I. Executive Summary
Pharmaceutical patents are not legal formalities. They are the single most important determinant of whether a drug generates $50 million or $50 billion in revenue over its commercial life. A compound patent filed one year too early can expire before peak sales materialize. A poorly constructed secondary patent on a formulation can be invalidated in 90 days of inter partes review. A missing Paragraph IV certification from a generic challenger can unlock billions in uncontested exclusivity that no one planned for.
This guide maps the full architecture of pharmaceutical patent strategy, from first-in-class composition-of-matter claims through the layered secondary patent portfolios that now define every major biologic on the market. It covers the mechanics of Hatch-Waxman, the structured information exchange of the Biologics Price Competition and Innovation Act (BPCIA), the post-Myriad landscape for gene therapy patenting, and the still-unsettled question of AI inventorship under 35 U.S.C. § 101.
The core argument running through all of it: the shift from protecting a single active pharmaceutical ingredient to managing a portfolio of 70-plus overlapping claims is not just a legal tactic. It is the dominant business model in branded pharmaceuticals, and understanding it precisely is a prerequisite for anyone valuing a drug asset, planning a generic launch, or structuring a licensing deal.
Key Takeaways — Executive Summary
- A drug’s effective market exclusivity averages 7 to 8 years after launch, versus 20 years from first patent filing — the gap is the strategic problem the entire patent lifecycle management industry exists to solve.
- Secondary patents on formulations, methods of use, polymorphs, and delivery devices have become the primary mechanism for extending revenue beyond primary compound expiry.
- Biosimilar interchangeability designations and the BPCIA patent dance create negotiation leverage that pure litigation cannot replicate.
- AI-assisted drug discovery is breaking the legal concept of human inventorship, and patent offices have not resolved this yet — which creates both risk and competitive opportunity.
- Jurisdiction-specific claiming is not optional for any drug with global revenue expectations. A method-of-treatment claim valid in the U.S. is excluded subject matter in the EU, Japan, and China without careful reformulation.
Investment Strategy Note — Portfolio Analysts
- When modeling loss-of-exclusivity (LOE) dates, do not anchor to the primary compound patent. Map every Orange Book-listed patent, every PTAB challenge filed in the prior 24 months, and every pending ANDA Paragraph IV certification.
- A single successful inter partes review (IPR) petition can collapse 3 to 5 years of assumed exclusivity in a single decision. Price that optionality into your DCF.
- AbbVie’s Humira playbook — over 132 secondary patents extending U.S. exclusivity to 2023 versus European biosimilar entry in October 2018 — produced roughly $75 billion in additional U.S. revenue that the European market did not generate. That is the value ceiling for a maximally executed secondary patent strategy on a blockbuster biologic.
II. Foundational Concepts: Anatomy of a Pharmaceutical Patent Claim
What a Patent Claim Actually Does
A patent grants its owner the right to exclude others from making, using, selling, offering for sale, or importing the claimed invention for 20 years from the application’s filing date. That right is geographically limited: a USPTO patent stops at the U.S. border, and a granted European Patent is enforceable only in the member states where it has been validated and maintained with ongoing annuity fees. The claims — the numbered sentences at the end of every patent — define the legal boundary of that exclusion right.
Patent attorneys describe effective claim drafting as capturing the broadest defensible embodiment of an invention. This is not abstract. Claim breadth directly determines whether a generic company can design around a patent by introducing an element not listed, whether a biosimilar applicant can avoid infringement by using a slightly different manufacturing process, and whether an ANDA filer’s Paragraph IV certification will survive a 30-month litigation stay. In pharmaceuticals, a single transitional word — “comprising” versus “consisting of” — can mean a difference of hundreds of millions of dollars in contested revenue.
The Three Transitional Phrases That Define Claim Scope
Comprising is an open-ended, inclusive term. A composition claim reciting elements A, B, and C using “comprising” covers any composition that contains A, B, and C even if it also contains D, E, and F. This breadth is generally preferred for primary composition claims because it prevents circumvention through minor additive modifications. The tradeoff is that it can be harder to distinguish from prior art that also contains A, B, and C in a different context.
Consisting of closes the claim entirely. A formulation claim using “consisting of” covers only the exact elements listed. A competitor who introduces a single additional excipient, even a pharmacologically inert one, may avoid infringement entirely. This language appears most often in Markush claims defining a finite chemical genus and in formulation patents where the exact composition is the point of differentiation — for instance, a proprietary pH buffer system.
Consisting essentially of occupies the middle position. It permits additional elements only when they do not materially affect the basic and novel characteristics of the invention as defined in the specification. The practical enforceability of a “consisting essentially of” claim depends heavily on how precisely the specification defines those “basic and novel characteristics.” A vague definition invites indefiniteness challenges under 35 U.S.C. § 112. A well-defined one creates a claim scope that is narrower than “comprising” but far more resistant to design-around than “consisting of.”
For pharmaceutical IP teams, the transitional phrase selection at claim drafting is not a stylistic choice. It is a strategic commitment that ripples through every future litigation, IPR proceeding, and licensing negotiation touching that patent.
Claim Structure: Preamble, Transitional Phrase, Body
Every patent claim has three components. The preamble names the category of invention — a compound, a pharmaceutical composition, a method of treatment. The transitional phrase defines the open or closed nature of the claim body. The body enumerates the essential elements or steps of the invention. Independent claims stand alone; dependent claims incorporate all elements of the parent claim and add limitations. A strong pharmaceutical patent typically has one or two broad independent claims covering the compound or core method, with multiple dependent claims adding specific dosing ranges, formulation parameters, or patient subpopulations. This layered structure means that even if an independent claim is invalidated in litigation, dependent claims can sometimes survive and retain commercially meaningful protection.
III. The Seven Core Claim Types and Their Strategic Utility
Pharmaceutical products rarely rely on a single patent. Companies build what practitioners call a multi-layered web of protection — a portfolio covering every commercially significant aspect of a product. The architecture of that web is built from the claim types below.
Composition of Matter (Product) Claims
These are the broadest and most valuable claims in pharmaceutical IP. A composition-of-matter patent covers the active pharmaceutical ingredient itself, preventing any party from making, using, or selling that compound regardless of the indication, formulation, or delivery route. The drug can be claimed by its chemical name, its structural formula, or within a Markush genus covering a range of structurally related compounds. Empagliflozin, marketed as Jardiance by Eli Lilly and Boehringer Ingelheim, illustrates the layered approach: the compound is protected by a foundational composition claim, with additional patents covering its specific crystalline Forms I and II, which themselves carry IP value separate from the base molecule.
IP Valuation Spotlight — Empagliflozin / Jardiance (Eli Lilly / Boehringer Ingelheim)
Jardiance generated approximately $6.9 billion in revenue in 2023, with continued growth driven by its expanded cardiovascular and chronic kidney disease indications. The foundational composition patent, covering the empagliflozin molecule, is the anchor asset. The polymorph patents on crystalline Forms I and II extend protection into the early 2030s and represent a meaningful independent IP layer because a generic seeking to avoid the compound patent would still need to use one of these crystal forms in any commercially viable tablet formulation. The method-of-use patents covering heart failure reduction (the EMPEROR-Reduced trial basis) add further years of clinical-indication exclusivity. Analysts modeling LOE should map all three patent layers — compound, polymorph, and method of use — before projecting generic entry timing.
Process (Manufacturing) Claims
Process claims cover the specific method used to manufacture a pharmaceutical. They protect proprietary synthesis routes, purification techniques, and production conditions. Their commercial value is highest when the process produces a material improvement in yield, purity, or cost that competitors cannot replicate without the claimed steps. Process claims are also strategically important in biologics manufacturing, where the specific cell line culture conditions, glycosylation control parameters, and downstream purification train can be claimed independently of the biologic molecule itself. This is one reason why biosimilar manufacturers must demonstrate manufacturing equivalence through extensive analytical characterization, not just structural similarity — a competitor’s biologic that uses the innovator’s patented fermentation conditions may infringe a process patent even if the resulting protein is analytically indistinguishable.
Method of Use Claims
Method-of-use patents cover a specific therapeutic application of a known compound. They are the primary vehicle for indication expansion and repurposing. Finasteride was originally approved for benign prostatic hyperplasia; method-of-use patents later secured protection for its androgenic alopecia indication. Bupropion was marketed for depression before method-of-use patents covered smoking cessation. Pfizer’s sildenafil (Viagra) began as a cardiovascular candidate before method-of-use patents on its phosphodiesterase-5 inhibition in erectile dysfunction generated revenues that far exceeded what the original cardiovascular indication would have produced.
Method-of-use claims carry a specific enforceability asymmetry. A generic manufacturer approved for the original indication cannot be sued for direct infringement for practicing the new use — but the brand company can pursue induced infringement claims if the generic’s product labeling encourages physicians or patients to use the drug for the patented indication. This has led to so-called “skinny label” strategies, where generics seek approval for a subset of indications, deliberately carving out the patented one from their label to avoid inducement liability.
Formulation Claims
Formulation patents cover the pharmaceutical composition as a whole: the specific combination of active ingredient, excipients, carriers, pH modifiers, and dosage architecture. Extended-release formulations are the most commercially significant subtype. AstraZeneca’s Seroquel XR and Bristol-Myers Squibb’s Glucophage XR are textbook examples of brand companies successfully shifting patient populations to patent-protected modified-release versions before the immediate-release compound patent expired. Extended-release formulations can carry genuine clinical benefits — reduced peak plasma concentration variability, improved tolerability, once-daily dosing compliance — which strengthens both the commercial and the patent arguments for the new dosage form.
Formulation patents require careful prosecution. A generic challenger filing a Paragraph IV certification against a formulation patent must argue either that the formulation is obvious over the prior art (usually because the specific excipients and release mechanisms were known) or that the generic’s formulation does not meet each element of the claimed formulation. Innovator companies strengthen formulation patents by tying the specific excipient combination to a quantifiable clinical benefit, making an obviousness argument harder to sustain in litigation.
Combination Claims
Combination patents protect fixed-dose combinations of two or more active ingredients. They are particularly prevalent in HIV, oncology, and cardiovascular indications where combination therapy is the clinical standard of care. The commercial logic is straightforward: a combination product that contains two agents whose individual compound patents have different expiry dates can be protected as a unified dosage form under a combination patent that runs independently of either component patent. The combination patent must demonstrate that the combination produces an effect — whether synergistic, pharmacokinetic, or compliance-based — that is more than additive, to survive an obviousness challenge.
Product-by-Process Claims
These claims define a product by the process used to make it, rather than by the product’s intrinsic structural characteristics. They are used when a product’s structure cannot be fully characterized through existing analytical tools or when the product is inherently difficult to define in structural terms alone. The key legal point: patentability of a product-by-process claim rests on the novelty of the product, not the process. A product that is structurally identical to a prior art compound is not rendered novel simply because a new process is used to make it. In biologics, product-by-process claims appear in situations where a specific manufacturing process produces a biologic with a glycosylation profile or aggregate content that is functionally distinct from prior art forms but analytically difficult to fully characterize in structural terms alone.
Markush Claims
Named for chemist Eugene Markush, these claims describe a chemical genus using a core scaffold with variable substituents. The standard format claims a compound of a given formula wherein a specific position is “selected from the group consisting of” a defined list of alternatives. Markush claims are the primary tool for claiming a genus of pharmacologically active compounds in early drug discovery, before a lead candidate has been identified. A well-constructed Markush claim filed at the time of initial synthesis can cover thousands of structural variants, blocking competitors from making minor structural modifications to design around the core scaffold. The prosecution tradeoff is that a very broad Markush claim must be supported by adequate experimental data across the claimed genus — a weak genus claim can be restricted by the USPTO or invalidated in litigation for lack of enablement across its full scope.
A single well-constructed Markush claim can cover thousands of structural variants from a single filing date. The genus protection it provides is why lead optimization programs generate IP value before any single candidate reaches IND.
Table 1: Pharmaceutical Patent Claim Types — Scope, Strength, and Litigation Profile
| Claim Type | What It Protects | Primary Strategic Value | Key Litigation Vulnerability |
|---|---|---|---|
| Composition of Matter | The API itself — structure, formula, or chemical genus | Broadest exclusion right; blocks all uses of the compound | Prior art compounds; obviousness of structural modifications |
| Process / Manufacturing | Method of synthesis, purification, or production conditions | Protects proprietary manufacturing efficiency and biologic production parameters | Alternative routes; reverse engineering of process from product |
| Method of Use | Specific therapeutic indication or dosing regimen | Enables indication expansion; supports skinny-label avoidance claims | Obviousness over known pharmacology; induced infringement proof |
| Formulation | Excipient combination, release mechanism, dosage form architecture | Extends exclusivity after compound patent expiry; tied to branded product differentiation | Prior art excipient combinations; lack of unexpected results |
| Combination | Fixed-dose combination of two or more active ingredients | Protects combination therapy standard of care; independent from component patent terms | Obviousness of combining individually known agents; lack of synergy |
| Product-by-Process | A product defined by the process producing it | Covers biologics or complex molecules with difficult structural characterization | Structural identity with prior art product; novelty must lie in product, not process |
| Markush | A genus of compounds sharing a core scaffold with variable substituents | Blocks design-around by competitors during lead optimization; broad genus protection from single filing | Enablement across full genus scope; written description requirement for full range |
Table 1. Claim types ranked by scope breadth, with primary litigation attack vectors for generic and biosimilar challengers.
IV. Historical Evolution: From Venetian Statute to Hatch-Waxman
The Origins: Process Patents, Not Product Patents
The modern pharmaceutical patent system did not begin with drugs. The Venetian Statute of 1474 — often cited as the first codified patent law — protected industrial processes, not chemical substances. England’s Statute of Monopolies (1624) narrowed patent rights to “new manufactures,” laying groundwork that American law would inherit through the Patent Acts of 1790, 1793, and 1836. For most of the 19th century, pharmaceutical compounds could be patented in the U.S. only as processes, not as product compositions. Direct product patents on chemical compounds were legally contestable and commercially unreliable.
Three decisions reshaped the landscape. Hotchkiss v. Greenwood (1850) introduced non-obviousness as a formal patentability requirement, establishing that an invention must represent more than what a person skilled in the relevant art would have found routine. O’Reilly v. Morse (1853) restricted patent eligibility for claims that were effectively abstract ideas untethered to any particular machine or process — a doctrine that would resurface more than 160 years later in AI-related patent disputes. And Diamond v. Chakrabarty (1980) was the ruling that opened biotechnology: the U.S. Supreme Court held that a live, human-made microorganism is patentable subject matter, with the now-famous statement that Congress intended patentable subject matter to include “anything under the sun that is made by man.” That ruling created the legal foundation for the entire modern biopharmaceutical industry.
In Europe, the European Patent Convention (EPC), in force from 1977, took a different path on pharmaceutical product patents. Unlike earlier national laws that limited protection to manufacturing processes, the EPC allowed claims covering chemical compounds directly — though with explicit carve-outs for methods of treatment of the human body, which remain excluded from patentability across EPC member states to this day. This divergence between U.S. and European patentability for treatment methods has shaped global patent strategy ever since.
The Pre-Hatch-Waxman Problem: Testing That Could Not Happen
Before 1984, U.S. patent law had no mechanism allowing generic manufacturers to conduct the testing required for FDA approval before a brand-name drug’s patents expired. The practical result was that even after patent expiry, it could take several more years for a generic product to reach the market, because the testing process could only legally begin at the moment of expiry. The Roche Products v. Bolar Pharmaceutical case (1984) made this problem explicit: the Federal Circuit held that Bolar infringed Roche’s patent by using the patented compound in bioequivalence testing before expiry, even though the purpose was solely to prepare an FDA submission. Congress responded the same year with the Hatch-Waxman Act, which resolved the Bolar problem and remade the competitive structure of the entire pharmaceutical industry in the process.
Key Takeaways — Historical Evolution
- Direct product patents on pharmaceutical compounds only became legally reliable in the U.S. in the 20th century; the pre-Hatch-Waxman system left generics in an impossible position — unable to test before expiry, unable to enter the market after.
- Diamond v. Chakrabarty (1980) was not just a biotech ruling. It was the legal prerequisite for the entire era of biopharmaceutical investment that followed.
- The EPC’s exclusion of treatment methods from patentability created a structural difference between U.S. and European patent strategy that every global pharma IP team must navigate today.
- Hotchkiss v. Greenwood‘s non-obviousness standard remains the most heavily litigated patentability doctrine in pharmaceutical cases — and its application to AI-generated molecules is still being worked out.
V. Hatch-Waxman Deep Dive: The Framework That Remade Pharma Competition
The Six Mechanisms
The Drug Price Competition and Patent Term Restoration Act of 1984 did not just streamline generic approvals. It created an entirely new competitive game in pharmaceuticals, with specific incentive structures that shaped how companies invest, litigate, and settle today.
The Abbreviated New Drug Application (ANDA) pathway allows a generic manufacturer to gain FDA approval by demonstrating bioequivalence to a Reference Listed Drug (RLD) without repeating the originator’s clinical trials. This single mechanism reduced the cost of generic market entry by an order of magnitude. The safe harbor provision (35 U.S.C. § 271(e)(1)) directly addressed the Bolar problem: activities conducted solely to generate data for an FDA submission, even before patent expiry, do not constitute infringement.
Patent Term Extension (PTE) compensates innovators for time lost during FDA regulatory review, up to a maximum of five additional years, with a ceiling that the total remaining patent life at approval cannot exceed 14 years. Patent Term Adjustment (PTA) separately compensates for USPTO prosecution delays. Both mechanisms require careful tracking from the moment of initial filing, and errors in calculating PTA adjustments have been the subject of litigation between applicants and the USPTO.
The Orange Book — formally the FDA’s “Approved Drug Products with Therapeutic Equivalence Evaluations” — became the central battlefield of pharmaceutical competition. Brand companies must list every patent covering an approved drug’s compound, formulations, and methods of use. Generic applicants must address each listed patent. The Orange Book listing requirement created transparency that simultaneously enabled generic challenges and allowed brand companies to use the listing process itself strategically, by listing patents late or listing patents of questionable scope to trigger additional 30-month stays.
The Paragraph IV Certification: Where Billions Are Won and Lost
When a generic manufacturer files an ANDA with a Paragraph IV certification, it is asserting that at least one Orange Book-listed patent is either invalid, unenforceable, or not infringed by the proposed generic product. That filing becomes a constructive act of patent infringement under the statute, giving the brand company the right to sue immediately without waiting for the generic to enter the market. If the brand company files suit within 45 days of receiving the Paragraph IV notice, a 30-month stay automatically halts FDA approval of the ANDA while litigation proceeds.
The 180-day exclusivity period awarded to the first Paragraph IV filer is the primary economic engine driving generic patent challenges. A first-filer generic that prevails in litigation — or reaches a settlement — can earn a 180-day window of market exclusivity before any other generic can enter. On a blockbuster drug with $5 billion in annual sales, 180 days of duopoly pricing (typically at 20 to 30 percent discounts rather than the 80 to 90 percent discounts that come with multi-source generic competition) can generate $300 to $600 million in generic revenue from a single product. That is why large generics companies maintain dedicated Paragraph IV litigation teams as core business functions.
Pay-for-Delay: The Unintended Consequence
The Hatch-Waxman framework inadvertently created the economic logic for “reverse payment” settlements, colloquially called pay-for-delay agreements. A brand company whose primary compound patent is facing a strong invalidity challenge has a straightforward calculation: paying a generic challenger $200 million to delay entry for five years is far cheaper than losing the patent and watching $4 billion in annual branded revenue evaporate to multi-source generic competition. The generic company, in turn, earns a guaranteed payment while preserving its 180-day exclusivity period for a later, still-protected entry.
The Supreme Court addressed this in FTC v. Actavis (2013), holding that reverse payment settlements are subject to antitrust scrutiny under a rule-of-reason analysis rather than per se legal. The FTC continues to track and challenge these agreements, publishing annual reports on their prevalence. The legal risk has not eliminated them — the economics remain too compelling in some cases — but it has changed how they are structured and disclosed.
IP Valuation Spotlight — Atorvastatin / Lipitor (Pfizer)
Lipitor was the best-selling drug in history at its primary patent expiry, generating over $130 billion in cumulative revenue. Pfizer’s patent strategy around atorvastatin illustrates the full toolkit: the base compound patent (US 4,681,893) anchored exclusivity, while secondary patents on atorvastatin’s calcium salt form, specific crystalline polymorphs, intermediate purification methods, and amorphous forms extended the defensible portfolio. The EU polymorph patent T 0777/08 (Warner-Lambert) addressed the crystalline versus amorphous form question at the European Patent Office. Multiple Paragraph IV challenges were filed by generic manufacturers beginning in the mid-2000s, and Pfizer settled several of them on terms that delayed generic entry. When Lipitor finally lost exclusivity in November 2011, its U.S. revenue dropped approximately 42 percent in the following quarter — a textbook patent cliff. The IP valuation lesson: even a portfolio of secondary patents cannot indefinitely defer the cliff when the compound patent is both dominant and eventually indefensible.
Investment Strategy Note — Hatch-Waxman Dynamics
- Track every Paragraph IV certification filed against portfolio drugs using FDA ANDA databases and court PACER filings. The filing date of the first Paragraph IV certification starts a clock that most equity models ignore until a press release announces it.
- Count the 30-month stay duration as protected revenue, not guaranteed revenue. Litigation outcomes can accelerate generic entry, and settlements can alter it in either direction.
- Pediatric exclusivity adds exactly six months to existing patents and exclusivities when a pediatric study is conducted per FDA Written Request. For a $5 billion drug, that is $2.5 billion in revenue for the cost of a pediatric clinical program — one of the highest ROI activities in pharmaceutical development.
- PTA adjustments compound over multi-year prosecution delays. A patent with substantial prosecution delays at the USPTO can have a calculated expiry date years after the nominal 20-year term. Always check the calculated expiry, not the filing date plus 20 years.
Table 2: Major U.S. Pharmaceutical Patent Law Milestones
| Year | Event | Mechanism or Ruling | Strategic Impact |
|---|---|---|---|
| 1850 | Hotchkiss v. Greenwood | Non-obviousness introduced as patentability requirement | Foundation of the most-litigated doctrine in pharmaceutical patent challenges |
| 1853 | O’Reilly v. Morse | Abstract ideas not tied to specific application are not patentable | Precursor to § 101 challenges now used against AI and diagnostic method claims |
| 1980 | Diamond v. Chakrabarty | Live, human-made organism held patentable | Opened the legal foundation for the entire biopharmaceutical industry |
| 1984 | Roche Products v. Bolar | Pre-expiry testing for FDA submission held infringing | Direct catalyst for Hatch-Waxman safe harbor provision |
| 1984 | Hatch-Waxman Act | ANDA pathway, safe harbor, PTE, Paragraph IV, 180-day exclusivity, 30-month stay | Created the modern generic drug industry and the Paragraph IV litigation economy |
| 2009 | Biologics Price Competition and Innovation Act (BPCIA) | Biosimilar approval pathway, patent dance framework, 12-year exclusivity for reference products | Established the legal and procedural framework for biologic competition |
| 2013 | Association for Molecular Pathology v. Myriad Genetics | Isolated naturally occurring DNA sequences held not patentable | Redirected gene-related patent strategy toward methods, applications, and modified sequences |
| 2013 | FTC v. Actavis | Reverse payment settlements subject to antitrust rule-of-reason analysis | Raised the legal risk of pay-for-delay agreements; changed settlement structuring |
| 2017 | Amgen v. Sandoz | Patent dance under BPCIA held not strictly mandatory | Converted the patent dance from a procedural requirement to a strategic choice |
Table 2. Key milestones in U.S. pharmaceutical patent law with strategic implications for IP teams and portfolio analysts.
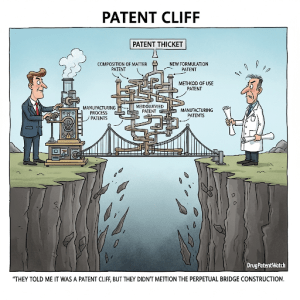
VI. Patent Thickets and Evergreening: Technology Roadmap and IP Valuation
What a Patent Thicket Actually Looks Like
A patent thicket is not a metaphor. It is a specific portfolio construction strategy in which a brand company systematically files patents on every commercial aspect of a product — compound, salts, polymorphs, enantiomers, prodrugs, formulations, release mechanisms, delivery devices, manufacturing processes, dosing regimens, patient subpopulations, combination therapies, and packaging — with overlapping claim scopes and staggered expiry dates. The goal is not necessarily to have 100 ironclad patents. The goal is to raise the cost of a comprehensive Paragraph IV challenge above the economic threshold that most generic companies can sustain.
Challenging a single formulation patent in U.S. district court costs between $5 million and $15 million in legal fees through trial. Challenging 30 patents simultaneously — which is what a first-filer generic may face on a major branded product — costs multiples of that, ties up litigation resources for years, and creates substantial risk that even one loss on one patent will delay market entry for the length of a damages award or injunction period. Most generic companies make a rational economic calculation to wait rather than challenge, which is precisely the outcome the thicket is designed to produce.
The Evergreening Technology Roadmap
Evergreening describes the process of extending effective market exclusivity through a sequence of incremental patent filings that individually may represent modest innovations but collectively add years of protected revenue beyond the primary compound patent. The roadmap is not uniform — it varies by drug class, regulatory history, and competitive environment — but the general sequence is well-established in industry practice.
Evergreening Tactical Sequence — Small Molecule
Phase 1 — Foundation (Year 0-2 post-IND): Composition-of-matter patent on API; Markush claims covering the chemical genus; process patents on the synthetic route.
Phase 2 — Salt and Polymorph Layer (Year 2-5): Patents on pharmaceutically acceptable salts (calcium, sodium, potassium, hydrochloride forms); patents on crystalline polymorphs and solvates; patents on amorphous forms and their stability profiles.
Phase 3 — Formulation Layer (Year 4-7): Immediate-release tablet or capsule formulation patents; modified-release (extended-release, delayed-release, sustained-release) formulation patents; patents on specific excipient combinations tied to bioavailability or stability data; patents on dosage strengths correlated with patient outcomes data.
Phase 4 — Clinical and Indication Layer (Year 5-12): Method-of-use patents for the primary indication; method-of-use patents for new indications discovered in Phase 3b or Phase 4 studies; dosing regimen patents tied to specific pharmacokinetic targets; combination patents with standard-of-care co-therapies.
Phase 5 — Device and Delivery Layer (Year 6-14): Patents on inhalers, auto-injectors, prefilled syringes, wearable delivery systems, or dissolution-modifying packaging tied to the product’s approved delivery mechanism.
Phase 6 — Manufacturing and Process Refinement (Ongoing): Patents on improved synthesis routes, impurity control processes, scalable manufacturing conditions; for biologics, patents on cell line selection, fermentation conditions, and purification train modifications.
Each layer adds years to the effective exclusivity horizon and raises the cost of a comprehensive challenge. The layers compound: a generic company seeking to launch a bioequivalent product may need to navigate the compound patent (or its polymorph variants), at least one formulation patent, and potentially a method-of-use patent on the primary indication — all simultaneously.
AbbVie / Humira: The Maximum-Execution Case Study
IP Valuation Spotlight — Adalimumab / Humira (AbbVie)
Humira is the most extensively documented case of patent thicket strategy in pharmaceutical history. AbbVie built a portfolio exceeding 250 patents around adalimumab, covering formulation (specifically the citrate-free, high-concentration formulation designed for subcutaneous self-injection), manufacturing processes (cell culture conditions, purification sequences, concentration steps), methods of administration (injection frequency, titration protocols), device features (prefilled syringe design, autoinjector mechanism), and multiple therapeutic indications across rheumatology, gastroenterology, and dermatology.
The IP valuation consequence was stark. Biosimilars entered the European market in October 2018, when AbbVie’s composition-of-matter patents expired there. In the U.S., AbbVie’s thicket held until January 2023 — 57 months longer — because biosimilar manufacturers faced credible infringement claims on formulation and manufacturing process patents that had no European equivalents, and AbbVie settled with each biosimilar entrant to control the entry timeline rather than lose in court. The revenue differential between the European and U.S. markets over those 57 months has been estimated at more than $50 billion in cumulative Humira sales that had no competitive pricing pressure in the U.S. For portfolio analysts: AbbVie’s market cap in 2022 was overwhelmingly a function of this IP strategy, not of adalimumab’s underlying molecular innovation.
The Policy Tension: Innovation Incentive vs. Access Barrier
Critics of patent thickets and evergreening argue — with legitimate empirical support — that the practice maintains drug prices at monopoly levels without delivering commensurate therapeutic advancement. I-MAK’s analysis of the top 12 selling drugs in the U.S. found an average of 140 patent applications per drug, with an average of 38 years of potential exclusivity from the filing of the first patent to the last-expiring secondary patent. A drug approved in 2000 on the basis of a compound patent filed in the early 1990s can thus have active patents reaching into the 2030s with no new clinical development required — only strategic IP filing.
The counterargument from innovators is also substantive: the cost of drug development has risen to an estimated $2.6 billion per approved drug (Tufts CSDD estimate, inclusive of the cost of failures), and the period of effective market exclusivity has been shrinking due to faster generic entry and more aggressive Paragraph IV challenges. Secondary patents, from this perspective, are the mechanism by which companies recover development costs that a single compound patent, eroded by regulatory delays, can no longer support.
Neither argument fully accounts for the other, which is why the policy debate persists — and why both sides need a precise understanding of the IP mechanics to engage it credibly.
Key Takeaways — Patent Thickets and Evergreening
- A patent thicket’s value lies not in the strength of individual patents but in the aggregate cost it imposes on challengers. A 30-patent portfolio where each patent has a 30 percent chance of surviving challenge is still extremely effective at deterring generic entry.
- Evergreening works best when secondary patents are tied to quantifiable clinical benefits — a formulation that demonstrably reduces adverse events is far harder to challenge as obvious than one that merely changes the excipient blend.
- The Humira precedent has set the benchmark for biologic thicket construction. Any biologic approaching blockbuster status without a comparable secondary patent program is leaving significant LOE protection on the table.
- The I-MAK data on 38-year effective exclusivity through patent stacking is the central ammunition for legislative action. Pharma IP teams should expect continued regulatory attention to this issue, including potential reforms to Orange Book listing eligibility requirements.
VII. Strategic Patenting Across the Drug Lifecycle
Discovery and Preclinical: Securing the Foundation
The patent clock starts at filing, not at approval. Every day of regulatory review burns through the 20-year patent term. A compound patent filed in Year 1 of a drug discovery program that takes 12 years to reach approval leaves only 8 years of patent-protected commercialization before the compound patent expires. This arithmetic is the foundational constraint that every subsequent patent strategy is designed to address.
Companies file provisional applications at the earliest point they can claim an actual reduction to practice — often after demonstrating activity in a primary biochemical assay and confirming structural identity of the lead candidate. The provisional gives 12 months to develop additional data and refine the full application without starting the 20-year term. The strategic calculus is deliberate: too early a provisional, and the 12-month window expires before the compound is fully characterized; too late, and a competitor may file on a structurally similar compound, establishing prior art that limits claim scope.
A patent landscape analysis before IND filing is not optional for any serious drug development program. It answers four questions: what patents cover the target space; what freedom-to-operate risks exist for the lead compound’s synthesis route; where are the white spaces in the existing IP landscape that the new program could occupy with strong, uncontested claims; and what does the competitive patent filing timeline suggest about which other companies are working in the same space. This analysis directly shapes both the R&D direction and the initial claim drafting strategy.
Clinical Development: Data Generation as Both Asset and Risk
Clinical trial data is a dual-use asset in pharmaceutical IP. On the asset side, Phase 2 and Phase 3 results provide the utility and efficacy data that supports patent claims for specific dosing regimens, patient subpopulations, and treatment outcomes. A claim that a specific dose of compound X reduces HbA1c by a specific percentage in patients with a specific baseline characteristic is supported — and made non-obvious — by the controlled trial data showing that outcome.
On the risk side, public disclosure of that data creates prior art. Clinical trial results published in a medical journal, presented at a conference, or posted on ClinicalTrials.gov can invalidate later-filed claims if those claims are not sufficiently distinguished from what the public disclosure already reveals. The U.S. grace period (35 U.S.C. § 102(b)) gives inventors 12 months to file after their own disclosure without that disclosure counting as prior art — but this grace period does not apply in most non-U.S. jurisdictions. A presentation at the American Society of Hematology annual meeting without a prior international filing can destroy patentability in Europe and Asia for everything disclosed in those slides.
Confidentiality agreements with clinical investigators and CROs must include specific provisions about what information can be presented or published and when. For major Phase 3 trials, IP counsel should be involved in deciding the timing of any abstract submission or data presentation that precedes a full patent filing.
Post-Launch: Managing Loss of Exclusivity
Loss of exclusivity (LOE) on a blockbuster drug is the most disruptive event in a pharmaceutical company’s revenue profile. When Lipitor lost exclusivity in November 2011, Pfizer’s quarterly revenue from that single drug fell by roughly $1.4 billion within three months. The “patent cliff” is not a figure of speech — it is a measurable, abrupt event driven by the structural dynamics of generic competition.
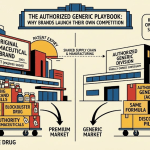
The defensive toolkit is extensive. Product-line extensions to patent-protected formulations — extended-release, once-daily, combination products — can shift a meaningful fraction of the prescriber base to a still-protected product before the cliff arrives. OTC switching, which eliminates the need for a prescription and thus disrupts the generic’s primary marketing channel, has been used successfully for products including omeprazole (Prilosec) and loratadine (Claritin). Authorized generic programs, where the brand company launches its own generic at a reduced price under the ANDA pathway, capture generic revenue while competing directly with the first-filer generic’s 180-day exclusivity period.
None of these strategies fully replaces the revenue from a fully protected branded product. They are damage-limitation mechanisms, not substitutes for the underlying IP protection.
Table 3: Patent Strategy Activities by Drug Lifecycle Stage
| Stage | Primary IP Activity | Key Risks to Manage | Regulatory Intersection |
|---|---|---|---|
| Lead Discovery | Markush claims on chemical genus; provisional applications for lead candidates | Competitor filings on same scaffold; enablement challenges on broad genus | None (pre-IND) |
| Lead Optimization / Preclinical | Narrowed compound claims; polymorph screening and filing; process patent drafting | Premature public disclosure; prior art from academic collaborators | IND preparation timing |
| Phase 1 | Formulation patents for clinical dose form; first method-of-use filings tied to mechanism | ClinicalTrials.gov registration as prior art; early results presentation | IND active; first human safety data |
| Phase 2 / Phase 3 | Dosing regimen patents; subpopulation method-of-use patents; combination therapy patents | Publication of interim results before claims filed; competitor combination filings | NDA/BLA preparation; Orange Book listing strategy |
| Regulatory Approval | Patent Term Extension (PTE) filing; Patent Term Adjustment (PTA) calculation; Orange Book listing | PTE calculation errors; Orange Book listing disputes | NDA/BLA approval; label finalization |
| Launch to Peak Sales | Secondary patent filing (device, new formulation, new indications); pediatric exclusivity applications | Paragraph IV certification; IPR petitions from generics or other challengers | FDA post-market commitments; label expansion filings |
| Pre-LOE / Post-Cliff Management | Product-line extension launches; authorized generic programs; OTC switch strategy | Paragraph IV settlements that accelerate generic entry; patent thicket challenge volume | OTC switching requires new FDA filing; authorized generic requires existing ANDA holder relationship |
Table 3. Patent activity sequenced against drug development and regulatory milestones.
VIII. Generic Entry, Biosimilar Interchangeability, and the Patent Dance
The ANDA Pathway as a Competitive Weapon
The ANDA pathway is not merely a regulatory mechanism. For large generic companies, it is an offensive business strategy. Teva, Viatris, Sandoz, and Sun Pharma maintain full-time Paragraph IV teams whose job is to identify weaknesses in branded companies’ Orange Book patent portfolios, file ANDAs with Paragraph IV certifications, and win the race to 180-day exclusivity. The financial returns are large enough that a successful Paragraph IV challenge on a major product can single-handedly define a generic company’s profitability for a given year.
The generic company’s challenge requires proving one of four things: the listed patent has expired, the listed patent is invalid, the listed patent will not be infringed by the proposed generic product, or some combination of these. The invalidity argument is most commonly pursued on grounds of anticipation (a prior art reference discloses every element of the claim), obviousness (the claimed invention is an obvious extension of prior art), or enablement failure (the patent specification does not adequately teach how to practice the full scope of the claimed invention). Each of these attack vectors requires a detailed claim-by-claim analysis that is essentially a reverse-engineering of the innovator’s prosecution strategy.
Biosimilar Interchangeability and the BPCIA Framework
The BPCIA (2009) created an abbreviated approval pathway for biosimilars — highly similar versions of reference biologic products — while recognizing that the complexity of biologics requires a different legal framework than the small-molecule ANDA process. Two approval designations exist: biosimilar (highly similar, with no clinically meaningful differences in safety, purity, and potency) and interchangeable (meeting additional standards sufficient to support substitution at the pharmacy level without prescriber intervention). Biosimilar interchangeability designation is the higher standard and the more commercially significant one, because pharmacist-level substitution dramatically accelerates market penetration.
The 12-year data exclusivity period for reference biologic products under BPCIA is entirely independent of patent protection. A biosimilar applicant cannot rely on the reference product’s safety and efficacy data for the first 12 years after the reference product’s first licensure in the U.S., regardless of when any individual patent expires. This creates a statutory floor beneath which biologic competition cannot begin, separate from and in addition to the patent thicket that a company like AbbVie constructs above it.
The Patent Dance: Structured Information Exchange as Strategy
The BPCIA patent dance is a legislatively mandated sequence of information exchanges between a biosimilar applicant and the reference product sponsor, designed to surface and resolve potential patent disputes before the biosimilar reaches the market. The process begins when the biosimilar applicant provides its complete application (including manufacturing information) to the reference product sponsor. The sponsor then identifies patents it believes could be infringed. The applicant responds with its non-infringement or invalidity positions. The parties negotiate a list of patents to litigate immediately versus a list to hold in reserve for later resolution.
The Supreme Court’s Amgen v. Sandoz (2017) decision confirmed that the patent dance is not mandatory — a biosimilar applicant can decline to provide the application to the reference product sponsor and proceed directly to market with 180 days notice, forgoing the dance’s structured resolution process. The strategic calculation for the biosimilar applicant is non-trivial. Participating in the dance forces disclosure of manufacturing information that the reference product sponsor can analyze for infringement claims, but it also compresses the litigation timeline and can narrow the universe of patents the applicant must deal with before launch. Opting out avoids that disclosure but shifts control of litigation timing to the reference product sponsor, who can file on any patent at any time after the biosimilar launches.
Investment Strategy Note — Biosimilar Market Entry
- The 12-year reference product exclusivity under BPCIA is a hard floor for biologic competition, but it runs from the date of reference product licensure, not from the biosimilar applicant’s filing date. A biologic licensed in 2012 reaches its 12-year exclusivity expiry in 2024 regardless of how many secondary patents AbbVie or Amgen or Regeneron has filed since then.
- Biosimilar interchangeability designation has a meaningful commercial premium in states with automatic substitution laws. A biosimilar that achieves interchangeability can be substituted at the pharmacy in most U.S. states without the prescriber’s explicit approval, which directly accelerates volume uptake compared to a biosimilar-only designation.
- The Humira biosimilar market in the U.S. (post-January 2023) has been slower to convert than the European biosimilar market was at analogous stage, primarily because U.S. payer contracting structures and rebate economics favor the branded originator in a way that the European reference pricing systems do not. Analysts should model U.S. biosimilar volume ramp at a slower rate than European experience suggests.
IX. Personalized Medicine, Gene Therapy, and the Post-Myriad Patent Landscape
What Myriad Genetics Actually Changed
The Supreme Court’s 2013 decision in Association for Molecular Pathology v. Myriad Genetics drew a clean line: naturally occurring DNA sequences, including isolated gene sequences identical to those found in the human genome, are products of nature and are not patentable. The cDNA (complementary DNA) sequences that do not occur naturally are patentable. The ruling invalidated Myriad’s BRCA1 and BRCA2 gene claims but left intact patents on the cDNA forms of those genes and, crucially, on the methods of testing and analysis using those sequences.
The downstream effect on precision medicine IP strategy was substantial. Companies filing patent applications on genetic sequences shifted from claiming the sequences themselves to claiming the methods of identifying those sequences, the methods of using sequence data to predict disease risk or therapeutic response, the algorithms that analyze sequence data, and the diagnostic tests (as combinations of method and apparatus) that implement those analyses. This is the IP architecture that governs precision oncology, pharmacogenomics, and liquid biopsy diagnostics today.
Precision Medicine Patenting: The Four Protectable Layers
Precision medicine inventions now cluster around four IP layers that survive post-Myriad scrutiny. First, novel biomarkers — genetic variants, protein expression patterns, or epigenetic signatures that have never been associated with a disease state or therapeutic response — remain patentable as non-obvious associations when supported by statistically robust clinical data. Second, companion diagnostic methods that use a specific biomarker to stratify patients for a particular therapy are patentable as method claims tied to a concrete therapeutic application, avoiding the abstract idea trap of 35 U.S.C. § 101. Third, algorithms that process genomic or proteomic data to generate a clinical decision — when claimed as part of a specific computing system or integrated with a particular hardware configuration — have succeeded in surviving § 101 challenges at the USPTO, though the legal landscape here remains fluid. Fourth, the therapeutic agents designed to target a specific molecular pathway identified through precision medicine approaches are themselves claimable as composition-of-matter or method-of-use patents in the traditional sense, as long as they are structurally novel compounds rather than naturally occurring molecules.
Gene Therapy: CRISPR, Vectors, and the Inventorship Wars
The CRISPR-Cas9 patent dispute between the Broad Institute (primarily represented by the Zhang lab) and the University of California Berkeley (the Doudna/Charpentier group) is the most expensive and consequential patent interference proceeding in the history of molecular biology. The dispute concerns which team reduced CRISPR-Cas9 gene editing to practice first in eukaryotic (human and other mammalian) cells — a distinction that determines who controls the foundational patents for a technology platform underlying hundreds of clinical programs.
The Patent Trial and Appeal Board (PTAB) ruled in February 2022 that the Broad Institute’s patents on CRISPR editing in eukaryotic cells were patentable over the University of California’s earlier filings on CRISPR in general — finding that it was not obvious from the UC work that the system would work in eukaryotic cells. The Federal Circuit affirmed. This means the Broad Institute holds foundational eukaryotic CRISPR patents in the U.S., while UC Berkeley holds rights to certain other applications. Companies building CRISPR-based therapeutics typically license from both groups or structure their programs to avoid the most contested claims — a familiar thicket navigation problem applied to a platform rather than a single drug.
Beyond CRISPR, gene therapy patenting must address viral vectors (particularly adeno-associated virus serotypes used for in vivo delivery), lipid nanoparticle (LNP) formulation compositions used for in vivo mRNA delivery (a category of intense current IP activity following the COVID-19 mRNA vaccine programs), therapeutic gene constructs (the promoter sequences, the transgene, the regulatory elements), and cell engineering methods for ex vivo gene therapy approaches. Each of these represents a separately patentable technical layer.
IP Valuation Spotlight — mRNA Delivery LNP Technology (Moderna / Alnylam / Arbutus)
The lipid nanoparticle delivery patent landscape is one of the most actively contested IP spaces in current pharma. Alnylam Pharmaceuticals built foundational LNP patents in the context of RNA interference delivery, covering ionizable lipid compositions, lipid ratios, and formulation parameters. Arbutus Biopharma (formerly Tekmira) holds separate LNP patents that have been the subject of IPR proceedings and inter partes challenges by Moderna. Moderna’s COVID-19 mRNA vaccine program — generating approximately $18 billion in revenue in 2021 alone — elevated the commercial stakes of LNP IP to a level not previously seen. Arbutus has claimed that Moderna’s mRNA-1273 vaccine product infringes its LNP patents. Moderna’s IPR petitions challenged those patents at the PTAB. The outcome will determine licensing economics across the mRNA therapeutics sector. For any investor holding a position in an mRNA-based therapeutic company, the LNP IP landscape is not background noise — it is a material valuation input.
Key Takeaways — Precision Medicine and Gene Therapy
- Post-Myriad, the protectable IP in genomics has shifted from sequences to methods, from discovery to application, and from products of nature to human-made tools. Teams filing on “biomarker associations” without a concrete diagnostic or therapeutic utility tied to the claim are wasting prosecution budget.
- CRISPR patent disputes have established that foundational platform technology can generate thickets as complex as any single drug — and that resolving inventorship in academic-industrial collaborative settings requires IP agreements drafted before research begins, not after.
- LNP delivery technology is the mRNA equivalent of the formulation patent layer for small molecules: an independently patentable technical stack that is separate from the active sequence it delivers, with its own portfolio construction logic and licensing economy.
- AAV capsid serotype patents and manufacturing process patents for gene therapies function analogously to biologic manufacturing patents. The cell line used, the production conditions, and the downstream purification method are each separately protectable and form the manufacturing IP layer of any gene therapy franchise.
X. AI in Drug Discovery: Inventorship, Non-Obviousness, and Claim Strategy
The Human Inventorship Problem
Current U.S. patent law requires at least one human inventor. The USPTO’s 2024 guidance on AI-assisted inventions confirmed that a machine cannot be named as an inventor, and that AI contributions to the inventive process do not, by themselves, satisfy the inventorship requirement. The legal question is not whether AI was used but whether identified human contributors made a “significant contribution” to the conception of the claimed invention — the standard articulated in Pannu v. Iolab Corp. (Fed. Cir. 1998).
For pharmaceutical discovery programs that use generative AI models to design novel molecules — platforms like Insilico Medicine’s Pharma.AI, Recursion Pharmaceuticals’ OS platform, or Exscientia’s AI-native drug design tools — the inventorship question requires specific documentation protocols. The humans who designed the training data, selected the generative parameters, defined the objective function (binding affinity, ADMET profile, synthetic accessibility), and validated the AI outputs through wet lab testing all have potential inventorship claims. The AI system itself does not, regardless of how generative its output appears.
The documentation that matters is not a general lab notebook. It requires a contemporaneous record of the human decisions made at each stage: what molecular targets were defined, what constraints were placed on the generative search, which AI-generated candidates were selected for synthesis and on what scientific basis, and what modifications to the AI-generated structure were made by human chemists before the final compound was synthesized. Each of these human decision points is a potential hook for inventorship claims and a basis for distinguishing the human contribution from what the AI alone generated.
Non-Obviousness in an AI-Assisted Discovery World
The non-obviousness doctrine under 35 U.S.C. § 103 asks whether the claimed invention would have been obvious to a person having ordinary skill in the art (PHOSITA) at the time of the invention, in light of the prior art. As AI drug discovery platforms become standard industry tools, the definition of what a PHOSITA would find routine is shifting.
If a generative AI model trained on the entire public patent and literature database can produce thousands of novel molecular candidates targeting a given protein in minutes, the question becomes: is any one of those candidates non-obvious? The USPTO has not yet issued definitive guidance on how AI capabilities affect the PHOSITA standard. Several scenarios are plausible. If the AI’s output is entirely expected — a straightforward variation on a known scaffold — the human who selects it for synthesis may have difficulty arguing non-obviousness. But if the AI discovers a structurally unexpected molecule in a chemical space that human chemists would not have searched, and if the experimental validation shows surprising properties, the non-obviousness argument is stronger, provided that the claim is directed to the specific compound and its demonstrated utility rather than to the AI-generated output in the abstract.
Subject Matter Eligibility: Avoiding § 101 Rejections
AI-related pharmaceutical inventions face heightened scrutiny under 35 U.S.C. § 101 for patent-eligible subject matter. The Alice/Mayo framework, which rejects patents on abstract ideas and natural phenomena that lack an “inventive concept” applying those abstract ideas in a practically useful way, is the primary tool USPTO examiners use to reject AI-related claims.
For pharmaceutical AI applications, the strategy for surviving § 101 examination requires tying the AI method to a specific, concrete technical improvement or application. A claim reciting “a method of identifying drug candidates using machine learning” is almost certainly an abstract idea under Alice. A claim reciting a specific computer-implemented system that trains a neural network architecture with defined parameters on a specific dataset of biochemical activity measurements to predict ADMET properties to a defined accuracy threshold, and then outputs molecular structures meeting those criteria, is a more concrete technical implementation with a better § 101 profile. The specificity of the technical implementation is what separates a patentable AI-assisted discovery method from an unpatentable abstract idea in the current USPTO and Federal Circuit landscape.
Investment Strategy Note — AI Drug Discovery IP
- When evaluating AI-native drug discovery companies, ask specifically about their IP documentation protocols. A company running generative AI discovery without contemporaneous records of human inventive contributions will have difficulty building defensible patent portfolios as their pipeline advances. This is a diligence item, not a hypothetical risk.
- The pipeline assets of AI drug discovery companies are often described in terms of their platform’s efficiency, not their IP position. But the patent coverage of specific clinical candidates — not the platform — is what generates revenue. A platform patent that covers the AI method may not cover the specific drug compound that platform generated. Compound-level IP coverage should be evaluated separately.
- DABUS litigation in multiple jurisdictions (the AI system claimed as inventor by Stephen Thaler) has uniformly failed, confirming that human inventorship is currently non-negotiable. Companies building AI discovery programs on the assumption that future legal reform will retroactively validate AI inventorship are taking on significant legal risk without current basis.
XI. Patent Analytics as Competitive Intelligence
Freedom-to-Operate Analysis: The Diligence Standard
A Freedom-to-Operate (FTO) analysis determines whether a proposed product or process can be commercialized without infringing unexpired patents in a target jurisdiction. In biopharmaceuticals, a rigorous FTO must cover the drug compound and all commercially relevant salt forms, formulations used in clinical or commercial presentations, the manufacturing process for the API and the finished dosage form, the primary therapeutic indication and any likely label expansions, and the delivery device if one is part of the commercial presentation. Omitting any of these layers creates gaps that a patent holder can exploit after commercial launch — when the consequences of an injunction or royalty obligation are far more expensive than the cost of a comprehensive FTO before clinical investment.
FTO analyses for biologics have unique complexity. Because biologics are manufactured through living cell systems, the manufacturing process patents of the reference product sponsor may cover conditions — cell culture temperature ranges, pH targets, glucose feeding strategies, downstream ultrafiltration parameters — that a biosimilar manufacturer cannot entirely avoid. Cell line patents, if unexpired, may restrict which host cell systems are available. Glycosylation pattern patents, if drafted broadly, may claim any biologic product with a defined range of glycan compositions regardless of how it is produced.
White Space Analysis: Where the Uncontested IP Lies
White space analysis maps the patent landscape to find areas of scientific and commercial relevance where no or minimal patent coverage exists. In pharmaceutical R&D, these gaps represent opportunities to file patents that will face fewer prior art obstacles, require less aggressive prosecution, and generate broader, more defensible claims. The identification of white spaces directly informs pipeline strategy decisions — a therapeutic area with a dense patent thicket around existing targets may warrant development of a program targeting a biologically adjacent but IP-uncrowded mechanism of action.
Machine learning patent analytics tools now automate much of the white space identification process, parsing millions of patent documents to map claim density, assignee concentration, filing velocity, and geographic coverage across defined technical domains. Platforms purpose-built for pharmaceutical patent intelligence — including DrugPatentWatch, Clarivate Derwent Innovation, PatSeer, and XLSCOUT — provide curated Orange Book linkages, ANDA filing alerts, and patent expiry timelines that are essential inputs for competitive intelligence in pharma.
Competitive Intelligence: Reading Competitor Pipelines Through Patent Filings
Patent applications become public 18 months after their priority date. A competitor’s patent filing on a novel compound targeting a specific receptor subtypes, filed 18 months ago, appears in the public patent database today — before any clinical trial registration, before any press release, and potentially before the company has decided whether to advance the program. For therapeutic areas where mechanism of action is the key differentiating variable, a systematic patent landscape review provides earlier visibility into competitive pipelines than any other publicly available data source.
Tracking competitor patent filings on a quarterly basis, mapped against known clinical programs, can reveal not only what molecules a company is developing but also how it is thinking about its commercial positioning. A competitor that begins filing formulation patents on a compound still in Phase 1 is signaling that it expects the compound to reach the market and intends to build a secondary IP layer from the outset. A competitor whose patent filings on a given program go silent after Phase 2 initiation may be experiencing clinical setbacks before any public announcement.
XII. Global Patentability: Jurisdiction-Specific Strategy
The Method-of-Treatment Divide
The U.S. allows patents on methods of treatment of the human body, subject to the § 271(e)(1) safe harbor that exempts medical practitioners from liability for infringing those methods in the course of patient care. Europe, Japan, and China explicitly exclude methods of treatment from patentability — in Europe under Article 53(c) EPC, in Japan under the industrial applicability requirement, and in China under Article 25 of the Patent Law. This is not a minor technicality. A portfolio of U.S. method-of-use patents protecting a specific dosing regimen or treatment protocol may have no equivalent protection in major ex-U.S. markets without deliberate reformulation of the claims.
The solutions are jurisdiction-specific. In Europe, “Swiss-type claims” — claiming “use of compound X in the manufacture of a medicament for treating condition Y” — were historically used to capture functional method-of-treatment protection in a form that satisfied EPC patentability requirements. The EPO has largely replaced Swiss-type claims with “purpose-limited product claims” under EPC Rule 54(5), which claim “compound X for use in treating condition Y.” Both approaches effectively protect the therapeutic application while avoiding the excluded method-of-treatment claim format. Japanese and Chinese prosecution requires analogous reformulations. Companies that file U.S. method-of-use claims without corresponding jurisdiction-specific reformulations in parallel PCT filings are leaving significant ex-U.S. exclusivity unprotected.
Polymorphs: China’s Higher Bar
Polymorph patents — covering specific crystalline forms of an API — are generally patentable in the U.S. and Europe when the specific form is structurally novel and meets the utility requirement. China’s State Intellectual Property Office (CNIPA) applies a materially stricter standard. Chinese patent examination requires quantifiable evidence that the claimed polymorph has superior properties — measurably better bioavailability, stability, solubility, or manufacturability — compared to prior art crystalline forms of the same compound. A polymorph patent supported only by structural characterization data (XRPD patterns, DSC profiles, solid-state NMR spectra) without comparative functional superiority data will typically be rejected during Chinese prosecution. This requires that polymorph screening programs in drug development include head-to-head comparison studies designed to generate the functional superiority data required for Chinese filing, not just the structural characterization data sufficient for U.S. or European prosecution.
Table 4: Regulatory Exclusivity Reference — U.S., EU, Japan
| Exclusivity Type | United States | European Union | Japan |
|---|---|---|---|
| Standard Patent Term | 20 years from earliest filing date (plus PTA) | 20 years from filing date | 20 years from filing date |
| Patent Extension (PTE / SPC) | Up to 5 years; maximum remaining life at approval = 14 years | SPC: up to 5 years; plus 6 months for pediatric indication | Up to 5 years regulatory extension |
| NCE / Data Exclusivity | 5 years (new active moiety); 3 years for new clinical investigations | 8 years data exclusivity + 2 years market exclusivity (8+2); +1 year for new therapeutic indication | 8 years data exclusivity (re-examination period) |
| Orphan Drug | 7 years from approval | 10 years market exclusivity | 10 years re-examination period |
| Pediatric Exclusivity | +6 months added to existing patents and exclusivities | +6 months added to existing exclusivity | No direct equivalent |
| Biologic Exclusivity | 12 years from BLA approval (reference product) | Covered under 8+2+1 regime; no separate biologic category | 8 years data exclusivity (re-examination period) |
| Method of Treatment Claims | Patentable; medical practice exemption applies | Not patentable; use purpose-limited product claims or Swiss-type equivalents | Not patentable; reformulate as purpose-limited composition claims |
| Polymorph Claims | Patentable with structural novelty and utility | Patentable with structural novelty and problem-solving utility | Patentable; inventive step interpretation can be strict |
| Polymorph Claims (China) | China (CNIPA): Requires quantifiable comparative superiority data over prior art forms. Structural characterization alone is insufficient for grant. | ||
Table 4. Cross-jurisdictional exclusivity reference for pharmaceutical portfolio planning. All durations subject to current applicable law and jurisdiction-specific calculation rules.
Key Takeaways — Global Patent Strategy
- Filing a PCT application does not constitute a global patent strategy. PCT national phase entry requires jurisdiction-specific claim adaptation, and a claim set optimized for U.S. prosecution will often need substantial reformulation for EU, Japanese, and Chinese examination.
- China’s polymorph patentability standard requires that clinical and formulation development teams generate head-to-head comparative data during the discovery phase, not as an afterthought before filing. This data must be built into the development program plan from the outset.
- The EU 8+2+1 regulatory exclusivity regime — 8 years of data exclusivity, 2 years of market exclusivity, plus 1 year for a new therapeutic indication — is often more commercially protective than the underlying patent position in Europe, particularly for drugs whose compound patents were filed early and have limited remaining term at approval.
- The EU Supplementary Protection Certificate (SPC) program is under active legislative reform. The proposed SPC manufacturing waiver, if enacted, would allow SPC-covered products to be manufactured in the EU for export to third countries during the SPC period — a material change for generic manufacturing capacity planning.
XIII. What Comes Next
The pharmaceutical patent system is under more concurrent pressure from more directions than at any point since Hatch-Waxman was passed. Proposed legislation in the U.S. (including the PREVAIL Act and various IRA provisions affecting drug negotiation) continues to test the boundaries of how patent exclusivity interacts with government pricing authority. The European SPC manufacturing waiver is moving through legislative stages. PTAB inter partes review proceedings continue to invalidate secondary pharmaceutical patents at meaningful rates — a check on the most aggressive evergreening strategies that patent offices and courts did not provide during the first 25 years of the post-Hatch-Waxman era.
Gene therapy and mRNA therapeutics are generating new categories of IP disputes — over LNP delivery formulations, AAV capsid designs, and CRISPR editing specificity — that do not map cleanly onto the small-molecule or biologic patent frameworks that courts have spent 40 years interpreting. AI drug discovery is creating a generation of compounds whose inventorship and non-obviousness status is genuinely ambiguous under current law. And biosimilar interchangeability markets are maturing in ways that are exposing the structural differences between U.S. and European payer systems, which affect how much of the theoretical exclusivity value of a biologic patent thicket actually translates into commercial revenue protection.
The companies that navigate this environment effectively will be those whose IP teams do not operate as a legal function separate from R&D and commercial strategy. Patent claims are not documents generated at the end of a discovery program. They are the commercial architecture of the program, built in parallel with the science, updated as clinical data emerges, and managed with the same analytical rigor that any other multi-billion-dollar asset demands. The gap between companies that understand this and companies that still treat IP as a compliance function is measurable in billions of dollars of revenue at risk per LOE event.
Investment Strategy Note — Forward-Looking Considerations
- The Inflation Reduction Act’s Medicare drug price negotiation provisions create a new dynamic for patent strategy: drugs subject to negotiation earlier in their commercial life (small molecules at 9 years, biologics at 13 years) now have a shorter effective period before government pricing intervention. This changes the NPV calculation for investing in secondary patent protection and may shift R&D investment toward biologic and gene therapy programs, which have the longer pre-negotiation window.
- IPR petition rates on pharmaceutical patents have increased consistently since PTAB was established in 2012. High-value secondary patents — particularly polymorph and formulation patents on major commercial products — attract petitions from generic companies, hedge funds holding short positions, and patent challengers acting on behalf of unnamed parties. Any patent in an active commercial portfolio should be assessed for IPR vulnerability as a routine matter, not only in response to an actual petition.
- The AI drug discovery IP landscape will crystallize around the first major district court case involving an AI-generated compound. That case has not yet been tried. When it is, it will set the practical standard for what “significant human contribution” means in AI-assisted pharmaceutical discovery — and that standard will directly affect the defensibility of every pipeline patent filed by an AI-native biotech between now and that ruling.
- Track CNIPA examination outcomes for polymorph and formulation patents on drugs with significant China revenue. An invalidation trend at CNIPA has outsized commercial impact because China’s pharmaceutical market scale means that lost exclusivity there is no longer a secondary consideration in global revenue modeling.
This analysis is prepared for informational purposes for pharmaceutical IP professionals, portfolio managers, and institutional investors. It does not constitute legal advice. Patent law is jurisdiction-specific and subject to change through litigation, administrative guidance, and legislation. Consult qualified patent counsel for advice on specific IP matters.