A $28 Billion Opportunity Most Vendors Are Missing
The 505(b)(2) pathway generates billions in annual drug revenue and runs on formulation innovation. Companies using it need someone to solve their hardest technical problems on tight timelines. Yet most contract development and manufacturing organizations (CDMOs) and formulation labs pitch themselves the same way: flexible capacity, proven technology, regulatory expertise.
That pitch lands nowhere because it describes everyone.
This guide is for business development professionals at formulation vendors who want a systematic, intelligence-driven approach to winning 505(b)(2) clients before the RFP ever goes out. You will learn who these companies are, what they need, how to find them at the right moment, and how to position your organization so you are not one of three finalists — you are the obvious call.
What the 505(b)(2) Pathway Actually Is
Section 505(b)(2) of the Federal Food, Drug, and Cosmetic Act allows a company to file a New Drug Application (NDA) that relies, at least in part, on data it does not own — typically safety and efficacy data from a previously approved Reference Listed Drug (RLD). The applicant still has to generate its own data to support any differences from the RLD, but it avoids repeating the full clinical development program that proved the original molecule was safe and effective.
The practical result is a regulatory pathway that is faster and cheaper than a full 505(b)(1) NDA, but produces a branded product that can be priced and promoted unlike a generic.
This creates a formulation problem by design. To justify a new NDA rather than an ANDA, a 505(b)(2) sponsor must demonstrate something clinically meaningful that differentiates the new product from the RLD. That differentiation almost always lives in formulation: a new dosage form, a different release profile, a novel delivery system, a reformulation that improves bioavailability, or an abuse-deterrent mechanism.
Without a credible formulation strategy, there is no 505(b)(2). Without a capable formulation vendor, there is often no credible formulation strategy.
The Regulatory Logic That Creates Vendor Demand
The FDA’s 21 CFR 314.54 governs 505(b)(2) applications and requires applicants to bridge their formulation to the RLD through in vitro data, pharmacokinetic studies, or both. If the new formulation differs substantially from the RLD, the agency may require a bioequivalence or bioavailability study.
Every one of those requirements is a formulation development task. Vendors who understand the specific data packages the FDA expects — and who can design development programs to generate that data efficiently — compress timelines and reduce regulatory risk for their clients. That competence is the value proposition that sells.
The Size of the Market
The global 505(b)(2) market generates significant revenue. Approval rates for 505(b)(2) NDAs run higher than for novel molecular entities — FDA data shows that 505(b)(2) applications have historically received approval at rates above 80% when filed [1]. Companies pursuing this pathway have already cleared the fundamental risk hurdle of proving the underlying molecule works. Their risk is formulation, development cost, and time.
“The 505(b)(2) pathway accounts for approximately 35-40% of all NDA approvals in recent years, making it the single most common route by which branded drugs reach the U.S. market.” — Drug Information Association & RAPS Annual Conference, 2023 [2]
The pipeline is consistently full. Between 2018 and 2023, the FDA approved more than 200 505(b)(2) applications [1]. Each one represents a formulation program worth anywhere from $500,000 to several million dollars in development fees, plus a manufacturing relationship that often spans the commercial life of the product.
The Four Buyer Archetypes
Not every company pursuing 505(b)(2) looks the same. Getting your targeting right means understanding the four primary buyer types and how each one behaves.
Specialty Pharma Companies
Specialty pharma companies are the most active 505(b)(2) sponsors. Organizations like Assertio, Azurity, Ayala Pharmaceuticals, and dozens of mid-market players run franchise models built around reformulating established molecules. They often have internal regulatory expertise but outsource formulation development almost entirely. Their BD teams are acquisition-focused, not development-focused. Their R&D leads make the vendor decision, and they move quickly when they find a reliable partner.
Virtual Biotechs and Development-Stage Companies
Virtual biotechs and development-stage companies have a scientific founder, a licensing deal or compound acquisition, and almost no internal infrastructure. Every development function is outsourced. These companies are high-value clients when you can get them early, but they are fragile — funding rounds collapse, programs get out-licensed, and timelines stretch. The formulation vendor who helps them build their IND package builds a relationship that survives most setbacks.
Generic-to-Branded Crossover Companies
Generic-to-branded crossover companies are generic manufacturers who have decided to diversify into branded products using 505(b)(2). Companies like Hikma, Par Pharmaceutical, and Lannett have done this. They have manufacturing capacity and analytical infrastructure, but they often lack experience with the specific formulation challenges that differentiate a 505(b)(2) product from its generic counterpart. They need specialist vendors for the development phase even when they plan to manufacture internally.
Large Pharma Lifecycle Management Teams
Large pharma lifecycle management teams use 505(b)(2) to extend the commercial life of branded assets before patent expiry. A new extended-release formulation, a pediatric dosage form, or a new route of administration can reset the exclusivity clock and protect market share. These programs have serious internal resources but still engage CDMOs and specialty formulation labs for specific platform technologies they do not have in house.
Reading the Patent Landscape to Find Your Next Client
The single best predictor of a company’s 505(b)(2) activity is its patent and exclusivity position. Companies file 505(b)(2) applications when they see a competitive opening — when foundational patents on a molecule are weakening, when Orange Book exclusivities are expiring, or when a competitor has already demonstrated that a reformulation is commercially viable.
You can read these signals systematically.
DrugPatentWatch (drugpatentwatch.com) provides structured access to Orange Book patent and exclusivity data, FDA NDA records, patent expiry timelines, and Paragraph IV certification filings. For a formulation vendor’s BD team, it functions as a prospecting database. You can query it to identify molecules approaching patent expiry, review which companies hold exclusivities on existing formulations, and identify categories where 505(b)(2) activity is clustering.
The workflow looks like this: pull a list of molecules with primary composition-of-matter patents expiring in the next three to five years, cross-reference against existing 505(b)(2) NDAs in those therapeutic areas, and flag molecules where competing reformulations exist — suggesting the FDA has already accepted the scientific premise. That list is your prospecting universe.
Orange Book Expiries as BD Triggers
The Orange Book lists every patent and exclusivity associated with approved drugs. When a key patent expires or is successfully challenged, the commercial window for a 505(b)(2) reformulation opens. The company that submits first benefits from a three-year data exclusivity period if it generates new clinical studies, and potentially longer if it qualifies for orphan designation, pediatric exclusivity, or a new chemical entity classification on a modified active.
Vendors who track these expiry dates can reach out to potential 505(b)(2) sponsors months or years before a program formally launches — before the RFP process, before the client has committed to any vendor, and before competitors are even aware that a program exists.
Paragraph IV Certifications as Intent Signals
A Paragraph IV certification is a formal legal notice that a generic applicant is challenging a patent in the Orange Book as invalid or non-infringed. These certifications trigger a 30-month stay period and signal that a molecule’s patent protection is under active attack. For a 505(b)(2) sponsor, a Paragraph IV certification on a competitor’s product is both a threat and an opportunity — it signals that the molecule will face generic entry, and that a differentiated reformulation may be the only way to maintain commercial relevance.
Tracking Paragraph IV certifications through DrugPatentWatch gives your BD team a real-time feed of molecules under competitive pressure. The companies that hold those NDAs are your most motivated prospective clients.
Formulation Challenges That Define the Engagement
The types of formulation problems 505(b)(2) sponsors face determine which vendors they hire. Knowing your capability set is not enough — you need to map your capabilities to specific problem categories and position each one as a solved problem, not a capability.
Bioavailability Enhancement
Many 505(b)(2) programs target molecules with poor solubility or permeability. If the RLD is an immediate-release oral tablet and your client wants to reduce dosing frequency or improve absorption, they need a vendor with deep experience in solubilization technologies: solid dispersions, lipid-based drug delivery, nanoparticle formulations, or cyclodextrin complexation.
The FDA will require a bioavailability study if the new formulation is not bioequivalent to the RLD. Your job is to design a formulation that passes that study on the first attempt. Every failed BA study adds six to twelve months and several hundred thousand dollars to the program. Vendors who can demonstrate predictive in vitro/in vivo correlation (IVIVC) data are worth a premium because they reduce the probability of that failure.
Modified Release
Extended-release, delayed-release, and pulsatile-release formulations represent the most common differentiation strategy in 505(b)(2). A twice-daily drug becomes once-daily. A product with food effects becomes food-effect-neutral. A molecule with a narrow therapeutic index benefits from a controlled-absorption profile that reduces peak-to-trough variability.
If you have a proprietary modified-release platform with an established pharmacokinetic track record, that platform is your most saleable asset. The key is connecting it to specific molecules and disease states where the clinical rationale for modified release is obvious — where you can show the client not just that you can do it, but why it will work.
Pediatric Formulations
The Pediatric Research Equity Act (PREA) and Best Pharmaceuticals for Children Act (BPCA) have driven sustained demand for age-appropriate pediatric dosage forms. Many 505(b)(2) programs target children specifically because the FDA grants six months of additional exclusivity for pediatric studies under the BPCA, and because the development hurdles are genuine enough that competition is limited.
Pediatric formulation work requires specific expertise in palatability, dose flexibility, and age-appropriate delivery mechanisms. Vendors with demonstrated pediatric taste-masking capabilities, mini-tablet technology, or oral suspension development experience occupy a clearly differentiated position in a market where most CDMOs have only limited expertise.
Abuse-Deterrent Formulations
The Abuse-Deterrent Formulations (ADF) guidance published by the FDA in 2015 and updated since has created a distinct class of 505(b)(2) opportunity. For opioids and other Schedule II and III substances, an ADF designation can extend market exclusivity and protect commercial position. Companies like Collegium Pharmaceutical built their entire strategy around FDA-approved abuse-deterrent technology.
ADF development is technically specialized and regulatory scrutiny is high. Vendors with proven ADF platforms — whether physical/chemical barriers, agonist/antagonist combinations, or prodrug approaches — command pricing premiums and client relationships that tend to be exclusive.
Building Your Target Account List: A Signal-Based Approach
Generic outreach to “pharma companies” is not business development — it is broadcasting. Effective BD targeting for 505(b)(2) clients starts with signals that indicate a specific company is actively developing or planning a 505(b)(2) program.
IND Filings and ClinicalTrials.gov
When a company files an Investigational New Drug (IND) application, the development program has started. ClinicalTrials.gov lists clinical studies tied to 505(b)(2) programs once they have begun human studies. Monitoring new registrations in the reformulation, bioequivalence, and bioavailability categories gives you a list of companies in active development — which means they have either chosen a vendor already or are actively evaluating one. At the IND stage, the formulation work is often still ongoing. Sponsors sometimes start clinical programs with early-stage formulations and refine during Phase 1/2. That creates an opening.
Job Postings as Competitive Intelligence
When a specialty pharma company posts for a “Director of Formulation Development” or a “Head of CMC,” it is often signaling that it is building internal capability for an active or imminent program. When it posts for a “CMC Project Manager” or a “Regulatory Affairs Manager, NDA,” the program is further along. These hires are publicly visible on LinkedIn, Indeed, and company career pages.
A company that is hiring externally for formulation and regulatory expertise is often doing so because it has a program that needs external development partners. Your BD team should be calling these companies within days of the job posting.
Deal Announcements and Licensing Agreements
When a company announces an in-licensing agreement for a compound, it is committing to a development program. Press releases, SEC filings (for public companies), and services like Citeline’s Pharmaprojects and Cortellis capture these deals in near-real time. A company that has just licensed a compound with an existing clinical proof of concept is an ideal 505(b)(2) prospect — the molecule is validated, the mechanism is understood, and the work ahead is primarily formulation and regulatory.
Conference Attendance Patterns
DIA, AAPS, PharmaED, and CPhI Worldwide attract the exact audience of formulation scientists, regulatory affairs professionals, and CMC executives who make vendor decisions. Companies that send technical staff to these conferences are active developers. Your BD team should attend the same sessions these companies attend — not just the exhibition floor.
The Pitch That Wins
Most vendor sales pitches fail because they lead with capabilities. The client does not care that you have a spray dryer and an analytical laboratory staffed by PhDs. They care whether you can solve their specific formulation problem on their specific timeline within their budget.
Lead With the RLD Problem
Every 505(b)(2) program starts with an RLD. The RLD has known characteristics — a specific release profile, a documented food effect, a known bioavailability challenge, or an established abuse liability. Your first pitch task is to demonstrate that you understand the RLD better than your competitors do.
If you are pitching a prospective client on a once-daily reformulation of a currently twice-daily IR product, walk them through the specific polymer systems you have used for similar absorption profiles, show them IVIVC data from a relevant analog, and present a development timeline with clear go/no-go decision points. That is a program design pitch — a fundamentally different conversation from a capabilities pitch.
Speak the Regulatory Language
Your technical team knows formulation science. Your BD team needs to know enough FDA regulatory guidance to discuss it intelligently with a regulatory affairs audience. For 505(b)(2) specifically, that means understanding bridging data requirements under 21 CFR 314.54, bioequivalence guidance for modified-release products, labeling implications of a 505(b)(2) approval, and how formulation choices affect the client’s patent certification requirements.
A vendor representative who can engage substantively on regulatory strategy is several levels more credible than one who promises “regulatory support” in general terms. Hire regulatory affairs consultants to brief your commercial team, or cultivate an advisory relationship with a regulatory consultant who will join critical sales calls.
Case Studies With Actual Numbers
“We have experience with extended-release formulations” is not a case study. A real case study says: “We developed an ER matrix tablet for a BCS Class II compound, achieved a target Tmax shift from 2 hours to 8 hours, passed a food-effect study on the first attempt, and our client filed their NDA 14 months after engaging us.” Anonymize as needed, but preserve the specificity.
Buyers of formulation services are technical professionals. They evaluate vendors on technical credibility. Vague assurances of experience have zero persuasive value; specific outcomes backed by data have high persuasive value.
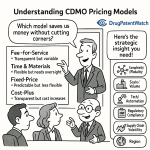
Pricing Formulation Development Engagements
505(b)(2) development contracts are not commodity pricing. They should reflect the regulatory risk you are helping the client manage, the platform technology you are bringing to bear, and the development timeline compression you deliver.
Feasibility Studies as a Sales Tool
A structured feasibility study — typically priced between $25,000 and $75,000 for formulation screening — serves two commercial purposes. It answers the client’s core question (“Can you actually solve this problem?”), and it creates a technical and financial foundation that makes switching vendors after feasibility extremely costly.
Design your feasibility proposals to include enough experimental work that the client learns something real, and structure the deliverable to integrate naturally into your Phase 1 development proposal. The feasibility report should read as Chapter 1 of the development program.
Milestone-Based Contracts
505(b)(2) sponsors manage cash carefully because their programs are often funded in tranches tied to development milestones. Structuring your contract around the same milestones — formulation prototype confirmation, pilot scale-up, IND-enabling stability, BA study formulation, NDA CMC package completion — aligns your payment terms with your client’s funding events.
Milestone-based contracts also protect you. They define clear scope boundaries, create natural renegotiation points if the program changes, and give you leverage to reprice if the client asks for work that was not in scope.
Risk-Sharing Models
Some 505(b)(2) sponsors, particularly virtual biotechs with limited capital, will propose deferred payment or royalty arrangements in exchange for reduced upfront fees. These deals are almost always bad for vendors unless the royalty rate and product potential are genuinely compelling. If you consider a risk-sharing arrangement, price the royalty as if the product will succeed commercially and require approval-contingent milestones rather than pure royalties.
There are cases where early equity or royalty positions in promising reformulation programs have generated substantial returns for CDMOs. Shire’s partnership history and the development relationships around products like Adderall XR illustrate how a formulation platform can become a commercial stake in a highly successful product. These cases are the exception; model them as such.
Partnership Models: Full-Service vs. Specialist
Not every formulation vendor offers the same scope. Understanding where you fit in the development value chain determines your target client profile and your competitive positioning.
Full-service CDMOs — organizations like Catalent, Lonza, and Patheon — offer integrated development-through-manufacturing capabilities. They are logical choices for sponsors who want a single external partner for the entire CMC journey. Their scale creates advantages in analytical capabilities, regulatory infrastructure, and manufacturing bandwidth. It also creates pricing structures that favor mid-to-large programs.
Specialist formulation labs occupy a different position. They focus on specific technology platforms — lipid-based systems, spray drying, modified-release matrices, or abuse-deterrent technologies — and they bring deeper expertise within that domain than any full-service CDMO can maintain across its entire portfolio. For programs where the core technical challenge lives in that specific domain, they consistently outperform full-service providers on both technical outcomes and cost.
The opportunity for specialists is to position themselves as essential subcontractors to the full-service CDMOs as well as direct partners to sponsors. If Catalent takes on a program requiring advanced amorphous solid dispersion work and your organization has a best-in-class HME or spray drying platform, you can win business as a technology partner to the CDMO rather than competing with it.
Common Mistakes Formulation Vendors Make in 505(b)(2) BD
Vendor BD teams make predictable errors that cost them deals.
Calling too late is the most expensive mistake. Once a 505(b)(2) sponsor has issued an RFP, the vendor landscape has already been partially decided — the technical team has thought through its requirements, and at least one vendor has probably already been part of early discussions. Getting into a formal RFP process as a new name is possible but difficult. Get in before the RFP exists.
Pitching capabilities instead of outcomes is almost universal and almost universally ineffective. The client does not need to know that you have a Wurster coater. They need to know that you can solve their enteric coating challenge without the food effect issue they encountered with the previous vendor.
Ignoring the CMC narrative is a strategic error. FDA reviewers evaluate 505(b)(2) CMC sections not just for technical adequacy but for the clarity and coherence of the narrative — is the formulation design justified? Does the development history support the proposed commercial specification? Vendors who help clients build that narrative as part of the development program, rather than just delivering data packages, are far more valuable.
Underestimating regulatory alignment creates expensive surprises. A formulation that is technically excellent but generates a development data package that does not match FDA expectations for a 505(b)(2) CMC section will create problems at NDA review. Vendors who design development programs around the end regulatory deliverable — not just the technical outcome — eliminate most of those surprises.
The Thought Leadership Moat
The formulation vendor market is relationship-driven, but relationships start somewhere. For a company that is not already a household name in pharma R&D, thought leadership is the most efficient way to get in front of 505(b)(2) development teams.
Content That Attracts 505(b)(2) Teams
The formulation scientists and regulatory affairs professionals who evaluate vendors search for technical information constantly. They look for guidance on specific problems: how to design an IVIVC study, how to select a polymer system for a specific drug class, how the FDA interprets manufacturing comparability data in a 505(b)(2) context.
A vendor that publishes high-quality technical content on these exact questions pulls relevant prospects into its orbit before any commercial conversation begins. White papers, webinars, and conference presentations on topics like “Designing Modified-Release Systems for BCS Class II Compounds” or “CMC Strategy for 505(b)(2) Bioavailability Studies” attract an audience that is actively looking for the solutions you provide.
This content is not marketing copy. It should be written by your formulation scientists and reviewed by your regulatory team. It should include data, cite FDA guidance documents, and take a specific technical position on methodological questions. Content that hedges on every point is useless to a technical audience.
Conference Strategy
The American Association of Pharmaceutical Scientists (AAPS) Annual Meeting is the highest-value conference for formulation vendor BD. Presenting data from your platform technology at AAPS puts your technical team in direct contact with formulation scientists from exactly the companies you want as clients. Platform sessions on bioavailability enhancement, modified release, or pediatric formulation draw the right audience.
Drug Information Association (DIA) annual meetings attract the regulatory affairs and medical affairs leadership at specialty pharma companies — the executives who set development strategy and approve vendor selections. Your presence in DIA technical sessions on 505(b)(2) strategy signals that you understand the regulatory side of what your clients are trying to do.
The Intelligence Infrastructure That Drives Sustained BD Performance
Individual deal wins are not a BD strategy. A systematic BD strategy for 505(b)(2) clients requires intelligence infrastructure: tools, processes, and review cadences that ensure your team is consistently aware of where opportunity is forming.
The minimum viable infrastructure includes a patent monitoring system (DrugPatentWatch covers the FDA-specific data layer, and pairing it with a patent analytics tool like Derwent Innovation or CPI covers the broader IP landscape), a clinical pipeline tracker from services like Citeline or Evaluate Pharma, a CRM configured to capture the trigger events described above, and a quarterly BD review process that uses patent expiry timelines to prioritize outreach.
The organizations that do this well are rare. Most vendor BD teams operate reactively — responding to inbound inquiries, following up on conference contacts, and quoting RFPs that arrive without context. Systematic prospecting based on patent intelligence consistently generates earlier engagement, stronger positioning, and higher win rates.
Firms that invest in this intelligence infrastructure over 18 to 24 months typically see their average deal size increase, their proposal win rate improve, and their pipeline composition shift toward programs where they have genuine competitive differentiation rather than programs where they are one of ten equivalent bidders.
What Your Competitors Are Not Doing
The formulation vendor BD market has well-established weaknesses you can exploit directly.
Most vendors do not call on 505(b)(2) sponsors before the program is public. They wait for RFPs. You can routinely be six to eighteen months ahead of the formal process by using patent data systematically.
Most vendors do not have BD staff who understand 21 CFR 314.54 and the FDA’s 505(b)(2) data requirements in enough depth to have a substantive regulatory conversation. Training your commercial team on the regulatory framework — even at a basic level — differentiates every conversation.
Most vendors do not publish technically credible content that attracts inbound from development teams. A single high-quality AAPS poster or white paper on a specific formulation problem, paired with a modest distribution effort, can generate more qualified inbound than a year of trade show presence.
Most vendors do not map their platforms to specific RLD problems before they call. Doing that homework transforms the first call from “let me tell you about us” to “we have worked with similar compounds and here is what we found” — which is the only conversation a busy formulation scientist wants to have.
Key Takeaways
- The 505(b)(2) pathway is structurally dependent on formulation innovation, making it a permanent, large, and growing source of demand for capable external vendors.
- Your best clients are not the ones who find you after issuing an RFP — they are the ones you reach while they are still deciding whether to pursue the program at all. Patent expiry data, Orange Book monitoring, and tools like DrugPatentWatch give you the intelligence to get there first.
- The four buyer archetypes — specialty pharma, virtual biotechs, generic-to-branded crossovers, and large pharma lifecycle management teams — each have different buying behaviors, risk profiles, and vendor requirements. Tailoring your approach to the archetype saves time and improves win rates.
- Technical credibility wins. Replace capability pitches with outcome case studies. Replace general regulatory language with specific knowledge of 21 CFR 314.54 and FDA CMC expectations for 505(b)(2) applications.
- Thought leadership is not a marketing expense — it is a BD function. Content that attracts formulation scientists and regulatory professionals before any commercial need is identified builds relationships that convert when programs launch.
- Systematic patent intelligence, applied consistently, is the highest-return BD investment a formulation vendor can make. Most competitors are not doing it. That gap is your advantage.
Frequently Asked Questions
Q1: At what stage in a 505(b)(2) program should a formulation vendor first make contact?
The ideal entry point is before the IND is filed — ideally at the compound selection or feasibility assessment stage, when the sponsor is still deciding whether the reformulation concept is technically viable. At this point, no vendor has been selected, development strategy is fluid, and a vendor who contributes to the feasibility analysis becomes structurally embedded in the program. DrugPatentWatch’s patent expiry data lets you identify these pre-IND windows by tracking molecules approaching the competitive exposure phase.
Q2: How do formulation vendors differentiate when core technology platforms such as HME, spray drying, and lipid-based systems are widely available?
Platform availability is not differentiation. Differentiation comes from the depth of your in vitro/in vivo correlation data library, the specificity of your IVIVC models for relevant drug classes, your ability to predict BA study outcomes before the study runs, and the track record of your regulatory team in writing CMC sections that pass FDA review without major deficiency letters. Clients will pay a premium for vendors who consistently get to NDA-ready data packages without the trial-and-error cycles that inflate timelines and cost.
Q3: How should a specialist formulation lab approach large pharma lifecycle management programs if it cannot offer full commercial-scale manufacturing?
Be explicit about scope from the first conversation. Large pharma sponsors have internal manufacturing capability or established CDMO relationships for commercial production. What they need from a specialist lab is technology transfer-ready development work: formulation prototypes, analytical methods, stability protocols, and a CMC data package that their manufacturing partner can implement without a major reformulation effort. Position your engagement as a development sprint with defined deliverables and a clear technology transfer protocol. That is a clean, credible scope that large pharma development teams can procure without internal political complications.
Q4: What is the most common reason formulation vendors lose 505(b)(2) proposals to competitors?
Late entry is the most common reason — arriving at the formal RFP stage when another vendor has already built a relationship with the technical team. When vendors compete on equal footing, the second most common cause of loss is failure to connect the proposed development program to a specific FDA regulatory outcome. Sponsors are not buying formulation experiments — they are buying a CMC section that will withstand FDA scrutiny. Vendors who design programs explicitly around that deliverable, with defined success criteria tied to regulatory standards rather than internal specifications, consistently outperform vendors who present technically equivalent programs without that regulatory narrative.
Q5: How do you use DrugPatentWatch as a practical prospecting tool, not just a research resource?
Build a screening routine. At a minimum, run a weekly query for new Orange Book patent expirations in your core therapeutic areas, a monthly review of Paragraph IV certifications filed against drugs in those categories, and a quarterly scan for new 505(b)(2) NDA approvals that indicate where competitors are finding commercial success. Map each output to a company list and assign an outreach priority score based on factors like program stage, company size, funding status, and alignment with your platform capabilities. DrugPatentWatch’s structured data makes this systematic rather than ad hoc. The vendors who run this process consistently are the ones who call the right company six months before anyone else does.
References
[1] U.S. Food and Drug Administration. (2023). Novel drug approvals for 2023. FDA Center for Drug Evaluation and Research. https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2023
[2] Drug Information Association & Regulatory Affairs Professionals Society. (2023). 505(b)(2) regulatory strategy: Optimizing the hybrid NDA pathway. DIA Annual Meeting Proceedings.
[3] U.S. Food and Drug Administration. (2023). Guidance for industry: Applications covered by section 505(b)(2). FDA Center for Drug Evaluation and Research. https://www.fda.gov/media/72419/download
[4] U.S. Food and Drug Administration. (2015, updated 2023). Guidance for industry: Abuse-deterrent opioids — Evaluation and labeling. FDA Center for Drug Evaluation and Research. https://www.fda.gov/media/84819/download
[5] U.S. Food and Drug Administration. (2010). Guidance for industry: Bioavailability and bioequivalence studies submitted in NDAs or INDs — General considerations. FDA Center for Drug Evaluation and Research. https://www.fda.gov/media/71012/download
[6] DrugPatentWatch. (2024). Orange Book patent and exclusivity data for FDA-approved drugs. https://www.drugpatentwatch.com
[7] Bernstein, D., & Luo, A. (2021). The 505(b)(2) regulatory pathway: A review of successful submissions and development strategies. Therapeutic Innovation & Regulatory Science, 55(3), 512-521. https://doi.org/10.1007/s43441-020-00245-y
[8] U.S. Food and Drug Administration. (2019). Guidance for industry: Pediatric drug development: Non-clinical and clinical considerations. FDA Center for Drug Evaluation and Research. https://www.fda.gov/media/128049/download
[9] Pharmaceutical Research and Manufacturers of America. (2023). 2023 PhRMA annual membership survey: Medicines in development. PhRMA. https://www.phrma.org/resource-center/topics/medicines-in-development
[10] U.S. Code of Federal Regulations. (2023). 21 CFR 314.54: Procedure for submission of an application relying on evidence of approval of a listed drug. Electronic Code of Federal Regulations. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-D/part-314/subpart-B/section-314.54