Introduction: The Patent Filing Is a Financial Document

The global prescription drug market cleared $1.6 trillion in annual revenue by end-2024. Every dollar of that figure traces, directly or indirectly, to a patent filing. Not to a Phase III readout. Not to an FDA approval letter. To the specific dates, claims language, and jurisdictional coverage of a set of intellectual property documents that most business professionals never read past the abstract.
That is a systematic analytical error, and it costs money.
A patent is not legal boilerplate. Read correctly, it is a revenue projection. The filing date sets the expiration clock. The claims language determines whether a generic challenger can design around the molecule or must litigate every claim. The geographic footprint signals how seriously management believes in the asset’s global commercial potential. The density and filing timeline of the surrounding patent family reveals whether the company intends to defend peak sales or is already preparing for a managed decline.
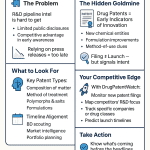
Between now and 2030, analysts at IQVIA project more than $236 billion in annual branded pharmaceutical revenue is exposed to Loss of Exclusivity (LOE). At the same time, patent thickets have grown so dense around several top-selling biologics that some primary composition of matter patents expired years ago while the drugs remain entirely insulated from biosimilar competition. These are not contradictions. They are two faces of the same analytical challenge: the naive LOE date and the real LOE date are often separated by years, and the difference between those two dates is where fortunes are made and lost.
This guide exists to close that gap. It covers the full analytical stack, from the foundational taxonomy of pharmaceutical monopoly rights through quantitative DCF construction, AI-driven predictive modeling, and the jurisdiction-specific legal environments that produce wildly different LOE dates for the same molecule in the U.S., EU, India, and China. Each section includes IP valuation spotlights on specific drugs and companies, a Key Takeaways summary, and an Investment Strategy note for institutional analysts.
Section 1: The Anatomy of Pharmaceutical Exclusivity
What Gives a Drug Its Monopoly, and How Long Does It Actually Last?
Market exclusivity in pharmaceuticals is not a single switch that flips on at FDA approval and off at patent expiration. It is a layered system of overlapping rights, each granted by a different authority under different statutory criteria, each expiring on a different date. Treating it as a monolith is the first and most common forecasting error.
Patent Protection vs. Regulatory Exclusivity: Know Your Monopoly Types
Patents and regulatory exclusivities are legally distinct instruments that can operate independently, run concurrently, or stack on top of each other, and the final LOE date is determined by whichever protection expires last.
A patent is a property right granted by a national patent office (the USPTO in the United States) based on criteria of novelty, non-obviousness, and utility. It blocks others from making, using, or selling the patented invention for a statutory term of 20 years from the application filing date. The scope of that block is defined entirely by the patent’s claims language, which means two patents covering the same drug can offer radically different levels of protection depending on how their claims were drafted and whether the USPTO examiner allowed broad or narrow claim language.
Regulatory exclusivity is a market right granted by the FDA upon approval of a drug product. It does not require a patent. Its purpose is to incentivize specific categories of innovation by guaranteeing a period during which the agency will not accept or approve competing generic or biosimilar applications, regardless of the underlying patent situation. A drug can lose all patent protection and retain full regulatory exclusivity. A drug can have robust patent coverage with no regulatory exclusivity. Each scenario produces a different revenue forecast.
The U.S. framework offers several distinct regulatory exclusivity types, and every analyst working in the space needs to know their precise parameters:
New Chemical Entity (NCE) exclusivity grants 5 years of FDA protection to drugs containing an active moiety never previously approved by the agency. It is the most commercially significant form of regulatory exclusivity for small-molecule drugs because it blocks the FDA from even accepting an Abbreviated New Drug Application (ANDA) for the first 4 years, eliminating the risk of early generic challenges for that window.
Orphan Drug Exclusivity (ODE) grants 7 years of market protection for drugs that treat diseases affecting fewer than 200,000 people in the United States. For drugs in rare disease categories, ODE is frequently the dominant exclusivity, often outlasting the primary composition of matter patent and proving decisive in shaping the LOE date. The rare disease pipeline has leaned hard on this provision: orphan designations granted by the FDA rose from roughly 200 per year in 2015 to more than 700 per year by 2023.
Biologics exclusivity under the Biologics Price Competition and Innovation Act (BPCIA) grants 12 years of market exclusivity from the date of first licensure, plus an additional 4-year data exclusivity period during which biosimilar applicants cannot even reference the originator’s clinical data. This 12-year period reflects the political bargain struck during the ACA negotiations and has been central to the enormous profitability of the biologic sector. It is also the primary reason that biosimilar penetration in the U.S. remained far lower than in Europe for the first decade after the BPCIA’s passage.
Pediatric exclusivity adds 6 months to any and all existing patents and regulatory exclusivities when a company completes FDA-requested pediatric studies. For a blockbuster drug generating $10 billion annually, 6 months of additional exclusivity has a present value that can easily exceed $1 billion after discounting, making it consistently worth pursuing.
The 3-year new clinical investigation exclusivity applies when a company obtains a new approval (new indication, new formulation, new combination) backed by new clinical studies. It protects only the specific change, not the underlying drug, but it is a standard tool in lifecycle management precisely because it can block generic substitution for the new indication while the rest of the patent thicket is being fought in court.
The Full Patent Arsenal: From Composition of Matter to Process Claims
A sophisticated reading of a drug’s patent portfolio requires understanding that every patent type within it has a different strategic function and a different vulnerability profile for generic challengers.
Composition of matter (COM) patents, also called base patents, cover the active pharmaceutical ingredient itself. They are the most powerful patents in the industry because a valid COM patent bars any competitor from selling the same API for any purpose during its term. Their Achilles heel is that they typically expire first, having been filed during early preclinical research when 10 to 14 years of patent life will be consumed by development and regulatory review before a single dollar of commercial revenue is collected.
Method-of-use patents protect specific therapeutic applications of a drug, not the molecule itself. A generic manufacturer can, in principle, sell a drug for off-patent indications while citing the remaining method-of-use patents only in the context of a “skinny label,” legally omitting the patented indication from the product labeling. However, skinny-label strategies create commercial risk for the generic because prescribers often prescribe by drug name regardless of indication, and brand manufacturers have successfully argued induced infringement in several litigations where the generic drug was widely used for the patented indication despite the skinny label.
Formulation and delivery system patents protect novel dosage forms, salt forms, polymorphic crystal forms, particle size specifications, proprietary excipient combinations, and drug delivery devices like autoinjectors, prefilled syringes, or inhalation platforms. These are the workhorses of lifecycle management for small-molecule drugs. They frequently survive patent challenges because they can meet the legal non-obviousness standard (a skilled formulation chemist would not have arrived at the same composition without undue experimentation) even when they offer only modest clinical differentiation.
Process patents cover manufacturing methods, including synthesis routes for small molecules and cell culture and purification processes for biologics. For biologics, manufacturing process patents are often the most durable defensive tool available because replicating a proprietary cell line and its associated purification train without infringing is technically demanding and legally risky. Many biologic manufacturers also hold manufacturing know-how as trade secrets rather than filing patents, a deliberate strategy that provides indefinite protection rather than the 20-year patent term.
Combination patents cover fixed-dose combinations of two or more active ingredients. They are particularly prevalent in HIV (where three-drug combination regimens are standard of care), cardiovascular disease, and respiratory medicine. Their vulnerability is that they often cannot block a competitor from selling the individual components separately once those components are off-patent; they only protect the specific combination.
Statutory Term vs. Effective Patent Life: The Core Economic Problem
The 20-year statutory patent term sounds generous until it is mapped against actual drug development timelines. A composition of matter patent for a new molecular entity is typically filed in the early discovery phase, often 12 to 14 years before the drug reaches commercial patients. By the time FDA approval is granted and first commercial sale occurs, the patent frequently has 6 to 8 years of statutory life remaining. This is the effective patent life, and it is the only period during which the patent actually translates into monopoly revenue.
Patent Term Extension (PTE) under the Hatch-Waxman Act partially addresses this problem by allowing brand manufacturers to recover a portion of the patent term lost to FDA regulatory review. The maximum PTE is 5 years, and the total remaining patent life after extension cannot exceed 14 years from FDA approval. In practice, PTE grants of 3 to 4 years are common for NCEs, meaningfully extending the effective patent life but not fully restoring it. Extensions require filing a PTE application within 60 days of approval, and the calculation of the eligible extension period involves detailed FDA and USPTO review of the clinical and regulatory timeline.
For biologics, the 12-year BPCIA exclusivity typically dominates the effective exclusivity period anyway, making PTE less critical than for small molecules. But for small-molecule drugs with large market opportunities, the delta between a 4-year PTE grant and the maximum 5 years can represent hundreds of millions in NPV, making the PTE application process a high-return legal exercise.
The average effective patent life for a marketed small-molecule drug in the U.S. runs approximately 11.5 years when the PTE is included, down from the statutory 20 years. For drugs with unusually long development programs, such as CNS drugs where Phase III trials frequently last 3 to 5 years, effective patent life can be as short as 7 years. This compression is the primary economic driver of every lifecycle management tactic discussed in Section 4.
IP Valuation Spotlight: How Patent Type and Portfolio Composition Determine Asset Value
The composition of a drug’s patent portfolio, not just the expiration date of any single patent, is the primary determinant of its IP valuation. Two drugs with identical peak sales forecasts can have radically different IP asset values depending on portfolio structure.
Consider a hypothetical drug generating $3 billion in annual peak sales with a COM patent expiring in 2029. If that COM patent is the drug’s only substantive protection, the IP asset value is the NPV of approximately 5 years of peak sales minus rapid post-LOE erosion, risk-adjusted for litigation outcomes. At a 10% discount rate and with standard generic erosion curves, that NPV might compute to $6 to $7 billion.
Now consider the same drug with an identical COM expiration but a surrounding portfolio of 35 formulation, method-of-use, and device patents extending through 2035. The expected LOE date shifts 6 years. The IP asset value, all else equal, increases to $11 to $13 billion. The difference is entirely attributable to the composition and strength of the secondary patent portfolio, not the underlying molecule’s clinical profile.
One empirical study of 847 FDA-approved drugs found that drugs with more than 50% of their portfolio consisting of method-of-use patents showed 18% higher peak sales than those anchored by a single COM patent. The mechanism is commercial, not legal: a method-of-use-heavy portfolio indicates an active indication expansion strategy, where each new approved indication adds a new patient population and extends the revenue curve beyond the original launch trajectory.
Key Takeaways: Section 1
The effective patent life for a marketed small-molecule drug averages roughly 11.5 years post-launch in the U.S. once PTE is factored in. Regulatory exclusivities, particularly BPCIA’s 12-year term for biologics and 7-year ODE for rare disease drugs, frequently determine the real LOE date rather than any patent expiration. Portfolio composition (the ratio of COM to secondary patents, and their individual strengths) is as material to IP valuation as peak sales projections. A forecaster who reads only the Orange Book patent expiration date without examining the full portfolio will produce a systematically optimistic LOE date for brand manufacturers and a systematically pessimistic generic entry date for challengers.
Investment Strategy Note
For portfolio managers evaluating pharma equities or licensing opportunities: build a custom LOE matrix that separately tracks (1) COM patent expiration, (2) latest-expiring secondary patent with substantive claims, (3) relevant regulatory exclusivity expiration, and (4) Paragraph IV filing activity. The LOE date to use in your DCF should be the latest of these four dates, weighted by litigation risk. Companies trading at multiples that implicitly assume an early LOE date are often mispriced relative to the full portfolio view.
Section 2: Three Valuation Methodologies That Actually Work
Turning Patent Data Into a Financial Figure
Identifying the LOE date is necessary but not sufficient. The analyst’s final deliverable is a financial value: how much revenue will this drug generate before competition arrives, what does that translate to in present value, and how does that present value compare to the asset’s acquisition or licensing price? Three methodologies answer that question, and using all three in combination produces more defensible forecasts than any single approach.
The Income Approach: Building a Patent-Based DCF Model
The Discounted Cash Flow model is the industry standard for pharma asset valuation. It is also the methodology most exposed to garbage-in-garbage-out risk, because its output is only as credible as its inputs, and its inputs span epidemiology, pricing strategy, competitive dynamics, and clinical risk, each of which carries its own uncertainty.
Step 1: Projecting the Revenue Baseline
The revenue projection starts with an epidemiological foundation. Prevalence (the total number of living patients with the condition) drives the market for chronic disease therapies. Incidence (new cases per year) drives acute and one-time-treatment markets. Both figures require sourcing from multiple databases (IMS/IQVIA, CDC, WHO, peer-reviewed epidemiology literature) because estimates can vary by 20 to 40% depending on the diagnostic criteria used, and the choice of estimate meaningfully affects the final forecast.
From the total patient population, the analyst builds a funnel: diagnosis rate (what fraction of patients receive a formal diagnosis), treatment rate (of those diagnosed, what fraction receive pharmacotherapy), market share (of those treated pharmacologically, what fraction receive this specific drug versus alternatives), and compliance/persistence (of those prescribed, what fraction take the drug as directed and remain on therapy long-term).
Each parameter in this funnel is a modeling choice that requires justification. Analysts who set market penetration at the drug’s eventual steady-state share without modeling the ramp-up period will overstate early-year revenues and inflate the NPV. Conversely, analysts who use a conservative penetration ceiling without modeling indication expansion or label changes will understate long-term revenue for drugs with active lifecycle management programs.
Net price, distinct from list price, is particularly complex in the U.S. market. Gross-to-net discounts have expanded substantially over the past decade, with specialty drugs in competitive classes frequently carrying gross-to-net discounts of 40 to 60% in practice. Using WAC (Wholesale Acquisition Cost) as the revenue assumption without applying a gross-to-net adjustment overstates net revenue and produces inflated NPV figures.
Step 2: Modeling the Patent Cliff
The patent cliff is the steepest single-year revenue decline most branded drugs ever experience, and its magnitude varies considerably by drug type, therapeutic area, route of administration, and the number of generic or biosimilar entrants.
For oral small-molecule drugs, the empirical data on post-LOE erosion is precise. In markets with six or more generic entrants at launch, branded price retains roughly 25 to 30% of pre-LOE WAC within 12 months, and branded volume can fall to single-digit percentage points of the total market within 24 months. The first generic entrant typically captures 50 to 60% of the generic market volume through the 180-day exclusivity period that Hatch-Waxman grants to first filers.
For injectable and biologic drugs, erosion curves are fundamentally different. Biosimilar uptake in the U.S. has been slower than early projections for three structural reasons: absence of automatic substitution at pharmacy level (unlike the small-molecule generic substitution mandated by most state pharmacy laws), physician hesitancy to switch stable patients, and formulary decisions by PBMs that often maintain brand-preferred status in exchange for substantial rebates. Real-world biosimilar penetration data from adalimumab biosimilars launched in January 2023 showed that by month 9, the combined biosimilar share reached approximately 15 to 18% of the anti-TNF market by volume, well below the 40% first-year penetration that most analyst models had projected. This slower erosion curve is a systematic upward bias in DCF models that use small-molecule erosion assumptions for biologic assets.
Step 3: Risk Adjustment, Discount Rates, and NPV
A DCF model without risk adjustment is a forecast of a world with no uncertainty. Risk adjustment can take two forms, which can be applied individually or in combination.
The first approach is probability-weighting the cash flows themselves. A Phase III asset facing an estimated 65% probability of approval (industry average for Phase III NCEs is approximately 55 to 65% depending on therapeutic area) has its post-approval cash flows multiplied by 0.65 before discounting. A drug approved but facing a 30% probability of a major safety label change in the next 3 years has those years’ cash flows haircut accordingly.
The second approach is reflecting risk in the discount rate. Development-stage pharmaceutical assets are typically discounted at rates of 10 to 15% for late-stage clinical assets and 20 to 30% for early-stage (Phase I or II) assets. Approved commercial-stage drugs are discounted at lower rates, typically 8 to 12%, reflecting the elimination of regulatory risk but the persistence of competitive, pricing, and patent litigation risk.
The Net Present Value (NPV) output of a well-constructed DCF for a marketed blockbuster with a remaining effective patent life of 10 years, $5 billion in peak annual sales, and standard U.S. biologic erosion assumptions might fall between $18 and $28 billion, varying significantly with the discount rate and erosion curve assumptions. The range, not just the point estimate, is the analytically useful output. Sensitivity tables showing NPV as a function of LOE date (column) and peak penetration (row) communicate the key variable interactions and allow the reader to stress-test the valuation against their own assumptions.
IP Valuation Spotlight: Pfizer/Lipitor, The Canonical Patent Cliff Case
Lipitor (atorvastatin) generated $13.6 billion in global revenues in 2006, its peak year, making it the best-selling drug in history at the time. Its U.S. primary composition of matter patent was set to expire November 30, 2011. Pfizer used a combination of PTE, method-of-use patents, and an Authorized Generic (AG) strategy to manage the cliff, but the magnitude of post-LOE erosion still exceeded most analyst projections.
Within 12 months of U.S. generic entry in November 2011, Lipitor’s U.S. revenue had declined approximately 78% from its pre-LOE baseline. Total revenue collapsed from $13.6 billion at peak to approximately $3.9 billion by fiscal year 2012. The present value of those lost future cash flows, which had been embedded in Pfizer’s stock price in 2010 and 2011, represented one of the largest single-drug LOE value destructions on record. Pfizer’s response, the $68 billion acquisition of Wyeth in 2009, was in part an attempt to replace the Lipitor revenue with a biologic portfolio (primarily Prevnar vaccines and Enbrel co-promotion rights) that would not face an equivalent generic cliff.
The Lipitor case demonstrates that even the most sophisticated LOE management for a small-molecule drug cannot materially alter the fundamental erosion curve after the COM patent expires and multiple generics enter the market. Lipitor’s secondary patent portfolio was not negligible, but it was not a thicket of the density that would define the next generation of biologics.
The Market Approach: Comparable Transactions and the CAR-T Benchmark
The Market Approach values an asset by reference to what informed buyers have paid for comparable assets in arm’s-length transactions. Its power lies in grounding the valuation in demonstrated market prices rather than modeled assumptions. Its limitation is finding transactions that are genuinely comparable along the dimensions that drive value: mechanism of action, therapeutic area, stage of development, patent portfolio strength, and competitive positioning.
For precedent M&A transactions, the most widely used metric is EV/Revenue or, for late-stage pipeline assets, EV per probability-adjusted peak sales dollar. For license deals, the standard metrics are upfront payment as a percentage of potential deal value, royalty rates as a percentage of net sales (typically 5 to 20% for late-stage assets depending on therapeutic area and exclusivity strength), and the structure and reachability of milestone payments.
IP Valuation Spotlight: Novartis/Kymriah and the CAR-T Market Comparables Problem
Novartis’s Kymriah (tisagenlecleucel) was the first CAR-T cell therapy to receive FDA approval, in August 2017 for relapsed/refractory pediatric ALL. Its IP position is built on foundational patents from the University of Pennsylvania covering the CAR construct design, licensed exclusively to Novartis, plus manufacturing process patents covering the ex vivo T-cell engineering workflow and the lentiviral vector technology used to transduce patient cells.
When analysts needed to value Kymriah’s IP independently for deal analysis and portfolio benchmarking, the only available market comparables were Gilead’s acquisition of Kite Pharma for $11.9 billion in August 2017 (pre-Yescarta approval) and Celgene’s $9 billion acquisition of Juno Therapeutics in January 2018. Both transactions implied a premium for novel CAR-T IP, but neither was a clean comparable: Kite’s Yescarta used a retroviral rather than lentiviral vector (different IP landscape), Juno’s bb2121 program was still early-stage, and all three companies faced the same unresolved question of whether outpatient CAR-T administration (which would dramatically expand addressable market) was achievable.
Adjusting for Kymriah’s superior clinical response rate in pediatric ALL (81% complete remission vs. 72% for Yescarta in a different indication), but discounting for Kymriah’s narrower approved indication at the time, analysts at the major investment banks converged on a standalone Kymriah IP valuation of approximately $8 to $11 billion. The range reflects the CAR-T market’s structural uncertainty at that moment, including reimbursement uncertainty (Kymriah launched at $475,000 per infusion) and manufacturing scalability questions.
What the CAR-T example illustrates is that the Market Approach performs poorly when a drug is a genuine first-in-class therapy with no direct biological or commercial analogue. In these cases, the approach requires extensive qualitative adjustment that reduces precision and can produce a false sense of comparability where none exists.
The Cost Approach: R&D Spend as a Valuation Floor
The Cost Approach takes the total R&D investment required to produce the asset and treats it as the baseline below which the asset should not logically sell. The average cost of developing a new molecular entity through FDA approval was estimated at $2.3 billion in a 2023 analysis by the Tufts Center for the Study of Drug Development, up from $802 million in 2000 (in 2000 dollars), reflecting both genuine cost escalation and increasing trial complexity.
The Cost Approach’s fundamental problem is that cost does not correlate with commercial success. A company can spend $3 billion developing a drug that receives a Complete Response Letter from the FDA and generates zero revenue. A company can license a compound for $50 million that becomes a $5 billion blockbuster. The market pays for future cash flows, not for past expenditures. For this reason, the Cost Approach functions primarily as a due diligence sanity check (“did we spend at least this much to justify this valuation?”) and as a floor in early-stage asset negotiations where future cash flows are too uncertain to project reliably.
Key Takeaways: Section 2
DCF models for pharma assets require explicit, data-grounded assumptions at each step: prevalence/incidence, penetration ramp, net (not list) price, gross-to-net discount, LOE date, and post-LOE erosion curve calibrated to drug type rather than using a generic template. Post-LOE erosion for biologics runs 40 to 60% slower in the U.S. than for oral small molecules; using the wrong curve produces a material NPV error. The Market Approach has limited precision for first-in-class biologics due to the absence of comparable transactions; analysts must document and defend every qualitative adjustment. The Cost Approach is a floor, not a measure of commercial value.
Investment Strategy Note
Analysts acquiring or licensing late-stage assets should run all three valuation methodologies and use them as a triangulation. The DCF establishes the upper bound under optimistic but defensible assumptions. Precedent transaction multiples provide market reality anchors. The R&D cost provides the floor. If the DCF and Market Approach outputs diverge by more than 30%, the source of divergence is where the analytic work should focus: it is almost always a difference in LOE date assumptions, erosion curve choices, or peak penetration estimates.
Section 3: The Algorithmic Edge: AI, ML, and Predictive Analytics
Why Traditional Models Consistently Fail New Drug Launches
The pharmaceutical sales forecasting track record is not good. An analysis of 50 U.S. prescription drug launches found that only 12% of first-year sales forecasts were accurate within a 25% margin of actual results. Roughly 32% of drugs were overestimated by more than double their actual first-year sales. Another 28% were underestimated by equivalent magnitudes. These are not rounding errors; they reflect systematic failures in the analytical frameworks used to build the forecasts.
The Limits of ARIMA and Traditional Time-Series Models
ARIMA models, Holt-Winters exponential smoothing, and related time-series approaches share a core structural weakness: they are backward-looking. They identify patterns in historical data and extrapolate them forward. For a drug launch, there is no historical data. The model is being asked to extrapolate from zero.
Even for drugs with several years of post-launch history, ARIMA models struggle with discontinuous events. The entry of a new competitor, a label change, a formulary restriction by a major PBM, a safety signal, or the launch of a patient assistance program can each create an immediate, non-linear shift in the revenue trajectory that no trend extrapolation model can anticipate. In the pharmaceutical market, these discontinuous events are not exceptional; they are routine features of the competitive environment.
ML-Driven Forecasting: What the Models Can Actually Do
Machine learning models, particularly ensemble methods (gradient boosting, Random Forest) and recurrent neural networks (LSTM), outperform ARIMA on pharmaceutical sales forecasting tasks because they can incorporate multiple heterogeneous data streams and capture non-linear relationships between inputs and outputs. One comparative study published in a peer-reviewed operations research journal found that LSTM models reduced mean absolute percentage error (MAPE) for pharmaceutical sales forecasting by 23 to 31% relative to ARIMA baselines across a dataset of 200 branded drugs.
The practical value of ML is not in the algorithm itself but in the data it can process. A well-constructed ML forecasting system for a new drug launch integrates prescription data from specialty physician cohorts during the pre-launch period, insurance claims from early access programs, patient advocacy and social media sentiment analysis, physician survey data on prescribing intent, clinical trial patient baseline characteristics (as a proxy for real-world patient population), and patent data including claim language, family size, and filing density.
The combination of these signals can produce a first-year launch forecast with materially better accuracy than any single-source model. A published study examining 78 new molecular entity launches found that an ML model combining NLP-processed patent claim language with Phase III clinical trial characteristics achieved 89% accuracy in predicting first-year sales within a defined confidence interval, a substantial improvement over industry baseline accuracy.
Patent Data as a Quantitative Input: NLP Signals and Predictive Indicators
The most analytically novel contribution of AI to pharmaceutical forecasting is its ability to extract quantitative signals from patent documents, converting what was previously a qualitative legal assessment into numerical inputs for predictive models.
Three specific patent signals have shown robust predictive correlation with commercial outcomes:
Patent family size and geographic breadth correlate with future sales at r = 0.71 in published analyses. The mechanism is straightforward: companies do not maintain large international patent families for assets they regard as marginal. Filing and maintaining patents across 30 or more jurisdictions costs $500,000 to $1.5 million annually. Companies do this for assets that management believes justify the investment. Family size is therefore a revealed-preference indicator of management’s commercial conviction.
Claims language complexity, measured by NLP analysis of claim count, average claim word count, and the linguistic specificity of independent claims, explains 67% of the variance in eventual sales volume (R-squared = 0.67) in one published study. The intuition is that broader, more carefully constructed claims indicate a well-resourced IP prosecution team working on an asset the company intends to fully commercialize. Narrow, quickly drafted claims suggest the opposite.
Post-approval patent filing rate is the most direct thicket indicator. Of the top 50 global pharmaceutical drugs by revenue in 2023, 72% of their total granted U.S. patents were filed after commercial launch. A high post-launch filing rate for a drug already on the market is a strong signal of active lifecycle management and directly correlates with a longer expected effective exclusivity period.
Predictive Due Diligence: How AI Is Reshaping M&A Analytics
Traditional pharmaceutical M&A due diligence asked one primary question: what does the target have, and what is it worth today? AI-powered due diligence asks a different question: what will the target’s assets become, and what is that future state worth today?
ML models trained on historical trial outcomes, drug characteristics, target biology, trial design features, and sponsor track records can estimate the probability of transitioning from Phase II to approval and from Phase III to approval with AUC metrics of 0.78 and 0.81 respectively in published research. These probability estimates, combined with patent-based revenue forecasts, allow acquirers to compute probability-weighted expected revenue for every asset in a target’s pipeline, not just the lead program.
This transforms the due diligence process. An acquirer can now surface hidden risks, such as a Phase III trial with a design feature that ML models flag as correlated with failure, or a patent portfolio with unusual vulnerability to IPR challenge based on its prosecution history. An acquirer can also surface hidden value, such as a mid-stage asset that the target has underinvested in but whose probability-weighted expected NPV exceeds the lead program under certain competitive assumptions.
The practical effect on deal pricing is that sophisticated acquirers using predictive due diligence models can walk into negotiations with a more granular view of asset quality than the seller’s own internal projections. That information asymmetry is a structural advantage in deal negotiations.
Key Takeaways: Section 3
ARIMA-based time-series models produce systematically unreliable new launch forecasts; ML models reduce MAPE by 23 to 31% by incorporating heterogeneous data inputs. Patent family size (r=0.71), claims language complexity (R-squared=0.67), and post-approval filing rate are independently measurable quantitative inputs that improve forecast accuracy. Predictive due diligence using ML approval probability models changes the question from asset inventory to probability-weighted pipeline NPV, creating an information advantage for analytical buyers. The 89% first-year sales prediction accuracy achieved in the best published ML models still leaves meaningful uncertainty; ensemble approaches that combine ML output with human expert judgment consistently outperform either in isolation.
Investment Strategy Note
For funds active in pharma M&A or licensing: build (or license access to) a patent NLP scoring system that flags post-approval filing density as an input to LOE date distributions in your DCF. Assets with high post-approval filing rates have wider LOE date distributions (more scenarios, more upside) than assets with minimal secondary portfolios. That optionality should be reflected in the valuation through a wider range of scenario weights, not a single-point LOE assumption.
Section 4: Lifecycle Management: The Complete Evergreening and Patent Thicket Technology Roadmap
The Systematic Extension of Pharmaceutical Monopoly
Pharmaceutical lifecycle management is not a fringe practice. It is a capital allocation strategy deployed systematically by every major branded drug manufacturer with a product generating over $1 billion in annual revenue. The financial logic is simple: the cost of secondary patent prosecution and new clinical studies is measured in tens to hundreds of millions of dollars. The revenue protected by a successful lifecycle management program is measured in billions. The return on investment is exceptional for any program that extends effective exclusivity by even one to two years.
The controversy surrounding these practices is also real, and it is now a material financial risk. Understanding lifecycle management requires understanding both the mechanics and the regulatory, legal, and political constraints that increasingly circumscribe it.
Why Lifecycle Management Is Financially Compulsory
A pharmaceutical company with a $10 billion revenue drug facing LOE in 3 years has a structural problem. Pipeline development takes 10 to 15 years. Even an aggressive R&D program will not deliver a commercial replacement for a loss of that magnitude within the replacement window. Every year of extended exclusivity bought through secondary patents or formulation strategies has an NPV of $1 billion or more for a drug of that scale. The cost of not pursuing lifecycle management, therefore, is not zero. It is the full NPV of that unprotected revenue.
This financial logic explains why the practice is universal rather than aberrational, and why companies make it a core function of their IP departments rather than an occasional tactical exercise.
The Evergreening Technology Roadmap for Small-Molecule Drugs
For small-molecule drugs, the lifecycle management playbook has evolved through four generations of tactics, each prompted by the legal constraints and court decisions that circumscribed the previous generation.
The first-generation tactic was new salt form and polymorph patents. Drug substances can exist in multiple crystalline forms (polymorphs) and salt forms, each with different solubility, stability, and bioavailability properties. Filing patents on specific, commercially advantageous polymorphs or salt forms can extend protection even after the base COM patent expires. The vulnerability of this tactic is the obviousness challenge: a skilled pharmaceutical chemist would routinely explore salt and polymorph variants, making it difficult to establish non-obviousness in litigation.
The second-generation tactic was modified-release formulation patents. Extended-release, delayed-release, and immediate-release/extended-release combination formulations allow once-daily dosing for drugs that required multiple daily doses in their immediate-release form. These formulations often provide genuine clinical benefit (improved compliance, smoother pharmacokinetic profiles) and therefore clear a higher quality bar for patentability. The strategic deployment is to launch the modified-release version, promote heavily, and then withdraw the original immediate-release formulation from the market just before generics can enter, forcing patients and physicians to the protected version. This tactic (often called “product hopping”) has faced antitrust scrutiny in the U.S. and has been challenged in several high-profile litigations.
The third-generation tactic was device and combination product patents. By packaging a drug in a proprietary delivery device (an autoinjector pen, a specialized inhaler, a transdermal patch system), brand manufacturers create patent protection for the device-drug combination that is separate from and typically later-expiring than the drug substance patents. Patients and physicians often prefer the proprietary device for convenience and dose accuracy, creating switching friction even when generic drug-only versions are available. Novo Nordisk’s GLP-1 portfolio and AstraZeneca’s respiratory inhaler franchise are both examples of device-patent lifecycle management executed at scale.
The fourth-generation tactic, now increasingly common, is indication expansion through method-of-use patents combined with pediatric exclusivity. A company systematically runs clinical trials for new indications, files method-of-use patents for each new approval, and requests pediatric exclusivity extensions for each approved indication. Each successful trial adds both a new revenue stream and a new layer of patent protection. For a drug with a broad biological mechanism, such as an anti-inflammatory biologic or a PCSK9 inhibitor, the addressable indication space can sustain multiple rounds of this tactic across different disease categories.
The Biologics Evergreening Roadmap: BPCIA, the Patent Dance, and Thicket Construction
The lifecycle management playbook for biologics is structurally different from small molecules for several reasons: the 12-year BPCIA exclusivity already provides a long baseline protection period; the complexity of biologic manufacturing creates inherent barriers to biosimilar entry that do not exist for chemical synthesis; and the “patent dance” mechanism built into the BPCIA creates a structured, multi-stage litigation framework that brand manufacturers have used to substantially extend the effective protection period.
The BPCIA patent dance is a mandatory information exchange and negotiation process between the biologic originator and a biosimilar applicant. It proceeds in defined stages: the biosimilar applicant provides its Biologics License Application (BLA) to the originator within 20 days of FDA filing. The originator then has 60 days to identify which of its patents it believes would be infringed by the biosimilar’s manufacture, use, or sale, forming the “Section 3(B)(1) list.” The parties then negotiate which patents to litigate in the first phase of litigation and which to hold in reserve.
Brand manufacturers have consistently used the Section 3(B)(1) list to identify dozens or hundreds of patents, forcing biosimilar applicants to engage with an enormous portfolio before launching. The litigation generated by the patent dance frequently pushes biosimilar entry dates years past the initial application timeline, compounding the protection effect of the 12-year BPCIA exclusivity.
The biologics thicket construction roadmap typically proceeds in five phases:
Phase one is the foundational IP phase, typically covering the period from initial discovery through Phase I/II, in which the company files COM patents on the protein sequence or antibody structure, method-of-use patents for the initial indication, and process patents on the initial cell line and expression system.
Phase two is the post-approval formulation phase, in which the company files patents on the specific formulation that received approval (concentration, excipients, pH, buffering system), any high-concentration versions that reduce injection volume, and the drug-device combination (autoinjector or prefilled syringe).
Phase three is the indication expansion phase, in which the company funds clinical trials for additional autoimmune, oncologic, or other indications, filing new method-of-use patents for each approval and requesting pediatric exclusivity for each.
Phase four is the manufacturing process evolution phase, in which the company systematically improves its production process (yield improvements, purification advances, analytical method refinements) and patents each material change, creating a library of process patents that a biosimilar developer would need to either license or design around.
Phase five is the device portfolio expansion phase, in which the company develops second- and third-generation delivery devices, including connectivity features, wearable injectors, and needle-free administration systems, each independently patented and promoted as a clinical advancement.
IP Valuation Spotlight: AbbVie and Humira
Humira (adalimumab) is the single most studied patent thicket in pharmaceutical history, and a quantitative examination of its IP architecture is instructive for anyone building a thicket model.
AbbVie’s Humira generated $21.2 billion in global revenue in 2022, its peak year, with U.S. revenue contributing approximately $16.5 billion. The primary COM patent covering adalimumab as a molecule was set to expire in the U.S. in December 2016. By standard analysis, 2017 should have been the U.S. LOE year.
AbbVie built 257 patent applications around Humira, resulting in 132 granted U.S. patents, with 90% of those patents filed after the drug received FDA approval in 2002. These patents covered 14 distinct formulations (including a citrate-free, high-concentration version specifically designed to reduce injection site pain and create a switching barrier), 20 device patents covering the Humira autoinjector and pen designs, multiple manufacturing process patents covering the cell culture conditions and purification methods, and method-of-use patents across all 11 FDA-approved indications.
The IP valuation consequence is measurable. The U.S. biosimilar LOE date, effectively January 2023, was 7 years after the primary patent expiration. At Humira’s $16.5 billion 2022 U.S. revenue run rate, each additional year of exclusivity had a net present value (using a 10% discount rate and standard biosimilar erosion curves) of approximately $5 to $6 billion. The full 7-year thicket extension generated an estimated $40 to $45 billion in additional U.S. revenue NPV above what a single-patent analysis would have predicted.
AbbVie settled with all seven major biosimilar manufacturers in a coordinated “patent detente,” licensing the thicket for U.S. entry no earlier than January 2023 in exchange for biosimilar manufacturers’ agreement not to challenge the patents. This strategy traded some long-term exclusivity in exchange for near-certainty of no early IPR invalidation, a rational risk management choice given the vulnerability of individual patents within the portfolio when scrutinized independently.
By 2023, biosimilar adalimumab products launched from seven manufacturers. By Q3 2023, the combined biosimilar market share reached approximately 15 to 20% of total adalimumab prescriptions, significantly below consensus analyst projections of 30 to 35% market share by year one, reflecting the structural factors in U.S. biologic markets discussed in Section 2.
IP Valuation Spotlight: Celgene/Revlimid and the 40-Year Monopoly
Revlimid (lenalidomide), Celgene’s multiple myeloma drug, generated $12.8 billion in global revenue in 2021. Its primary COM patent expired in 2019. Through a combination of a 96-patent thicket covering formulations, methods of use, and manufacturing processes, and a settlement structure with generic manufacturers that restricts entry volumes even after LOE, the effective U.S. LOE date for unrestricted generic entry is effectively 2026 for most entrants.
The IP asset value of Revlimid to Bristol Myers Squibb (which acquired Celgene in November 2019 for $74 billion) is partially captured by this detail: the deal was priced at a significant premium to Celgene’s then-current revenues precisely because the acquirer’s models reflected the thicket-extended exclusivity, not the primary patent expiration.
IP Valuation Spotlight: Amgen and Enbrel
Enbrel (etanercept), co-commercialized by Amgen and Pfizer in the U.S., generates approximately $4 billion in annual U.S. revenue. Its complex patent portfolio, which Amgen defends with particular tenacity, has successfully blocked U.S. biosimilar entry through approximately 2029 despite the primary biologic license being well past the BPCIA exclusivity period.
Amgen’s defense strategy for Enbrel is particularly notable because it relies heavily on manufacturing process patents and trade secret protections around its proprietary CHO cell line, making it technically challenging for biosimilar manufacturers to demonstrate that their manufacturing process does not infringe. Amgen has also filed multiple continuation patents in the U.S. that have been rejected by the USPTO but contested through multiple rounds of review, a litigation strategy that delays resolution even if individual patents are ultimately invalidated.
The contrast between Enbrel’s protected U.S. market and its fully biosimilar-competed European market (where Enbrel biosimilars launched in 2016) is a direct illustration of the jurisdictional differences covered in Section 5.
Pay-for-Delay and Reverse Payment Settlements: The Litigation Weapon
One of the most financially significant lifecycle management tools is the reverse payment settlement, commonly called “pay-for-delay.” When a generic manufacturer files a Paragraph IV certification challenging a brand’s patents, the brand manufacturer typically sues within 45 days, triggering the 30-month stay on FDA approval of the generic. Rather than litigating to judgment, brand manufacturers have historically settled these cases by paying the generic manufacturer a substantial sum (in cash, in supply agreements, or in authorized generic licenses) in exchange for the generic manufacturer agreeing to delay market entry until a negotiated date.
The U.S. Supreme Court’s 2013 FTC v. Actavis decision established that reverse payment settlements can constitute antitrust violations and must be evaluated under a “rule of reason” standard. This decision has reduced (though not eliminated) the frequency of cash-based pay-for-delay settlements. However, non-monetary reverse payment settlements, in which the brand grants the generic an authorized generic license or other commercial concession, continue and are harder to challenge under the Actavis framework.
For the forecaster, each Paragraph IV settlement creates a new data point: the settled entry date becomes the LOE date for that challenger. Tracking active Paragraph IV litigation and settlement activity in the FDA Orange Book and public court records is a real-time source of LOE date refinement.
Modeling the Thicket Effect in a DCF
Incorporating a patent thicket into a DCF requires abandoning the single-LOE-date model in favor of a probabilistic LOE distribution. For a drug with a 40-patent thicket, the analyst should construct at minimum three scenarios:
A maximum-thicket scenario where all patents are successfully defended or settled, producing an LOE date equal to the latest-expiring thicket patent, weighted at whatever probability the analyst assigns based on litigation history and patent quality.
A partial-invalidation scenario where two or three key formulation or method-of-use patents are invalidated in IPR (Inter Partes Review) proceedings, causing one or two earlier generic entrants while the remaining thicket still delays full competition, producing a partial erosion curve rather than a full cliff.
A catastrophic-challenge scenario where a core patent is invalidated by the PTAB and the invalidation survives Federal Circuit appeal, effectively exposing the full revenue base to competition years earlier than expected.
The weighted average LOE date across these three scenarios (each multiplied by its probability) is the LOE date to use in the base-case DCF. The scenario range defines the sensitivity table that communicates the valuation’s dependence on thicket durability.
The Regulatory and Antitrust Backlash: Quantifying the Headline Risk
Patent thickets and evergreening practices have become recurring targets for legislative and regulatory action in the United States, and the trajectory of that pressure has been consistently upward since 2019. The American Innovation and Choice Online Act, the Affordable Prescriptions for Patients Act, and multiple Senate and House drug pricing bills have all contained provisions targeting Orange Book patent listing practices, inter partes review reform, and pay-for-delay restrictions.
The FTC under Chair Lina Khan dramatically escalated enforcement activity against Orange Book patent listings, filing formal challenges against hundreds of listings it characterized as improper or anticompetitive between 2023 and 2024. While many of these challenges do not result in immediate patent invalidity, they impose litigation costs, create adverse press coverage, and signal regulatory intent in a way that can accelerate settlement negotiations with generic challengers.
For a drug whose valuation is substantially dependent on a dense thicket of secondary patents, this regulatory risk is a non-clinical, non-competitive financial risk that requires explicit quantification in any long-term model. A 10% probability of legislative reform that cuts the effective thicket duration by 2 years reduces the NPV of the drug’s IP portfolio by a material amount that should be reflected in the model’s scenario analysis.
Key Takeaways: Section 4
For biologics, the BPCIA patent dance has been used to substantially extend the period of biosimilar-free competition beyond the 12-year exclusivity baseline; modeling the patent dance’s litigation timeline is essential for accurate biologic LOE dates. AbbVie’s Humira thicket generated an estimated $40 to $45 billion in NPV beyond what a primary patent analysis would have predicted, demonstrating the financial scale of successful lifecycle management. Revlimid’s restricted-volume settlement structure and Enbrel’s process patent strategy show that thickets can take multiple forms, not all of which are visible from the Orange Book alone. Regulatory risk, specifically FTC patent listing challenges and legislative pay-for-delay reform, is now a material factor in thicket-dependent valuations.
Investment Strategy Note
Pharma equities trading at multiples that embed maximum-thicket LOE assumptions are disproportionately exposed to FTC challenge risk and legislative reform. Stress-testing these multiples against a 2-year acceleration in LOE under a partial-invalidation scenario frequently reveals 15 to 25% downside from the base case equity valuation. That asymmetric downside risk relative to limited upside from further thicket extension (most thickets are already near-fully exploited) argues for underweighting single-product biologic companies where thicket durability is the primary valuation driver.
Section 5: Global Patent Jurisdiction Analysis
The Same Drug, Different LOE Dates on Five Continents
A drug’s patent status is jurisdictionally specific. The LOE date in the United States, the European Union, India, and China for the same branded drug can differ by 5 to 10 years, driven by fundamental differences in patent law, patentability standards, regulatory exclusivity frameworks, and judicial practice. A global revenue forecast that applies the U.S. LOE date uniformly across all markets will systematically overstate the drug’s value in markets where LOE is earlier, and systematically understate it in markets where local thickets are equally dense.
United States: Hatch-Waxman, BPCIA, and the Paragraph IV Filing Process
The U.S. patent landscape for pharmaceuticals is the most litigated and the most extensively studied in the world. Its complexity is not accidental; it reflects decades of policy decisions that have attempted to balance innovator incentives with generic competition, often landing in places that serve the interests of well-resourced brand manufacturers.
The 30-Month Stay and Its Exploitation
The Hatch-Waxman Act’s patent linkage system creates a structured pathway for generic challenges, but brand manufacturers have used it to generate years of additional protection through a well-worn playbook. When a generic manufacturer files a Paragraph IV certification asserting that a listed Orange Book patent is invalid or will not be infringed, the brand manufacturer has 45 days to file a patent infringement suit. Filing that suit automatically triggers a 30-month stay on FDA approval of the generic, effectively providing a minimum 30-month delay regardless of the underlying merit of the patent.
The exploitation mechanism is Orange Book patent listing strategy. Brand manufacturers have listed patents of varying quality in the Orange Book, knowing that each listed patent that a Paragraph IV filer must address creates another potential 30-month stay trigger. The FDA has historically had limited authority to refuse listings, accepting brand manufacturers’ representations that listed patents cover the approved product. The FTC’s escalated Orange Book challenge program represents a direct challenge to this dynamic.
For the forecaster, the Paragraph IV filing date is a data point of high value. When a generic manufacturer files a Paragraph IV certification (which becomes publicly available in FDA records), it signals that a well-resourced challenger has reviewed the patent portfolio, concluded the patents are vulnerable, and made a capital commitment to litigation. Historical Paragraph IV success rates run approximately 75% for filers that see their cases to judgment (rather than settling), a striking figure that suggests the average challenged patent in the Orange Book is weaker than its listing implies.
The First Filer Advantage: 180-Day Exclusivity
The Hatch-Waxman Act grants the first generic filer a 180-day period of market exclusivity before additional generic entrants can launch. This incentive drives substantial first-to-file competition among generic manufacturers for high-revenue branded drugs. For a drug with $2 billion in annual U.S. sales, the 180-day first-filer exclusivity can be worth $300 to $500 million in gross profit to the first entrant, making the Paragraph IV race commercially significant in its own right.
First-filer exclusivity also affects the brand manufacturer’s competitive strategy. Some brand manufacturers have negotiated settlements that grant the first filer an Authorized Generic license (the brand’s product sold under a generic label), splitting the 180-day market with the filer in exchange for a delayed launch date. This increases the brand’s revenue during the exclusivity period at the cost of the first filer’s exclusivity premium.
European Union: Added Matter, SPCs, and the Strict Inventive Step Standard
Europe offers a fundamentally different patent environment for pharmaceutical lifecycle management, one that structurally limits the effectiveness of the dense thicket tactics that dominate U.S. strategy.
The European Patent Office applies a stricter “inventive step” standard than the USPTO for secondary patents. A new polymorphic form, a new salt, or a modified dosing regimen must demonstrate a technical effect that a skilled person would not have anticipated and that provides a genuine functional advantage. Minor modifications that pass USPTO examination regularly fail EPO examination, reducing the density of thickets that can be constructed under European patent law.
Article 123(2) of the European Patent Convention prohibits amending a patent application to include subject matter not directly and unambiguously derivable from the application as filed. This provision bars the U.S. practice of filing continuation applications that progressively broaden claims based on what competing products do, effectively capturing the competitor’s innovation under the original application’s umbrella. This continuation strategy is a major tool for broadening U.S. patent scope over time and is simply not available in Europe.
Supplementary Protection Certificates (SPCs) provide a single, predictable extension of up to 5 years for the specific product (active ingredient and approved combination) to compensate for regulatory review delays, with a maximum 6-month pediatric extension on top. SPCs are a streamlined, fit-for-purpose term extension mechanism that reduces the incentive to build broad secondary patent portfolios, because a well-designed SPC provides near-equivalent benefit with none of the litigation risk.
The practical consequence: Humira biosimilars launched in Europe in October 2018, a full 4 years and 3 months before any U.S. biosimilar competition. Enbrel biosimilars launched in Europe in 2016, more than a decade before they could reach the U.S. market. European LOE dates are consistently earlier than U.S. dates for drugs with dense thickets, and a global model must reflect this.
India: Section 3(d) and the Anti-Evergreening Regime
India’s patent law takes the most explicit position on evergreening of any major jurisdiction. Section 3(d) of the Indian Patents Act, amended in 2005 when India joined the TRIPs Agreement, specifies that a new form of a known substance (including salts, polymorphs, metabolites, isomers, and combinations) is not patentable unless it demonstrates significantly enhanced efficacy relative to the known substance. The Indian Supreme Court’s 2013 ruling in Novartis v. Union of India (the Gleevec case) interpreted “efficacy” to mean specifically therapeutic efficacy, not improved solubility or bioavailability unless those properties directly and demonstrably translate to better therapeutic outcomes in patients.
This interpretation creates a near-complete bar on the categories of secondary patents that form the core of most U.S. thicket strategies. Polymorphic forms, new salts, modified-release formulations, and dosage variants are presumptively non-patentable in India unless the applicant can demonstrate superior clinical outcomes, which is a high and expensive bar to clear.
The consequence for the pharmaceutical revenue forecast is that India’s LOE date for virtually every major branded drug is determined by the primary COM patent alone, with no meaningful thicket extension. For drugs where the COM patent has expired or was never filed in India (India did not recognize product patents for pharmaceutical substances before 2005), the drug may be commercially manufactured by domestic generic manufacturers with no patent constraint whatsoever.
India’s generic pharmaceutical industry generated approximately $24 billion in exports in fiscal year 2023, making it the world’s largest generic drug exporter by volume. Section 3(d) is structurally central to that industry’s competitive economics.
China: Patent Linkage, PTE, and the Accelerating IP Enforcement Landscape
China’s pharmaceutical patent environment has undergone a structural transformation over the past 5 years, moving from a historically weak enforcement regime toward a framework that increasingly resembles U.S. practice, with important local variations.
China implemented a pharmaceutical patent linkage system in June 2021, roughly analogous to Hatch-Waxman’s Paragraph IV framework. Under this system, a domestic generic manufacturer seeking approval for a drug listed in China’s patent register must notify the innovator company and provide a patent status declaration. The innovator can then initiate a patent dispute resolution proceeding that triggers a 9-month stay on NMPA (National Medical Products Administration) approval of the generic, a shorter stay than the U.S. 30-month period.
China simultaneously implemented a patent term extension mechanism in 2021, allowing brand manufacturers to recover up to 5 years of patent term lost to NMPA regulatory review, with the extended term capped at 14 years from NMPA approval. This PTE provision directly mirrors the U.S. Hatch-Waxman PTE and provides a meaningful incentive for innovators to file and maintain patents in China.
China’s National Reimbursement Drug List (NRDL) annual negotiations have also created a distinctive pricing dynamic. Drugs entering the NRDL accept substantial price reductions (often 60 to 80% from list price) in exchange for broad reimbursement coverage across China’s 1.4 billion person population. This creates a different revenue shape for Chinese forecasts than Western models: lower per-unit revenue but dramatically higher volume, producing total revenue that can rival or exceed European revenue for drugs with broad patient populations.
Jurisdictional Arbitrage: How Generic Manufacturers Use Favorable Rulings as Global Leverage
The differences between jurisdictions create strategic opportunities for generic and biosimilar manufacturers that are as sophisticated as the brand manufacturers’ lifecycle management playbooks.
A generic manufacturer that successfully challenges a key secondary patent at the EPO or before an Indian court has a legal ruling in hand that it can use in settlement negotiations in every other jurisdiction. While EPO or Indian court rulings are not legally binding on U.S. courts, they constitute persuasive evidence that the patent is vulnerable, and brand manufacturers facing aggressive generic challengers armed with foreign invalidation decisions frequently find that settlement terms become more favorable to the generic.
The reverse is also true. A brand manufacturer with an exceptionally strong U.S. thicket can use the litigation cost and uncertainty of the U.S. market as a deterrent in other, less-protected markets. A generic manufacturer that might otherwise launch in a European market where the patent position is weaker may be deterred by the knowledge that launching anywhere in the world could trigger a retaliatory patent infringement suit in the U.S., where the costs of litigation far exceed any European market revenue.
Building a Multi-Jurisdictional LOE Model
A credible global revenue forecast requires a jurisdiction-specific LOE framework that assigns separate LOE dates to at least the U.S., EU-5 (Germany, France, UK, Italy, Spain), Japan, China, and rest-of-world clusters. The LOE date in each jurisdiction reflects the local patent law, the specific patent family coverage in that country, the applicable regulatory exclusivities under local law, and any active litigation or settlement history in that jurisdiction.
Each jurisdiction’s post-LOE erosion curve also requires calibration to local market structure. Germany, which has a reference price reimbursement system and aggressive generic substitution policies, sees post-LOE branded market share collapse faster than any other major European market. France, which uses a committee-based tendering system, sees a more gradual transition. Japan, which retains a strong brand-loyal prescribing culture, historically sees slower brand erosion than European comparables.
Key Takeaways: Section 5
Europe’s LOE dates are consistently 3 to 10 years earlier than U.S. dates for drugs with dense secondary patent thickets, driven by Article 123(2) EPC restrictions on continuation applications and stricter EPO inventive step standards. India’s Section 3(d) creates a near-complete bar on secondary pharmaceutical patents, making primary COM patent expiration the operative LOE date in almost every case. China’s 2021 patent linkage and PTE reforms have substantially improved the IP enforcement environment for brand manufacturers but have not yet produced the dense thickets common in the U.S. market. Multi-jurisdictional LOE models should assign separate patent and exclusivity analysis to each major revenue market, not use the U.S. LOE date as a global proxy.
Investment Strategy Note
For drugs generating more than 40% of revenue in non-U.S. markets, the U.S.-centric patent analysis that dominates sell-side research systematically overstates global revenue duration. Build the non-U.S. LOE dates from first principles for each major geography rather than applying a U.S. offset rule-of-thumb. The revenue difference between a 2026 European LOE and a 2029 European LOE for a $3 billion European franchise is a $3 to $5 billion NPV delta that frequently swings a buy/sell recommendation.
Section 6: The Intelligence Stack
Databases, Platforms, and Analytical Workflows for Patent-Based Forecasting
Accurate patent-based forecasting is impossible without access to accurate and comprehensive data. The analytical workflow moves through several distinct layers, from primary public sources that provide authoritative legal data to commercial platforms that aggregate, contextualize, and analyze that data at scale.
The FDA Orange Book: Structure, Limitations, and Forensic Usage
The Orange Book (“Approved Drug Products with Therapeutic Equivalence Evaluations”) is the definitive U.S. source for the intersection between approved drug products and the patents that their manufacturers have listed as covering those products, along with all applicable regulatory exclusivities. Every U.S. pharmaceutical patent-based forecast starts here.
The Orange Book search returns patent numbers, expiration dates (including any PTE), patent use codes that specify the method of use covered (if applicable), and regulatory exclusivity codes with their expiration dates. Analysts should run searches by active ingredient, trade name, and application number to ensure complete coverage.
The Orange Book has four documented limitations that every analyst should internalize. First, it lists only patents that brand manufacturers have chosen to list, and listing requires only a certification that the patent claims the drug substance, drug product, or method of use. The FDA does not independently verify these claims. Second, it does not include process patents, which can be equally important for biologic thickets. Third, it does not include metabolite patents or patents covering unapproved (off-label) uses. Fourth, the expiration dates listed reflect any PTEs claimed by the manufacturer, but do not reflect patent term adjustments (PTAs) granted by the USPTO for examination delays, which can extend individual patent terms by months to years.
The FTC’s challenge program (initiated under Chair Khan) has resulted in the FDA removing dozens of listed patents from the Orange Book that were challenged as improperly listed. Analysts should cross-reference Orange Book listings against the FTC’s public challenge database to identify listed patents that may be removed or challenged before the listed expiration date.
USPTO Patent Public Search: Prosecution History and Claims Analysis
The USPTO’s Patent Public Search (PPS) tool provides access to the full text, prosecution history, and legal status of every U.S. patent. For patent-based valuation, PPS provides three capabilities that the Orange Book cannot.
First, full claims analysis. The claims section of a patent defines the legal boundaries of protection. An analyst who reads only the abstract and title of a patent will miss critical claim scope information. Independent claims (unnumbered or numbered 1, and any claims that do not reference other claims) define the core scope; dependent claims add specificity. The breadth of the independent claims and the presence of means-plus-function claim limitations (which courts have historically interpreted narrowly) are key indicators of patent strength.
Second, prosecution history (file wrapper) review. The prosecution history records every communication between the patent applicant and the USPTO examiner, including amendments to the claims made during prosecution to overcome rejections. Under the doctrine of prosecution history estoppel, claim amendments made to overcome prior art rejections can limit the patent’s scope in litigation, even if the literal language of the final claims appears broader. A patent that required significant claim narrowing to overcome a prior art rejection is more vulnerable than one that issued with its original broad claims.
Third, continuation and continuation-in-part tracking. The PPS displays the priority chain of a patent, showing every earlier application to which it claims priority and every later application that claimed priority to it. A large family of continuation applications filed by a single assignee is the clearest visible indicator of a thicket construction strategy.
WIPO PATENTSCOPE and EPO Espacenet: Building the Global Patent Family Map
WIPO’s PATENTSCOPE database and the EPO’s Espacenet are the primary gateways to international patent data. Both databases allow patent family reconstruction, which is the analytical process of identifying all national and regional patent filings that claim priority to a single original application.
Patent family analysis is the basis for the quantitative predictive signal discussed in Section 3: family size and geographic breadth correlate with future commercial success at r = 0.71. Building a complete patent family map for a drug or pipeline asset requires searching both databases, as each has different coverage strengths: PATENTSCOPE covers PCT (Patent Cooperation Treaty) applications and national filings in WIPO member states comprehensively; Espacenet has particularly strong coverage of European patent filings.
WIPO’s Pat-INFORMED initiative deserves specific mention for analysts working in global health or procurement contexts. It provides a country-by-country summary of patent status for essential medicines and is maintained in cooperation with major pharmaceutical companies, making it the fastest path to a reliable answer on whether a specific drug has active patent protection in a specific lower-income country.
Commercial Intelligence Platforms: The Case for Specialized Aggregators
Public databases provide the authoritative raw data, but converting that data into a usable analytical input for forecasting requires substantial processing: cross-referencing Orange Book listings with USPTO patent text, building global patent families across multiple databases, tracking Paragraph IV filing and litigation histories, monitoring expiration dates across hundreds of drugs simultaneously, and receiving real-time alerts when new secondary patents are listed or challenged.
Platforms purpose-built for pharmaceutical patent intelligence, with DrugPatentWatch as a prominent example, address this processing gap. DrugPatentWatch aggregates patent, regulatory, clinical trial, and competitive supplier data into a unified interface specifically designed for the business and analytical questions that forecasters and IP teams face: when is the LOE date for a specific drug in the U.S., which generics have filed Paragraph IVs, what is the status of pending litigation, which drugs are approaching patent expiry in the next 3 years within a given therapeutic area.
The platform’s patent alerts, expiration dashboards, and biosimilar/generic pipeline tracking tools replace hours of manual database work with structured data views that can be exported into financial models. For portfolio managers tracking LOE dates across dozens of assets, or for generic manufacturers identifying entry opportunities, this represents a material efficiency gain.
The Analyst’s Workflow: A Step-by-Step Protocol
A complete patent-based LOE analysis for a single drug in the U.S. market follows this sequence: start with the Orange Book to identify all listed patents and regulatory exclusivities; retrieve each patent’s full text and claims from USPTO PPS; build the patent family map using PATENTSCOPE and Espacenet; review the prosecution history of the highest-risk (most-likely-challenged) patents for claim narrowing or enablement issues; check public court records for any active Paragraph IV litigation; and cross-reference FTC challenge listings for any Orange Book listing disputes.
This workflow produces a complete patent status map from which a probabilistic LOE date distribution can be constructed. In practice, a commercial platform accelerates the first three steps substantially, leaving the analyst’s time for the more judgment-intensive prosecution history review and litigation tracking steps.
Key Takeaways: Section 6
The Orange Book is the necessary starting point but not a complete source: it omits process patents, PTA adjustments, and patents challenged by the FTC. USPTO prosecution history review is the highest-value analysis that public sources enable; claim narrowing during prosecution is the most frequently exploited vulnerability in patent litigation. Patent family size (from PATENTSCOPE and Espacenet) is both a valuation input and a predictive commercial signal. Specialized commercial platforms that aggregate and cross-reference these sources into analytical dashboards materially reduce the analyst’s time burden and reduce the risk of missing a critical patent or Paragraph IV filing.
Section 7: Forecasting Failure Modes, the IRA’s New Variables, and the Future of the Field
What the Models Get Wrong and How to Build a More Resilient Framework
Even a sophisticated analyst using every tool described above will produce forecasts that turn out to be wrong. The question is not whether forecasts will be wrong but which dimensions of error are systematic, how large they tend to be, and which are reducible through better methodology versus genuinely irreducible uncertainty.
Why Most First-Year Launch Forecasts Are Wrong
The Simon-Kucher analysis referenced in Section 3 found that only 12% of pharma launch forecasts were accurate within 25%. The distribution of errors is not random; it reflects specific, predictable analytical biases.
Analysts consistently overestimate penetration speed for drugs in competitive therapeutic areas where physician prescribing habits are already established and payers use formulary tools to favor established alternatives. New drugs entering saturated markets like Type 2 diabetes or rheumatoid arthritis face formulary positioning challenges that are known in advance but difficult to model with precision.
Analysts consistently underestimate the time required to establish specialist physician awareness and prescribing habits for truly novel mechanisms or first-in-class drugs. Drugs entering markets without established treatment protocols require physician education, treatment guideline updates, and patient identification system development, all of which take longer in practice than in forecast models.
Gross-to-net discount expansion is structurally difficult to forecast. PBMs negotiate rebates confidentially, and the magnitude of those negotiations for newly launched drugs depends on formulary competition that is resolved after launch, not before. Analysts typically apply a gross-to-net assumption derived from comparable drugs in the same class, but for genuinely novel drugs with no direct comparable, this assumption carries wide uncertainty.
The IRA’s Price Negotiation Mechanism: Rewriting the DCF
The Inflation Reduction Act’s Medicare drug price negotiation provision, which selected its first 10 drugs for negotiation in August 2023 with prices effective January 2025, introduces a structural new variable into pharmaceutical DCF models that requires explicit treatment in any post-2025 forecast.
The IRA’s negotiation framework applies to small-molecule drugs more than 9 years post-approval and biologics more than 13 years post-approval that have no generic or biosimilar competition and represent high Medicare expenditure. The negotiated prices for the first 10 drugs were set at reductions of 38 to 79% from list price for the Medicare Part D market, a range that exceeded most analyst expectations for the first negotiation round.
For DCF modeling, the IRA introduces three distinct effects. First, for drugs selected for negotiation, it reduces the effective net price during the post-negotiation period, compressing the revenue forecast in years 9 and beyond for small-molecule drugs. A 50% price reduction in year 10 for a drug generating $3 billion annually in Medicare revenue reduces that year’s net revenue by $1.5 billion, which at a 10% discount rate reduces the NPV by approximately $500 million.
Second, the IRA creates an incentive distortion for small-molecule versus biologic drug development. The 9-year pre-negotiation window for small molecules versus 13 years for biologics effectively reduces the expected commercial life for small molecules relative to biologics, a structural change in the economics of small-molecule drug development that has already prompted multiple major drug companies (Bristol Myers Squibb, AstraZeneca, among others) to shift R&D investment toward biologic modalities.
Third, the IRA potentially compresses the economic motivation for biosimilar development in certain markets. If the originator biologic’s Medicare price has already been negotiated down to 80% of list price, the price differential that a biosimilar manufacturer can offer to payers narrows, reducing the revenue opportunity for biosimilar entry. This effect is most pronounced in markets where the originator’s Medicare exposure is high and where biosimilar entry would primarily displace the negotiated originator rather than the full list-price product.
Data Quality, the Black-Box Problem, and the Interpretability Barrier
ML models for pharmaceutical forecasting face two related limitations that constrain their applicability in regulated, decision-accountable contexts. The first is data quality. Electronic health record data is fragmented across thousands of health systems, lacks standardization in diagnostic coding, and contains substantial missing data for the drug exposure, outcome, and adherence fields most relevant to forecasting. Models trained on biased or incomplete data will produce biased forecasts, often with no visible warning signal in the model’s performance metrics on its training dataset.
The second is interpretability. Deep learning models, particularly LSTM networks and transformer architectures, produce accurate predictions in backtesting but cannot articulate why they predict a given outcome in terms that a business decision-maker can evaluate, audit, or challenge. In the pharmaceutical industry, where regulatory submissions, board presentations, and M&A due diligence all require that analytical conclusions be explainable and defensible, a black-box model that says “this drug will achieve $2 billion in year 3 sales” without a traceable reasoning chain is difficult to use as a primary decision-making input.
Gradient boosting models (XGBoost, LightGBM) occupy a useful middle ground: they produce accuracy metrics comparable to deep learning models on many pharmaceutical forecasting tasks while generating feature importance rankings that allow analysts to understand which inputs are driving the predictions. This interpretability advantage makes them more practical for regulated and high-accountability forecasting contexts than opaque neural networks.
The Future Trajectory: Real-World Evidence, Generative AI, and Quantum Computing
The next generation of pharmaceutical forecasting models will incorporate data streams that are largely excluded from current practice. Real-world evidence (RWE), derived from insurance claims, patient registries, and anonymized EHR data, is becoming a regulatory-grade source of efficacy and safety data and a first-order input into market opportunity models. FDA’s increasing acceptance of RWE in label expansions and post-marketing commitments means that real-world data will influence not just forecast inputs but the actual patent and exclusivity landscape for drugs with active RWE programs.
Generative AI is being deployed in pharmaceutical intelligence primarily as an accelerant for the research and synthesis process. Large language models fine-tuned on patent, regulatory, and clinical literature can summarize patent family prosecution histories, compare claim scope across a competitive landscape, and flag potential validity risks in patent portfolios at a speed and scale not previously achievable. McKinsey’s analysis estimated that generative AI could unlock $60 to $110 billion in annual productivity value for the pharmaceutical industry across R&D, clinical, commercial, and regulatory functions.
Quantum computing remains on a longer horizon but holds specific relevance for patent landscape analysis. The computational problem of mapping all pairwise relationships in a 500-patent thicket (assessing overlapping claim scope, litigation risk correlations, and invalidity contagion across the portfolio) is an NP-hard optimization problem that classical computers cannot solve at scale. Quantum computers, when available at practical scale, could perform this analysis in hours rather than the weeks it currently requires, enabling real-time portfolio valuation updates as new patents are issued, challenged, or settled.
Human-AI Symbiosis: The Non-Replaceable Analyst Function
The accumulation of AI capability described above might suggest that the human patent analyst is a transition technology, to be replaced by autonomous ML systems within a defined number of years. This view underestimates the irreplaceable functions that human judgment performs in the forecasting process.
AI models identify correlations. Human analysts establish causation and strategic intent. An ML model can report that a drug’s patent family has grown by 40% in the past 18 months, which the model flags as a positive commercial signal based on historical correlation. A human analyst reads those new patents, recognizes that three of them cover a manufacturing process for a subcutaneous administration route, connects that to a public statement by the CEO about improving convenience in the maintenance patient population, and concludes that the company is setting up a formulation pivot to defend against a competitor’s subcutaneous version. That inferential leap from data to strategy is not a task that current AI systems perform reliably.
Human analysts also perform the political and regulatory risk assessment that is structurally outside the training data of any historical ML model. A legislative change, a new FTC commissioner with a specific enforcement agenda, or a judicial appointment to the Federal Circuit Court of Appeals (which hears all patent appeals) can shift the probability landscape for an entire class of lifecycle management strategies in ways that have no historical precedent in the training data. These judgments require human contextual reasoning that is qualitatively different from pattern recognition over historical datasets.
The optimal analytical structure, and the one that best-performing pharma intelligence teams are converging on, pairs ML models for data processing, pattern detection, and quantitative signal extraction with human analysts who provide strategic context, causal reasoning, political judgment, and ethical oversight. The AI produces the “what.” The analyst provides the “so what” and “what next.”
Key Takeaways: Section 7
First-year launch forecast inaccuracy is systematic and large: only 12% of forecasts are within 25% of actuals, with overestimation errors concentrated in competitive market entries and underestimation errors concentrated in first-in-class launches. The IRA’s Medicare price negotiation compresses revenue in years 9 and 13+ for small-molecule and biologic drugs respectively, and this compression should be modeled as a probability-weighted revenue reduction in DCFs for any drug with significant Medicare exposure. ML interpretability limitations constrain the use of deep learning models in high-accountability forecasting contexts; gradient boosting models with feature importance outputs are more practically deployable. Human analyst judgment on political risk, regulatory intent, and competitive strategy is structurally irreplaceable by current AI systems.
Investment Strategy Note
For funds evaluating companies with late-cycle assets (7 or more years post-approval with significant Medicare exposure), apply the IRA negotiation probability as an explicit haircut to post-year-9 or post-year-13 revenue in your DCF. The probability of selection is not 100% for any given drug but is non-trivial for high-spend drugs in competitive markets. A 30% probability of negotiation with a 50% price reduction for the Medicare portion of revenue (which may represent 30 to 40% of total U.S. revenues for broadly prescribed drugs) produces a non-trivial NPV reduction that is not yet consistently reflected in consensus estimates.
Frequently Asked Questions
How should a small biotech with limited resources approach the patent thicket of a large-pharma competitor?
Litigation is expensive, and fighting every patent in a large thicket to judgment is rarely economically rational for an emerging company. The more efficient approach starts with patent quality stratification: not all 100 patents in a thicket are equally defensible. Some were filed opportunistically and have prosecution histories showing significant claim narrowing; some cover formulations or processes the company is not using in its biosimilar manufacturing approach; some may be invalid over newly located prior art. An Inter Partes Review petition at the PTAB costs approximately $40,000 to $80,000 plus legal fees to file, compared to the $5 to $10 million required for full federal district court litigation. IPR proceedings have historically resulted in patent claim invalidation rates of approximately 70% for instituted petitions in pharmaceutical cases, making them a cost-effective first tool for thicket thinning. A favorable PTAB ruling also provides global negotiating leverage, even though it is binding only in U.S. proceedings.
What is the generic paradox, and why does it matter for brand revenue forecasting?
The generic paradox is the empirically documented phenomenon in which a branded drug’s list price increases after initial generic entry rather than declining. The mechanism reflects market segmentation: generic entry captures the most price-sensitive market segments (managed care formularies, PBMs with mandatory substitution policies) while leaving behind a brand-loyal segment composed of patients and physicians who continue using the brand regardless of generic availability. With a smaller but less price-sensitive customer base, the brand manufacturer may increase the list price to maximize revenue from the remaining brand franchise. This dynamic is most pronounced for oral solid dosage drugs in categories where brand loyalty is high (some cardiovascular, neurological, and psychiatric drugs). For forecasting purposes, the practical implication is that brand revenue may not decline as steeply in the first 12 months post-generic entry as standard erosion curves suggest, but the total revenue will be lower due to the volume loss, and the higher list price accelerates the shift of remaining volume to generics over subsequent years as payer formularies catch up.
How does biosimilar interchangeability designation affect U.S. market forecasting?
A biosimilar designated as “interchangeable” by the FDA can be substituted for the reference biologic at the pharmacy counter without physician intervention in all states that permit pharmacy-level substitution (currently most U.S. states). This is analogous to the automatic substitution for small-molecule generics that drives rapid post-LOE brand erosion for oral drugs. Historically, no biosimilar had achieved interchangeability status until Semglee (insulin glargine) in July 2021. Since then, several additional interchangeable biosimilar designations have been granted, and the FDA has streamlined the switching study requirements, reducing the clinical development burden. For drugs where biosimilar interchangeability is granted, the post-LOE erosion curve will more closely resemble the small-molecule pattern (steep and rapid) than the standard biologic pattern (gradual, physician-driven). Analysts building erosion models for biologic LOE events should flag interchangeability status as a binary variable with a significant impact on the shape of the post-LOE revenue curve.
How does Section 3(d) of the Indian Patents Act affect global pharmaceutical deal valuations?
Section 3(d) has a direct impact on the Indian market valuation of any drug whose commercial life in Western markets is substantially extended by secondary patents. If a drug’s COM patent has expired and its U.S. revenue is sustained by a formulation or polymorph thicket, that thicket provides zero protection in India. The Indian market value of the drug is the NPV of revenue under generic competition, not under exclusivity, and that value may be substantially lower than a pro-rata share of the U.S. multiple would imply. For global M&A deals where Indian market revenue is a material component of the valuation, this jurisdictional difference requires explicit treatment rather than the application of a uniform global exclusivity assumption.
How should analysts model the impact of FDA Complete Response Letters on patent-based valuation?
A Complete Response Letter (CRL) does not terminate a drug’s development but does impose a timeline delay and may require additional clinical data, which has both a direct cost (additional trial expense) and an indirect cost (patent term consumed during the additional development period). For a drug where effective patent life is already compressed, a CRL that adds 2 years to the development timeline can eliminate $1 to $3 billion in NPV from a mid-tier product simply through the time-value-of-money effect on pushed-out revenue, independent of any change in commercial peak sales assumptions. PTE applications can partially recover this time, but PTE calculations include only the FDA regulatory review period, not any additional clinical trial time required by the CRL. The analyst’s practical approach is to update the probability of approval downward and extend the commercial launch date by the expected additional development period, then recalculate NPV under the revised timeline.
Conclusion: The Patent Portfolio as a Balance Sheet
The pharmaceutical patent portfolio is not a legal filing cabinet. It is a balance sheet item, a financial asset with a quantifiable present value, a defined duration, and a specific risk profile. Reading it requires combining legal literacy (claims language, prosecution history, Paragraph IV strategy), financial modeling (DCF construction, scenario analysis, discount rate calibration), regulatory knowledge (Orange Book mechanics, BPCIA exclusivity, IRA negotiation probability), and geopolitical judgment (jurisdiction-specific patent law, global arbitrage dynamics, legislative risk).
The drugs that dominate today’s pharmaceutical revenue landscape, the biologics and specialty drugs that generate billions in annual sales, are protected not by simple, single-date patent expirations but by layered portfolios of secondary patents, regulatory exclusivities, and device protections that can extend effective monopoly periods by years or decades beyond what a primary patent analysis would project. Modeling those extensions accurately is the difference between a naive forecast and a credible valuation.
At the same time, the legal and regulatory environment is tightening around the practices that make those extensions possible. FTC enforcement, legislative reform efforts, the IRA’s pricing intervention, and the increasing sophistication of generic and biosimilar challengers all apply downward pressure on the duration and economic value of thicket-based exclusivity extensions. The long-run expected LOE date for a drug with a dense thicket is earlier, in expectation, than the latest-expiring thicket patent would suggest. Quantifying that regulatory risk premium is as important as mapping the patent landscape itself.
The analysts who do this work well, who read the claims, map the family, model the litigation scenarios, calibrate the erosion curves to drug type and geography, and stress-test the DCF against the IRA and FTC headwinds, possess a genuine informational advantage in an industry where the difference between the right LOE date and the wrong one is measured in billions of dollars.
Patent expirations, exclusivity dates, and legislative provisions are subject to change. Consult current FDA Orange Book listings, USPTO records, and relevant legal counsel before making investment or operational decisions based on specific patent data.