The drafting choices made in a Chinese prosecution specification should be made with U.S. secondary considerations in mind. The launch sequencing decision that determines U.S. FDA priority should be made with Chinese PTE optionality in its financial model. The antibody claims too broad for U.S. prosecution post-Amgen should be evaluated for Chinese filing suitability on their own terms.
This simultaneous, mutually informed approach to global pharmaceutical IP strategy is no longer a best practice recommendation. Given the financial stakes — where a single IP decision can swing a drug asset’s value by billions of dollars — it is a baseline operational requirement.
This article reflects publicly available regulatory guidance, court decisions, and commercial intelligence current as of March 2026. Patent prosecution costs, PTAB statistics, and Chinese patent grant data are subject to change. This article is for informational purposes and does not constitute legal advice.