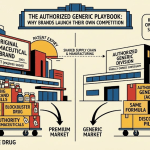
Atorvastatin lost its primary patent on November 30, 2011. What followed was the most studied loss-of-exclusivity response in pharmaceutical history. This pillar page dissects the IP assets, pricing mechanics, authorized generic economics, evergreening architecture, and commercial tactics behind Pfizer’s defense — and draws the operational lessons that apply to any brand facing generic erosion today.
1. The Patent Cliff: Mechanics and Market Economics

Every blockbuster drug has an expiration date printed in the Orange Book. When that date arrives, the loss of exclusivity (LOE) event compresses years of pricing power into a single quarter of brutal erosion. The average brand-name drug loses between 80% and 90% of its volume to generics within six months of LOE, with price erosion in that window averaging 80% to 85% below the pre-generic brand price. For drugs with annual revenues above $1 billion, the projected revenue loss over 24 months post-LOE often exceeds the combined R&D spend on the next two pipeline candidates.
The economic engine here is the Hatch-Waxman Act of 1984, which created the Abbreviated New Drug Application (ANDA) pathway. A generic filer submits bioequivalence data rather than a full clinical trial package, slashing development costs. The first ANDA filer that challenges a listed patent under a Paragraph IV certification gets 180 days of generic exclusivity — a right with enormous commercial value, since the first-to-file exclusivity period typically captures 50% to 75% of the eventual generic market volume before multi-generic competition drives prices to marginal cost.
The resulting dynamic is structurally predictable: brands know their cliff date years in advance. The strategic question is not whether generic entry will happen, but how much of the brand’s revenue stream — through pricing, authorized generics, reformulations, or direct-to-consumer (DTC) loyalty — can be preserved across a 12- to 36-month transition window.
| Metric | Figure |
|---|---|
| Typical price erosion vs. brand in first year post-LOE | 80–85% |
| Market share erosion at 6 months for average brand | 89% |
| Days of exclusivity granted to first Paragraph IV filer | 180 |
| Estimated average cost to bring a new drug to market (2014) | $2.6B |
How the Orange Book and Patent Term Extensions Shape LOE Timing
The FDA’s Orange Book lists all patents associated with an approved drug: compound patents, formulation patents, method-of-use patents, and metabolite patents. Each carries its own expiration date, and a generic filer must either wait for all listed patents to expire or challenge them individually under a Paragraph IV certification, asserting invalidity or non-infringement. The brand holder then has 45 days to sue, triggering a 30-month stay on ANDA approval.
Patent Term Extensions (PTEs) under 35 U.S.C. § 156 can add up to five years to the life of a compound patent to compensate for time spent in regulatory review. Separately, the FDA grants non-patent exclusivities: five years for new chemical entities (NCE), three years for new clinical investigations supporting a supplemental application, seven years for orphan drugs, and pediatric exclusivity that tacks six months onto both patent and non-patent exclusivities. A sophisticated lifecycle management team layers these exclusivities with deliberate overlap, creating a patent thicket that forces generic filers to litigate multiple patents simultaneously rather than challenging a single compound claim.
Key Takeaways — Patent Cliff Mechanics
- LOE events are structurally deterministic; their timing is visible in the Orange Book years before the cliff date arrives.
- The Hatch-Waxman 180-day exclusivity for first Paragraph IV filers creates a concentrated competitive entry that brands can model precisely.
- PTEs and pediatric exclusivity add to compound patent duration and should be factored into any LOE model as potential upside to projected revenue.
- Brands with a single compound patent and no formulation or method-of-use coverage face the steepest and fastest post-LOE erosion curves.
2. Lipitor’s Commercial and IP Architecture
Atorvastatin calcium, marketed as Lipitor by Pfizer, received FDA approval in January 1997. It is a synthetic HMG-CoA reductase inhibitor that produces greater LDL-C reductions at equivalent doses than any statin that preceded it. By the time the primary compound patent expired in November 2011, the drug had generated cumulative worldwide sales exceeding $125 billion — a figure that makes it the highest-revenue prescription drug in pharmaceutical history. At its 2006 peak, Lipitor produced $13.7 billion in annual global sales, representing roughly 23% of Pfizer’s total revenue.
The clinical foundation supporting those revenues was unusually robust. Pfizer ran more than 400 clinical trials involving approximately 80,000 patients across indications including hyperlipidemia, mixed dyslipidemia, familial hypercholesterolemia, and secondary prevention of cardiovascular events. The ASCOT-LLA and TNT trials established atorvastatin’s superiority over lower-intensity statins at high doses — a clinical differentiation that Pfizer’s medical affairs team translated into a durable physician preference story, particularly among cardiologists managing high-risk patients.
The Statin Market Competitive Map at LOE
By 2011, the statin class was crowded. Simvastatin (Zocor, Merck) had already lost exclusivity in 2006. Rosuvastatin (Crestor, AstraZeneca) was still on patent and positioning aggressively against generic atorvastatin with claims of superior HDL raising and pleiotropic effects. Pravastatin and lovastatin were long genericized. Pfizer’s task was to defend Lipitor not just against generic atorvastatin, but against a competitive narrative that rosuvastatin would fill the premium brand statin niche that Lipitor was vacating.
That competitive context shaped the architecture of Pfizer’s response. The company needed to price Lipitor low enough to compete with $4 generic atorvastatin at pharmacy level, while preserving enough premium identity to prevent AstraZeneca from claiming the entire physician preference market with Crestor.
3. Atorvastatin’s IP Valuation as a Portfolio Asset
Valuing Lipitor’s Patent Estate Before and After LOE
IP valuation in pharmaceuticals uses three primary methods: income-based (discounted cash flows tied to remaining exclusivity), market-based (comparable licensing transactions), and cost-based (replacement cost of equivalent clinical data). For Lipitor, the income-based method dominated because the revenue profile was large and the LOE date was fixed.
- The compound patent (US 4,681,893) covering the atorvastatin molecule expired November 30, 2011. At that date, the NPV of patent-protected cash flows fell to zero for that specific asset.
- A pediatric exclusivity grant tacked six additional months onto certain patent claims, granting Pfizer a period to prepare its LOE commercial response before multi-generic entry fully commoditized the molecule.
- The authorized generic agreement with Watson Pharmaceuticals effectively monetized Pfizer’s manufacturing IP and brand recognition inside the generic market, capturing approximately 70% of generic-segment profits that Pfizer would otherwise have lost entirely to third-party filers.
- Method-of-use patents covering cardiovascular indications provided limited defensive value post-LOE because generic labels can carve out patented indications — but they still required litigation to enforce, creating settlement leverage.
- Formulation patents on the calcium salt form and specific tablet coatings, while secondary, contributed to the negotiating architecture of the Ranbaxy settlement by adding uncertainty about ANDA approval timelines.
The residual IP estate — manufacturing know-how, brand equity codified in physician prescribing habits, and data exclusivity for specific pediatric labeling updates — retained real economic value even after compound patent expiry, precisely because Pfizer’s co-pay and authorized generic programs converted that residual value into cash during the 180-day exclusivity window.
Royalty Rate Benchmarking for Mature Small-Molecule Assets
For licensing and deal purposes, a mature statin at LOE would benchmark at a royalty rate of 2% to 5% on net sales for manufacturing technology transfers, reflecting the commodity nature of atorvastatin synthesis. The drug’s active pharmaceutical ingredient (API) synthesis pathway — a multi-step Paal-Knorr pyrrole synthesis and subsequent side-chain assembly — had been off-patent as manufacturing art well before the compound patent itself expired. That manufacturing commoditization is why generic entry was so rapid: several ANDA filers had qualified manufacturing processes ready before the LOE date, with at least six generic versions approved within months of November 2011.
The more durable IP value post-LOE sat in Pfizer’s production scale advantage, regulatory approval dossiers for pediatric and secondary prevention labeling, and the brand equity stored in physician and patient trust — none of which is captured in a standard patent valuation model but all of which drove the commercial outcomes Pfizer achieved in 2012.
Key Takeaways — Lipitor IP Valuation
- Lipitor’s compound patent (US 4,681,893) was the primary income-generating asset; its expiry on November 30, 2011 extinguished roughly $10B+ in annual protected revenue.
- Pediatric exclusivity, method-of-use patents, and formulation claims created a secondary IP layer that supported settlement negotiations and delayed multi-generic competition.
- The authorized generic deal with Watson converted residual manufacturing and brand IP into a revenue-sharing structure worth approximately 70% of generic-segment profits.
- Residual IP value after LOE (brand equity, manufacturing know-how, labeling data) is systematically underweighted in standard DCF valuations and should be explicitly modeled in LOE planning.
4. Patent Expiration, Ranbaxy, and the Paragraph IV Landscape
The Ranbaxy Paragraph IV Challenge
Ranbaxy Laboratories, then a subsidiary of Japan’s Daiichi Sankyo, filed a Paragraph IV ANDA against Lipitor in 2003, asserting that the compound patent was invalid and that Ranbaxy’s generic did not infringe. That filing gave Ranbaxy first-filer status and the 180-day generic exclusivity reward. The subsequent patent litigation consumed five years and produced one of the most commercially consequential pharmaceutical settlements of the decade.
In June 2008, Pfizer and Ranbaxy settled worldwide Lipitor patent litigation. The terms allowed Ranbaxy to begin selling generic atorvastatin in the United States on November 30, 2011 — the compound patent expiration date — with the 180-day exclusivity period running from that date. Other ANDAs from manufacturers including Apotex, Mylan, Sandoz, Teva, and Greenstone were blocked during Ranbaxy’s exclusivity window. This settlement gave Pfizer a defined six-month window before multi-manufacturer generic price collapse, and the company used that window as the commercial centerpiece of its LOE response.
Multi-Filer ANDA Landscape
By November 2011, the FDA had received ANDA submissions for generic atorvastatin from at least eight manufacturers spanning 10mg, 20mg, 40mg, and 80mg doses. The 180-day Ranbaxy exclusivity meant that none of these received final approval until May 2012, after which atorvastatin prices dropped sharply again as the generic market moved toward multi-player equilibrium. Understanding this two-phase entry — single-generic exclusivity followed by multi-generic competition — is essential for LOE financial modeling, because the revenue erosion curve is non-linear: moderate in the first six months, then steep after multi-generic entry.
| Phase | Timeline | Key Competitors | Avg. Generic Price vs. Brand | Pfizer Market Share (Volume) |
|---|---|---|---|---|
| Pre-LOE Brand Peak | 2006–2010 | Crestor (rosuvastatin) only on-patent statin competitor | N/A | ~40% statin market |
| Single-Generic Exclusivity | Dec 2011–May 2012 | Ranbaxy (authorized generic via Watson also active) | ~20–30% of Lipitor list price | ~40% of atorvastatin Rx through co-pay program |
| Multi-Generic Competition | May 2012 onward | Apotex, Mylan, Sandoz, Teva, Greenstone, others | ~5–15% of original Lipitor list price | Declining rapidly; brand effectively commoditized |
5. Pfizer’s Post-LOE Commercial Playbook
Pfizer’s response to the Lipitor LOE is studied precisely because it was pre-planned, multi-layered, and executed against a defined timeline. The commercial team had three objectives: retain as many brand prescriptions as possible through the 180-day exclusivity window, monetize generic demand through the authorized generic deal, and protect brand equity for any future lifecycle extensions. The tactics mapped against those objectives fall into five categories.
Co-Pay Card Mechanics: The $4-per-Month Strategy
Pfizer launched a co-pay assistance program that reduced patient out-of-pocket costs for Lipitor to $4 per 30-day supply for commercially insured patients. A co-pay card program reimburses the gap between a patient’s insurer co-pay and the pharmacy’s cash price. Pfizer was effectively subsidizing patients’ co-pays to match the $4 generic atorvastatin price available through programs like Walmart’s Pharmacy Savings program.
The economic logic: Pfizer’s wholesale acquisition cost (WAC) for Lipitor was far above $4, but the rebate contracts with pharmacy benefit managers (PBMs) and the co-pay card subsidy together allowed Pfizer to maintain prescription volumes while collecting net revenue above generic atorvastatin’s net pricing. The margin per prescription on branded Lipitor, even with the subsidy, exceeded the margin Pfizer earned on its authorized generic because the brand still commanded a higher WAC from which rebates were calculated. Analysts estimated that the co-pay program alone retained 40% of brand Lipitor prescriptions through the exclusivity window, against a typical post-LOE retention of 10% to 15%.
Direct Delivery and Adherence Infrastructure
The ‘Lipitor for You’ program, launched in 2010 and extended through 2012, combined the co-pay card with direct prescription delivery and automated refill reminders. Direct delivery bypassed the pharmacy counter — the point of care where pharmacists, operating under state substitution laws, default to dispensing generic equivalents. By routing prescriptions through a specialty pharmacy channel, Pfizer short-circuited the generic substitution mechanism that operates at most retail chains. The adherence data from the program also served a secondary function: it allowed Pfizer to demonstrate to payers that brand Lipitor patients showed higher refill rates, building a health economics argument for preferred formulary positioning.
PBM and Formulary Negotiation
Pfizer’s managed markets team negotiated formulary positioning with PBMs and health plans, offering rebate structures that kept Lipitor at Tier 2 or Tier 3 rather than non-formulary status. This was critical because formulary tier determines the co-pay differential that drives generic switching at the pharmacy level. A Tier 2 brand co-pay of $40 against a Tier 1 generic co-pay of $10 is a strong switching incentive; that differential narrows substantially when the co-pay card reduces the brand’s effective patient cost to $4. Some PBMs and payers, however, restricted or disallowed co-pay card use for patients covered under government programs (Medicare Part D, Medicaid), which limited the program’s reach among elderly and low-income populations.
6. Authorized Generic Economics: The Watson Deal Deconstructed
The Pfizer-Watson authorized generic arrangement is one of the clearest case studies in how branded pharmaceutical companies convert LOE events from pure revenue destruction into partial monetization. An authorized generic (AG) is a drug manufactured by the brand holder, using the brand’s NDA, but sold under a third-party generic label. The AG is not subject to the 180-day generic exclusivity granted to the first Paragraph IV filer, because it is distributed under the NDA rather than an ANDA.
Pfizer supplied Watson with branded atorvastatin product and Watson distributed it under its own label as a competing generic to Ranbaxy’s authorized generic. Pfizer retained approximately 70% of the profits from Watson’s generic atorvastatin sales. The economic consequence: during Ranbaxy’s 180-day exclusivity window, Pfizer was effectively competing against itself (Ranbaxy generic) with its own authorized generic (Watson label), while also defending the brand (Lipitor) with co-pay support. That three-party structure in the atorvastatin market during the exclusivity window was deliberate and produced a market where Pfizer’s combined brand-plus-AG share significantly exceeded what any single-track strategy would have achieved.
Why Authorized Generics Are Controversial
The FTC has periodically argued that authorized generics undermine the incentive structure Hatch-Waxman created for first-filer Paragraph IV challengers. When a brand launches an AG during the first-filer’s exclusivity window, the first-filer’s 180-day period generates far lower profits because the AG takes market share that the first-filer had modeled as monopoly generic revenue. Some legal scholars argue this dampens future Paragraph IV challenges by making the first-filer reward unpredictable. Pfizer’s Watson arrangement survived regulatory scrutiny, but the FTC’s 2011 study on authorized generics recommended that Congress address the issue. The debate remains active, and portfolio teams modeling generic entry should account for the possibility that AG-enabled competitive structures will receive further regulatory attention.
Investment Strategy
- When evaluating a brand with an approaching LOE, assess whether the company’s manufacturing infrastructure supports a viable authorized generic launch. AG economics are strongest during first-filer 180-day exclusivity windows and become marginal after multi-generic entry collapses prices.
- Model the AG profit-share percentage explicitly. At 70% profit retention (as in the Watson deal), an AG arrangement can contribute 15% to 25% of the combined brand-plus-generic revenue stream during the exclusivity window.
- Discount AG earnings heavily in models for drugs where the FTC or DOJ has signaled concern about anti-competitive AG use, particularly in therapeutically narrow categories where the first Paragraph IV filer is a smaller generic house dependent on the exclusivity premium.
7. Evergreening Tactics and Formulation IP
Evergreening is the practice of filing additional patents on modifications to an existing drug — formulations, dosing regimens, polymorphs, metabolites, enantiomers, or new indications — to extend effective market exclusivity beyond the compound patent’s expiration. The FDA lists these secondary patents in the Orange Book, requiring ANDA filers to address them with Paragraph IV certifications or carve-out statements. Each additional listed patent creates both a potential litigation trigger (30-month stay) and a negotiating chip in settlement discussions.
Atorvastatin’s Evergreening Architecture
Pfizer’s secondary patent filings on atorvastatin covered the crystalline calcium salt form (as opposed to the sodium salt of the active acid), specific tablet formulations, and method-of-use claims tied to secondary cardiovascular prevention indications established in the ASCOT-LLA and TNT trials. The calcium salt patent was commercially important because atorvastatin calcium is the pharmaceutical form used in Lipitor; the synthesis and tableting of the calcium salt gave Pfizer additional patent coverage on the exact product generic filers needed to replicate.
Generic filers responded by filing Paragraph IV certifications against these secondary patents and initiating inter partes reviews (IPR) at the USPTO. The Ranbaxy settlement resolved both the compound patent and secondary patent disputes simultaneously, which is why the settlement’s significance extended beyond the LOE date: it set terms for the entire atorvastatin IP estate rather than leaving secondary patent disputes to produce unpredictable ANDA approval delays.
Technology Roadmap: Formulation-Based Evergreening Strategies
For teams planning lifecycle extensions on a small-molecule asset, the formulation evergreening roadmap has a defined sequence. The first-generation effort targets modified release: extended-release (XR) or controlled-release versions that reduce dosing frequency, improve tolerability, or enable new patient populations. A successful XR or sustained-release formulation qualifies for three years of NCE-equivalent data exclusivity on the new clinical investigation supporting the supplemental NDA. The second-generation effort covers fixed-dose combinations (FDCs), which combine the anchor molecule with a complementary agent to reduce pill burden and qualify for a fresh set of data exclusivities. Pfizer pursued this pathway with Caduet, combining atorvastatin with amlodipine (Norvasc), another Pfizer asset. Caduet received FDA approval in 2004, creating an FDC with its own Orange Book listings and a separate LOE date that extended Pfizer’s atorvastatin franchise past the primary compound patent cliff.
The third-generation formulation effort targets device-drug combinations or novel delivery mechanisms (orally disintegrating tablets, transdermal patches, or nanoparticle formulations) that qualify for device-related IP and potentially 510(k) or PMA regulatory pathways. For statins specifically, this pathway was of limited relevance due to the class’s oral bioavailability and patient adherence profile. For biologics and complex molecules, nano-formulation or device-combination IP represents a significant evergreening runway.
Key Takeaways — Evergreening and Formulation IP
- Secondary patent filings on the crystalline calcium salt form and specific formulations created litigation leverage that supported the Ranbaxy settlement’s favorable terms for Pfizer.
- Fixed-dose combination products (Caduet) represent the most commercially durable evergreening strategy for small-molecule assets because they address a real clinical need and qualify for independent Orange Book listings.
- The Commonwealth Fund and FTC have both documented evergreening as a practice that extends effective exclusivity by an average of 3.5 to 6.5 years on the 12 highest-revenue drugs studied; portfolio teams should model this extension as a probability-weighted upside scenario, not a base case.
- IPR proceedings at the USPTO have increased the vulnerability of secondary formulation patents, with invalidation rates for Orange Book-listed secondary patents higher than for compound patents. Build this risk into exclusivity extension valuations.
8. Detailing Economics Post-Generic Entry
The decision about whether to maintain, reduce, or redirect physician detailing spend after LOE is one of the most consequential commercial calls a brand team makes. The academic literature on this question is more definitive than industry practice might suggest.
A 2023 study published in the International Journal of Research in Marketing examined 72 branded drugs facing generic entry and tracked detailing expenditure and returns for three years post-LOE. The core finding: brands that reduced detailing to 57% of pre-LOE levels on average — the typical industry response — saw standard post-generic erosion curves. Brands that maintained or increased detailing while redirecting it specifically to physicians with demonstrated brand loyalty (high historical share-of-voice responsiveness) saw detailing ROI increase from $1.20 per dollar spent pre-LOE to $3.20 per dollar spent post-LOE. The improvement in ROI occurs because the physician universe has been naturally segmented by LOE: price-sensitive prescribers have already switched to generics, leaving behind a concentrated group of physicians who require specific clinical justification to prescribe brand, and who respond more efficiently to targeted detailing than the pre-LOE heterogeneous physician pool did.
The Loyal Prescriber Segmentation Model
Pfizer’s medical affairs team developed prescriber segmentation data from claims and sales force analytics to identify cardiologists and primary care physicians who were most likely to maintain brand Lipitor prescriptions given adequate clinical justification and co-pay support. These high-loyalty prescribers represented a minority of total Lipitor prescribers but a disproportionate share of post-LOE brand volume. Detailing resources were concentrated on this segment, with sales representatives carrying co-pay card supply, clinical reprints of TNT and ASCOT-LLA trial data, and support materials for the ‘Lipitor for You’ adherence program.
This segmentation approach has become standard LOE practice: the pre-LOE physician universe of several hundred thousand prescribers narrows to a targeted post-LOE list of several thousand high-value prescribers. The contraction in the effective sales territory size reduces absolute detailing cost while the ROI per call increases, improving the economics of maintaining any sales force presence at all in the post-LOE period.
Investment Strategy
- Track companies’ sales force headcount disclosures in the quarters immediately before and after LOE dates. A premature reduction (more than 40% below prior-year levels before LOE) is a signal that management has written off the brand prematurely, which accelerates erosion and destroys residual brand equity that could support a lifecycle extension.
- Companies that maintain a targeted specialty physician sales force post-LOE typically show 15% to 25% higher brand revenue retention at 12 months versus those that eliminate detailing entirely.
- Detailing ROI improvement post-LOE (documented at $1.20 to $3.20 in published research) means that the per-dollar return on maintaining a reduced, segmented sales force post-LOE is substantially higher than any pre-LOE period. Model this when evaluating whether a management team is making rational LOE commercial decisions.
9. Digital and DTC Strategy: The Recipes 2 Go Case Study
In May 2012, Pfizer partnered with EatingWell Media Group to launch the ‘Recipes 2 Go’ mobile application on iOS and Android. The app provided cardiovascular-healthy recipes, a shopping list builder, a cooking timer, and a digital co-pay card for Lipitor. The timing — just before Ranbaxy’s 180-day exclusivity began driving multi-pharmacy generic substitution — was deliberate: Pfizer embedded the brand within a health behavior platform to create a persistent patient touchpoint that existed outside the pharmacy dispensing decision.
The Recipes 2 Go strategy reflected an important insight about DTC in the post-LOE context. Pre-LOE DTC advertising for chronic disease drugs targets awareness and prescription initiation; the audience is undiagnosed or undertreated patients. Post-LOE DTC targeting shifts to existing patients already on the drug, because they are the ones whose prescription could be switched to generic at refill. The mobile application was designed for retention, not acquisition. It created a branded interaction at the moment of medication refill reminder — a behavioral trigger point where patient inertia toward the existing brand is strongest and generic switching is most preventable.
CRM and Patient Engagement Infrastructure
The ‘Lipitor for You’ program built a CRM infrastructure that was genuinely novel for pharmaceutical DTC at the time. Pfizer collected patient consent, prescription data, and refill histories to send periodic communications — health information emails, refill alerts, and co-pay card renewal notifications. The program ran through 2012 and maintained engagement with a patient cohort that might otherwise have drifted to generic at routine refill without awareness that the co-pay card made brand Lipitor cost-competitive.
The privacy and consent architecture of these programs has since evolved significantly. FDA guidelines on patient support programs and the FTC’s enforcement of deceptive practices in pharmaceutical communications have tightened the compliance requirements for CRM-based patient engagement. Teams running equivalent programs today must navigate HIPAA’s marketing exception requirements, state prescription data privacy laws (including California’s CMIA), and FDA’s Office of Prescription Drug Promotion guidance on branded patient communications.
Key Takeaways — Digital and DTC Post-LOE
- Post-LOE DTC strategy should shift from awareness-building to patient retention; the target audience is the existing patient base, not new diagnosis cohorts.
- Mobile applications embedded in health behavior (diet, exercise, monitoring) create branded touchpoints outside the pharmacy dispensing decision, where generic substitution pressure is highest.
- CRM-based refill reminder programs outperform passive co-pay card distribution because they activate at the behavioral moment of highest switching risk.
- Compliance with prescription data privacy laws and FDA promotional guidance is substantially more complex for CRM-based patient programs than for traditional DTC advertising; build compliance review timelines into launch planning.
10. Comparative LOE Responses: Zocor, Cymbalta, and Beyond
Pfizer’s Lipitor defense does not exist in a vacuum. Two contemporaneous cases — Merck’s Zocor and Eli Lilly’s Cymbalta — provide direct comparisons that reveal why some LOE responses produce durable brand retention and others accelerate erosion.
Merck’s Zocor: The Price-Capitulation Strategy
Simvastatin (Zocor) lost patent protection in June 2006. Merck responded by dropping Zocor’s price sharply and negotiating preferred formulary placement for the brand with several major health plans and PBMs. The strategy rested on a different insight than Pfizer’s later Lipitor defense: Merck recognized that simvastatin would be rapidly genericized at very low prices (the $4 Walmart list was for simvastatin, not atorvastatin, initially) and that the brand’s only realistic retention lever was preferred formulary positioning at parity pricing. Merck did not launch an authorized generic or a loyalty co-pay program of the same scale as Pfizer’s later effort. The brand’s market share eroded faster than Lipitor’s would five years later, but Merck’s approach minimized commercial investment in a terminal asset, reallocating resources to the pipeline. The Zocor LOE generated significant volume for generic simvastatin manufacturers and played a role in driving down statin costs broadly — which, indirectly, compressed the commercial opportunity for Lipitor’s later co-pay program by normalizing low statin prices in pharmacy benefit plan designs.
Eli Lilly’s Cymbalta: The Premature Drawdown
Duloxetine (Cymbalta) lost exclusivity in December 2013. Lilly’s response was studied in the IESE Insight research as an example of premature resource reallocation. Lilly began reducing its Cymbalta sales force and marketing investment before the LOE date, redirecting resources toward the pipeline. The outcome was a steeper-than-expected brand erosion curve in the first year post-LOE. The lesson from the Cymbalta case is not that brands should always maintain high spending post-LOE, but that the timing of drawdown matters as much as the magnitude. Reducing detailing and DTC spend three to six months before LOE — when the brand still has exclusivity and prescribers are still making prescribing decisions — accelerates the pre-LOE market share loss that then compounds into a lower base for post-LOE brand retention.
“Lipitor was the first drug to exceed $10 billion a year in sales, and accounted for almost one-quarter of Pfizer’s revenue in the last decade.” — The New York Times, November 29, 2011
Broader Industry Pattern: The LOE Response Matrix
| Drug | Company | LOE Date | Primary Strategy | 12-Month Brand Retention | Authorized Generic |
|---|---|---|---|---|---|
| Lipitor (atorvastatin) | Pfizer | Nov 2011 | Co-pay card + AG + CRM loyalty program | ~40% | Yes (Watson) |
| Zocor (simvastatin) | Merck | Jun 2006 | Price reduction + preferred formulary | ~15% | No |
| Cymbalta (duloxetine) | Eli Lilly | Dec 2013 | Premature sales force reduction | ~8% | Yes (Lilly) |
| Plavix (clopidogrel) | BMS/Sanofi | May 2012 | Co-pay assistance + indication messaging | ~12% | Yes (BMS) |
The pattern across the LOE matrix is consistent: brands that combine co-pay card programs with authorized generic monetization and targeted detailing outperform those using any single strategy in isolation. The Lipitor case remains the performance benchmark precisely because Pfizer deployed all three simultaneously, executing each against a defined 180-day exclusivity timeline.
11. Regulatory Scrutiny, FTC Oversight, and Litigation Risk
Pfizer’s LOE strategy generated legal exposure on multiple fronts. In July 2012, a group of retail pharmacies filed suit against Pfizer alleging that the company had conspired with Medco Health Solutions (now Express Scripts) and CVS Caremark to restrict generic atorvastatin dispensing during the Ranbaxy exclusivity period. The retailers alleged that agreements between Pfizer and PBMs created economic incentives for pharmacies to dispense brand Lipitor rather than Ranbaxy’s generic, effectively extending the practical duration of brand-level pricing beyond what the patent settlement had intended. The litigation was settled out of court, but it established the template for FTC scrutiny of the intersection between co-pay card programs and PBM contracting in the post-LOE period.
Reverse Payment Settlements and Actavis
The Ranbaxy settlement is often discussed alongside the broader debate over reverse payment (pay-for-delay) settlements. In a reverse payment structure, the brand pays the first-filer Paragraph IV challenger to accept a later generic entry date, delaying competition in exchange for cash or value. The U.S. Supreme Court’s 2013 decision in FTC v. Actavis, Inc. held that reverse payment settlements are subject to antitrust scrutiny under the rule of reason, rather than being presumptively legal. The Pfizer-Ranbaxy settlement predated Actavis and its terms — allowing generic entry on the patent expiration date rather than earlier — did not constitute a classic pay-for-delay arrangement. But the settlement’s broader architecture (including terms restricting third-party generic dispensing and the coordinated launch of the Watson authorized generic) sat adjacent to conduct that the FTC has since characterized as potentially anti-competitive.
Post-Actavis, reverse payment settlements have declined in frequency, and generic entry dates in patent settlements have moved closer to the compound patent expiration date. Portfolio teams evaluating branded assets near LOE should assume that any settlement structure offering the generic challenger significant value (whether monetary or non-monetary) will receive FTC scrutiny, and should build litigation reserve assumptions into LOE financial models.
Key Takeaways — Regulatory and Litigation Risk
- FTC v. Actavis (2013) eliminated the presumptive legality of reverse payment settlements; any settlement granting the first Paragraph IV filer material value for delaying entry is now subject to rule-of-reason antitrust analysis.
- Co-pay card programs that interact with PBM formulary positioning may attract antitrust scrutiny if the combined effect is to restrict generic dispensing beyond what the patent estate alone justifies.
- Retailer suits against brands for post-LOE generic delay tactics have become a recurring litigation risk; build legal reserve assumptions into LOE commercial models for drugs above $2B annual revenue.
- The intersection of co-pay cards, authorized generics, and PBM rebate contracting is the highest regulatory-risk zone in post-LOE commercial strategy; involve antitrust counsel in program design before launch.
12. Patent Intelligence Platforms and Strategic LOE Planning
Effective LOE management depends on knowing the exact structure of your own patent estate and your competitors’ filing activity years before the cliff date. Patent intelligence platforms — DrugPatentWatch being the most widely cited in the pharmaceutical industry — aggregate Orange Book listings, ANDA filing data, litigation dockets, Paragraph IV certification notices, and generic approval timelines into a queryable database that supports both proactive planning and competitive intelligence.
What Patent Intelligence Actually Enables
The practical utility of a platform like DrugPatentWatch spans several distinct use cases across the enterprise. IP teams use it to audit their own Orange Book listings for accuracy, identify secondary patents that may have been inadvertently omitted from Orange Book submission, and track competitor ANDA filings that signal imminent Paragraph IV challenges. Commercial teams use generic entry timeline data to trigger LOE commercial planning gates — typically 36 months before the compound patent expiration date for a drug above $500M annual revenue, and 24 months for smaller assets.
Portfolio managers and institutional investors use patent expiration data as the primary input to LOE financial models, with the ANDA filing activity and litigation docket data providing probability-weighted adjustments to the base LOE date. A drug with ten active Paragraph IV challenges and no strong secondary patent coverage will have a different expected LOE date distribution than a drug with one challenge and a dense formulation patent thicket. The litigation docket data captures the settlement history and pending outcomes that drive that probability distribution.
Integration with Commercial Planning Gates
Best-practice LOE planning uses patent intelligence data to define commercial planning gates. At 36 months before projected LOE, the brand team completes a patent estate audit, authorized generic feasibility assessment, and preliminary co-pay card budget modeling. At 24 months, the lifecycle management team submits supplemental NDA filings for any formulation improvements or new indication data that could support additional Orange Book patent listings or data exclusivities. At 18 months, the authorized generic partner negotiation is finalized and the co-pay card program is designed. At 12 months, physician segmentation data is built, detailing redeployment plans are finalized, and PBM formulary negotiation is active. At six months, the co-pay card is launched and the DTC retention campaign begins.
This gate model is standard at Tier 1 pharmaceutical companies with dedicated lifecycle management functions. Smaller companies and biotech licensors often lack the infrastructure to execute all six gates, which is where the outcome diverges from the Lipitor benchmark: the brand either fails to launch co-pay infrastructure in time, misses the authorized generic partner negotiation window, or enters the LOE date without a segmented physician target list.
13. Investment Strategy for Analysts
The following framework applies to institutional investors and buy-side analysts evaluating pharmaceutical companies with material LOE events within a 36-month forecast horizon.
- Identify the compound patent expiration date and any PTEs from the Orange Book. Then identify all secondary patent listings and their expiration dates. The gap between compound patent expiration and the last secondary patent expiration is the effective IP runway, but only if the secondary patents are defensible — check the litigation docket for IPR filings and Paragraph IV challenges against each listed secondary patent.
- Model three LOE scenarios: base (compound patent expiration date, all secondary patents settled or expired), early (Paragraph IV challenge succeeds, generic entry 24 months early), and late (pediatric exclusivity or secondary patent litigation delays multi-generic entry by 6 to 18 months). Weight by litigation probability. The spread between early and late scenarios is the IP risk-adjusted valuation range.
- Check whether the company has executed authorized generic arrangements historically. Companies with AG infrastructure and existing generic distribution partnerships can deploy an AG within 90 days of LOE decision. Companies without that infrastructure face a 12- to 18-month lead time, meaning they will miss the first-filer 180-day exclusivity window entirely.
- Track sales force headcount disclosures in the four quarters before the LOE date. A premature reduction (greater than 40% below prior-year levels before LOE) is a signal that management has written off the brand prematurely, which accelerates erosion and destroys residual brand equity that could support a lifecycle extension.
- Evaluate whether the company has a fixed-dose combination product or reformulation in the NDA pipeline that extends the effective franchise past the primary compound LOE. Caduet (atorvastatin/amlodipine) generated independent revenue that offset a portion of Lipitor’s erosion. This type of franchise extension is underweighted in consensus models because it requires analysts to separately value the combination product’s LOE date from the primary compound’s.
- For biologics rather than small molecules: replace ANDA/Paragraph IV analysis with 351(k) biosimilar interchangeability applications. The biosimilar market has slower erosion dynamics than small-molecule generics — typically 30% to 50% volume loss in the first year versus 80% to 89% for small molecules — because biosimilar interchangeability designations require additional switching studies, and prescriber inertia is stronger. Adjust LOE erosion curve assumptions accordingly.
14. FAQ
What is the patent cliff in pharmaceutical investment analysis?
The patent cliff is the revenue discontinuity that occurs when a drug’s primary compound patent expires and generic versions enter the market under the Hatch-Waxman ANDA pathway. The average brand loses 80% to 89% of its prescription volume within six months of generic entry. For drugs above $1B annual revenue, this loss is material enough to move company-level earnings per share by 10% to 40% in the LOE year, which is why patent expiration dates are primary inputs to pharmaceutical equity models.
What is an authorized generic, and how does it affect the first-filer’s 180-day exclusivity?
An authorized generic is a product marketed by the brand holder under a third-party label, using the brand’s NDA rather than an ANDA. Because it is distributed under the NDA, it is not subject to the 180-day generic exclusivity granted to the first Paragraph IV filer. The AG therefore competes directly with the first filer during the exclusivity window, reducing the first filer’s monopoly profits. This has been documented to reduce first-filer exclusivity period revenues by 40% to 60%, which the FTC argues dampens incentives for future Paragraph IV challenges.
What is evergreening, and how do regulators constrain it?
Evergreening is the filing of additional Orange Book-listed patents on modifications to an approved drug — formulations, salts, polymorphs, metabolites, or new indications — to extend effective market exclusivity past the compound patent’s expiration. The FTC and academic researchers have documented that the 12 top-selling drugs in the United States averaged 125 patent applications each, with median secondary patent terms extending effective exclusivity by 3.5 to 6.5 years. The principal regulatory constraints are the ‘listing’ requirements for Orange Book patents (patents must claim the drug or a method of using it, not merely manufacturing processes) and the IPR procedure at the USPTO, which allows generic filers to challenge secondary patent validity with a lower burden of proof than district court litigation.
How does physician detailing ROI change after generic entry?
Published research on 72 branded drugs found that detailing ROI increases from approximately $1.20 per dollar spent pre-LOE to $3.20 per dollar spent post-LOE when detailing is redirected to loyal prescribers rather than distributed across the full pre-LOE physician universe. The improvement occurs because LOE naturally segments the prescriber market: price-sensitive physicians have switched to generic, leaving a concentrated pool of brand-loyal prescribers who are efficiently targeted with clinical-differentiation messaging and co-pay support.
What did the Ranbaxy settlement with Pfizer actually allow?
The June 2008 settlement resolved worldwide Lipitor patent litigation between Pfizer and Ranbaxy (then a subsidiary of Daiichi Sankyo). Ranbaxy received the right to sell generic atorvastatin in the United States beginning November 30, 2011 — the compound patent expiration date — with 180-day first-filer exclusivity. The settlement also covered secondary patents and resolved litigation in multiple international jurisdictions. Pfizer used the defined 180-day window to launch its co-pay card program, authorized generic with Watson, and CRM loyalty infrastructure before multi-generic price collapse in May 2012.
How should biosimilar LOE be modeled differently from small-molecule generic entry?
Biosimilar market entry under the 351(k) pathway produces slower and shallower erosion curves than small-molecule ANDA generics. Biologics competition shows 30% to 50% volume erosion in the first year in most categories, versus 80% to 89% for small molecules. The slower erosion reflects higher prescriber switching inertia, the additional clinical evidence required for biosimilar interchangeability designation (which enables pharmacy-level substitution), and payer formulary management practices that often support a gradual rather than immediate transition. LOE analysts should apply category-specific erosion assumptions rather than using small-molecule curves as defaults for biologic assets.
References
- DiMasi JA et al., ‘Innovation in the pharmaceutical industry: New estimates of R&D costs,’ Journal of Health Economics, 2016.
- Harvard Health Blog, ‘Consumers could save as generic Lipitor hits the market,’ December 2011.
- PM360, ‘Pfizer’s 180-Day War for Lipitor,’ 2012.
- Journal of Ethics, AMA, ‘For Me There Is No Substitute: Authenticity, Uniqueness, and the Lessons of Lipitor,’ October 2010.
- Health Populi, ‘Statins, Food, and a Mobile App,’ May 2012.
- The New York Times, ‘Facing Generic Lipitor Rivals, Pfizer Battles to Protect Its Cash Cow,’ November 29, 2011.
- Pfizer press release, ‘Pfizer and Ranbaxy Settle Lipitor Patent Litigation Worldwide,’ June 16, 2008.
- GaBI Online, ‘Pfizer continues to fight for Lipitor,’ 2011.
- Patent Docs, ‘Pfizer’s Lipitor: A New Model for Delaying the Effects of Patent Expiration,’ November 30, 2011.
- PMLiVE, ‘Pfizer taps brand loyalty to protect Lipitor from generics,’ November 29, 2011.
- Health Populi, ‘Statins, Food, and a Mobile App,’ May 23, 2012.
- Pfizer-Ranbaxy settlement documentation, June 2008.
- Reuters, ‘Pfizer interested in over-the-counter Lipitor: report,’ August 4, 2011.
- International Journal of Research in Marketing / ScienceDirect, ‘Branded response to generic entry: Detailing beyond the patent cliff,’ 2023.
- CNBC, ‘Facing Generic Lipitor Rivals, Pfizer Battles to Protect Its Cash Cow,’ November 30, 2011.
- NPR, ‘Merck to Drop Price as Zocor Patent Lapses,’ June 23, 2006.
- IESE Insight, ‘To promote or not to promote? The mixed fate of branded drugs when generics arrive,’ January 13, 2025.
- DrugPatentWatch, https://www.drugpatentwatch.com/
- The Commonwealth Fund, ‘Strategies That Delay Market Entry of Generic Drugs,’ September 2017.
- Shrank et al., ‘The implications of choice,’ Archives of Internal Medicine, 2006.
- CBS News, ‘Retailers sue Pfizer over generic Lipitor delay,’ July 6, 2012.
- FTC v. Actavis, Inc., 570 U.S. 136 (2013).
- FTC Study: Authorized Generic Drugs: Short-Term Effects and Long-Term Impact, August 2011.