There is a certain type of pharmaceutical executive who still thinks of patent searching as something lawyers do—a back-office function that happens somewhere between the R&D floor and the courtroom, insulated from the real business of developing drugs. That view is expensive.
Companies that treat patent intelligence as a strategic function, not a legal chore, get to market faster, spend less on avoidable litigation, and identify acquisition targets before the banker pitch decks arrive. The ones that don’t tend to rediscover these facts the hard way: a surprise blocking patent eighteen months before launch, a competitor’s biosimilar that nobody saw coming, or a billion-dollar acquisition that turns out to rest on a patent the USPTO should never have granted.
This piece is for business professionals who want to stop treating the patent database as a legal archive and start using it as a real-time intelligence feed. We’ll cover the full picture: what drug patents actually are, how they work economically, the specific types of searches you need to run and when, the tools that do the job properly, and how to apply all of it to the situations where it matters most. Think of it as a working manual, not a primer.
Part I: What a Drug Patent Is—and Why the Economics Make It Unavoidable
The Core Bargain
A pharmaceutical patent is, at its simplest, a government-backed deal. The inventor gets a time-limited monopoly; the public gets a detailed technical disclosure it can eventually use. The statutory term is 20 years from the date of filing [1]. After that, anyone can manufacture the drug, the price competes down, and patients generally benefit.
This bargain exists because pharmaceutical R&D has an unusual economic problem. The cost of the first pill—counting all the failed compounds, all the clinical attrition, all the regulatory work—runs to something between $1 billion and $2.6 billion by most estimates, depending on the therapeutic area and how you count [2]. The cost of the second pill is a few cents of manufacturing. A competitor who waits for clinical proof and then copies the molecule would outcompete the innovator on price without bearing any of the development cost. No rational company would invest in drug discovery under those conditions. The patent, by preventing that outcome for a defined period, makes the investment viable.
That logic is uncontroversial among economists of almost every stripe. What is contested is whether the current system is calibrated correctly—whether the periods of exclusivity are too long, whether secondary patents extend protection in ways that far exceed the original investment rationale, and whether the prices that result from monopoly protection are justified. These are real debates, and they have real consequences for the regulatory environment you operate in. Pressure from Congress, the FTC, and large payers has intensified steadily over recent years. A patent strategy that ignores this political reality is a strategy with blind spots [3].
To obtain a patent, an invention must meet three core criteria. It must be novel—not already disclosed in prior art. It must be non-obvious to a person of ordinary skill in the relevant field. And it must be useful. For pharmaceutical patents, ‘useful’ typically means demonstrating some pharmacological activity, though the bar varies by the type of claim. Composition of matter claims for new molecular entities face relatively modest utility requirements at filing; the deep safety and efficacy data comes later in clinical development. Method-of-use claims must be supported by credible evidence that the recited use actually works [10].
Why Patent Searching Is a Business Function
The reason patent searching has historically been siloed in legal departments is historical, not logical. When patent prosecution was primarily about filing your own patents and occasionally checking for infringement, lawyers were the right people for the job. The analytical work involved reading legal documents and making legal judgments.
The job has changed. Modern patent intelligence involves tracking thousands of documents across multiple jurisdictions, connecting patent filings to clinical trial data, predicting when a competitor’s exclusivity will lapse, identifying white space in a crowded technological field, and assessing whether an acquisition target’s IP can survive a challenge. That is business analysis, not legal review. It requires different skills, different tools, and a different organizational home.
The companies that have internalized this tend to sit their competitive intelligence teams directly alongside business development and R&D strategy, not inside the legal function. Patent data flows into portfolio decisions, pricing models, licensing negotiations, and M&A due diligence. The ones that haven’t made this shift often have excellent lawyers and mediocre strategic visibility [4].
There is also a timing argument for treating patent intelligence as a business function. Lawyers typically engage with patents reactively: a filing deadline arrives, a lawsuit is threatened, a license negotiation begins. Business-oriented patent intelligence is proactive by design. You don’t wait for a competitor’s press release to learn about their oncology pipeline; you pull their PCT applications every quarter. You don’t learn about a blocking patent the week before your NDA submission; you run FTO analyses at every development milestone. That time advantage compounds significantly across a product lifecycle.
The Multi-Layer Fortress: Types of Pharmaceutical Patents
Before you can search for drug patents effectively, you need to know what you’re looking for. A single drug product can carry dozens of patents of different types, each protecting a different aspect of the commercial asset.
Composition of Matter Patents
These are the foundational patents—sometimes called compound patents or API patents. A composition of matter patent covers the chemical entity itself. If your molecule is in a product, the patent covers it, regardless of formulation, route of administration, or indication [5]. This is the broadest and strongest form of pharmaceutical patent protection. From a competitive intelligence standpoint, these are the first patents you look at when assessing a drug’s market exclusivity position.
For small molecules, the composition of matter patent typically covers the specific compound, often claimed within a broad Markush structure that encompasses structurally related analogs. For biologics, it covers the molecular structure of the biologic—the specific amino acid sequence of a monoclonal antibody, for instance. The breadth of composition of matter protection in the biologic space has been a major battleground: claims that cover entire classes of antibodies targeting the same epitope have been challenged as insufficiently disclosed under the ‘written description’ and ‘enablement’ requirements, with the Supreme Court’s 2023 Amgen v. Sanofi decision narrowing the permissible scope of functional genus claims.
Formulation and Delivery Patents
These patents cover how the drug is put together and delivered to the patient: extended-release tablets, particular excipient combinations, transdermal patches, injectable formulations, and so on [6]. From a pure science standpoint, many of these innovations are genuine improvements. Extended-release formulations reduce dosing frequency and improve adherence. Specific excipient combinations can improve stability or reduce side effects. From a lifecycle management standpoint, they also provide additional years of exclusivity after the core compound patent expires, which is why critics describe the practice as ‘evergreening’ [7].
The legal validity of formulation patents as an extension strategy has been tested in litigation repeatedly. Courts have found many secondary formulation patents valid; they have also found many obvious in light of prior art, particularly when the claimed formulation differs only modestly from known approaches. The outcome depends heavily on the specific facts: how different is the formulation from what was previously known, and was that difference predictable to a person of ordinary skill?
Whether you call it lifecycle management or evergreening depends largely on whether you’re the innovator or the generic competitor. From a patent intelligence standpoint, what matters is that you identify all of them and assess each individually. A patent thicket that looks impenetrable on paper may contain several patents of questionable validity. The gap between ‘patent exists’ and ‘patent is enforceable’ is where competitive strategy is won and lost.
Method-of-Use Patents
A method-of-use patent protects a specific way of using a drug to treat a particular condition [8]. The drug itself may not be patentable—it might already be known—but a novel therapeutic application can be. These patents are particularly relevant in two scenarios: first, when an innovator discovers a new indication for an existing drug; second, when a generic company is trying to determine whether it can launch for an established indication without infringing a patent on a newer, still-protected use.
Skinny labeling—launching a generic drug with a label that carves out the patented indication—is the generic industry’s standard response to method-of-use patents. Whether it works as a legal strategy depends on specifics that have evolved through litigation. The Caraco Pharmaceutical Laboratories v. Novo Nordisk case established that generic companies can use a counterclaim mechanism to require innovators to correct overbroad patent use codes in the Orange Book—an important procedural tool in disputes over method-of-use patent scope.
One practical complication: method-of-use patents are not always listed in the Orange Book. Under Hatch-Waxman, only patents that claim the drug itself or a method of using it for a condition claimed in the NDA are listable. A company that develops a new indication after original approval and files a method-of-use patent may not be able to list it if the new indication is covered by a separate supplemental NDA rather than the original. This creates situations where a valid, in-force method-of-use patent exists but doesn’t appear in the Orange Book—and won’t trigger the Paragraph IV certification process. Comprehensive FTO analysis must look beyond the Orange Book listings [9].
Process and Manufacturing Patents
Process patents cover how you make the drug, not the drug itself [6]. For small molecules, these can sometimes be designed around: if the end product is the same, you may be able to reach it through a different synthetic route. For biologics, process patents are often the most critical layer of protection. The manufacturing process for a monoclonal antibody is extraordinarily complex—cell line selection, fermentation conditions, purification steps, post-translational modifications, formulation—and the process defines the product in ways that are genuinely difficult to replicate.
Biosimilar developers must demonstrate their product is ‘highly similar’ to the reference biologic despite knowing that the innovator’s exact process is protected by patents they cannot use. That creates a substantial technical barrier to entry on top of the legal one. The FDA’s totality-of-evidence standard for biosimilar approval means that a biosimilar developer must often conduct extensive analytical comparability studies and at least some clinical testing to bridge from their process to the reference product’s clinical data—a requirement that doesn’t exist for small molecule generics, which simply need to demonstrate bioequivalence [10].
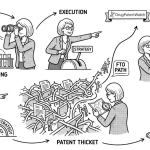
The Patent Thicket
When you layer composition of matter patents, formulation patents, method-of-use patents, and process patents around a single drug, you get what academics and litigators call a patent thicket: a dense web of overlapping protections that any would-be competitor has to navigate [7]. The classic example cited in academic literature is Humira (adalimumab), which at its peak carried over 130 distinct patents covering various aspects of the drug, its manufacturing, its formulations, and its uses. AbbVie’s patent strategy around Humira is credited with—or blamed for, depending on your perspective—delaying biosimilar competition in the United States by several years relative to the European market, where the patent landscape was managed differently. That delay represents tens of billions of dollars in branded revenue.
Understanding the full extent of a thicket—not just the primary patent—is the core task of any serious FTO analysis. Mapping a thicket comprehensively requires pulling the complete Orange Book listing, searching for non-listed patents that might still be infringed, checking for pending applications that could issue as additional blocking patents, and verifying the legal status and ownership of each patent identified. This is not a two-hour job; done properly, it requires a systematic multi-database search process and expert analysis of the results.
Part II: The Patent Lifecycle—What the Numbers Actually Mean
The 20-Year Fiction
The 20-year patent term sounds like a substantial commercial window. In practice, for pharmaceuticals, it rarely is. Here is why.
The clock on a drug patent starts when the application is filed, typically early in the preclinical research phase. What follows is a decade-plus process: preclinical safety studies, Phase I dose escalation, Phase II proof-of-concept, Phase III efficacy and safety trials, regulatory submission, and FDA review. The average time from initial discovery to first approval runs around 12 to 15 years [11]. By the time a drug reaches the market, it has consumed 10 to 15 years of its 20-year patent term in development. The remaining window of exclusivity—the period during which the company can actually sell the patented drug before generic competition enters—averages 7 to 10 years for most small molecules [12].
That compressed commercial window is the single most powerful force shaping pharmaceutical business strategy. It explains high launch prices (you need to recover multi-billion-dollar investments in a short window), aggressive lifecycle management (extending that window by any legitimate means), the premium placed on indication expansions, and the ferocity of patent litigation when generics attempt early entry.
For oncology drugs, the effective patent life tends to be shorter than average, because phase III trials are large, expensive, and long even for drugs targeting validated mechanisms. For rare disease drugs, the effective life may be longer, partly because development is sometimes faster when placebo-controlled trials aren’t feasible and surrogate endpoints are accepted.
The investor community focuses on the primary patent expiry date when modeling revenue trajectories. What they should focus on is the loss of exclusivity (LOE) date—a different number that accounts for extensions, secondary patents, and regulatory exclusivities. Getting this calculation right is where real financial modeling value comes from, and getting it wrong is a recurring source of earnings surprises and valuation errors in the sector.
Clawing Back Time: Patent Term Extensions
The Hatch-Waxman Act of 1984 recognized the effective patent life problem and created Patent Term Extension (PTE) as a partial remedy [13]. PTE lets the patent owner restore a portion of the time lost to FDA review. The extension calculation combines half the time spent in the IND-to-NDA clinical testing period with the full regulatory review period from NDA submission to approval.
The statutory limits are significant:
- Maximum extension: five years
- Maximum total remaining term after extension: 14 years from FDA approval
- Application deadline: 60 days following marketing approval
- Only one patent per drug approval is eligible for PTE
That last point makes the selection of which patent to extend a genuine business decision, not a legal formality. If you have a compound patent expiring in three years and a formulation patent expiring in six years post-approval, choosing which to extend requires modeling the competitive landscape and the likely lifecycle strategy. A generic competitor analyzing your LOE position needs to know which patent you selected and whether the extension was granted, because the unextended patent expires on schedule while the extended one gets additional time.
PTE data is publicly accessible through USPTO databases, but cross-referencing it with FDA approval dates, clinical development timelines, and Orange Book data is time-consuming manually. DrugPatentWatch integrates these data streams, making LOE calculations for marketed drugs far faster and more reliable than doing it by hand across multiple government databases [14].
There is also a European equivalent worth understanding for companies operating globally. The Supplementary Protection Certificate (SPC) mechanism in the EU serves a similar function to PTE but operates through national patent offices and can add up to five years of protection beyond the patent term, capped at 15 years of total protection from first EU marketing authorization. The SPC calculates its term differently from PTE, and the two mechanisms interact with global patent families in ways that require jurisdiction-by-jurisdiction analysis.
Regulatory Exclusivities: The Layer Most Financial Models Get Wrong
Entirely separate from the patent system, the FDA grants regulatory exclusivities that operate as independent barriers to generic entry. These are statutory prohibitions on FDA approvals—meaning even if all patents have expired or been invalidated, the FDA cannot approve a competing generic while an exclusivity is in force [15].
The major categories:
New Chemical Entity (NCE) exclusivity grants five years of protection for drugs containing active ingredients never before approved by the FDA. During the first four years of this window, the FDA won’t accept a generic application at all. In year five, a generic can file with a Paragraph IV certification, but the 30-month stay triggered by an innovator lawsuit means FDA approval still can’t happen until roughly year five plus time for the litigation to resolve, or until the exclusivity expires. For many drugs, NCE exclusivity functions as a de facto five-to-six year protection floor.
Orphan Drug Exclusivity (ODE) provides seven years of market exclusivity for drugs approved for rare diseases affecting fewer than 200,000 U.S. patients. This has become increasingly relevant as pharmaceutical companies pursue rare disease indications, sometimes for drugs that also have broader potential applications. If a drug receives orphan designation for one indication and then the company pursues a much larger indication separately, the orphan exclusivity applies only to the specific designated indication—it doesn’t block generic entry for the non-orphan use. The interaction between orphan exclusivity and method-of-use patents covering the broader indication creates strategic complexity that rewards careful analysis.
Pediatric Exclusivity deserves special attention because of how it operates. Unlike other exclusivities, it is an add-on: six months tacked onto all existing patents and exclusivities for that active ingredient [16]. This means a Paragraph IV challenger who has won in court—defeating the innovator’s patent—can still face six additional months of exclusivity if the innovator completed pediatric studies in the interim. At blockbuster revenue levels—$3 billion to $5 billion annually—six months of additional exclusivity is worth $1.5 billion to $2.5 billion in protected revenue. That’s why innovators nearly always pursue pediatric studies and why generic companies factor the pediatric exclusivity timeline into their litigation and market entry strategies.
New Clinical Investigation Exclusivity provides three years of protection for supplements to existing approvals—new indications, dosage forms, or strengths—when new clinical investigations were essential to the approval. The ‘essential to approval’ standard requires that the new clinical work was actually necessary for the supplemental approval, not merely supportive. This exclusivity is narrower than NCE exclusivity: it blocks approval of competitors using the innovator’s new clinical data, but doesn’t prevent a competitor from submitting their own application with their own clinical data.
The critical business point: the true LOE date for a drug is the later of the relevant patent expiration (including any PTE) and the relevant regulatory exclusivity expiration. Analyzing only patents without checking exclusivity status leads to systematic underestimation of the protected period. This is among the most common errors in competitive intelligence work, and it has cost companies real money: a generic entry forecast that assumes LOE at patent expiry without checking for an in-force orphan exclusivity will be systematically early.
The Hatch-Waxman Framework: Rules of the Adversarial Game
The 1984 Hatch-Waxman Act created a structured, adversarial game between innovators and generic companies. Understanding the rules matters for patent intelligence across both sectors.
Under Hatch-Waxman, innovators must list their patents in the FDA’s Orange Book if those patents claim the drug or a method of using it [17]. When a generic company files an Abbreviated New Drug Application (ANDA), it must certify its position on each listed patent. The four certification options range from Paragraph I (no patent listed) to Paragraph IV (patent is invalid or not infringed by the generic product). Paragraph IV filings trigger the most commercially significant litigation in the pharmaceutical sector.
Filing a Paragraph IV certification is defined under the statute as a technical act of patent infringement, which gives the innovator the right to sue within 45 days. If the innovator sues within 45 days, the FDA cannot approve the generic for 30 months—a critical protection period that gives the innovator time to litigate. If the innovator wins the litigation before the 30-month stay expires, the generic is blocked. If the generic wins, or if the innovator doesn’t sue within 45 days, the generic can get approved immediately.
The first generic company to file a successful Paragraph IV certification generally receives 180 days of exclusive generic marketing before other generics can enter—a powerful financial incentive for first-mover advantage in the generic industry. This 180-day exclusivity can be worth tens of millions of dollars for major products, which is why generic companies invest heavily in FTO and invalidity analysis to position themselves as first filers.
Every step in this framework depends on patent intelligence: which patents are listed in the Orange Book, whether they are valid, whether the generic product infringes them, what prior art might be used to challenge them, and how similar litigation has resolved for related patents. DrugPatentWatch maintains comprehensive data on ANDA filings, Paragraph IV certifications, and related litigation outcomes—including both district court decisions and PTAB IPR outcomes—that makes tracking this landscape manageable [18].
Part III: The Mechanics of Search—What to Run and When
Defining the Search Before You Start
The most common mistake in patent searching is firing up a database before knowing exactly what question you’re trying to answer. Different business questions require fundamentally different search approaches, databases, and interpretations. Let’s be specific.
Patentability and Novelty Searches
Question being answered: Can we patent this invention?
A patentability search looks for prior art—existing patents, published applications, journal articles, conference presentations, or any other public disclosure—that might prevent your invention from being granted a patent. The two grounds for rejection are lack of novelty (someone already disclosed the same thing) and obviousness (the invention is a predictable combination or modification of what’s already known) [19].
These searches should happen before you file, obviously, but the more valuable discipline is running them before you commit significant R&D resources. Finding that a particular synthetic route was disclosed in a 2015 paper is far less costly when you discover it in month two of a project than month thirty. Companies that run patentability searches at the concept stage—before initiating synthesis campaigns—avoid the problem of spending two years generating data for an unpatentable compound. The search takes days; the wasted development work takes years.
The scope of a patentability search is intentionally broad: all relevant jurisdictions, all types of documents including non-patent literature. A journal article from 1988 describing your target compound structure is prior art regardless of whether it was ever patented. This is why text-based database searching must be combined with literature searches (SciFinder, Reaxys) and structure-based chemical searches that reach across both patent and non-patent databases.
Freedom-to-Operate Searches
Question being answered: Can we commercialize this product without infringing anyone else’s active patents?
FTO analysis is the highest-stakes routine patent search in pharmaceutical development. The consequences of getting it wrong range from expensive to catastrophic. An FTO search focuses specifically on the claims of unexpired, in-force patents in the jurisdictions where you plan to sell the product [20]. It asks: does our product, as we plan to make and market it, fall within the scope of anyone else’s patent claims?
FTO is not a one-time event. It’s a recurring process tied to development milestones. Your drug candidate changes during development—new formulations, new synthesis routes, new indications. Each change alters the FTO picture. Companies that run a single FTO check at the start of development and consider the box checked are creating risk, not managing it. The standard practice is FTO analysis at IND filing, Phase II initiation, Phase III initiation, and NDA submission, with ad hoc analyses triggered by any material change in the product or the patent landscape.
An FTO search that identifies a potentially blocking patent doesn’t necessarily mean the program is dead. The blocking patent may be invalid (triggering an invalidity analysis). Your product may be designable around the claims without compromising efficacy. A license may be negotiable on acceptable terms. But you need to know about the issue early enough to exercise these options—not three months before your planned launch.
A key nuance: FTO analysis is specific to the product being commercialized in the jurisdiction being analyzed. A formulation patent that blocks your tablet product may not apply if you switch to a capsule. A U.S. process patent doesn’t block manufacturing in Ireland. Good FTO analysis is precise about the specific product, process, and geography under evaluation—generalizations produce results that are neither accurate nor useful.
Invalidity Searches
Question being answered: Is this patent weak enough to challenge?
When you have a blocking patent—one that stands between your product and a clear path to market—an invalidity search becomes your offensive tool. You are looking for prior art that the patent examiner didn’t find: publications, earlier patents, or other disclosures that demonstrate the invention wasn’t novel or was obvious to a person of ordinary skill in the relevant field [19].
The standard for invalidity in U.S. district court litigation is ‘clear and convincing evidence’—a high bar that Congress established explicitly to protect issued patents from being easily undone. At the USPTO’s Patent Trial and Appeal Board (PTAB), the standard is lower: ‘preponderance of the evidence.’ This difference has made Inter Partes Review (IPR) proceedings at PTAB a preferred venue for invalidity challenges in the pharmaceutical sector. Generic companies routinely file IPR petitions as a complement to Hatch-Waxman litigation—sometimes before the ANDA is even filed, sometimes in parallel with district court proceedings.
PTAB has a relatively fast schedule—final written decisions are typically issued within 12 to 18 months of institution—and the petition fee is far lower than full district court litigation. For patents of questionable validity, an IPR can resolve the question faster and more cheaply than a trial. The IPR process has limitations, though: estoppel rules mean that grounds raised in an IPR petition (and some that weren’t raised) cannot be reasserted in district court, which requires careful strategic planning before filing.
Landscape and Competitive Intelligence Searches
Question being answered: What is everyone working on, where is technology heading, and where can we establish a defensible position?
Landscape analysis operates at a higher altitude than the other search types. Rather than analyzing a single patent or product, it maps an entire technology space. The output is a visualization and analysis of all patents within a defined area, organized by technology type, assignee, and filing date—revealing who holds the dominant positions, which targets are heavily contested, and which areas show low patenting activity relative to scientific publication activity [21].
Competitive intelligence searches focused on specific companies—tracking what a rival is filing, in which therapeutic areas, targeting which biological mechanisms—can give you 18 to 24 months of visibility into their pipeline before clinical trial disclosures appear in public registries. Patent applications publish 18 months after filing, so for a program that files a patent at the start of discovery and enters Phase I two years later, you have six months of advance notice from the patent publication alone. For programs that take longer to develop, the lead time is even greater.
These searches are most valuable when run continuously, not periodically. The value of a monitoring alert that fires when a competitor files a new patent in your core therapeutic area is the time it gives you to respond strategically—accelerating your own program, adjusting your IP filing strategy, or starting a due diligence process on the competitor’s asset. A quarterly landscape review catches the same information three months late.
Part IV: The Tools—Choosing the Right Database
Free Public Databases: What They Do Well and Where They Fall Short
The major public patent databases—USPTO Patent Public Search, the European Patent Office’s Espacenet, and WIPO’s PATENTSCOPE—are genuinely powerful resources that have improved substantially over the past decade. Espacenet covers over 150 million patent documents worldwide, with machine translation capabilities that make non-English patents accessible to English-speaking searchers [22]. PATENTSCOPE provides early visibility into PCT applications, often giving you a look at global filing strategies before regional patents are granted [23].
For basic lookups, document retrieval, and exploratory searches, these tools work. Their limitations become apparent when you need to answer complex business questions. They present raw, uncurated data. Assignee names are inconsistent—’Merck’ means different companies depending on jurisdiction, and the same company can appear under dozens of slightly different name variants as a result of mergers, acquisitions, subsidiary naming conventions, and simple data entry errors. Legal status information is often unreliable or significantly lagged. You can find the patent document, but determining whether it is currently in force, who currently owns it, or how it connects to the FDA’s Orange Book requires substantial additional work.
For pharmaceutical-specific business analysis, the free databases lack the integration layer that converts patent data into actionable intelligence. A patent document on its own is a legal text. A patent connected to the Orange Book listing, the ANDA filing history, the litigation docket, and the clinical trial record is a piece of business intelligence. Public databases provide the former. Specialized commercial platforms provide the latter, and the difference matters for the quality of decisions that flow from the analysis.
Commercial Databases: General vs. Pharmaceutical-Specific
Commercial patent databases fall into two groups: general-purpose platforms (Clarivate’s Derwent World Patents Index, PatSnap, Questel Orbit) and pharmaceutical-specific intelligence platforms like DrugPatentWatch.
General-purpose commercial platforms solve the data quality problems of free databases. They standardize assignee names, maintain current legal status, support advanced search syntax including chemical structure and biologic sequence searching, and provide analytics and visualization tools. For a technology company doing patent landscaping across multiple industries, these platforms are the right choice. For pharmaceutical-specific work, they still require extensive manual bridging between patent data and the regulatory and commercial context that drives business decisions.
DrugPatentWatch occupies a different category. Rather than being a patent database that pharmaceutical professionals can use, it is a pharmaceutical intelligence platform with patents as one of its core data layers. The key feature is deep integration across data sources that are otherwise siloed: when you pull a drug’s profile, you see the relevant patents, the Orange Book listings, the regulatory exclusivities, the ANDA filing history, the Paragraph IV certification history, the litigation record across both district courts and PTAB, and the clinical trial pipeline—all in a unified interface [18]. Answering the question ‘What is the true LOE date for this drug and what is its litigation risk profile?’ requires data from all of these sources simultaneously. Manual cross-referencing across USPTO, FDA, PACER, and ClinicalTrials.gov is a half-day project per drug. On DrugPatentWatch, it’s a ten-minute query.
For business professionals who must justify subscription costs, the ROI argument is clear. The relevant comparison is not the subscription cost versus zero; it is the subscription cost versus the cost of decisions made with incomplete data. In the pharmaceutical sector, the decisions at stake—whether to pursue an ANDA, how to price an acquisition target, which development programs to accelerate—routinely involve hundreds of millions of dollars. The information advantage from specialized tools is not a luxury at those stakes. <blockquote> ‘Companies that use patent data for trend forecasting are 2.3 times more likely to be market leaders in their respective fields.’ — McKinsey & Company [24] </blockquote>
Choosing the Right Tool: A Framework
Here is a practical guide to tool selection based on the task:
Basic document retrieval or a specific patent lookup: USPTO Patent Public Search or Espacenet. Free, fast, and adequate for the purpose.
Patentability or FTO search for a small molecule: you need a commercial database with chemical structure search capability. CAS STNext, Questel Orbit Chemistry, or PatSeer. Text-based keyword searching will miss structurally similar compounds claimed in Markush structures, which can be the most commercially significant prior art.
Pharmaceutical competitive intelligence, LOE forecasting, ANDA tracking, or lifecycle management analysis: DrugPatentWatch or a comparable pharmaceutical intelligence platform. General-purpose databases require extensive manual work to reach the same result, with substantially more opportunity for error on the regulatory and litigation data.
Biologic sequence searching: Clarivate’s Derwent SequenceBase, STN. Keyword searching is not capable of finding patents covering a biological sequence that shares 94% identity with yours but is described with different nomenclature or embedded in a figure without a standardized Sequence Listing.
The framework is not about budget or prestige. It is about whether the tool can actually answer your question. Using USPTO Patent Public Search to assess the LOE position of a blockbuster biologic is like using a paper map to navigate a subway system: technically a navigation tool, wrong for the job.
Part V: Search Methodology in Depth
Keyword Searching: Where to Start and Where to Stop
Keyword searching is the entry point for most patent searches because it requires no specialized training—you type terms related to your subject into a database and review the results. For pharmaceutical patent work, useful starting keywords include active pharmaceutical ingredient names (INN and brand names), CAS registry numbers, chemical synonyms and alternate nomenclature, therapeutic class descriptors, target or mechanism descriptions, and disease and condition terms.
The problem with keyword searching alone is systematic. It is simultaneously too broad and too narrow. Too broad: pharmaceutical patents use technical language, and searching for common disease terms retrieves thousands of tangentially related documents that have to be manually triaged. Too narrow: if you don’t know the right synonym, you miss the document. A compound your team calls ‘Compound 12c’ internally might be described in a competitor’s patent with its IUPAC name, its CAS number, a proprietary code name, and a structural description in the figures. None of these strings matches the others. Any search built solely on one of them will miss the rest.
The correct role of keyword searching in a comprehensive strategy is as a starting point that identifies the most relevant CPC classification codes and key assignee names for subsequent, more structured searching. The first pass with keywords gives you 20 to 30 highly relevant documents. You then extract the most common classification codes from those documents and the names of the most active assignees. Those outputs feed the next, more systematic search passes. Keyword searching alone is not a complete methodology—it is the first step in an iterative process.
Classification Searching: The Foundation of Systematic Coverage
The Cooperative Patent Classification (CPC) and International Patent Classification (IPC) systems assign hierarchical codes to patents based on their technical content. These systems are the closest thing the patent world has to a language-independent controlled vocabulary, which matters enormously for comprehensive searching across jurisdictions [25].
For pharmaceutical patents, the relevant CPC sections include A61K (preparations for medical purposes, including drug formulations), A61P (therapeutic activity of chemical compounds, organized by disease area), C07D (heterocyclic compounds, covering a large proportion of small molecule drugs), C07K (peptides and proteins), and C12N (microorganisms, enzymes, and biologics). Within each section, the codes become increasingly granular—A61K 31/ covers organic active ingredients generally, while A61K 31/505 covers specifically pyrimidines (a substructure present in many kinase inhibitors and antivirals).
Searching by classification code retrieves all patents within a specific technology domain regardless of the specific keywords used by the inventors. A German patent, a Japanese patent, and a U.S. patent on the same compound will all receive the same CPC classification codes if they describe the same technology. This makes CPC-based searching genuinely cross-jurisdictional in a way that keyword searching—constrained by language—is not.
The practical workflow: use keywords to find a small set of highly relevant patents, examine their CPC codes to identify the most relevant classification buckets, then run targeted classification searches to retrieve everything in those buckets. Combine the classification results with your keyword results, remove duplicates, and you have a substantially more complete starting set than either method would produce alone.
Assignee and Inventor Searching: The Competitive Intelligence Core
Tracking the patent filings of specific companies or research groups is one of the most direct competitive intelligence techniques in the patent intelligence toolkit. An assignee search retrieves all patents and applications owned by a specific entity, revealing its R&D focus, key technologies, and strategic direction over time [4].
When you run assignee searches quarterly against your top five competitors, you get a near-real-time view of their R&D priorities—often a year or more before clinical trial disclosures appear in public registries. A company that begins filing patents around a specific biological target in Q1 is probably advancing a discovery program. If those filings cluster around particular chemical scaffolds or formulation approaches, you can often infer what the molecule looks like and what the development strategy is.
Practical points about assignee searching that experienced analysts know and beginners learn the hard way: assignee names must be standardized before searching, because the same company can appear under dozens of name variants in raw patent databases. Subsidiaries, acquired entities, joint ventures, and simple data entry variations all create fragmentation. Commercial platforms that have already done this cleaning work will give you materially more complete results than free databases for any major company with a complex corporate history.
Inventor searching adds another dimension. Key scientists carry their research programs with them when they change employers. A principal investigator who spent ten years developing a therapeutic modality at one company and then joins a startup is probably continuing related work. Their patent filings at the new employer often reveal the new company’s direction six to twelve months before any public announcement. Monitoring the publication records of known key opinion leaders and drug discovery scientists in your therapeutic area, combined with assignee monitoring of the companies they join, creates a human-intelligence layer on top of the documentary intelligence.
Citation Searching: The Intellectual Genealogy of Innovation
Every patent cites prior documents—backward citations—that the examiner and applicant considered relevant to the scope of the invention. Every later patent that cites a key document—forward citations—indicates that document’s influence in the subsequent technology development.
Backward citation analysis gives you a pre-curated map of foundational prior art. The patent examiner and the applicant’s attorneys have already identified documents relevant to the claimed subject matter. Starting from a known highly relevant patent and following its backward citations outward is faster than starting from scratch and often surfaces documents that keyword or classification searching would miss [26].
Forward citation analysis serves a different purpose. The forward citation count on a patent is a rough proxy for its technical importance and commercial significance. A foundational platform patent might accumulate hundreds of forward citations over a decade as subsequent inventors build on it. A narrow formulation patent covering one specific excipient combination might have a handful. Citation frequency is a useful triage signal when you have a large result set and need to prioritize which documents to analyze in depth.
One specific application of forward citation analysis that business professionals often overlook: if you have already been granted an important patent, monitoring its forward citations over time tells you who is building on your technology, whether anyone is attempting to design around it, and whether competitors are filing improvement patents that might create their own blocking positions in adjacent space.
Boolean Logic and Search Syntax: Speaking the Database’s Language
Patent databases use Boolean logic to combine search terms, and precision in query construction directly determines the quality of results. The three Boolean operators are AND, OR, and NOT [27].
AND narrows the search by requiring all connected terms to appear in a retrieved document. (kinase AND inhibitor) retrieves only patents discussing both concepts. Adding more AND terms narrows the set further—useful when a broad initial search retrieves thousands of irrelevant results.
OR broadens the search by accepting either connected term. (erlotinib OR ‘OSI-774’ OR Tarceva) ensures you catch all naming variants for the same compound. This is where synonyms, trade names, INN names, and code numbers need to be consolidated—linked by OR operators within a single clause, then connected to other concepts with AND.
NOT excludes terms. Use it carefully: (antibody NOT bispecific) retrieves antibody patents but excludes any document mentioning bispecific antibodies, which could inadvertently exclude relevant patents that discuss both standard and bispecific antibody formats in the same document.
Parentheses control the logical order of operations, just as in mathematics: (erlotinib OR gefitinib OR osimertinib) AND (resistance OR ‘acquired resistance’ OR progression) is fundamentally different from erlotinib OR (gefitinib AND resistance) OR osimertinib.
Truncation symbols extend keyword coverage to word variations. The asterisk stands for any number of characters: immuno* captures immunology, immunotherapy, immunomodulatory, and immune. Database-specific syntax varies: some platforms use $ for truncation, others use ?. Check the database’s documentation before assuming the standard symbol applies.
Proximity operators—NEAR, ADJ, WITHIN—require connected terms to appear within a specified number of words of each other, in any order (NEAR) or in a specific order (ADJ). (extended NEAR3 release) finds documents where ‘extended’ and ‘release’ appear within three words of each other. This avoids the problem of retrieving documents where ‘extended’ appears in a methods section and ‘release’ appears in the claims section, with no substantive relationship between them. Proximity operators significantly improve precision in complex multi-concept searches [27].
Constructing the Full Search Strategy
An effective pharmaceutical patent search combines these methods in a systematic sequence:
Start with a keyword search using all relevant synonyms for the key concepts, connected with appropriate Boolean operators. This retrieves an initial set of highly relevant documents.
From those documents, extract the most frequently assigned CPC codes. Run targeted classification searches on those codes.
Identify the most active assignees in the retrieved set. Run assignee searches to retrieve their complete relevant portfolio, then filter for the specific technology domain.
Run forward and backward citation searches on the five to ten most relevant documents identified so far. This surfaces related art that the earlier searches missed.
Combine all results, de-duplicate, and apply relevance screening. For a thorough FTO or invalidity analysis, this final set then goes into detailed claim-by-claim review.
This iterative approach takes longer than a single keyword search, but it substantially improves recall—the proportion of relevant prior art actually found. For FTO analysis, where missing a blocking patent has direct commercial consequences, recall is the critical metric. For patentability analysis, where missed prior art can result in an invalid patent that gets invalidated later during litigation, recall is equally critical.
Part VI: Chemical Structure and Biologic Sequence Searching
Why Keyword Searching Fails for Novel Compounds
If you are developing a new small molecule drug, the most important question in patentability and FTO analysis is whether structurally similar compounds have already been claimed. Text-based searching cannot answer this question reliably, and no amount of synonym generation closes the gap.
A composition of matter patent for a small molecule claims a specific chemical structure, typically extended through Markush notation—a generic claim format that uses variable substituents to cover families of related compounds [28]. A Markush claim might read: ‘A compound of formula (I), wherein R1 is selected from hydrogen, alkyl C1-C6, or halogen; R2 is selected from hydroxyl, alkoxy, or amino; and R3 is aryl or heteroaryl.’ This claim covers millions of specific compounds without naming any of them individually. Your compound may be a specific instance of this claim without the compound’s name, CAS number, or common synonym appearing anywhere in the patent text.
The only reliable method to assess whether a compound is novel and not covered by existing Markush claims is a chemical structure search. For any serious patentability or FTO analysis involving a chemical entity, structure searching is not optional.
Types of Chemical Structure Searches
Commercial databases with indexed chemical structures support four search types, each with a different purpose:
Exact match searches retrieve patents covering precisely the compound you drew. These confirm whether a specific compound has been explicitly claimed and are the fastest to run.
Substructure searches find larger molecules that contain your drawn structure as a fragment. If your compound is a specific substituted pyrimidine, a substructure search finds all patents claiming compounds that contain the pyrimidine ring system with your specific substitution pattern, regardless of what other functional groups they may add. This is relevant because a patent claiming a broader scaffold may cover your specific compound as one of many specific embodiments.
Similarity searches retrieve compounds within a defined structural distance from your query structure. The similarity threshold is adjustable—90% similarity returns fewer, more closely related results than 70% similarity. Similarity searching is the primary tool for assessing the prior art landscape around a compound structure: a prior art compound at 85% structural similarity that shows the same biological activity could be the basis for an obviousness rejection.
Markush searches are the most computationally intensive and commercially important structure search type. They determine whether your specific compound falls within the scope of a generic Markush claim in an issued patent. Running a comprehensive Markush search against a major database requires specialized software and can take hours even on optimized hardware. For drugs targeting validated mechanisms with extensive prior patent art—kinase inhibitors, GPCRs, nuclear receptors—Markush searching against the relevant claim universe is essential for any credible FTO analysis.
Biologic Sequence Searching: The Special Challenge
For biologic drugs—monoclonal antibodies, fusion proteins, antibody-drug conjugates, siRNA, antisense oligonucleotides, gene therapies—the equivalent of structure searching is sequence searching. The patent claim for a biologic is typically defined by a specific amino acid sequence (for proteins) or nucleotide sequence (for nucleic acid-based therapies). Finding whether a competitor has already claimed your biologic, or a variant with sufficient similarity to create FTO risk, requires running your sequence against patent databases using alignment algorithms [29].
Biologic sequence searching uses BLAST (Basic Local Alignment Search Tool) or proprietary alignment algorithms to find patents containing sequences with significant identity to your query. The threshold for relevance varies by context: an antibody VH sequence with 98% identity to a claimed sequence is an obvious candidate for FTO concern; a sequence with 65% identity in the framework regions but different CDRs may present different considerations depending on how the claims are drafted.
The challenge unique to biologics: not all patent applications that disclose sequences do so in machine-readable form. Standardized Sequence Listing files (in XML or the older ST.25 text format) enable automated database indexing. But sequences embedded in specification text, in figures, or in tables—without a formal Sequence Listing—are not automatically indexed by standard databases. Clarivate has reported that approximately 18% of biosequence patent applications filed in 2021 lacked a formal Sequence Listing [30]. This represents a meaningful coverage gap in any automated sequence search.
The practical implication for biosimilar developers and biologic innovators: automated sequence database searches should be supplemented with manual review of the most relevant application sets, particularly applications from competitors working on the same biological target or therapeutic class. A sequence that escaped automated indexing because it was embedded in a figure can still be prior art; missing it in an FTO analysis can have significant commercial consequences.
Part VII: Reading What You Find—Patent Analysis for Non-Lawyers
The Anatomy of a Patent Document
Once you have run your searches and retrieved the relevant documents, the next task is extracting the business intelligence from them. A patent document has a defined structure, and knowing where to look saves time.
The front page contains the filing date, the priority information (which can establish an earlier effective date for prior art purposes), the assignee, the inventors, the abstract, and the cited references. The filing date drives the 20-year term calculation. Priority information matters when a patent claims the benefit of an earlier provisional application or a foreign filing—the effective filing date for prior art purposes is the priority date, not the date the U.S. application was filed. The assignee tells you who owns it, though ownership may have changed since grant through subsequent assignments.
The specification—the detailed technical disclosure of the invention—must teach a person of ordinary skill in the relevant field how to make and use the invention. It contains the background (contextualizing the problem the invention addresses), the summary, the detailed description, and the examples. The examples in pharmaceutical patents are particularly valuable for competitive intelligence: they often describe specific compounds synthesized, specific biological assays run, and specific in vivo results obtained. Reading the examples in a competitor’s patent gives you a view of their actual experimental work that goes well beyond what their press releases reveal.
The specification also functions as the definitional dictionary for the patent. Technical terms used in the claims must be interpreted in light of how they are defined or used in the specification. When a claim says ‘effective amount,’ the specification’s definition of that term, and the examples illustrating it, inform how broadly that limitation should be read. Courts use this principle—reading claims in light of the specification—to resolve ambiguities in claim language [31].
Why the Claims Are All That Legally Matter
The claims are the only part of the patent that defines the legal monopoly. They are numbered sentences at the end of the document that describe the precise boundaries of what the patent owner has the exclusive right to prevent others from doing. Infringement is determined claim by claim, element by element: a product or process infringes a claim if, and only if, it meets every single limitation of that claim. Missing any one limitation means no infringement of that particular claim [32].
Two distinctions in claim structure have major practical consequences for FTO and invalidity analysis:
Independent vs. dependent claims. An independent claim stands alone, containing all elements necessary to define the invention. A dependent claim refers back to an independent claim and adds further, narrowing limitations. Dependent claims are always narrower than the independent claims they depend from. A product that infringes a dependent claim necessarily infringes the independent claim it depends on, because the dependent claim incorporates all of the independent claim’s limitations plus additional ones. In FTO analysis, you must analyze independent claims first—they define the broadest protection—and then dependent claims if the independent claim analysis raises concerns.
Transitional phrase scope. The word connecting the preamble to the body of a claim defines how broad or narrow the claim is. ‘Comprising’ is open-ended: the claim covers inventions that include the listed elements and may include additional, unlisted elements. This is the broadest and most common transitional phrase. ‘Consisting of’ is closed-ended: the claim covers inventions that have only the listed elements, nothing more. A formulation patent with ‘consisting of’ language is potentially vulnerable to design-around by adding an unlisted excipient. A patent with ‘comprising’ language is not. ‘Consisting essentially of’ falls between the two: the claim covers inventions with the listed elements plus elements that don’t materially affect the basic and novel characteristics of the invention—a phrase that has generated extensive litigation over what ‘materially affect’ means in specific contexts [33].
What a Patent Document Won’t Tell You
A granted patent is a snapshot of what was true at the time of grant. It doesn’t tell you whether the patent is currently in force—maintenance fees must be paid at 3.5, 7.5, and 11.5 years from grant for U.S. patents, and many patents lapse before their expiration date because the owner let them go. It doesn’t tell you whether ownership has changed. It doesn’t tell you whether the patent has been challenged, partially invalidated, or found not infringed in litigation.
All of this dynamic post-grant information is essential for accurate FTO and competitive intelligence work. Checking USPTO assignment records, PACER for district court litigation, PTAB for IPR proceedings, and the Orange Book for pharmaceutical-specific listing status are all required steps beyond reading the patent document itself. This is where specialized platforms like DrugPatentWatch provide the most direct time savings—consolidating these post-grant monitoring data streams into a single queryable environment rather than requiring sequential manual lookups across multiple government systems [18].
Part VIII: Applied Patent Intelligence—Three Scenarios Where It Pays Off
Scenario 1: The Generic Entry Decision
A generic company is evaluating whether to target a $4 billion branded drug. The composition of matter patent expires in four years, but the Orange Book lists eleven additional patents with varying expiration dates, and the brand company has been actively filing new applications in the relevant CPC classes.
This is fundamentally a patent intelligence question. The answer requires a systematic multi-step analysis.
First, map the full patent portfolio. Using DrugPatentWatch, pull the complete Orange Book listing and verify the expiration dates of all listed patents, checking specifically for PTE grants that may have extended individual patents beyond their nominal expiry. Identify any non-Orange Book patents that could still be asserted against a generic product—particularly method-of-use patents covering the primary indication.
Second, conduct an FTO analysis of the proposed generic formulation against all in-force patents. The goal is to determine whether a generic can be formulated to avoid the brand’s secondary patents. If the brand’s extended-release formulation patents use ‘comprising’ language, a different excipient combination may not constitute a viable design-around. If they use ‘consisting of’ language, the path may be clearer.
Third, run an invalidity search on the highest-risk secondary patents. A formulation patent filed in 2016 covering an extended-release version of a small molecule may have been anticipated by 2008 publications from academic groups who described similar excipient combinations for related compounds. Finding prior art that was not before the USPTO during prosecution is the generic company’s most powerful legal tool.
Fourth, pull the complete litigation history. If the brand company has already litigated this drug in Hatch-Waxman proceedings against earlier ANDA filers, the district court opinions and any PTAB decisions are primary research. Those proceedings have already generated judicial analysis of claim scope and prior art arguments that the current team can build on or avoid repeating [18].
None of this work is theoretical. It directly drives the business decision: whether to file the ANDA, which specific Paragraph IV certifications to include, which patents to challenge, and what litigation budget to reserve. A generic company that skips or shortcuts this process is making a multi-million-dollar bet without understanding the odds.
Scenario 2: The In-Licensing and White Space Search
A mid-sized biotech wants to build an oncology pipeline but lacks the resources to compete in the checkpoint inhibitor space, where the dominant PD-1/PD-L1 patents are held by Merck, Bristol Myers Squibb, and a handful of other major players with extensive secondary patent coverage. The scientific team has identified a specific mRNA regulatory mechanism in tumor microenvironments as therapeutically promising but underexplored.
They commission a patent landscape analysis. Using classification codes targeting mRNA-based therapeutics (A61K 31/7105 for RNA preparations), keyword searches incorporating the specific biological mechanism and target class, and assignee analysis across academic institutions and early-stage biotechs, they generate a filing activity visualization across ten years of patent history.
The landscape confirms that the PD-1/PD-L1 space is densely covered—a result that could have been predicted, but now the density is quantified and the specific claim boundaries are mapped. More importantly, the analysis reveals that the mRNA regulatory mechanism has generated substantial academic publication volume (indicating scientific community validation) but has attracted relatively sparse commercial patent filings. The ratio of publications to patents is high—an indicator that scientific knowledge has outpaced commercial IP exploitation.
A follow-on inventor search on the key publications identifies a research group at a major university whose work covers the most relevant aspects of the mechanism. A search of USPTO provisional applications reveals a recent provisional filing from the same group. Moving quickly to negotiate an exclusive license before the provisional publishes and attracts wider commercial attention becomes the business development priority [21].
This outcome depends entirely on the quality and timeliness of the patent intelligence. A team working from static, quarterly landscape reports would have arrived at the licensing negotiation three to six months late—potentially after the university’s technology transfer office had already talked to two competitors.
Scenario 3: M&A Due Diligence
A large pharmaceutical company is considering a $1.5 billion acquisition of a clinical-stage company whose entire value rests on a single Phase III oncology asset. Impressive Phase II data, but Phase III is the real test, and the deal is being structured as if the drug will succeed.
Before a term sheet is signed, the acquiring company’s business development and IP team conducts a systematic due diligence process on four tracks simultaneously.
Track one: independent validity analysis. The acquiring company’s IP team runs a comprehensive prior art search against the target’s core composition of matter patents—not just the searches that produced the claims examiner’s allowance, but an independent analysis designed to find art the examiner missed. The standard they’re using is ‘clear and convincing evidence’ of invalidity, because that’s the standard a future generic challenger will have to meet. If they can find prior art that would meet that standard, the patent’s durability is questionable.
Track two: FTO assessment of the full commercial program. The drug candidate’s proposed commercialization—specific formulation, manufacturing process, target indication, dosing regimen—is analyzed against all third-party patents. If there’s a blocking patent in the formulation space owned by a competitor who is unlikely to license on acceptable terms, the acquisition price needs to account for the cost of either litigation or design-around work.
Track three: portfolio lifecycle assessment. What is the target’s long-term exclusivity strategy? The composition of matter patent may expire in 2031, but has the company filed secondary patents on specific polymorphic forms, specific delivery devices, or specific dosing regimens that would extend protection? Has the company completed pediatric studies, locking in six additional months of exclusivity at the back end of the patent term? Has it pursued orphan designation for any related indication? These questions directly affect the revenue model beyond year seven.
Track four: litigation history and third-party risk assessment. Are there pending IPR petitions against any of the target’s patents? Have any of the Orange Book-listed patents been challenged by generic filers who haven’t yet become public? Are there any third-party patents that the target’s legal team considers potentially problematic but hasn’t disclosed? The answers to these questions can materially change the acquisition price or deal structure.
The IP due diligence output is not a legal opinion filed in a folder. It is a probability-weighted financial model that quantifies the likelihood that key patents will survive challenge, the estimated LOE date under different scenarios, and the cost range of resolving any identified FTO issues. That model is a primary input to the valuation [34].
Part IX: The FDA Orange Book—The Patent Intelligence Resource Most Professionals Underuse
What the Orange Book Is and Why It Matters
The FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, universally called the Orange Book, is the authoritative public database linking FDA-approved drug products to the patents and exclusivities that protect them. For pharmaceutical patent intelligence, it is the single most important regulatory reference resource. Yet many business professionals who work with drug market data every day have never opened it, relying instead on secondary sources that may lag or summarize it imperfectly.
The Orange Book has two primary functions. First, it provides the list of patents that an ANDA filer must certify against—the legally required disclosures that trigger the Hatch-Waxman patent dispute framework. Second, it lists the regulatory exclusivities that the FDA has granted for each approved product, providing the baseline for LOE calculations that is independent of the patent analysis.
Understanding how the Orange Book is populated—and where its limitations lie—is essential for using it accurately.
What Gets Listed in the Orange Book
Patent listing in the Orange Book is both a right and a strategic obligation. Innovator companies submit patents for listing; the FDA does not independently verify whether submitted patents actually meet the statutory listing criteria. The criteria are specific: a patent must claim the drug product (the active ingredient or an approved formulation) or a method of using the drug for an FDA-approved indication. Process patents, patents claiming metabolites, and patents claiming intermediates in synthesis are not eligible for listing, though companies have historically tested these boundaries.
The Orange Book lists, for each approved drug product:
- The NDA number and the specific drug product (dosage form and strength)
- Each listed patent number, expiration date (including any PTE), and use code
- Applicable regulatory exclusivities with their expiration dates
Use codes are a critical and often misunderstood feature. Each method-of-use patent listed in the Orange Book carries a use code—a brief description of the specific use claimed. Generic companies use these use codes to determine which Paragraph III or IV certifications to submit. A generic that will not be labeled for the patented use submits a Paragraph III certification (that it is not seeking approval for the patented indication). The accuracy and scope of use codes have been contested extensively in litigation, with some innovators submitting overly broad use codes that require certifications against method-of-use patents for uses the generic doesn’t even claim.
Orange Book Limitations That Matter for Patent Intelligence
Several important Orange Book limitations create risks for analysts who treat it as a complete picture of a drug’s patent protection.
First, not all relevant patents are listed. Patents that do not meet the statutory listing criteria—including patents on manufacturing processes, intermediate compounds, and chemical starting materials—may still be asserted in litigation even though they don’t appear in the Orange Book. A comprehensive FTO analysis must look beyond the Orange Book listings to the full patent portfolio of the innovator company, including non-listed patents that might be infringed by the generic product’s manufacturing process.
Second, Orange Book listings may be incorrect or outdated. Companies sometimes list patents that don’t actually meet the statutory criteria. The FDA’s review of listing submissions is limited in scope—the agency takes the position that it is not the appropriate body to adjudicate patent eligibility questions and largely accepts submissions at face value. Generic companies that believe a patent is improperly listed have a mechanism to challenge the listing (a ‘172 petition’), but the listing remains in force during the challenge.
Third, the Orange Book is a U.S.-only resource. It tells you nothing about the patent situation in other jurisdictions. The EU, Japan, and other major pharmaceutical markets each have their own regulatory and patent databases that must be checked separately. For global LOE analysis, the Orange Book is the starting point for the U.S. analysis and requires supplementation with jurisdiction-specific resources elsewhere.
DrugPatentWatch integrates Orange Book data with the broader patent universe, including non-listed patents that may still be commercially relevant, and connects the Orange Book data to ANDA filing history and litigation outcomes. This integration converts the raw Orange Book data into a more complete picture of a drug’s actual protection status than the Orange Book alone provides [14].
Reading the Orange Book for LOE Forecasting
The practical LOE calculation for a U.S. drug product using Orange Book data proceeds in two steps.
First, identify the latest patent expiration among all listed patents, accounting for any PTE grants. This is the patent-based LOE date—the date after which no listed patent would block generic entry.
Second, identify the latest exclusivity expiration among all applicable regulatory exclusivities. This is the exclusivity-based LOE date.
The true LOE date is the later of these two. For many drugs, particularly recently approved molecular entities with NCE exclusivity, the exclusivity-based LOE date is later than the patent-based LOE date for the first several years post-approval. For drugs with extensive patent thickets of secondary patents, the patent-based LOE date may extend years beyond the NCE exclusivity.
One common error in LOE modeling: treating pediatric exclusivity as if it only applies to the specific patent or exclusivity it was added to. Pediatric exclusivity attaches to all patents and exclusivities for the active moiety, including those that might otherwise expire before the pediatric grant. If a composition of matter patent expires in December 2028 and the company earns pediatric exclusivity, the effective expiry of that patent’s protection extends to June 2029. All downstream LOE calculations must reflect this addition.
A second common error: failing to distinguish between the expiry of patent protection and the expiry of data exclusivity. A generic company cannot reference the innovator’s clinical data during an applicable data exclusivity period regardless of patent status. But it can potentially conduct its own clinical trials and seek approval through a non-ANDA pathway if it has its own clinical data. For high-value products where a competitor might fund independent clinical work, this distinction can matter for LOE timing.
Part X: Global Patent Searching and the Jurisdictional Dimension
Why Geography Matters
A U.S. patent provides exclusivity only in the United States. A European patent provides exclusivity only in the specific European countries where it has been validated and maintained. A Japanese patent provides exclusivity only in Japan. For any drug with global commercial ambitions—or for any competitive analysis of a company with global operations—single-jurisdiction searching produces an incomplete and often misleading picture.
For competitive intelligence purposes, the global picture matters even when your immediate commercial focus is the U.S. market. A competitor who holds robust patent protection in the EU and Japan but has weak or expired U.S. coverage creates a fundamentally different strategic situation than one with the inverse profile. A drug that loses U.S. exclusivity in 2027 but maintains strong EU protection until 2031 will face a very different competitive dynamic in each market. Understanding the global patent family structure—how the same underlying invention has been prosecuted across national and regional offices—is essential for accurate competitive modeling.
The PCT System and Early-Warning Signals
The Patent Cooperation Treaty (PCT) provides a single-application pathway for seeking patent protection in over 150 contracting states. A PCT application, filed through WIPO, publishes 18 months after the earliest priority date—typically before the applicant has entered the national or regional phase in any individual country. This publication timing makes PCT applications a valuable early-warning signal for competitive intelligence.
A drug discovery program that files a U.S. provisional patent in January 2024 and a PCT application in January 2025 will have that PCT application published in July 2026—before most companies would expect to see clinical trial registrations for a discovery-stage program. Monitoring PCT applications in your therapeutic area provides visibility into international filing strategies roughly 18 months after they’re initiated and often two to four years before any clinical disclosure [35].
For business intelligence purposes, the PCT application also reveals the international filing strategy itself. The list of countries designated in a PCT application signals where the applicant intends to seek protection—and where they don’t. A company that seeks protection in North America, Europe, and Japan but omits China is revealing something about their commercial strategy or their assessment of Chinese patent enforcement. These signals accumulate into a picture of the competitor’s global market strategy.
Key International Patent Offices and Resources
Espacenet covers over 150 million documents from patent offices worldwide and includes a ‘Global Dossier’ feature that shows the prosecution history across patent families in different jurisdictions [22]. For competitive intelligence on how a competitor’s claims were shaped during examination—what was argued, what was narrowed, what was abandoned—the prosecution history is essential reading. Claims often narrow during prosecution in ways that create ‘prosecution history estoppel,’ limiting the patent owner’s ability to recapture scope through the doctrine of equivalents.
PATENTSCOPE is the primary resource for PCT application monitoring and supports full-text searching with machine translation across multiple languages [23].
For Japan, J-PlatPat provides searchable access to Japanese patent documents with machine translation tools. Japan has historically been a significant pharmaceutical patent market, and Japanese patent applications filed by both domestic and international pharmaceutical companies are an important part of any complete competitive landscape.
China’s CNIPA database has grown substantially in importance as China’s domestic pharmaceutical industry has scaled. Both international companies seeking Chinese market protection and Chinese innovators seeking global protection are generating significant patent activity. Any competitive analysis that omits Chinese patent filings is systematically incomplete.
Part X: AI, Automation, and the Evolution of Patent Intelligence
What AI Is Actually Doing in Patent Search Today
Artificial intelligence has moved from marketing language to operational deployment in commercial patent intelligence platforms. Being specific about what it changes—and what it doesn’t—is important for setting accurate expectations.
AI improves semantic search quality. Traditional keyword searching treats documents as bags of words: it finds documents containing specific strings. Semantic search understands conceptual meaning. A search for ‘kinase inhibitor for cancer treatment’ can retrieve documents describing ‘ATP-competitive enzyme blocking agent for tumor suppression’ without exact keyword matches. This improves recall—the proportion of relevant documents actually found in a search—which directly matters for completeness of FTO and invalidity analyses.
AI accelerates document classification and clustering. Large patent sets that previously required weeks of manual review to categorize can now be automatically clustered by technology sub-area, assignee, and claim type in hours. This compresses what’s feasible in landscape analysis: comprehensive landscape studies that took months now take weeks, making it economically viable to commission landscape analyses more frequently and at earlier stages of decision-making.
AI assists with claim element parsing. Some platforms now offer automated identification of the key elements within patent claims, facilitating faster creation of claim charts for FTO analysis. This doesn’t replace legal judgment about whether a specific product element meets a specific claim limitation, but it reduces the mechanical preparation work that precedes that judgment.
What AI is not doing reliably: strategic interpretation remains a human function. A tool can detect that a competitor filed 47 patent applications in a specific CPC class over 24 months, representing a 340% increase from the prior period, and flag this as a significant filing trend. What that trend means for your R&D strategy, your licensing posture, and your competitive timeline requires an analyst with domain expertise, access to clinical and commercial context, and genuine business judgment. The AI provides the data; the human provides the meaning [36].
The practical advice for teams evaluating AI patent tools is to test them on retrospective cases where the answer is already known. Run the AI-assisted search on a past FTO analysis where the relevant patents have already been identified, and measure recall and precision against that known-correct result set. Vendors claim high recall rates in general benchmarks; the relevant metric is recall on the specific type of patent that matters for your specific search problem—Markush claims in your chemical class, biologic sequences in your therapeutic target, method-of-use claims in your indication. General benchmarks don’t translate directly to performance on your specific use case.
Monitoring Programs: The Continuous Intelligence Practice
A patent landscape conducted once is a photograph. A monitoring program is a live feed. The difference in business value is substantial, particularly in fast-moving therapeutic areas where the competitive landscape evolves continuously.
Every major commercial platform, including DrugPatentWatch, offers configurable monitoring and alerting. You define the search parameters once—specific assignees, specific CPC classes, specific drug names, specific ANDA filing patterns—and receive alerts when new documents matching those parameters are published. For competitive intelligence, this means you learn about a competitor’s new patent filing in the week it publishes, not six months later when it appears in an analyst report or a competitor’s press release [18].
The events worth monitoring systematically in pharmaceutical patent intelligence include new patent publications by key competitors in your core therapeutic areas (both U.S. applications and PCT publications), new ANDA filings against your own products, Paragraph IV certifications filed against your Orange Book-listed patents, new IPR petitions filed at PTAB targeting patents in your space, and litigation outcomes in related cases that set precedents affecting your patent claims.
The organizational infrastructure required to extract value from monitoring is worth addressing directly. Monitoring alerts are only useful if reviewed, triaged, and routed to relevant decision-makers on a consistent schedule. The alert about a competitor’s new PCT application in your core therapeutic area needs to reach the head of business development and the IP lead within 24 hours of receipt if it’s going to drive a competitive response. Without defined owners, review cadences, and escalation paths, even excellent alerting systems generate noise that analysts learn to filter—which defeats the entire purpose.
The companies with the most effective competitive intelligence practices treat monitoring as an operational process, not a research project. Alerts feed a weekly or biweekly intelligence briefing that surfaces the highest-priority developments to business and R&D leadership with enough context to act. The analytical work that translates raw alerts into actionable intelligence is where investment in trained analysts pays off most directly. A monitoring program without the analytical infrastructure to process and synthesize its outputs is a data collection exercise, not an intelligence function.
Part XI: Building Patent Intelligence Into the Organization
Where the Function Should Live
There is no single right answer to the organizational question, but common failure modes are worth identifying. The most prevalent: housing patent intelligence entirely within the legal function, where output is optimized for legal review rather than business decision-making, and where the function’s relationship is primarily with the general counsel rather than R&D and business development leadership.
The more effective model places a dedicated patent intelligence capability directly adjacent to R&D strategy and business development. The team produces work product for lawyers and for business executives, but its primary orientation is toward informing business decisions rather than supporting legal proceedings. Access to business and R&D leaders—and the credibility to influence their decisions—requires that orientation to be genuine, not cosmetic.
In smaller companies, this function may be a single person with strong analytical and technical skills who works with external search firms on the more complex assignments. In larger companies, it may be a team of six to twelve analysts organized by therapeutic area or search type. The size matters less than the clarity of purpose and the quality of the connections between patent intelligence output and business decision processes.
The Skills Required
A patent intelligence team in a pharmaceutical company needs a combination of capabilities that doesn’t map neatly onto traditional job descriptions.
Scientific literacy is the foundation. A team member who can’t read a medicinal chemistry patent well enough to understand what the claims actually cover, or who can’t distinguish between a Markush claim covering a narrow structural series and one covering a broad chemical class, will produce unreliable analysis. This doesn’t require a PhD in every relevant discipline, but it does require genuine familiarity with pharmaceutical science at a level that most business school graduates don’t have.
Search methodology is a specialized skill, not a generic analytical capability. Constructing a comprehensive search strategy that combines classification, keyword, structure, and citation methods—and knowing when each is the right tool—takes training and experience. Many companies build this capability through internal training programs combined with relationships with specialized patent search firms for the most demanding assignments.
Database fluency means knowing which tool answers which question, and knowing the limits of each. Understanding that DrugPatentWatch integrates regulatory and litigation data in ways that general patent databases do not, or knowing when chemical structure searching is mandatory rather than complementary—this tool knowledge translates directly into the quality of the analysis produced.
Business acumen closes the loop. The best technical searcher who cannot translate findings into a business recommendation that a non-specialist can act on is delivering half the value of one who can. The goal is not a list of patents; it is a clear, evidence-based recommendation about what those patents mean for a specific business decision.
Conclusion: The Competitive Advantage Is in the Execution
Drug patent searching, done well, is one of the highest-ROI analytical practices available to pharmaceutical business professionals. It provides early visibility into competitor strategies, quantifies the durability of market exclusivity positions, surfaces licensing opportunities before they become widely known, and reduces the cost of patent-related surprises across the full product lifecycle.
Done poorly, or not done systematically, it creates structural blind spots that competitors exploit. The generic company that files first wins 180 days of exclusive generic marketing worth hundreds of millions of dollars. The innovator that catches a competitor’s PCT application early has time to adjust its filing strategy and accelerate its own program. The acquirer who misses a fatal FTO problem in due diligence writes a check that buys someone else’s litigation risk.
The tools available today are better than they have ever been. Public databases have improved dramatically. Specialized platforms like DrugPatentWatch have made pharmaceutical-specific intelligence—connecting patent data with regulatory, litigation, clinical, and commercial information—far more accessible than it was a decade ago. AI-assisted search is reducing the time required for complex landscape analyses from months to weeks. The technical barriers to good patent intelligence work have fallen.
The remaining barriers are organizational: the commitment to build the function properly, to invest in appropriate tools and trained personnel, and to integrate the output into business decisions from early development through commercialization. That is a leadership choice. The companies that make it gain a durable competitive advantage. The ones that don’t will continue to be surprised by the patent landscape—and surprised is not a good position to be in when hundreds of millions of dollars are at stake.
Key Takeaways
Drug patents are time-limited financial assets, not permanent monopolies. The 20-year statutory term is substantially consumed by development and regulatory timelines, leaving an average effective commercial exclusivity window of 7 to 10 years for most small molecules.
Loss of exclusivity is not the same as patent expiration. The true LOE date is the later of the primary patent expiration (accounting for Patent Term Extension) and applicable FDA regulatory exclusivity. Analyzing only patents produces systematic LOE underestimation that has direct financial consequences.
A drug’s IP position is a portfolio, not a single patent. Composition of matter, formulation, method-of-use, and process patents layer into a thicket that must be mapped completely for accurate competitive analysis. Mapping only the primary patent misses most of the strategic picture.
The type of search must match the business question. Patentability, FTO, invalidity, and landscape searches are fundamentally different in scope, methodology, and output. The wrong search type produces results that answer the wrong question, no matter how well it is executed.
Chemical structure and biologic sequence searching are not optional. Text-based keyword searches cannot identify compounds covered by Markush claims or biologic sequences disclosed without standardized Sequence Listing files. For life sciences FTO and patentability work, these specialized methods are required.
Specialized pharmaceutical intelligence platforms justify their cost through integration. Connecting patent data with Orange Book listings, regulatory exclusivities, ANDA filings, and litigation records in a single environment—as DrugPatentWatch does—eliminates the manual bridging that accounts for most of the time and most of the error in pharmaceutical-specific patent analysis.
Continuous monitoring outperforms periodic analysis. Structured alerting programs provide competitive visibility when events occur rather than months after the fact. The time advantage compounds across a product development lifecycle.
The strategic value comes from interpretation, not search volume. AI and improved databases reduce the mechanical work of patent searching. The competitive advantage accrues to teams who translate findings into specific, actionable business recommendations that non-specialists can act on.
FAQ
Q1: We use Google Patents for most of our drug patent searches. What are we actually missing?
Google Patents retrieves patent documents reasonably well for basic lookups. What it doesn’t provide is the pharmaceutical-specific integration layer that makes patent data actionable for business decisions. It doesn’t connect to the FDA Orange Book, so moving from a patent to its regulatory exclusivity status requires a separate manual step. It doesn’t track ANDA filings, Paragraph IV certifications, IPR proceedings, or district court litigation outcomes. Assignee data is inconsistent. Legal status information is often lagged or incorrect for non-U.S. documents. For casual lookups, Google Patents is adequate. For business questions like ‘What is the true LOE date for this drug and what is its litigation risk profile?’—questions that require synthesizing patent, regulatory, and litigation data simultaneously—Google Patents requires extensive manual bridging across multiple government systems. Platforms built specifically for pharmaceutical intelligence, DrugPatentWatch being the most purpose-built example, integrate these data sources in ways that convert a multi-hour manual task into a ten-minute query. At the decision-making level that pharmaceutical executives operate at, the accuracy improvement from curated integrated data matters as much as the time savings.
Q2: How often should we update FTO analyses for assets in active development?
At minimum, at each major development milestone: IND filing, Phase II initiation, Phase III initiation, and NDA or BLA submission. You should also update whenever the product formulation or manufacturing process changes materially, whenever a competitor files a relevant new patent that appears in your monitoring alerts, whenever a major litigation outcome occurs in a related product space, and whenever your geographic target markets expand. The pharmaceutical patent landscape changes continuously: new patents issue, pending applications publish, existing patents lapse for non-payment of fees, and PTAB decisions change the validity status of key patents. An FTO analysis from 18 months ago may be materially outdated. Treating it as a living document updated at structured intervals, rather than a historical record reviewed once, is the difference between active risk management and false confidence.
Q3: Our general counsel says we have a ‘strong’ patent portfolio, but we’ve never done a comprehensive landscape analysis of the competitive space. How much should we trust that assessment?
A general counsel’s assessment of portfolio strength typically reflects the internal quality of your own patents: well-drafted claims, clean prosecution histories, appropriate claim scope. It usually doesn’t reflect a systematic assessment of the competitive environment—how your portfolio compares to competitors’, whether there are third-party blocking patents in your freedom-to-operate path, or whether competitors have filed improvement patents that design around your protection. These are different questions, and they require different analyses. A portfolio can be technically excellent by internal metrics while being commercially vulnerable because competitors have developed non-infringing alternatives or because an FTO analysis would reveal third-party blocking patents in critical formulation or method-of-use areas. If you’ve never done a comprehensive landscape study, you have an incomplete picture of both your offensive strength and your defensive gaps. The landscape analysis is what converts an internal portfolio assessment into a competitive competitive intelligence picture.
Q4: We’re about to sign a term sheet for an in-licensing deal. The licensor’s IP team says their patents are ‘rock solid.’ What should our independent due diligence actually cover?
The licensor’s self-assessment is not due diligence—it’s advocacy. Independent due diligence should cover four areas. First, validity: run an independent prior art search against the core composition of matter patents and the most commercially important secondary patents, looking for art the USPTO examiner didn’t consider. Second, FTO: assess whether the licensed product’s commercial development path would infringe any third-party patents. Licensing rights to use someone’s IP doesn’t automatically clear third-party blocking patents—those are separate rights requiring separate analysis. Third, portfolio completeness: verify that the specific patents being licensed actually cover the product and indication you plan to develop, check for gaps in geographic coverage relevant to your target markets, and assess whether the portfolio has the secondary patent layers needed for lifecycle management. Fourth, litigation and challenge history: check whether any patents in the portfolio have been challenged at PTAB or in district court, and what the outcomes were. Prior unsuccessful challenges can reveal vulnerabilities that future challengers may exploit more effectively with better prior art or stronger legal arguments.
Q5: How is the search challenge different for biologics versus small molecules?
The strategic framework applies equally—patentability, FTO, invalidity, and landscape searches are all relevant for both. The technical execution differs significantly. For small molecules, chemical structure searching is the foundational non-negotiable, supplementing keyword and classification searches. For biologics, biologic sequence searching plays the equivalent role. The databases required differ: CAS STNext and Questel Orbit Chemistry for small molecule structures; Clarivate Derwent SequenceBase and STN for biologic sequences. The patent types that matter most differ too: for biologics, manufacturing process patents are often more durable competitive barriers than compound patents, because the process defines the product in ways with no small-molecule analog, and biosimilar developers face extensive analytical work to demonstrate comparability without access to the innovator’s protected process. The regulatory exclusivity baseline also differs: biologics receive 12 years of reference product exclusivity under the BPCIA, compared to five years of NCE exclusivity for new small molecule drugs under Hatch-Waxman. Any competitive analysis of a biologic that ignores this extended exclusivity baseline will systematically underestimate the protected period and overestimate the timing of biosimilar competition.
References
[1] U.S. Patent and Trademark Office. (2024, October 28). Understanding the statute on patent term extension. IPWatchdog. https://ipwatchdog.com/2024/10/28/understanding-statute-patent-term-extension/id=182598/
[2] Simson Pharma Limited. (n.d.). Why are patents important to drug development? https://www.simsonpharma.com/blog-details/why-are-patents-important-to-drug-development
[3] Congressional Research Service. (2021). The role of patents and regulatory exclusivities in drug pricing (R46679). https://www.congress.gov/crs-product/R46679
[4] GeneOnline News. (n.d.). Drug patent searching crucial for pharma companies to understand patent landscape and avoid infringement. https://www.geneonline.com/drug-patent-searching-crucial-for-pharma-companies-to-understand-patent-landscape-and-avoid-infringement/
[5] O’Brien Patents. (n.d.). Types of pharmaceutical patents. https://www.obrienpatents.com/types-pharmaceutical-patents/
[6] Patsnap Synapse. (n.d.). What are the types of pharmaceutical patents? https://synapse.patsnap.com/blog/what-are-the-types-of-pharmaceutical-patents
[7] AJMC. (n.d.). How drug life-cycle management patent strategies may impact formulary management. https://www.ajmc.com/view/a636-article
[8] DrugPatentWatch. (n.d.). The value of method of use patent claims in protecting your therapeutic assets. https://www.drugpatentwatch.com/blog/the-value-of-method-of-use-patent-claims-in-protecting-your-therapeutic-assets/
[9] Minesoft. (n.d.). The role of patents in the pharmaceutical sector. https://minesoft.com/the-role-of-patents-in-the-pharmaceutical-sector/
[10] Number Analytics. (n.d.). Pharmaceutical patents: A comprehensive guide. https://www.numberanalytics.com/blog/ultimate-guide-pharmaceutical-patents
[11] News-Medical.net. (n.d.). Drug patents and generic pharmaceutical drugs. https://www.news-medical.net/health/Drug-Patents-and-Generics.aspx
[12] DrugPatentWatch. (n.d.). Drug patent life: The complete guide to pharmaceutical patent duration and market exclusivity. https://www.drugpatentwatch.com/blog/how-long-do-drug-patents-last/
[13] Wikipedia. (n.d.). Drug Price Competition and Patent Term Restoration Act. https://en.wikipedia.org/wiki/Drug_Price_Competition_and_Patent_Term_Restoration_Act
[14] DrugPatentWatch. (n.d.). The basics of drug patent searching. https://www.drugpatentwatch.com/blog/the-basics-of-drug-patent-searching/
[15] U.S. Food and Drug Administration. (n.d.). Frequently asked questions on patents and exclusivity. https://www.fda.gov/drugs/development-approval-process-drugs/frequently-asked-questions-patents-and-exclusivity
[16] Merck. (2023, December). Patent term extensions. https://www.merck.com/wp-content/uploads/sites/124/2023/12/Patent-Term-Extensions_MRK_DEC11.pdf
[17] Sterne Kessler. (n.d.). Patent term extension. https://www.sternekessler.com/news-insights/insights/patent-term-extension/
[18] DrugPatentWatch. (n.d.). Drug patent litigation and Patent Trial and Appeal Board (PTAB) cases. https://www.drugpatentwatch.com/p/litigation/
[19] Number Analytics. (n.d.). Pharmaceutical patent litigation guide. https://www.numberanalytics.com/blog/ultimate-guide-pharmaceutical-patent-litigation
[20] DrugPatentWatch. (n.d.). How to conduct a drug patent FTO search. https://www.drugpatentwatch.com/blog/how-to-conduct-a-drug-patent-fto-search/
[21] Dilworth IP. (n.d.). Patent landscape analysis: A vital strategy. https://www.dilworthip.com/resources/news/patent-landscape-analysis/
[22] European Patent Office. (n.d.). Espacenet – patent search. https://www.epo.org/en/searching-for-patents/technical/espacenet
[23] World Intellectual Property Organization. (n.d.). PATENTSCOPE. https://www.wipo.int/en/web/patentscope
[24] McKinsey & Company, as cited in DrugPatentWatch. (n.d.). The basics of drug patent searching. https://www.drugpatentwatch.com/blog/the-basics-of-drug-patent-searching/
[25] World Intellectual Property Organization. (n.d.). International Patent Classification (IPC). https://www.wipo.int/en/web/classification-ipc
[26] PatentPC. (n.d.). How to use citation analysis in patent searches. https://patentpc.com/blog/how-to-use-citation-analysis-in-patent-searches
[27] PatentPC. (n.d.). Advanced patent search techniques for experienced researchers. https://patentpc.com/blog/advanced-patent-search-techniques-for-experienced-researchers
[28] CAS. (n.d.). Chemical structure patent search. https://help.patentexplorer.cas.org/cur/Searching_in_Patent_Explorer/Patent_Search/Find_Patents_by_Chemical_Structure.htm
[29] Effectual Services. (n.d.). Biological sequence search. https://www.effectualservices.com/patent-services/patent-searches/bio-sequence-search/
[30] Clarivate. (n.d.). Finding biological sequences in patents without sequence listing. https://clarivate.com/intellectual-property/blog/finding-patented-biological-sequences-for-patents-without-a-sequence-listing/
[31] U.S. Patent and Trademark Office. (n.d.). 2111 – Claim interpretation; broadest reasonable interpretation. https://www.uspto.gov/web/offices/pac/mpep/s2111.html
[32] Minesoft. (n.d.). An expert guide to understanding patent claims. https://minesoft.com/an-expert-guide-to-understanding-patent-claims/
[33] Henry Patent Law Firm. (n.d.). Understanding the parts of a patent. https://henry.law/blog/the-anatomy-of-a-patent/
[34] DrugPatentWatch. (n.d.). Leveraging drug patent data for strategic investment decisions: A comprehensive analysis. https://www.drugpatentwatch.com/blog/leveraging-drug-patent-data-for-strategic-investment-decisions-a-comprehensive-analysis/
[35] World Intellectual Property Organization. (n.d.). The PCT applicant’s guide. https://www.wipo.int/en/web/pct-system/guide/index
[36] Dennemeyer. (n.d.). The secret to IP success: AI-driven patent search and analysis. https://www.dennemeyer.com/ip-blog/news/the-secret-to-ip-success-ai-driven-patent-search-and-analysis/