The Architecture of Resilience: A Comprehensive Analysis of Premarin’s Sustained Market Dominance Without Patent Protection
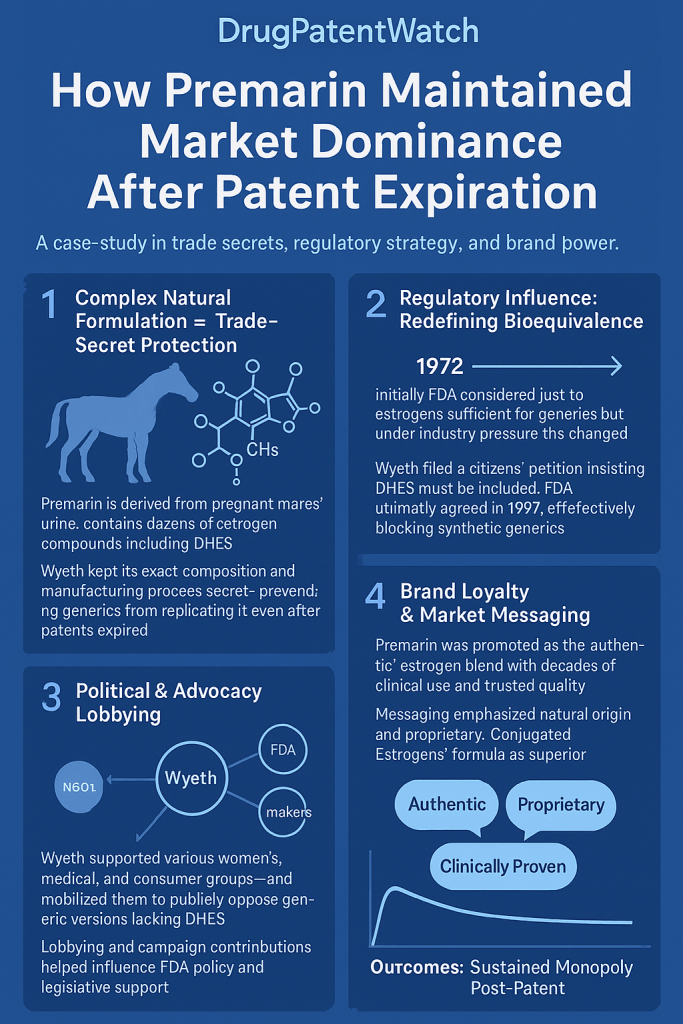
The pharmaceutical industry operates on a fundamental economic premise: the patent lifecycle. Typically, an innovator company secures a composition-of-matter patent, providing a twenty-year window of exclusivity to recoup multi-billion-dollar research and development investments before facing the “patent cliff”.1 Upon the expiration of these patents, generic competitors enter the market, leading to a rapid erosion of market share and a precipitous decline in pricing.1 However, the case of Premarin (conjugated equine estrogens) represents one of the most significant anomalies in the history of biopharmaceutical regulation and market dynamics. Originally approved by the United States Food and Drug Administration (FDA) in 1942, Premarin maintained a dominant market position for over eighty years, despite the expiration of its core patents in the mid-20th century.1 This sustained dominance was not the result of a single strategic maneuver but rather a sophisticated, multi-layered fortress built upon biological complexity, trade secret protection, astute regulatory navigation, and aggressive brand management.1
By the late 1990s, Premarin had achieved “blockbuster” status, with gross sales in the United States growing from $500 million in 1992 to over $2 billion by 2001.1 Even after the catastrophic impact of the 2002 Women’s Health Initiative (WHI) study, which linked the drug to increased risks of breast cancer and cardiovascular events, Premarin demonstrated a level of resilience that defied conventional industry trends.1 This report provides an exhaustive analysis of the non-patent barriers that allowed Wyeth (now Pfizer) to maintain market exclusivity and explores the scientific, legal, and regulatory mechanisms that transformed a 1940s-era hormone therapy into a generational asset.1
The Biological Barrier: Natural Complexity and Compositional Ambiguity
The primary obstacle to generic entry for Premarin was the inherent complexity of the drug itself. Unlike modern synthetic drugs that consist of a single, well-characterized chemical entity, Premarin is a naturally derived mixture of dozens of components extracted from the urine of pregnant mares (PMU).1 This biological origin created a scientific vacuum that the FDA struggled to address for decades, effectively granting the innovator “natural exclusivity”.1
Chemical Fingerprinting and the “Witches’ Brew”
Premarin is not a single chemical entity but a highly complex mixture of conjugated estrogens (CEEs).1 While the United States Pharmacopeia (USP) historically required the identification of only a small subset of components—primarily sodium estrone sulfate and sodium equilin sulfate—the actual composition includes over 50 to 60 distinct steroidal components.9 The manufacturer successfully argued that the therapeutic effect of Premarin was a result of the entire mixture, rather than any individual component or subset of components.5
| Composition Category | Description and Component Details | Impact on Generic Development |
| Primary Estrogens | Sodium estrone sulfate and sodium equilin sulfate; the two most abundant components. | Standardized by USP but insufficient for “sameness”.9 |
| Secondary Estrogens | $\Delta^{8,9}$-dehydroestrone sulfate (DHES), sodium equilenin sulfate, and $17\alpha$-estradiol sulfate. | Acted as “required” active ingredients in later FDA rulings.11 |
| Minor Steroids | Over 50 identified steroidal components, many present at levels $\leq 0.1\%$. | Created a nearly impossible target for synthetic replication.5 |
| Non-Steroidal Molecules | Various organic compounds naturally occurring in equine urine. | Contribution to efficacy remains largely unknown, creating regulatory doubt.5 |
The lack of a definitive characterization meant that the FDA could not establish a reproducible standard for “sameness”.1 In pharmaceutical regulation, the “burden of proof” for an Abbreviated New Drug Application (ANDA) rests on the applicant to demonstrate that their product is identical in active ingredient to the reference listed drug (RLD).14 Because the RLD (Premarin) was not fully defined, proving “sameness” was a scientific impossibility for generic manufacturers.5
Pharmacokinetic and Bioequivalence Challenges
Traditional bioequivalence (BE) is established by showing that the rate and extent of absorption of the generic drug are not significantly different from the brand-name drug.1 For Premarin, this process was complicated by its slow-release bioavailability pattern.1 Early generic versions in the 1970s and 1980s were often immediate-release, leading to disparities in blood and urine concentrations.10
Furthermore, the estrogenic potency of the mixture could not be clearly defined relative to its individual constituents.12 For years, the tablet strengths (such as the 0.625 mg dose) were controlled using a rat bioassay rather than a chemical assay, with estrone as the reference standard.14 This meant a 0.625 mg tablet was not necessarily 0.625 mg of a specific chemical, but rather a mixture that produced an estrogenic response in a rat model equivalent to 0.625 mg of sodium estrone sulfate.14 Generic manufacturers, attempting to use modern chemical synthesis, found it nearly impossible to match this biological “fingerprint” across multiple lots and years of production.1
Trade Secret Defense: The Brandon Process
While patents provide a twenty-year monopoly in exchange for public disclosure, trade secrets can provide indefinite protection if the information remains confidential.1 Wyeth utilized this legal framework to protect the “Brandon Process,” the proprietary chemical extraction and purification method used at its facility in Brandon, Manitoba.1
The Value of Process Secrecy
The Brandon Process was the result of decades of experimentation and refinement, allowing Wyeth to consistently produce a therapeutically equivalent mixture from a variable raw material (horse urine).1 Because the process was never patented, its details were never disclosed to the public.17 This created a formidable barrier: even if a competitor could obtain the same raw materials, they lacked the “recipe” to process those materials into a finished drug that would meet the FDA’s stringent requirements for consistency and purity.1
| Defense Mechanism | Strategic Application | Economic Impact |
| Confidentiality Agreements | Strict internal protocols for employees and researchers. | Prevented legal “leakage” of manufacturing know-how.17 |
| Facility Security | Physical and digital security at the Brandon, Manitoba plant. | Described by courts as being at the “upper end” of corporate reasonableness.17 |
| Long-Term Evolution | Continuous refinement over 60+ years. | Created a “time moat” that competitors could not easily bridge.17 |
Case Study: Wyeth v. Natural Biologics
The robustness of this trade secret protection was tested in the landmark litigation Wyeth v. Natural Biologics.17 Natural Biologics, a startup company, claimed to have developed a generic version of Premarin using only public records and expired patents.17 However, the court found that the company’s CEO had conspired with a former Wyeth senior research chemist, Dr. Douglas Irvine, to misappropriate the Brandon Process.17
The evidence presented in the case was overwhelming. Natural Biologics’ research protocol shifted overnight from a non-viable method based on public records to a process that was “essentially the same” as the Brandon Process—without any intervening testing or development.17 Phone records revealed over 1,000 minutes of calls and faxes between the CEO and Dr. Irvine.17 Consequently, the court issued a permanent injunction, ordering Natural Biologics to be shut down and all of its research and drug supplies destroyed.17 This ruling reaffirmed the Brandon Process as a core pillar of Premarin’s market dominance, effectively deterring other competitors from attempting to replicate the extraction process.1
Regulatory Warfare: The Citizen Petition and Administrative Delay
Wyeth demonstrated a masterful ability to navigate and influence the regulatory environment, primarily through the strategic use of the citizen petition process.1 By introducing scientific doubt into the FDA’s review process, the company successfully delayed generic approvals for decades.13
The DHES Contention
In 1994, Wyeth submitted a citizen petition requesting that the FDA recognize $\Delta^{8,9}$-dehydroestrone sulfate (DHES) as an essential component of Premarin.10 At the time, DHES was not part of the standard USP definition for conjugated estrogens.11 Wyeth subsequently amended the petition in 1996 to request that DHES be recognized as an active ingredient.13
This maneuver was highly effective. By arguing that DHES contributed to the drug’s therapeutic profile, Wyeth forced the FDA to re-evaluate what constituted “sameness” for the entire class of conjugated estrogens.10 While the FDA identified deficiencies in Wyeth’s studies, the petition introduced enough scientific ambiguity to stall the approval of synthetic generics that lacked DHES.10 The Office of Inspector General (OIG) later noted that this petition process was extended for an “excessive period of time,” during which Premarin faced no generic competition.13
The 1997 Woodcock Memorandum
The most significant regulatory victory for Premarin came in May 1997, when Janet Woodcock, then-director of the CDER, issued a memorandum stating that synthetic generic versions of Premarin could not be approved at that time.5 The memorandum concluded that Premarin was not adequately characterized and that the contribution of its various estrogens to its overall potency was not well understood.5
This ruling established a high scientific bar for generic entrants:
- Synthetic exclusion: The FDA stated it was “hard to imagine” a synthetic product being found equivalent to a naturally derived mixture.10
- Inversion of proof: Generic manufacturers were tasked with proving that their products would work identically to the complex mixture, even though the mixture itself remained a “black box” of pharmacological activity.10
- Indefinite delay: As long as the scientific community lacked the tools to fully define the components of pregnant mare urine, no generic could satisfy the legal requirement for “sameness”.1
The 2018 Pfizer Method: The Modern Analytical Moat
As analytical technologies improved, Pfizer (which acquired Wyeth in 2009) proactive moved to create a new, even higher standard for bioequivalence.5 This was formalized as the “Pfizer Method,” an ultra-high-performance liquid chromatography and high resolving power mass spectrometry (UHPLC-HRMS) platform.5
Technical Specifications of the Pfizer Method
The Pfizer Method represents a shift from the 10-component USP standard to a 154-component characterization standard.5 It is capable of identifying and measuring steroidal and non-steroidal components down to the $0.1\%$ level.5
| Parameter | Pfizer Method Requirement | Traditional USP Requirement |
| Characterization Depth | 154 component peaks | 10 steroidal components.5 |
| Detection Limit | $0.1\%$ concentration | Undefined.5 |
| Statistical Rigor | $90\%$ confidence interval across all 154 peaks | Not required for all components.5 |
| Sensitivity | Superior to previous FDA suggested methods | Standard GC methods.5 |
By submitting this method to the FDA’s public docket, Pfizer effectively established a new technical moat.5 Any generic competitor would not only need to source PMU but also prove that their specific batch-to-batch consistency matched Pfizer’s internal standard across 154 different chemical parameters.5 This requirement moved the goalposts for potential entrants, making the “sameness” demonstration more statistically and scientifically demanding than ever before.1
Market Disruption and the Women’s Health Initiative (WHI)
The most significant external challenge to Premarin’s dominance was not competition, but a radical shift in medical guidelines following the 2002 Women’s Health Initiative (WHI) study.7 The study, which included over 16,000 women, was stopped early after finding that the combination of estrogen and progestin increased the risk of heart disease, stroke, and breast cancer.7
Reputation Management and the “Timing Hypothesis”
Following the WHI, prescriptions for hormone therapy plummeted by nearly $45\%$.7 Pfizer responded with a massive campaign to re-educate physicians and patients on the “timing hypothesis”.7 This hypothesis suggested that for women who start hormone therapy closer to the onset of menopause (ages 50-59), the absolute risks are low and the benefits for symptoms like hot flashes outweigh the potential harms.7
| Health Outcome (WHI) | Relative Risk (Estrogen + Progestin) | Clinical Response |
| Coronary Heart Disease | $1.29$ (29% increase) | Use lowest dose for shortest duration.7 |
| Invasive Breast Cancer | $1.26$ (26% increase) | Increased monitoring and black box warnings.7 |
| Stroke | $1.41$ (41% increase) | Shift toward younger, symptomatic women.7 |
| Hip Fracture | $0.66$ (34% decrease) | Maintained indication for osteoporosis.7 |
To stabilize sales, Pfizer pursued and received FDA approval for new indications, such as the prevention of postmenopausal osteoporosis in 2020.1 They also focused on non-oral formulations, like Premarin Vaginal Cream, which were perceived as having less systemic absorption and therefore lower risk.1 In 2012, Pfizer implemented a $68\%$ price increase for the cream, leveraging its lack of generic competition to offset the declining volume of tablet prescriptions.1
Case Studies in Competitive Failure: Cenestin and Enjuvia
Several companies attempted to launch synthetic alternatives to Premarin, but none were able to achieve “AB” rating, which allows for automatic generic substitution at the pharmacy.10
Cenestin (Duramed)
Cenestin was a synthetic mixture of nine plant-derived estrogens.10 While it was approved by the FDA in 1999 for the treatment of hot flashes, it was approved as a New Drug Application (NDA), not a generic (ANDA).10 Because it was not a “generic equivalent” to Premarin, pharmacists could not substitute it for Premarin prescriptions without a doctor’s permission.10 This lack of interchangeability meant that Cenestin failed to capture significant market share, as it could not benefit from the automatic cost-saving mechanisms that drive generic adoption.1
Enjuvia (Teva)
Enjuvia was another synthetic conjugated estrogen product that sought to compete with Premarin.16 In 2016, Teva notified the FDA that Enjuvia was being discontinued for commercial reasons.16 The product was eventually moved to the “Discontinued Drug Product List” in the Orange Book.16 The failure of Enjuvia highlighted the difficulty of marketing a “branded generic” in a space where the innovator (Premarin) had established deep brand loyalty and where the regulatory path for true generic substitution remained blocked.1
Supply Chain as a Structural Barrier
A critical but often overlooked aspect of Premarin’s dominance is the vertical integration of the raw material supply chain.28 Sourcing pregnant mare urine is not a simple commodity purchase; it requires a specialized infrastructure that Wyeth spent decades building.29
The Equine Ranching Infrastructure
By 2009, Pfizer (via Wyeth) managed contracts with approximately 71 PMU farms in Western Canada and North Dakota.29 This network involved approximately 5,600 broodmares.29 The collection process is highly regulated and requires significant logistical coordination to ensure the consistency of the hormone concentrations in the raw material.1
| Ranching Region | Number of PMU Farms | Livestock Volume |
| Manitoba | 53 | ~4,200 mares.29 |
| Saskatchewan | 15 | ~1,200 mares.29 |
| North Dakota | 3 | ~200 mares.29 |
For a new competitor to enter the market, they would need to either build their own equine infrastructure—a process fraught with logistical and animal-welfare challenges—or convince existing ranchers to break their long-term contracts with Pfizer.1 The ethical controversies surrounding the PMU industry, including the fate of “byproduct” foals, also acted as a reputational barrier that deterred many large generic companies from entering the space.28
Financial Performance and Lifecycle Metrics
The success of Premarin’s non-patent strategy is clearly visible in its financial trajectory.1 Even eighty years after its launch, the drug remains a significant revenue contributor for Pfizer.23
Revenue Analysis (2018–2025)
As Pfizer pivoted toward oncology and COVID-19 products, Premarin provided a stable base of legacy revenue with very high margins.31
| Year | Pfizer Total Revenue ($B) | Premarin Sales Performance / Market Context |
| 2018 | $40.8$ | Sustained blockbuster status in the HRT market.1 |
| 2020 | $41.7$ | Osteoporosis indication approval; price stabilization.1 |
| 2021 | $81.3$ | Legacy brand resilience despite COVID-19 product focus.33 |
| 2022 | $101.2$ | Peak pandemic revenue; Premarin accounts for 40-50% of North American estrogens.23 |
| 2023 | $59.6$ | Global sales stabilized at approximately $800M.1 |
| 2024 | $63.6$ | Sustained 12% operational growth in non-COVID portfolio.30 |
| 2025 (Proj) | $62.0$ | Anticipated negative impact of $1.5B from various LOEs, including potential generic entry.35 |
The ability of a drug to maintain a $40-50\%$ share of its therapeutic class eight decades after launch is an anomaly in modern medicine.23 It reflects the success of a strategy that utilized “evergreening” through non-patent means: developing new formulations (creams), pursuing new indications (osteoporosis), and implementing support programs like “Her Source” and “Simplefill” to maintain patient adherence.1
The 2025 Breakthrough: The First Generic Approval
The fortress that Wyeth and Pfizer built around Premarin finally saw its first major breach on November 13, 2025, when the FDA approved the first-ever generic equivalent to Premarin tablets.37
Ingenus Pharmaceuticals and Novast Laboratories
The approval was granted to Ingenus Pharmaceuticals for Conjugated Estrogens Tablets, USP, manufactured by Novast Laboratories.37 The generic is available in all five FDA-approved strengths ($0.3$ mg, $0.45$ mg, $0.625$ mg, $0.9$ mg, and $1.25$ mg).38
| Generic Milestone | Detail |
| Approval Date | November 13, 2025.37 |
| Marketing Status | Immediate nationwide distribution.38 |
| Exclusivity | Competitive Generic Exclusivity through April 15, 2026.39 |
| Regulatory Compliance | Includes full boxed warnings for endometrial cancer and cardiovascular risk.38 |
The arrival of this generic marks the end of an 84-year monopoly.4 While technical details regarding how Ingenus finally proved “sameness” have not been fully disclosed, the approval suggests that the analytical hurdles (such as the 154-peak Pfizer Method) have finally been cleared by a competitor.1 This development is expected to significantly expand patient access to affordable hormone therapy and may lead to a permanent shift in the market dynamics of conjugated estrogens.38
The Socio-Equine Loophole: Supply Chain Ethics
A unique factor in Premarin’s dominance was the ethical and logistical “no-go zone” created by the equine supply chain.28 Large generic manufacturers, which are sensitive to ESG (Environmental, Social, and Governance) scores and public image, were often hesitant to engage in the PMU industry.1
The Byproduct Problem
The PMU industry requires mares to be kept in a state of continuous pregnancy to maximize urine collection.28 This results in thousands of foals born each year who are essentially byproducts of the industry.28 If adoptive homes are not found, many of these foals are slaughtered for profit.28 For decades, Wyeth and Pfizer managed the public relations surrounding this issue by emphasizing the medical necessity of Premarin and implementing welfare guidelines for ranchers.28
For a competitor like Teva or Lupin to enter this space, they would have had to account for these ethical risks.1 The “structural monopoly” of the supply chain was therefore not just about having the horses, but about having the stomach for the industry’s controversial practices.1 This provided an additional layer of protection that a synthetic drug—manufactured in a clean, high-tech lab—would never possess.1
Economic Implications: The rNPV of Longevity
From a financial analysis perspective, Premarin represents the ultimate “cash cow” asset.2 In the pharmaceutical sector, analysts use Risk-Adjusted Net Present Value (rNPV) to evaluate assets.2 While most drugs have an rNPV that peaks shortly after launch and then decays rapidly, Premarin’s rNPV was sustained by its indefinite exclusivity.2
Return on Research Capital (RORC)
The Return on Research Capital for Premarin in the 21st century was exceptionally high because the innovator was no longer spending heavily on the core molecule’s development.1 Instead, investments were shifted toward “secondary patents” (polymorphs, delivery mechanisms) and regulatory defense.1
| Financial Metric | Premarin Context | Industry Standard |
| Market Exclusivity | 84 years.4 | 12-14 years (avg post-launch).2 |
| Annual Maintenance R&D | Minimal (primarily regulatory).1 | 15-20% of revenue.31 |
| Peak-to-Trough Decay | ~50% drop post-WHI; stabilized for 20+ years.1 | 80-90% drop within 12 months of LOE.1 |
This financial profile allowed Wyeth and Pfizer to use Premarin’s cash flow to fund other, more risky ventures, such as the acquisition of biotech firms or the development of the COVID-19 vaccine.31 The drug’s longevity effectively turned it into a biopharmaceutical utility, providing steady, predictable returns decades after its scientific “prime”.1
Synthesis: The Mechanics of an 84-Year Monopoly
The sustained dominance of Premarin without patent protection is the definitive case study in “Non-Patent Exclusivity.” It demonstrates that when a product is sufficiently complex, the regulatory and manufacturing barriers can be more formidable than a patent itself.1
The strategy can be synthesized into a four-stage defense model:
- Complexity as a Shield: By utilizing a naturally derived mixture, the innovator ensured that the drug was “un-characterizable” by 1940s-1990s standards.1
- Trade Secret as a Lock: By keeping the extraction “Brandon Process” secret, the innovator ensured that even if the mixture were identified, it could not be legally replicated.1
- Regulatory Doubt as a Delay: By utilizing citizen petitions (DHES) and influencing bioequivalence standards (Woodcock Memo), the innovator turned scientific uncertainty into legal delay.10
- Brand Resilience as a Moat: By re-educating physicians after the WHI study and focusing on high-priced niche formulations (vaginal cream), the innovator maintained its economic base even as volumes declined.1
The 2025 approval of a generic version by Ingenus Pharmaceuticals represents the ultimate scientific triumph over these barriers, but it does not diminish the effectiveness of the strategy that preceded it.37 For eighty-four years, Premarin was the “exception that proved the rule” of pharmaceutical lifecycles, showing that a combination of biological ambiguity and astute corporate strategy can defy the gravitational pull of the patent cliff.1
Works cited
- How Premarin Maintained Market Dominance Without Patent Protection – DrugPatentWatch, accessed January 28, 2026, https://www.drugpatentwatch.com/blog/how-premarin-maintained-market-dominance-without-patent-protection/
- How Financial Analysts Use Drug Patent Expiry Data to Predict Pharma Stock Movements, accessed January 28, 2026, https://www.drugpatentwatch.com/blog/how-financial-analysts-use-drug-patent-expiry-data-to-predict-pharma-stock-movements/
- The Myth of the “Clean” Patent Expiry in Pharmaceuticals: Strategic Analysis of Loss of Exclusivity, Patent Thickets, and Market Entry Dynamics – DrugPatentWatch, accessed January 28, 2026, https://www.drugpatentwatch.com/blog/the-myth-of-the-clean-patent-expiry-in-pharmaceuticals-strategic-analysis-of-loss-of-exclusivity-patent-thickets-and-market-entry-dynamics/
- Premarin’s 84-year hold on the market ends as FDA approves a generic version, accessed January 28, 2026, https://cancerletter.com/clinical/20260123_1/
- Global Research & Development August 29, 2018 … – Regulations.gov, accessed January 28, 2026, https://downloads.regulations.gov/FDA-2018-P-3325-0001/attachment_1.pdf
- The History of Menopausal Hormone Therapy (MHT) – Women Living Better, accessed January 28, 2026, https://womenlivingbetter.org/the-history-of-hormone-therapy/
- Women’s Health Initiative Comments on FDA Panel on Menopause and Hormone Replacement Therapy for Women (Docket: FDA: 2025-N-25, accessed January 28, 2026, https://www.whi.org/doc/banner/WHI_response_to_FDA_Expert_Panel_on_MHT_use_09.19.2025.pdf
- Estrogens, Conjugated Topical, Vaginal Cream – accessdata.fda.gov, accessed January 28, 2026, https://www.accessdata.fda.gov/drugsatfda_docs/psg/PSG_020216.pdf
- Contains Nonbinding Recommendations – Draft Guidance on Conjugated Estrogens – accessdata.fda.gov, accessed January 28, 2026, https://www.accessdata.fda.gov/drugsatfda_docs/psg/Conjugated_estrogens_004782_RC12-14.pdf
- The Strange Case of Premarin – ACS Publications, accessed January 28, 2026, https://pubs.acs.org/subscribe/archive/mdd/v03/i08/html/kling.html
- Marketplace Analysis of Conjugated Estrogens: Determining the Consistently Present Steroidal Content with LC-MS – PMC, accessed January 28, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC4627451/
- Information by Drug Class > FDA Backgrounder on Conjugated Estrogens Page 1 of 2 – Regulations.gov, accessed January 28, 2026, https://downloads.regulations.gov/FDA-2005-P-0319-0020/attachment_12.pdf
- Review of the Food and Drug Administration’s Handling of Issues …, accessed January 28, 2026, https://oig.hhs.gov/reports/all/1997/review-of-the-food-and-drug-administrations-handling-of-issues-related-to-conjugated-estrogens/
- Information by Drug Class > Conjugated Estrogens – Letter from Dr. Janet Woodcock – Regulations.gov, accessed January 28, 2026, https://downloads.regulations.gov/FDA-2007-D-0369-0289/attachment_31.pdf
- Bioequivalence Studies for Generic Drug Development | FDA, accessed January 28, 2026, https://www.fda.gov/media/166152/download
- Determination That ENJUVIA (Estrogens, Conjugated Synthetic B) Tablets, 0.625 Milligrams and 1.25 Milligrams, Were Not Withdrawn From Sale for Reasons of Safety or Effectiveness – Federal Register, accessed January 28, 2026, https://www.federalregister.gov/documents/2017/09/05/2017-18693/determination-that-enjuvia-estrogens-conjugated-synthetic-b-tablets-0625-milligrams-and-125
- Trends Special Edition_Wyeth Case.indd – Faegre Drinker, accessed January 28, 2026, https://www.faegredrinker.com/webfiles/Trends_Special_Edition_WyethCase.pdf
- From discovery to debate: The history of menopausal hormone therapy and its impact on cardiovascular health – PubMed, accessed January 28, 2026, https://pubmed.ncbi.nlm.nih.gov/40812163/
- NJ Personal Injury cases related to Prempro – Simon Law Group, LLC, accessed January 28, 2026, https://www.simonattorneys.com/blog/nj-personal-injury-prempro
- Millions spent on marketing hormone therapies despite the risks, Stanford study shows, accessed January 28, 2026, https://med.stanford.edu/news/all-news/2004/millions-spent-on-marketing-hormone-therapies-despite-the-risks-stanford-study-shows.html
- Pfizer Medical Education Group Appropriate use of Hormone Therapy in Postmenopausal Women Request for Proposals, accessed January 28, 2026, https://cdn.pfizer.com/pfizercom/rfp_hormone_therapy.pdf?VersionId=9qTgfJxvmS.xphcKQg46AAw_pKXk5krU
- Pfizer ordered to pay $103m over hormone drugs and breast cancer – The BMJ, accessed January 28, 2026, https://www.bmj.com/content/339/bmj.b5085
- PREMARIN historic drug sales, accessed January 28, 2026, https://www.drugpatentwatch.com/p/drug-sales/drugname/PREMARIN
- What’s the difference between Premarin (conjugated estrogens) vaginal cream and estradiol vaginal cream? – Dr.Oracle, accessed January 28, 2026, https://www.droracle.ai/articles/185496/whats-the-difference-between-premarin-conjugated-estrogens-vaginal-cream
- PREMARIN Generic Name: conjugated estrogens Sponsor – accessdata.fda.gov, accessed January 28, 2026, https://www.accessdata.fda.gov/drugsatfda_docs/nda/2008/020216Orig1s060.pdf
- How does Premarin compare to bioidentical hormones or generic alternatives for hormone therapy? – Israel Pharmacy, accessed January 28, 2026, https://www.israelpharm.com/faq/how-does-premarin-compare-to-bioidentical-hormones-or-generic-alternatives-for-hormone-therapy/
- Generic Enjuvia Availability & Release Date – Drugs.com, accessed January 28, 2026, https://www.drugs.com/availability/generic-enjuvia.html
- Just Say Neigh: A Call for Federal Regulation of By-Product Disposal by the Equine Industry, accessed January 28, 2026, http://nationalaglawcenter.org/publication/download/craig-just-say-neigh-a-call-for-federal-regulation-of-by-product-disposal-by-the-equine-industry-12-animal-l-193-216-2006/
- Pfizer Wyeth merger worries PMU producers (US/CAN) – Tuesday’s Horse, accessed January 28, 2026, https://tuesdayshorse.wordpress.com/2009/02/24/pfizer-wyeth-merger-worries-pmu-producers-uscan/
- Pfizer 2024 Annual Report: Pfizer’s year in review, accessed January 28, 2026, https://annualreview.pfizer.com/
- Pfizer Inc. – Current Students – The University of Iowa, accessed January 28, 2026, https://students.tippie.uiowa.edu/sites/students.tippie.uiowa.edu/files/2025-08/s25_PFE.pdf
- PFIZER REPORTS FOURTH-QUARTER AND FULL-YEAR 2020 RESULTS AND RELEASES 5-YEAR PIPELINE METRICS, accessed January 28, 2026, https://s206.q4cdn.com/795948973/files/doc_financials/2020/q4/Q4-2020-PFE-Earnings-Release.pdf
- Pfizer’s year in review | Pfizer 2023 Annual Report, accessed January 28, 2026, https://www.pfizer.com/sites/default/files/investors/financial_reports/annual_reports/2023/
- Pfizer Revenue 2012-2025 | PFE – Macrotrends, accessed January 28, 2026, https://www.macrotrends.net/stocks/charts/PFE/pfizer/revenue
- Pfizer Reaffirms Full-Year 2025 EPS Guidance and Provides Full-Year 2026 Guidance, accessed January 28, 2026, https://www.pfizer.com/news/press-release/press-release-detail/pfizer-reaffirms-full-year-2025-eps-guidance-and-provides
- Deconstructing Lifecycle Management and Filing Strategies of Pharmaceutical Blockbusters, accessed January 28, 2026, https://www.drugpatentwatch.com/blog/deconstructing-lifecycle-management-and-filing-strategies-of-pharmaceutical-blockbusters/
- Ingenus Pharmaceuticals Announces FDA Approval and Launch of …, accessed January 28, 2026, https://www.biospace.com/press-releases/ingenus-pharmaceuticals-announces-fda-approval-and-launch-of-first-and-only-generic-equivalent-to-premarin-tablets-conjugated-estrogens-tablets-usp
- Ingenus launches first FDA-approved generic for Premarin tablets, accessed January 28, 2026, https://chaindrugreview.com/ingenus-launches-first-fda-approved-generic-for-premarin-tablets/
- Pharmacy Bulletin – VativoRx, accessed January 28, 2026, https://vativorx.com/pharmacy-bulletin-12-08-2025/
- Ingenus Pharmaceuticals Launches First Generic Version of Premarin Tablets After FDA Approval – MedPath, accessed January 28, 2026, https://trial.medpath.com/news/f926b9ff00b1d12c/ingenus-pharmaceuticals-launches-first-generic-version-of-premarin-tablets-after-fda-approval


























