Part I: The Strategic and Financial Imperative of FTO
The $2.6 Billion Bet and the One Analysis That Protects It
Developing a new drug costs, on average, $2.6 billion and consumes ten to fifteen years of a company’s resources, according to the widely cited Tufts Center for the Study of Drug Development analysis. Some estimates, when accounting for the full cost of capital and the cascade of failed candidates required to generate one approval, push that figure past $5 billion. In 2023, global pharmaceutical R&D spending crossed $250 billion. These numbers frame the only relevant financial question about a Freedom-to-Operate (FTO) analysis: what fraction of a $2.6 billion investment is it rational to spend confirming that the asset can actually reach patients without triggering a patent injunction?
The answer, calculated against the median patent damages award in U.S. pharmaceutical cases of $8.7 million in 2023 — and against the top awards, which have exceeded $2 billion — is that FTO spending at any figure between $50,000 and $500,000 is one of the better-returning risk-mitigation expenditures in all of corporate finance. Yet IP teams routinely fight with CFOs over FTO budgets. That argument gets easier once willful infringement enters the discussion.
Under 35 U.S.C. § 284, when a court finds willful infringement — meaning the infringer had actual knowledge of the patent and continued infringing with reckless disregard for the patent holder’s rights — it can award up to three times actual damages. A documented, timely, written FTO opinion from qualified patent counsel is the primary affirmative defense against that finding. No FTO opinion: treble damages are live. A well-reasoned FTO opinion issued before commercialization: treble damages are effectively neutralized. That calculus alone justifies treating FTO as a line item in every stage-gate budget from IND-enabling studies forward.
The pharmaceutical industry accounted for 25% of all U.S. patent damages awarded in 2023. Patent litigation is not an edge case in this business; it is a structural feature of how incumbents defend market share against biosimilar sponsors, generics filers, and competing innovators.
Key Takeaways — Part I
FTO costs between 0.002% and 0.02% of the R&D investment it protects. Without a written FTO opinion, treble damages under 35 U.S.C. § 284 remain available to a plaintiff. The pharmaceutical sector routinely generates the largest patent damage awards in U.S. federal courts, making FTO due diligence an economic necessity, not a legal formality.
Investment Strategy — Part I
For institutional investors evaluating a pre-commercial biotech, the absence of a documented, stage-appropriate FTO analysis is a red flag equivalent to missing clinical data. Ask the target to produce the written opinion. If they cannot, haircut your valuation to account for unquantified IP risk, and require a representation and warranty in any term sheet that FTO clearance will be obtained before closing or disbursement.
Part II: FTO vs. Patentability vs. Validity — The Framework That Saves Deals
The Distinction That Kills Startups
The single most dangerous misconception in biopharmaceutical IP strategy is treating patentability and freedom to operate as equivalent. They are not. A company can own a valid, issued patent on its drug and simultaneously have zero legal right to sell that drug. This is not a theoretical edge case; it describes a significant proportion of second-generation formulation and combination-product programs.
A patent grants a negative right: the right to exclude others from making, using, or selling the claimed invention. It grants nothing affirmative. The patent owner can stop others from copying their invention, but if practicing that invention requires using a broader, earlier patent owned by someone else, the patent owner still needs a license or clearance to sell their own product.
The practical example: a company develops a novel extended-release matrix for a known GLP-1 receptor agonist, files a patent application on the formulation, and receives issuance. The underlying GLP-1 peptide compound is covered by a separate, in-force patent held by a competitor with a decade of remaining term. The formulation patent is valid and enforceable. The company cannot sell the product without infringing the compound patent. Patentability confirmed; freedom to operate: zero.
Three Searches, Three Questions
IP professionals must keep three distinct search types — FTO, patentability, and validity — operationally separate, because conflating them produces the wrong intelligence at the wrong time.
An FTO search asks: ‘Can we commercialize this specific product in these specific markets without infringing in-force patent claims?’ It looks only at active patents and published applications in the target commercial jurisdictions. Pre-grant applications matter because they can issue during development and cover a product that enters the market years later. The temporal scope is the last twenty to twenty-five years, because older patents have expired and pose no infringement risk.
A patentability search asks: ‘Is this invention novel and non-obvious? Can we get a patent on it?’ It covers all public disclosures worldwide regardless of age or legal status. A 1987 journal article describing a structurally similar compound can destroy novelty just as effectively as a 2023 patent. The goal is finding prior art that the USPTO or EPO would use to reject claims, so the search must be exhaustive across literature, conference proceedings, and patent databases.
A validity search asks: ‘Is this blocking patent actually enforceable? Can we find prior art to kill its claims?’ The target is a specific, identified patent. The search reconstructs the prior art landscape as it existed before that patent’s priority date, hunting for ‘killer prior art’ that would render the claims anticipated or obvious. This search type is triggered by the FTO analysis, once a high-risk blocking patent has been identified.
Table 1: FTO vs. Patentability vs. Validity Search
| Feature | FTO Search | Patentability Search | Validity Search |
|---|---|---|---|
| Core Question | ‘Can I sell this without being sued?’ | ‘Can I patent this?’ | ‘Is this blocking patent valid?’ |
| Scope | In-force patents and published apps in commercial jurisdictions | All public disclosures worldwide, any age | All prior art before the target patent’s priority date |
| Key Documents | Active claim language only | Full disclosure of any prior art reference | Full disclosure of any prior art reference |
| Timeframe | Last 20-25 years (since expired patents are irrelevant) | No time limit | Any date before the blocking patent’s priority date |
| Primary Focus | Infringement risk against a defined product or process | Novelty and non-obviousness of a new invention | Finding art to invalidate specific claims of a specific patent |
| Typical Trigger | Pre-commercialization, M&A due diligence, new market entry | Early R&D, before filing a patent application | After FTO identifies a high-risk blocker; in response to litigation |
Key Takeaways — Part II
Owning a patent on a drug does not authorize selling that drug. Three functionally distinct search types address three distinct questions, and deploying them in the wrong context produces useless intelligence. Every stage-gate review should specify which search type is required and why.
Part III: The Hatch-Waxman Machine — Orange Book, Paragraph IV, and the Generic FTO Battlefield
Why Hatch-Waxman Is Inseparable from Small-Molecule FTO
For any company developing a small-molecule drug in the United States — whether as an originator, a generic applicant, or an acquirer of either — the Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman) is the operating system of the IP landscape. A complete FTO analysis for a U.S. small-molecule asset cannot be done without understanding the Orange Book, the 30-month stay mechanism, the Paragraph IV pathway, and patent term extensions (PTEs).
The Orange Book, formally the FDA’s ‘Approved Drug Products with Therapeutic Equivalence Evaluations,’ lists every patent covering an approved NDA drug that an NDA holder has certified to FDA. These patents fall into two functional categories: drug substance patents covering the active ingredient, and drug product patents covering formulations, delivery devices, and dosage forms. Method-of-use patents covering therapeutic indications are also listable. As of 2024, the Orange Book lists more than 17,000 active patent-NDA associations across roughly 4,500 drug products.
An originator company’s primary competitive weapon under Hatch-Waxman is the 30-month stay. When a generic applicant files an ANDA with a Paragraph IV certification — asserting that a listed Orange Book patent is invalid, unenforceable, or not infringed by the generic formulation — the originator has 45 days to sue for infringement. Filing that lawsuit triggers an automatic 30-month stay of FDA approval. The generic cannot launch while the stay is active. For a drug generating $1 billion or more per year in revenue, a 30-month stay is worth roughly $2.5 billion in protected sales, assuming the litigation is ultimately settled or decided in the originator’s favor. This explains why originators fight hard to maintain maximum Orange Book listings and why patent term extension strategy is inseparable from launch strategy.
Patent Term Extension: The Mechanics of Regulatory Capital Recovery
Under 35 U.S.C. § 156, an NDA holder can apply for a patent term extension to restore some of the patent life lost during the regulatory review period. The calculation: the PTE equals the sum of half the IND testing period plus the full NDA review period, subject to a ceiling of five years. The extended patent term cannot extend more than fourteen years beyond the product’s date of first commercial marketing. In practice, most FDA-approved drugs receive PTEs of two to three years.
European equivalents, called Supplementary Protection Certificates (SPCs), operate under similar logic but provide up to five years of additional exclusivity beyond the basic patent term, with a further six-month pediatric extension available. For a drug with $3 billion in annual European sales, each additional year of SPC protection is worth approximately $3 billion in revenue, net of manufacturing costs. Getting the SPC application right — identifying the correct ‘basic patent’ and filing within six months of the first EU marketing authorization — is therefore a multi-billion-dollar administrative task.
An FTO analysis for any asset approaching late-stage development must map the PTE and SPC landscape of potential blocking patents. A patent that appears to expire in three years might actually have five to eight years of remaining enforceability when PTE and SPC terms are accounted for. Failing to model these extensions produces a materially wrong picture of when a product can enter the market free of infringement risk.
The Paragraph IV Landscape as Competitive Intelligence
The public record of Paragraph IV certifications is one of the most underutilized sources of competitive intelligence in pharmaceutical IP. Every time a generic company files a Paragraph IV certification against an Orange Book patent, it is making a public declaration that it believes that patent is invalid, unenforceable, or not infringed by the generic version of the drug. That declaration, which becomes part of the public ANDA record once litigation commences, reveals which Orange Book patents the generic industry considers weak and which it is avoiding.
A company conducting FTO on a drug in a therapeutic class where multiple generic Paragraph IV filings have already occurred should pull the complaint and counterclaim filings from the associated district court dockets. Those documents contain detailed claim charts prepared by the generic applicant’s attorneys mapping every element of every Orange Book claim against the generic product. These claim charts are, in effect, free invalidity analyses conducted by highly motivated litigants who have already identified the weakest arguments against the blocking patents. They can meaningfully accelerate the FTO analysis and sharpen the validity challenge strategy.
Key Takeaways — Part III
The Orange Book is the first stop for any U.S. small-molecule FTO analysis. PTE and SPC calculations can add years to a blocking patent’s effective life that a raw expiration date will miss. Public Paragraph IV litigation records contain plaintiff-funded claim charts that are directly useful to anyone conducting an invalidity or design-around analysis.
Investment Strategy — Part III
When analyzing a branded pharmaceutical company with a major product approaching generic challenge, model the Orange Book patent strength quantitatively. Specifically: how many listed patents will require separate Paragraph IV certifications, what is the litigation track record of each patent family (IPR history, prior litigation outcomes), and what is the median time-to-resolution of prior Paragraph IV suits by those same patents. That analysis produces a probabilistic launch window for generics that is substantially more precise than simply reading the last Orange Book expiration date.
Part IV: The BPCIA’s ‘Patent Dance’ and Biosimilar Interchangeability FTO
The Purple Book and the 12-Year Clock
The Biologics Price Competition and Innovation Act of 2010 (BPCIA) created a regulatory and IP framework for biosimilars that is structurally analogous to Hatch-Waxman but operationally far more complex. The Purple Book — the FDA’s equivalent of the Orange Book for reference biological products — lists the exclusivity dates for approved biologics, including the twelve-year data exclusivity period that bars FDA from approving a biosimilar application until twelve years after the reference product’s first U.S. licensure.
That twelve-year clock is a core IP asset. For a biologic like Humira, which generated over $21 billion in global revenue at its peak, twelve years of data exclusivity was worth more on a discounted cash flow basis than most of the secondary patents in AbbVie’s 247-patent portfolio, because exclusivity is not litigated — it runs automatically.
An FTO analysis for a biosimilar program must therefore begin with the Purple Book to confirm the data exclusivity status of the reference product. Launching before the twelve-year exclusivity window closes triggers automatic FDA rejection of the 351(k) application. Launching after the window but before all asserted patents are cleared triggers BPCIA litigation.
The Patent Dance: Information Exchange as Litigation Strategy
The BPCIA’s patent exchange mechanism, colloquially known as the ‘patent dance,’ governs how biosimilar applicants and reference product sponsors identify and resolve patent disputes before biosimilar launch. It is simultaneously a transparency mechanism and a strategic battleground.
Under the dance, after FDA accepts the 351(k) application, the biosimilar applicant provides a detailed description of the manufacturing process for the proposed biosimilar. The reference product sponsor then has sixty days to identify a list of patents it believes the biosimilar infringes and would be willing to license. The biosimilar applicant then provides its non-infringement and invalidity positions. This information exchange process can span months and generates the formal patent lists that define the scope of any subsequent litigation.
The strategic asymmetry is significant. The reference product sponsor controls the first patent list and can include its entire portfolio. The biosimilar applicant’s non-infringement and invalidity contentions, filed under the dance, are treated as admissions in subsequent litigation if they are inconsistent with the applicant’s later litigation positions. Companies that treat the dance as a pro forma information exchange rather than a litigation-shaping exercise routinely find that their early contentions constrain their later legal arguments.
For FTO purposes, a biosimilar applicant conducting analysis before filing the 351(k) application needs to model not just which patents will appear on the reference product sponsor’s first list, but which patents might appear on the supplemental list — the second list the sponsor can provide after reviewing the manufacturing process description. Manufacturing process patents, cell line patents, and formulation patents that were not apparent from published literature often surface on the supplemental list because the manufacturing description triggers new sponsor contentions. An FTO analysis that does not anticipate the supplemental list underestimates the patent exposure of the biosimilar program by potentially dozens of patents.
Biosimilar Interchangeability and Its FTO Implications
FDA’s designation of a biosimilar as ‘interchangeable’ — meaning a pharmacist can substitute it for the reference biologic without prescriber intervention — confers substantial commercial value. The first biosimilar approved as interchangeable for a given reference product gets a one-year exclusivity period on that designation. But interchangeability also creates additional IP exposure.
The clinical and analytical data required to demonstrate that a biosimilar can be repeatedly switched with the reference product often requires use of proprietary analytical methods patented by the reference product sponsor. Demonstrating immunogenicity equivalence across switching conditions can require specific assay protocols that are themselves IP-protected. A biosimilar company pursuing interchangeability designation needs a separate FTO analysis on the analytical methodologies required to generate the switching study data, not just on the final drug product itself.
Key Takeaways — Part IV
BPCIA’s twelve-year data exclusivity is frequently worth more than any single secondary patent in a reference product sponsor’s portfolio. The patent dance is litigation strategy, not paperwork — contentions filed under the dance bind the applicant in subsequent proceedings. Biosimilar interchangeability programs require FTO analysis on switching-study methodologies, not just on the biosimilar drug product.
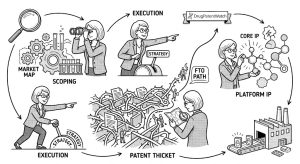
Part V: A Step-by-Step FTO Execution Framework
Phase 1: Scoping — The Technology Document
Every FTO analysis lives or dies by its scoping. A poorly defined scope produces one of two equally dangerous outcomes: the analysis is too broad and burns budget analyzing patents with no relevance to the actual product, or it is too narrow and misses a blocking patent on a feature the analyst assumed was standard rather than proprietary.
The Technology Document is the formal instrument that prevents both failures. It is a technical and strategic blueprint prepared collaboratively by R&D, legal, and business development before the first patent database is queried. For a small-molecule drug, the document should capture the active ingredient’s chemical structure (including stereochemistry and salt forms), the proposed formulation including all excipients and their functional roles, the manufacturing synthetic route, all intended therapeutic indications, and the proposed dosing regimen. Each element is a potential claim target, and each must be separately analyzed in the FTO.
For a biologic, the document expands considerably. It must specify the amino acid sequence, the gene construct, the host cell line, the fermentation and purification process at a unit operation level, post-translational modification profile (especially glycosylation), the formulation buffer and stabilizers, and the delivery device if applicable. A mAb program that fails to document its Protein A affinity chromatography purification step will miss an entire class of potential blocking patents on downstream processing.
The Technology Document also captures what the product is not — features that the company has already confirmed it will not use. This ‘negative specification’ is valuable because it can quickly clear large blocks of patents covering approaches the product does not take.
Phase 2: Timing — The Iterative FTO Calendar
FTO is not a pre-launch event. It is a continuous process mapped to product development milestones. Companies that wait until Phase 3 to conduct their first serious FTO analysis have already committed the majority of their R&D capital against a patent landscape they have not systematically analyzed. The ability to redesign a compound or reformulation is highest and cheapest in discovery. It is lowest and most expensive in Phase 3.
A rational FTO calendar for a small-molecule program runs roughly as follows. At target identification, conduct a high-level landscape analysis of the therapeutic area to flag obvious blocking patents on the mechanism of action. At lead optimization, commission a focused FTO on the lead compound class, particularly on the scaffold and key pharmacophores. At IND-enabling studies, conduct a formal written FTO on the specific IND candidate. Update the analysis at Phase 2 entry, when clinical proof-of-concept data drives commitment to a specific dosing regimen and formulation. Commission a comprehensive pre-Phase 3 FTO covering the final intended commercial product. Conduct a final FTO update immediately before NDA filing.
Each of these analyses is not a repeat of the prior one. It is an update that adds coverage of newly published applications (which publish eighteen months after filing), newly issued patents, and new product features added during development. The cumulative investment across all six stages for a typical small-molecule program runs between $200,000 and $400,000 — less than 0.02% of the cost of Phase 3 alone.
Phase 3: Jurisdictional Strategy — Where Rights Run and Where They Don’t
Patent rights are territorial. A USPTO-issued patent covers the United States. It has no effect in Germany, Japan, or Brazil. A company planning a global launch must conduct FTO in each commercially significant territory, and the scope of that analysis must account for where the product will be manufactured, where it will be sold, and where key raw materials and intermediates will be produced.
Manufacturing jurisdiction is often overlooked. If a biosimilar’s active ingredient is produced at a contract manufacturing organization in South Korea for sale in the European Union, the FTO analysis must cover both South Korea (manufacturing acts) and the EU (importation and sale). The ‘safe harbor’ exemption for research activities, which exists in U.S. law under 35 U.S.C. § 271(e)(1) and in EU law under Bolar exemption provisions, does not protect commercial manufacturing runs.
The practical jurisdictional priority order for most programs entering global commercialization is: United States first, EU (particularly Germany, France, and the United Kingdom) second, Japan third, China fourth, and other markets as the commercial opportunity justifies. India requires separate analysis because Section 3(d) of the Patents Act creates a materially different patentability standard for secondary pharmaceutical patents, which in turn changes the blocking patent landscape dramatically. A secondary formulation patent that creates a five-year blocking position in the United States may have no corresponding protection in India.
Phase 4: Multi-Pronged Search Strategy
Keyword searching alone will miss blocking patents. Inventors and their attorneys use proprietary terminology, coined terms, and functional language that does not map cleanly to standard chemical nomenclature or therapeutic descriptors. A robust FTO search requires at least four distinct search methodologies run in parallel and cross-referenced.
The first is keyword searching across titles, abstracts, and claims, using a comprehensive synonym set developed with the product’s R&D team. The second is classification-based searching using Cooperative Patent Classification (CPC) codes specific to the compound class, formulation type, and therapeutic indication. CPC codes capture patents regardless of the language used in the claims, which is particularly important for older patents filed before standardized nomenclature.
The third methodology is assignee and inventor searching. Key competitors’ patent portfolios should be systematically reviewed, not just searched for the target compound. A competitor may have filed relevant blocking patents under the names of subsidiary entities, university collaborators, or individual inventors, all of which require direct portfolio queries. The fourth methodology is citation-based expansion. Once a highly relevant patent is identified, its backward citations reveal the prior art the patent examiner considered, and its forward citations reveal all later patents that claim a relationship to it. Patents with high forward citation counts are statistically five times more likely to be litigated, making them priority targets for detailed analysis.
For biologics and CGTs, a fifth methodology is mandatory: sequence searching. This is covered in detail in Part VIII.
Phase 5: Database Selection — Free vs. Commercial vs. Specialized
The selection of search platforms has a direct impact on search comprehensiveness. Free platforms (USPTO Patent Full-Text and Image Database, Espacenet, PATENTSCOPE) provide broad global coverage and are appropriate for initial surveys and citation tracking. They are not sufficient for a high-stakes, late-stage FTO analysis because their data normalization, assignee name standardization, and legal status tracking are significantly inferior to commercial alternatives.
Commercial platforms (Questel Orbit, LexisNexis TotalPatent One, Clarivate Derwent Innovation, PatSeer) offer cleaner data, faster updates, more powerful Boolean and semantic search capability, and integrated collaboration tools for multi-analyst projects. LexisNexis TotalPatent One covers more than 155 million patent documents with daily updates and is generally regarded as the most comprehensive source for pharmaceutical FTO. Questel Orbit has particular strengths in claim highlighting and annotated claim charts that accelerate the analysis phase.
Specialized pharmaceutical intelligence platforms add a layer of business and legal context that pure patent databases cannot provide. DrugPatentWatch integrates patent data with FDA drug approvals, regulatory exclusivity timelines, Orange Book and Purple Book listings, litigation dockets, and generic and biosimilar entry forecasts. For an FTO analyst trying to understand not just whether a patent covers a target drug but when it expires, whether it has been challenged, and what the competitive entry timeline looks like, that integration is operationally essential. It transforms a technical patent search into a commercial intelligence product.
Table 2: Patent Database Comparison for Biopharma FTO
| Database | Provider | Biopharma-Specific Strengths | Best Use Case |
|---|---|---|---|
| Espacenet | EPO | 110M+ documents, legal status, family viewer | International initial searches |
| PATENTSCOPE | WIPO | PCT applications, cross-lingual search, chemical structure search | PCT application search, international families |
| Google Patents | Fast, full-text search, Lens integration | Quick preliminary screens | |
| DrugPatentWatch | DrugPatentWatch, Inc. | Drug-to-patent linking, Orange/Purple Book, litigation tracking, exclusivity modeling | Competitive intelligence, M&A diligence, lifecycle management |
| Questel Orbit | Questel | Advanced analytics, claim highlighting, automated charts | Professional FTO analysis, team projects |
| LexisNexis TotalPatent One | LexisNexis | 155M+ documents, daily updates, normalized assignees | Comprehensive global FTO for high-stakes assets |
| Derwent Innovation | Clarivate | Derwent abstracts with enhanced chemistry indexing | Chemistry-focused FTO, structure searching |
Phase 6: Claim Analysis — The Only Thing That Determines Infringement
Infringement is determined by claims, and only by claims. This sounds obvious, but the ‘Feature Fallacy’ — reading a patent specification, finding a described embodiment that resembles the product under analysis, and concluding infringement risk exists — is one of the most common errors made by non-attorney technical staff conducting preliminary FTO screens. A feature described in the specification but not recited in any independent claim creates no infringement liability.
The correct process requires mapping each element of each independent claim in a patent against the features of the product under analysis. If every element of a claim is present in the product, literal infringement exists for that claim. If even one element is absent, there is no literal infringement, though the doctrine of equivalents requires a separate analysis.
Three transitional phrases in claim drafting have specific, legally defined scope implications that FTO analysts must apply consistently. ‘Comprising’ is open-ended: a product that has all listed elements plus additional elements still infringes. ‘Consisting of’ is closed: a product with additional unlisted elements does not infringe. ‘Consisting essentially of’ is intermediate: additional elements are permissible only if they do not materially affect the invention’s basic and novel characteristics. For pharmaceutical formulation claims, this distinction is dispositive: a ‘comprising’ claim covering a formulation with polymer X and surfactant Y will be infringed by a product containing those elements plus additional excipients, while a ‘consisting of’ claim will not.
Ambiguous terms of degree (‘about,’ ‘substantially,’ ‘effective amount’) require prosecution history analysis to determine meaning. The prosecution history, the complete record of communications between the applicant and the patent office, reveals how the applicant defined or distinguished the term to overcome rejections. If an applicant argued during prosecution that ‘about 5%’ means within a specific narrow range to distinguish prior art, that narrowing argument limits the claim’s scope through prosecution history estoppel and cannot be walked back in litigation. A thorough FTO analysis reviews the prosecution history for every high-risk claim to determine whether prosecution history estoppel has constrained scope beyond what the claim language alone suggests.
Phase 7: Risk Stratification
The output of claim analysis is not a binary infringement verdict. It is a risk spectrum that must be stratified to focus response resources correctly.
High-risk patents require immediate escalation. These are in-force patents with broad claims that appear to cover a core product feature through literal infringement, owned by an entity known to litigate or assert patents aggressively, with substantial remaining term in major commercial jurisdictions. The response options for high-risk patents are limited and expensive: license-in, invalidate through IPR, design around, or abandon the product feature.
Medium-risk patents require deeper analysis and monitoring. These are patents where infringement turns on claim interpretation of an ambiguous term, or where infringement would arise only under the doctrine of equivalents. For medium-risk patents, the path forward often involves getting a qualified attorney’s written opinion on the specific claim scope question, monitoring the patent for any continuation applications that might broaden the claims, and documenting the basis for the non-infringement position.
Low-risk or cleared patents are those where one or more claim elements are clearly absent from the product, where the patent has expired or been abandoned, or where the patent has been found invalid in a prior proceeding. These can be resolved and documented but require minimal ongoing attention unless they are part of a family that has active continuation applications.
Table 3: Risk Stratification Matrix
| Factor | High Risk | Medium Risk | Low Risk |
|---|---|---|---|
| Technical Overlap | Direct literal mapping of all elements to core product feature | Ambiguous; infringement depends on claim interpretation or doctrine of equivalents | At least one element clearly absent |
| Legal Strength | Broad claims; survived prior challenge; long remaining term | Narrow claims; potential validity issues; short remaining term | Expired, abandoned, or previously invalidated |
| Owner Behavior | Known to litigate; directly competing in the market | Non-practicing entity or passive licensor | Non-competitor; no known assertion history |
| Design-Around | Not feasible without major product change | Feasible but requires moderate R&D investment | Easy and inexpensive |
| Commercial Impact | Blocks non-negotiable core feature | Affects secondary feature | Affects minor feature only |
Phase 8: The Written FTO Opinion
A formal written FTO opinion from a qualified patent attorney is both a legal document and a strategic asset. It is the primary defense against willful infringement. It is the central IP document in any due diligence process. It provides R&D and commercial leadership with the definitive basis for go/no-go decisions at each development gate.
The opinion should explicitly reference the Technology Document as its scope definition. For each high-risk patent identified, it should include a detailed claim chart, a discussion of the prosecution history relevant to claim scope, a conclusion on infringement risk, and a specific set of recommended next steps. The conclusion need not be a clean ‘no infringement’ finding — an opinion that honestly characterizes a ‘material question of infringement’ on a specific claim, documents the analysis, and recommends a response strategy is legally and strategically valuable even when the answer is not favorable.
Key Takeaways — Part V
FTO analysis executed across eight sequential phases — Technology Document, timing calendar, jurisdictional mapping, multi-methodology search, database selection, claim analysis, risk stratification, and written opinion — is a different product from a patent search. Each phase requires its own resource allocation and its own expertise. Conflating them produces gaps.
Part VI: Evergreening Technology Roadmaps and Patent Thicket Navigation
What Evergreening Actually Is — and What It Costs Generic Competitors
‘Evergreening’ is the colloquial term for pharmaceutical lifecycle management strategies that extend effective market exclusivity beyond the primary compound patent term through the systematic accumulation of secondary patents. Academic analyses have estimated that add-on patents cost U.S. consumers an additional $52.6 billion in drug expenditures over a specific study period. The term carries a pejorative connotation in policy circles but describes commercially rational behavior: companies invest billions in R&D and seek to maximize the return on that investment through all legally available IP tools.
From an FTO perspective, evergreening strategies are what create patent thickets, and understanding the specific mechanisms used in lifecycle management is essential for any company seeking to enter a market with a competitive product, a biosimilar, or a generic.
The Evergreening Technology Roadmap
A complete originator lifecycle management program for a successful drug product typically deploys patents in four or more distinct waves, each targeting a different aspect of the product and its use.
Wave 1 is the compound patent, filed at or near the time of compound synthesis and claiming the active pharmaceutical ingredient by structure. This is the foundational blocking patent. For a small molecule, the compound patent typically runs twenty years from filing date, with a PTE of two to three years added after approval. For a biologic, the equivalent is the sequence or structural patent covering the protein, antibody, or nucleic acid.
Wave 2 covers formulation and pharmaceutical composition. These patents claim specific excipients, salt forms, polymorphic crystal structures, particle size distributions, coating systems, and drug delivery mechanisms. Polymorph patents on crystalline forms of APIs are among the most commonly granted and most frequently litigated in this category. A single API can have multiple commercially relevant polymorphic forms, each with different stability and bioavailability properties, and each potentially separately patentable. The most aggressive programs file polymorph patents on all physically accessible crystal forms of the API, whether or not those forms will be used in the commercial product, to prevent generic companies from using alternative polymorphs as design-arounds.
Wave 3 covers methods of treatment and dosing regimens. These patents claim specific therapeutic uses, patient populations, dosing schedules, and combination therapies. Method-of-use patents can be filed long after the compound patent and can cover new indications discovered through clinical development. They are listable in the Orange Book if they claim the approved use of the product, which means they can trigger 30-month stays against generic Paragraph IV challenges even after the compound patent has expired.
Wave 4 covers manufacturing processes. For small molecules, this includes specific synthetic routes, purification methods, and analytical control methods. For biologics, this category expands to cover fermentation conditions, cell culture media compositions, chromatography methods, viral inactivation protocols, and fill-finish processes. Manufacturing patents are particularly important for biosimilars because they can block the specific manufacturing process even if the biosimilar’s active ingredient is structurally identical to the reference product.
A fifth wave applies specifically to biologics and consists of device patents covering the autoinjectors, pens, and prefilled syringes used to administer the product. Device patents can extend the effective exclusivity of a biologic into the administration mechanism even after all biological sequence and formulation patents have expired.
Humira’s 247-Patent Fortress: A Detailed IP Valuation
AbbVie’s adalimumab (Humira) is the most analyzed pharmaceutical patent portfolio in the history of the industry. By 2022, AbbVie had accumulated what researchers at the Initiative for Medicines, Access & Knowledge (I-MAK) identified as 247 patent applications in the United States covering Humira. This fortress consisted of 167 granted patents plus 80 pending applications at the time of analysis.
The primary compound patent (U.S. Patent No. 6,090,382) expired in December 2016 in the United States. AbbVie’s subsequent strategy was to populate the Orange Book with formulation, manufacturing, and method-of-use patents with expiration dates extending to 2034. AbbVie also reached settlement agreements with every U.S. biosimilar sponsor in the initial wave of challengers, licensing each company to enter the U.S. market no earlier than 2023. The biosimilar sponsors that agreed to these settlements obtained U.S. market access approximately four to five years earlier than if they had litigated every Orange Book patent to final judgment, but seven years after the primary compound patent had already expired. In the EU, where AbbVie’s secondary patent portfolio was considerably thinner due to stricter inventive step requirements and the absence of an Orange Book analog, multiple biosimilars launched in 2018.
The IP valuation implication is direct. The gap between the 2016 primary patent expiration and the 2023 U.S. biosimilar entry represents approximately $7 billion per year in Humira U.S. net revenue that AbbVie retained through secondary patent litigation and settlement strategy. The net present value of that seven-year extension, discounted at a pharmaceutical sector cost of capital of approximately 10%, is roughly $35 billion — more than the total acquisition cost of many mid-size pharmaceutical companies. This is the concrete IP valuation of a well-executed lifecycle management program.
For any biosimilar sponsor or institutional investor modeling the Humira biosimilar opportunity, the relevant FTO analysis was not the 2016 compound patent expiration but the map of all 167 granted secondary patents, their U.S. litigation status, and the realistic probability of clearing each. Companies that modeled only the compound expiration systematically underestimated the blocking power of AbbVie’s secondary portfolio.
Keytruda’s IP Landscape: What $25 Billion in Annual Revenue Looks Like in Patent Form
Merck’s pembrolizumab (Keytruda) is the world’s best-selling drug as of 2024, with net sales approaching $25 billion annually. Its primary IP protection rests on several interlocking patent families covering the anti-PD-1 antibody sequence, its humanized structure, methods of treating specific tumor types, and combination therapies with other checkpoint inhibitors and chemotherapy agents.
The foundational humanized antibody patents were filed in the early 2000s and are scheduled to expire in the 2026-2028 timeframe, depending on jurisdiction, with PTE and SPC adding additional years in key markets. Merck has filed aggressively in the methods-of-treatment space, with patents covering pembrolizumab’s use in more than fifteen oncology indications, each with its own filing date and expiration timeline. Because each approved indication generates a separate Orange Book listing, a biosimilar or follow-on competitor must separately address method-of-use patents across all approved indications to achieve complete U.S. FTO.
The broader PD-1/PD-L1 space involves parallel patent portfolios from Bristol-Myers Squibb (nivolumab/Opdivo) and AstraZeneca/Merck (durvalumab/Imfinzi), creating a cross-cutting thicket of checkpoint inhibitor patents that any new entrant in this mechanism class must navigate. FTO for a new anti-PD-1 antibody requires analysis not just of Merck’s Keytruda portfolio but of the full universe of PD-1/PD-L1 blocking patents, many of which claim the receptor-binding mechanism rather than specific sequences and therefore have potential applicability to structurally distinct antibodies targeting the same pathway.
Key Takeaways — Part VI
Evergreening operates through at least five sequential patent waves, each covering a distinct product or process feature. The Humira case proves that secondary patents are worth tens of billions of dollars in NPV terms when used to delay biosimilar entry. FTO analysis for a competitive or biosimilar program must treat every wave of the originator’s lifecycle management portfolio as a potential blocking position, not just the primary compound patent.
Investment Strategy — Part VI
When valuing a company with a major biologic approaching its primary patent expiration, the correct model is not ‘drug goes generic at primary expiration.’ The correct model is a probabilistic simulation of secondary patent litigation outcomes across all Orange Book-equivalent listed patents, calibrated against the owner’s historical settlement behavior and PTAB IPR success rates for each patent family. Analysts who build this model — rather than using simple primary expiration dates — will produce valuations that are closer to what a strategic acquirer’s IP team would compute.
Part VII: Eliquis and the Compound-Formulation Patent Interaction
Apixaban (Eliquis): A Case Study in Layered Exclusivity
Bristol-Myers Squibb and Pfizer’s apixaban (Eliquis) illustrates how compound patents, formulation patents, and method-of-use patents interact to create layered exclusivity that is more durable than any single patent could provide.
The primary compound patent covering apixaban’s chemical structure (U.S. Patent No. 6,967,208) ran until 2023. Before that expiration, BMS and Pfizer had listed formulation patents in the Orange Book covering the specific tablet composition — including a spray-dried dispersion technology that improves dissolution — with expirations extending to 2026. Method-of-use patents covering apixaban’s approved indications (atrial fibrillation stroke prevention, VTE treatment and prevention, post-surgical prophylaxis) extend into the late 2020s in some jurisdictions.
Generic sponsors filing ANDAs for apixaban faced Paragraph IV challenges against both the formulation and method-of-use Orange Book patents after the compound patent expired. Each Paragraph IV certification against a listed patent triggered a new 30-month stay opportunity, sequentially resetting the clock. The practical result was that even after the primary compound patent’s PTE-extended expiration, BMS/Pfizer retained automatic stay protection for the formulation patents, extending commercial exclusivity.
For an FTO analysis on a proposed apixaban generic as of 2024, the operative question was not the compound patent expiration — that event had passed — but the specific formulation claims and method-of-use claims remaining in the Orange Book. A generic formulation that achieves bioequivalence through a different dissolution mechanism might avoid the spray-dried dispersion patent. A generic labeled only for indications no longer covered by in-force method patents might avoid those listings. The FTO analysis directly drove the product design and label strategy for the ANDA.
Part VIII: FTO for Biologics — Manufacturing, Formulation, and Sequence Claims
Why Sequence Analysis Is Just the Beginning
More than 80% of biopharmaceutical patents contain amino acid or nucleic acid sequence claims. This creates a common but analytically inadequate approach: biosimilar FTO teams that query sequence databases, confirm no direct sequence conflict with their proposed product, and declare early FTO clearance. That approach misses the substantial majority of the patent risk for a biologic drug.
For a monoclonal antibody, the sequence is the skeleton. The commercially relevant product is a glycoprotein with specific post-translational modifications, produced in a specific host cell line under specific culture conditions, purified through a specific multi-step chromatography process, and formulated in a specific buffer system with specific stabilizing excipients. Each of these product attributes and process steps is independently patentable, and all have been actively patented by originator companies.
Cell Line Patents: The CHO Cell Problem
The vast majority of commercial monoclonal antibodies are produced in Chinese hamster ovary (CHO) cell lines. The fundamental CHO cell technology is old enough to be unpatented. However, specific genetically engineered CHO cell derivatives — modified for enhanced expression yield, glycosylation profile control, or productivity under serum-free conditions — carry active patent protection. Lonza’s GS (glutamine synthetase) knockout CHO cell system, which improves selection efficiency and expression levels, has been the subject of extensive licensing. Using a licensed expression system for clinical material does not automatically extend that license to commercial manufacturing. CDMOs and biosimilar sponsors operating under research-stage licenses that do not cover commercial use are a common source of late-stage FTO surprises.
The FTO analysis for a biosimilar program must specifically confirm whether the host cell line being used for commercial manufacturing requires a license, under what terms that license was granted, and whether those terms cover commercial-scale manufacturing and all target markets. A cell line license restricted to the EU does not provide FTO for U.S. commercial manufacturing.
Formulation Patents: The Stabilizer Problem for Biologics
Biologic drugs are structurally complex proteins prone to physical degradation (aggregation, denaturation, adsorption) and chemical instability (deamidation, oxidation, disulfide scrambling). Achieving a commercially acceptable shelf-life of eighteen to twenty-four months under refrigerated storage requires a formulation system of buffers, sugars, surfactants, and amino acids that is drug-specific and often requires substantial formulation development investment. That investment produces patentable innovations.
Originator companies patent their formulation solutions. Patent claims in this area cover specific buffer-surfactant-sugar combinations, amino acid stabilizer concentrations, pH ranges, protein concentration windows, and container systems. A biosimilar formulated identically to the reference product to simplify analytical characterization may directly infringe formulation patents. A biosimilar formulated with a different buffer system to avoid those patents must demonstrate bioequivalence and stability for its specific formulation — a development investment that non-trivially increases both cost and timeline.
The FTO analysis must therefore evaluate not just whether the proposed biosimilar formulation infringes the reference product’s formulation patents, but what design-around formulation options exist and what development work each requires to demonstrate equivalence. This analysis belongs at the pre-IND stage of biosimilar development, when formulation design decisions are still being made, not at Phase 3 when the formulation is locked.
Analytical Method Patents and Their Hidden Blocking Power
Regulatory agencies require extensive analytical characterization of biosimilar products to demonstrate structural and functional similarity to the reference product. Many of the gold-standard analytical methods for biologics characterization — particular mass spectrometry methods for glycoform profiling, hydrogen-deuterium exchange methods for higher-order structure analysis, and surface plasmon resonance methods for binding kinetics — are subject to active patent protection.
A biosimilar sponsor that must use a patented analytical method to generate FDA-required characterization data has an FTO problem even if its drug product itself is completely unencumbered. The method patent covers the use of the analytical process, and using it during commercial regulatory submissions is typically considered commercial activity beyond research safe harbor. Several analytical platform companies have begun asserting method patents against biosimilar sponsors specifically in the context of regulatory submissions, creating a new category of FTO exposure that was not previously on most companies’ radar.
Key Takeaways — Part VIII
Biologic FTO requires analysis across at least six distinct patent categories: sequence, manufacturing process, host cell line, formulation, delivery device, and analytical methods. Each category has independent blocking power. Missing any one can halt commercialization after substantial late-stage development investment.
Part IX: FTO for Cell and Gene Therapies — Platform-Level Analysis
Why Product-Level FTO Is Insufficient for CGTs
For a small-molecule drug, the FTO analysis can be substantially product-focused: define the chemical entity, map the manufacturing process, and clear both against the patent landscape. The active ingredient and the manufacturing process are conceptually separable.
For cell and gene therapies, this separation does not exist. The therapeutic payload (a transgene, a CAR construct, an edited gene) cannot be administered to patients without a delivery platform (AAV vector, lentiviral vector, lipid nanoparticle), and that delivery platform is inextricably tied to manufacturing processes (transient transfection, stable producer lines, plasmid production, viral purification) that are subject to independent patent coverage. Each component of the vein-to-vein process for an autologous cell therapy — leukapheresis, T-cell isolation, ex vivo activation, viral transduction, expansion, cryopreservation, quality release, and infusion — involves technologies that may be independently patented by entities unrelated to the CGT developer.
This creates what can accurately be called ‘platform-level’ FTO exposure: the product cannot be made, tested, released, or administered using any available manufacturing platform without potentially infringing someone’s IP. The FTO analysis must therefore begin when platform technology selection decisions are being made — typically in the IND-enabling research phase — not when the product itself is defined.
AAV Vector Patents: The Delivery Problem
Adeno-associated virus (AAV) vectors are the dominant delivery system for in vivo gene therapy. The foundational patents covering recombinant AAV production were largely held by the University of Florida and its commercial partner, with broad licensing programs that created workable access to the technology for the first generation of AAV gene therapy developers. However, the landscape has become substantially more complex.
Specific AAV serotypes with clinically relevant tropism profiles — AAV5, AAV8, AAV9, AAVrh10 — carry their own patent estates owned by different entities, many of which maintain different licensing policies and fee structures. Engineering AAV capsids for enhanced transduction efficiency or reduced immunogenicity has generated a new layer of patents on modified capsids that cover the specific constructs now used in most clinical-stage programs. A program using an engineered AAV9 variant for CNS delivery needs FTO clearance on the capsid engineering patents, not just the basic AAV9 serotype.
The ITR (inverted terminal repeat) sequences that flank the therapeutic payload and are essential for AAV replication and packaging are also subject to patent claims. Specific promoter sequences, poly-A signals, and intron elements incorporated into the expression cassette to optimize transgene expression each carry their own IP considerations. An AAV gene therapy FTO analysis conducted at the product level — treating the vector as a black box — will miss this entire category of exposure.
The CRISPR Patent War: A Live FTO Case Study
No area of CGT IP has generated more legal complexity than CRISPR-Cas9 gene editing. The foundational CRISPR-Cas9 technology was the subject of an eight-year patent interference and inter partes review proceeding between the University of California, Berkeley (UC Berkeley, inventors Doudna and Charpentier) and the Broad Institute of MIT and Harvard (inventor Zhang). The dispute centered on whether Broad’s patents covering CRISPR-Cas9 use in eukaryotic (including human) cells were patentably distinct from UC Berkeley’s patents covering CRISPR-Cas9 use in any setting.
The Patent Trial and Appeal Board and, subsequently, the Federal Circuit confirmed that Broad’s eukaryotic cell claims were patentably distinct from UC Berkeley’s earlier work. Broad holds the dominant patents for therapeutic applications of CRISPR-Cas9 in human cells, which is the commercially relevant application for almost all CRISPR-based medicines in development.
Any company developing a CRISPR-based therapeutic must have Broad Institute license coverage. The Broad licenses CRISPR technology through its commercialization partner, Editas Medicine, for human therapeutic applications, and through its MilliporeSigma partnership for research and industrial applications. The structure of the Broad licensing program — which field is covered, whether sublicensing is permitted, and how royalties are calculated — is directly material to the FTO analysis for any CRISPR program.
The CRISPR patent landscape has further complicated as base editing (BE) and prime editing (PE) technologies have generated their own patent estates, many of which were also filed by Broad-affiliated inventors and licensed through the same Editas commercialization channel. A program using cytosine base editing to correct a point mutation needs FTO clearance on both the CRISPR-Cas9 nickase and the base editor fusion protein, which are covered by separate patent families with different inventors and potentially different licensors.
For institutional investors evaluating CRISPR-based biotech companies, the IP stack analysis is directly predictive of licensing cost structure: a company with Broad license coverage for its lead indication has a quantifiable royalty obligation, while a company operating without that coverage has an injunction risk that is not fully reflected in standard DCF models.
LNP Patents: The mRNA Delivery Problem
The success of mRNA-based vaccines and therapeutics has created fierce competition for lipid nanoparticle (LNP) delivery technology. The dominant LNP technologies for mRNA delivery trace back to foundational work at Alnylam Pharmaceuticals (for siRNA) and to University of British Columbia patents licensed through Arbutus Biopharma.
Moderna’s COVID-19 vaccine (mRNA-1273) was the subject of patent litigation brought by Arbutus, which claimed that Moderna’s proprietary LNP technology infringed Arbutus patents on ionizable lipid formulations. Moderna challenged those patents through IPR proceedings, with mixed success across different claims. The litigation outcome continues to influence LNP FTO strategy for the entire mRNA therapeutics field.
Any company developing an mRNA therapeutic must conduct FTO analysis not just on its mRNA sequence and target antigen, but on its specific LNP formulation — the ionizable lipid structure, the helper lipid composition, the PEGylation chemistry, and the manufacturing process for LNP assembly. These are separate patent families with separate owners, separate licensing requirements, and separate IPR challenge histories that must each be analyzed and cleared.
Key Takeaways — Part IX
CGT FTO is platform-level, not product-level. AAV capsid engineering, expression cassette components, CRISPR editing tools, and LNP delivery system compositions each carry independent patent exposure that must be cleared alongside the therapeutic payload itself. Platform selection decisions made in pre-IND research directly determine the FTO landscape for the entire program, making early analysis essential and late analysis expensive.
Investment Strategy — Part IX
When evaluating a cell or gene therapy company, the FTO analysis should map IP exposure across three layers: therapeutic target and payload, delivery platform, and manufacturing process. For each layer, identify the patent holder, the licensing status (granted, pending, or refused), the royalty structure if disclosed, and the company’s plan if the license is unavailable. A compelling program with unresolved platform-level IP exposure should carry a material discount to its risk-adjusted NPV, and representations regarding licensing status should be included in any financing or M&A documentation.
Part X: AI-Augmented FTO — Semantic Search, Triage, and Degenerate Sequences
What AI Does in FTO — and What It Cannot Do
AI-powered tools have materially improved FTO workflow efficiency. The honest characterization of the state of the technology in 2025 is that AI is an excellent first-pass tool and a poor substitute for attorney judgment. Understanding what AI can and cannot do prevents both underinvestment and overreliance.
AI excels at semantic search: querying patent databases using natural language descriptions of a technology concept rather than Boolean keyword strings. Graph AI approaches (used by platforms such as IPRally) represent patents as nodes in a network, where edges encode conceptual relationships between claims, and then find patents that occupy the same conceptual neighborhood as the input description. This approach captures patents that use different terminology to describe similar concepts, which keyword search systematically misses. In benchmarking studies, semantic search has been shown to recover 15-30% more relevant patents in a given technology area compared to keyword search alone, with the additional recovery concentrated among older patents filed before standardized nomenclature.
AI-powered triage tools can score large result sets by relevance, automatically generate preliminary claim charts, flag patents with specific structural features (broad claims, high forward citation count, recent prosecution activity), and identify the subset of results that warrant attorney review. In a typical biopharma FTO with an initial result set of 3,000-10,000 patent documents, AI triage can reduce the set requiring detailed attorney analysis to 200-400 documents — a 10-20x reduction in manual review time. Platforms such as Clearstone FTO+AI quantify a 40% reduction in project timelines for FTO analyses incorporating AI-assisted review.
What AI cannot do is interpret claim scope, apply prosecution history estoppel, assess the doctrine of equivalents under case-specific legal standards, or provide the written opinion that constitutes a willful infringement defense. These require legal judgment exercised by a qualified attorney. A company that uses AI triage output as its final FTO determination and proceeds to market without an attorney opinion has not reduced its willful infringement exposure — it has created a paper trail suggesting awareness of potentially relevant patents while failing to obtain legal clearance.
Degenerate Sequence Patents: The 30% Blind Spot
Degenerate sequence claims are among the most dangerous and most commonly missed patent risks in biopharmaceutical FTO. Patent applicants seeking broad protection for a protein or nucleic acid sequence frequently claim the invention using degenerate notation — representing variable positions in the sequence with ambiguity codes rather than specific residues. In protein sequence claims, this is done using Markush group language listing amino acid options at specific positions (‘wherein X3 is Ala, Ser, or Thr’). In nucleic acid claims, standard IUPAC ambiguity codes (‘R’ for A or G; ‘Y’ for C or T) represent positions where multiple nucleotides are acceptable.
These degenerate claims can cover millions or billions of specific sequences within a single claim. Standard sequence alignment tools designed for genomic research (BLAST, FASTA) perform exact matching or identity-percentage matching against query sequences. They do not parse degenerate claim language, enumerate all sequences covered by a degenerate specification, and then check the query sequence against each enumerated variant. The result is a systematic blind spot: a biosimilar developer whose candidate sequence exactly matches one of the millions of sequences covered by a degenerate patent claim will receive no match flag from a BLAST search.
Studies of biopharmaceutical patent databases suggest that degenerate sequences appear in approximately 30% of all biopharma patent claims. AI-powered platforms built specifically for biopharmaceutical FTO — including Derwent’s sequence analysis tools and specialized offerings from contract search firms — address this problem by parsing the claim language, generating the full set of sequences covered by the degenerate specification, and then performing similarity searching against that enumerated set. The use of these tools can reduce the probability of missing a degenerate-sequence blocking patent by up to 40%.
Predictive Analytics: Forward-Looking FTO
Machine learning models trained on historical patent litigation data can identify structural features of patents that correlate with litigation risk: claim breadth scores, forward citation velocity, assignee litigation frequency, claim language overlap with other active portfolios, and examination history metrics. Platforms incorporating predictive litigation risk scoring can flag patents in an FTO result set that, based on their structural characteristics, are historically elevated litigation risks — even if a claim-by-claim analysis does not yet confirm infringement.
This capability shifts FTO from retrospective (analyzing the current landscape) to prospective (anticipating where litigation risk will crystallize as the patent matures and is forward-cited). For a company planning a ten-year development program, the most strategically valuable FTO intelligence is not the landscape as it exists today but the landscape as it will exist at the time of launch.
Key Takeaways — Part X
AI delivers a 10-20x reduction in manual patent review time through semantic search and triage. It does not produce a willful infringement defense. Degenerate sequence searching requires specialized tools beyond BLAST/FASTA and is an essential component of biologic and CGT FTO. Predictive analytics enable prospective rather than retrospective risk modeling for long-horizon development programs.
Part XI: Global Jurisdictional Analysis
United States: Broad Patent Scope, High Litigation Risk
The United States remains the world’s highest-stakes FTO jurisdiction because of the combination of relatively permissive patentability standards (particularly for formulations and methods of treatment), high litigation frequency, and the availability of treble damages for willful infringement. The U.S. also offers mechanisms that create commercial leverage unavailable elsewhere: the 30-month stay under Hatch-Waxman and the BPCIA patent dance each allow originator companies to delay generic and biosimilar competition through legal mechanism rather than technical barriers alone.
Key post-KSR considerations: the Supreme Court’s 2007 decision in KSR International v. Teleflex raised the obviousness bar for patents generally, and subsequent Federal Circuit decisions have applied this heightened standard to pharmaceutical formulation and combination therapy patents. Patents on specific dosage regimens that were routinely granted pre-KSR now face tougher examination. An FTO analysis for a method-of-treatment claim must assess not just whether the claim covers the proposed use but whether the claim would survive a post-KSR validity challenge — because a high-risk blocking patent that is also vulnerable to an IPR validity challenge represents a fundamentally different strategic situation than the same patent with bulletproof prosecution history.
The one-year grace period under 35 U.S.C. § 102(b)(1) means that a U.S. inventor’s own public disclosure does not destroy their right to file a patent application within one year. For FTO purposes, this means that a pre-filing public disclosure by the blocking patent’s inventor does not constitute prior art against that inventor’s own patent — it only counts against others seeking to file on the same invention.
European Union: The Unified Patent Court and Its FTO Implications
The Unified Patent Court (UPC), which became operational in June 2023, represents the most significant structural change to European patent litigation in decades. Before the UPC, a patent holder seeking to enforce a European patent had to litigate in each national court separately — a German action did not extend to France, and vice versa. A single invalidity finding in one national proceeding did not automatically invalidate the patent in other EU member states.
Under the UPC, a Unitary Patent (UP) covers all participating EU member states through a single grant, and a single UPC action can enforce that patent across all participating states simultaneously. This dramatically increases the stakes of UPC litigation: a successful injunction in a UPC central division proceeding can block the defendant from the entire participating EU market in a single action. Conversely, a successful invalidity finding in a UPC proceeding invalidates the patent across all participating states simultaneously.
For FTO purposes, the UPC changes the risk calculus in two material ways. First, a patent holder with a Unitary Patent can now obtain EU-wide injunctive relief in a single proceeding, making the potential damage of a missed blocking patent substantially higher than under the pre-UPC national litigation model. Second, an IPR-equivalent challenge at the UPC can eliminate a blocking patent across the entire EU market in a single proceeding, making UPC invalidity actions a more efficient clearing mechanism than the prior multi-jurisdiction approach.
Companies conducting FTO for EU commercial plans as of 2025 must evaluate whether identified blocking patents are traditional European patents (enforced nationally) or Unitary Patents (enforced EU-wide through the UPC), because this changes both the infringement exposure and the invalidity challenge strategy.
India: Section 3(d) and Its Structural Impact on FTO
India’s Patent Act Section 3(d) prohibits the grant of patents on ‘new forms of known substance’ unless those new forms show significantly enhanced therapeutic efficacy compared to the known substance. The provision was drafted specifically to prevent pharmaceutical evergreening strategies based on polymorph patents, new salt form patents, and formulation patents for drugs already known in their basic compound form.
In practice, Section 3(d) means that the secondary patent portfolio that creates a ten-year exclusivity extension in the United States — polymorphs, hydrates, solvates, particle size specifications, new formulations — typically does not exist in India. Indian patent offices routinely reject these applications under Section 3(d) unless applicants can produce clinical data demonstrating superior therapeutic efficacy, not merely improved pharmacokinetics or physical stability.
For an FTO analysis covering India, the relevant blocking patent landscape is therefore substantially thinner than the U.S. or EU landscape for any given drug. A company that can navigate the primary compound patent period in India faces little residual blocking risk from secondary patents that would dominate the FTO analysis in other jurisdictions. For a biosimilar or generic company prioritizing emerging markets, India’s Section 3(d) environment dramatically shortens the effective blocking patent timeline compared to U.S. and EU projections.
Table 4: Key Jurisdictional Differences for Biopharma FTO
| Feature | United States | EU (UPC Era) | China | Japan | India |
|---|---|---|---|---|---|
| Methods of Treatment | Patentable | ‘Second medical use’ format required | Patentable | ‘Second medical use’ format required | Not patentable |
| Grace Period | 12 months for inventor’s own disclosure | Effectively none | 6 months, limited circumstances | 12 months with formal requirements | 12 months, limited circumstances |
| Secondary Patent Environment | Permissive; evergreening common | Moderate; stricter inventive step | Increasingly scrutinized | High inventive step bar | Severely restricted by Section 3(d) |
| Data Exclusivity (NCE) | 5 years | 8+2+1 years | 6 years | 8 years | 4 years |
| Patent Enforcement Mechanism | Federal district court; ITC available | National courts OR UPC (for Unitary Patents); UPC injunctions EU-wide | CNIPA and courts; enforcement improving | Courts; strong protection | Courts; Section 3(d) limits portfolio |
| Key Strategic Risk | Treble damages for willfulness; Paragraph IV/30-month stays | UPC enables EU-wide injunctions; SPCs extend term significantly | Trade secret protection; local working requirements | High inventive step bar for follow-on patents | Section 3(d) blocks secondary patent filing; thin FTO landscape for originator |
Key Takeaways — Part XI
The UPC’s EU-wide injunction capability materially raises the stakes of unresolved blocking patents in the EU compared to the pre-2023 national litigation model. India’s Section 3(d) creates a fundamentally different blocking patent landscape for secondary pharmaceutical patents. Any global commercialization model that applies U.S. or EU secondary patent timelines to Indian revenue projections overstates the duration of Indian market exclusivity.
Part XII: When a Blocker Appears — IPR, Licensing, and Design-Around Playbooks
Inter Partes Review: Statistics, Strategy, and Limitations
Inter Partes Review (IPR) at the PTAB is the primary U.S. mechanism for challenging the validity of a blocking patent outside of district court litigation. IPR proceedings are faster than district court (final written decisions issue within one year of institution), cheaper (typically $300,000-$800,000 for petitioner’s costs compared to $3-$10 million for district court patent litigation through trial), and statistically favorable to petitioners in the pharmaceutical and biotech sectors.
PTAB statistics for pharmaceutical patent IPRs as of 2024: institution rate approximately 65%, meaning roughly two-thirds of filed petitions proceed to a full review. Of instituted petitions, approximately 75-80% result in at least some claims being cancelled or narrowed. The overall ‘kill rate’ for pharmaceutical patent claims challenged through IPR is therefore roughly 50% when calculated from petition filing.
These statistics inform IPR strategy significantly. A high-risk blocking patent with relatively narrow, clearly-scoped claims and a clean prosecution history has a lower IPR success probability than a broad patent with close prior art that the examiner did not clearly consider. The FTO analysis should therefore evaluate IPR candidates not just on whether they technically block the product but on the probability of a successful IPR challenge, which depends on the quality of the prior art available, the breadth of the claims, and the petitioner’s ability to construct an obviousness argument that PTAB’s particular interpretive standards will find persuasive.
One critical limitation: filing an IPR petition creates an estoppel. Under 35 U.S.C. § 315(e), a petitioner who participates in an IPR cannot later assert in district court that a claim is invalid on any ground that was raised or ‘reasonably could have been raised’ in the IPR. This estoppel provision means that filing an IPR is a strategic commitment, not just a tactical option. If the IPR fails, the petitioner’s validity arguments in subsequent district court litigation are restricted to grounds not covered by the IPR petition.
Licensing Strategy: Structuring for FTO Efficiency
When a blocking patent cannot be designed around and IPR has an insufficient probability of success, licensing is the direct path to FTO. License structure matters enormously for financial modeling and competitive positioning.
An exclusive license removes the blocking patent from the landscape entirely for the licensee’s field. A non-exclusive license grants FTO but allows the patentee to license competitors. A field-of-use limitation can create a license covering only specific indications or markets, which lowers upfront cost but requires careful drafting to ensure the commercial uses actually planned are covered. A cross-license can resolve a mutual blocking situation efficiently when two companies each hold patents that restrict the other’s activities.
Royalty structures for pharmaceutical licensing typically fall into three models: running royalties as a percentage of net sales (typically 1-5% for a secondary patent, 5-15% for a foundational platform patent), lump-sum buyouts calculated based on NPV of expected future sales, and combination structures with upfront payments and tiered running royalties. The choice between models depends on the licensee’s cash position, the revenue visibility for the licensed product, and the tax treatment of royalty payments in the relevant jurisdictions.
A critical and frequently overlooked element of license negotiation is the FTO scope of the licensed rights. A license to ‘use and sell the patented invention’ may not explicitly cover manufacturing the patented compound at a contract manufacturing site, importation of finished goods, or sublicensing to co-development partners. Every commercial use contemplated in the business plan must be explicitly covered by the license language, or the FTO analysis that identifies ‘licensed’ as the resolution for a high-risk patent is incomplete.
Design-Around: Engineering Your Way to FTO
A design-around modifies the product or process to avoid infringing a specific claim, typically by eliminating one or more claim elements from the product. The feasibility and cost of a design-around depends entirely on the specifics of the blocking claim.
For a formulation patent with claims requiring a specific excipient at a specific concentration range, a design-around might involve substituting a structurally different excipient or adjusting the concentration outside the claimed range. The development cost is the work required to demonstrate that the reformulated product retains bioequivalence and stability — typically several months of formulation development and stability studies. For a manufacturing process patent claiming a specific purification step, the design-around might involve a different chromatography method or a different process order, with corresponding process validation costs.
Design-around analysis must precede regulatory development commitments. Reformulating after Phase 2 to avoid a newly identified blocking patent means generating a new comparability dataset, potentially requiring additional clinical bridging studies, and resetting part of the regulatory clock. The development cost of a design-around is orders of magnitude lower when it is identified and implemented in pre-IND research than when it is forced on a late-stage program.
Table 5: Blocking Patent Response Playbook
| Strategy | Description | Key Advantages | Key Disadvantages | Optimal Condition |
|---|---|---|---|---|
| License-In | Negotiate paid right to use the patented technology | Certain FTO; avoids litigation and R&D delays | Ongoing royalty obligation; may fund a competitor | Essential technology, strong patent, design-around not feasible |
| Cross-License | Exchange patent rights with a blocking patent holder who is also blocked by your patents | Can resolve complex thickets; lower cash cost | Requires a portfolio with value to trade; complex negotiations | Mutual blocking situation exists |
| IPR Challenge | PTAB validity challenge on prior art grounds | If successful, eliminates the threat entirely | 315(e) estoppel; costly; uncertain; can provoke litigation | Strong prior art available; claim is broad and arguably anticipated or obvious |
| Design-Around | Modify product/process to avoid specific claim elements | Avoids royalties; may produce new patentable innovations | R&D delay and cost; may result in suboptimal product design | Blocking claim has narrow scope; technical alternative exists |
| Acquire the Patent | Purchase the patent or its owner | Complete control; no ongoing royalty | Most expensive option; requires integration | Blocking technology is core to long-term strategy; owner is acquirable |
| Await Expiration | Monitor the patent as it approaches end of term | No current cost | Leaves the program exposed; launch is delayed | Patent has very short remaining term; product launch can wait |
| Abandon Feature | Remove the infringing feature from the product | Eliminates the legal risk entirely | Loss of planned product capability | Feature is non-essential and the patent cannot be cleared economically |
Part XIII: FTO in M&A Due Diligence and Capital Raising
Why You Cannot Rely on the Target’s FTO Opinion
When a pharmaceutical company acquires a biotech with a clinical-stage asset, the target company’s existing FTO opinion is a useful starting document. It is not a substitute for independent diligence. This is not a procedural formality; it is a substantive risk management requirement with specific legal and commercial justifications.
An FTO opinion is provided to a specific client. The legal protection against willful infringement damages flows to the entity that received and relied on the opinion, not to subsequent holders of the asset. An acquirer that relies solely on the target’s FTO opinion as its own due diligence cannot definitively assert that it possessed a good-faith non-infringement belief based on competent counsel’s advice, because it did not commission that advice. Obtaining an independent opinion, or at minimum a formal reliance letter from the original opinion’s authors confirming that the acquirer can rely on the opinion, is the correct procedure.
The target’s FTO analysis may also have gaps relative to the acquirer’s commercial plans. A biotech that developed its product for a single indication in the United States may have conducted FTO only for U.S. commercialization in that indication. An acquirer planning a global launch across five indications needs FTO coverage for all five indications in all five markets. The scope mismatch between the target’s analysis and the acquirer’s business plan is a common source of post-close IP surprises.
Valuing IP Assets in M&A: The Patent Landscape Overlay
The combination of FTO analysis (microscope) and patent landscape analysis (telescope) produces the only rigorous basis for valuing a pharmaceutical IP portfolio in an M&A context. A patent count is meaningless without strategic context. Twenty patents in a technology area owned by six major competitors with broad foundational patents and active litigation histories is a portfolio of contested claims that will require sustained licensing or litigation spend to deploy commercially. Two patents in an unoccupied ‘white space’ identified through landscape analysis may represent the foundational blocking position for an entire new product category.
The specific patent landscape metrics that translate into IP asset value are: whitespace density (percentage of the technology sub-areas around the target’s patents that are unoccupied by third-party patents), forward citation velocity (rate at which other patents are citing the target’s patents, which correlates with technology importance), claim scope index (breadth of the independent claims relative to the prior art, which determines how easy it is to design around), and remaining effective patent life accounting for PTE and SPCs.
Bristol-Myers Squibb’s $74 billion acquisition of Celgene in 2019 was, at its core, a transaction about the depth and breadth of Celgene’s oncology patent portfolio across its marketed products (revlimid, pomalyst, abraxane) and its pipeline. A central element of BMS’s valuation was modeling the effective patent life remaining for revlimid in key markets after accounting for patent challenge litigation that was already active at deal signing. Getting that model wrong would have materially mispriced the deal — revlimid accounted for approximately $12 billion in annual net sales at the time of acquisition.
Key Takeaways — Part XIII
Independent FTO analysis is always required in M&A due diligence, regardless of the quality of the target’s existing opinion. The patent landscape overlay transforms a patent count into a strategic asset valuation. IP valuation in major pharmaceutical M&A transactions requires probabilistic patent term modeling that accounts for secondary patents, PTEs, SPCs, and active litigation status — not just primary compound expiration dates.
Investment Strategy — Part XIII
For institutional investors in pre-commercial biotech, the IP diligence framework should include four specific data requests: a copy of the most recent written FTO opinion (not a summary), the Technology Document that defines the FTO scope, a list of all known blocking patents and the company’s response plan for each, and the company’s pending patent application portfolio with priority dates. These four documents, read together, tell the story of whether the company’s IP position is genuinely protected or whether it has latent risks that will materialize as expensive surprises at the worst possible time.
Part XIV: Continuous Monitoring — Converting FTO from a Snapshot to a System
Why the Patent Landscape Changes Around Your Product
The patent landscape for any active pharmaceutical or biotechnology program shifts continuously. New patents issue every week. Continuation applications that were pending and invisible (unpublished) at the time of the original FTO analysis publish eighteen months after filing and may contain broadened claims not present in the parent application. Patents in a blocking portfolio may be challenged by third parties through IPR, reducing their scope or eliminating them. A competitor may file a new formulation or method-of-use patent specifically designed to create a blocking position against an anticipated market entrant.
A company that conducts FTO once at IND filing and then operates on that single analysis through commercialization has a document that was accurate on one specific date and has decayed in relevance every day since. The longer the development timeline, the greater the gap between the original FTO’s conclusions and the current landscape.
Building an Automated Monitoring System
Commercial patent platforms allow users to save search strategies as persistent saved searches with automated alert configurations. When a new patent document is published that matches the saved search criteria, the system sends an automated notification. Most systems allow alerts to be configured for new publications, for status changes on previously identified patents (grant, abandonment, IPR filing, Federal Circuit decision), and for new filings by specified assignees.
For a biopharmaceutical program in active development, a monitoring configuration should maintain at minimum four categories of saved searches: a classification-based search covering the product’s technology area, an assignee-based search covering each known competitive patent filer, a keyword/semantic search tuned to the specific product features identified in the Technology Document, and a citation-based alert on each high-risk patent previously identified in the FTO.
This system produces a stream of new documents requiring review, which can be integrated into the company’s IP committee or patent steering committee review cycle. The effort required to maintain the system is substantially lower than the cost of discovering a significant new blocking patent late in development — after clinical and manufacturing investments have made design-around or program abandonment enormously expensive.
Part XV: Investment Strategy for Analysts
Using FTO Intelligence to Improve Biotech Valuation Models
Standard DCF valuation models for clinical-stage biotech companies discount future cash flows by a probability of technical success, which is typically estimated from historical clinical phase transition rates by indication. This framework captures clinical risk but systematically underweights IP risk. A compound with 70% probability of clinical success but material unresolved FTO exposure does not have a 70% probability of commercial success — it has something substantially lower, depending on the severity of the IP exposure.
Incorporating FTO intelligence into a biotech valuation model requires assigning probability-weighted resolution costs to each identified high-risk blocking patent. The resolution cost for a high-risk blocking patent includes the probability-weighted cost of each available resolution path: licensing (expected royalty NPV at applicable deal rates), IPR challenge (cost times probability of failure times post-failure litigation cost), design-around (expected R&D cost and timeline penalty), and abandonment (probability times asset NPV). Summing these probability-weighted costs and subtracting from the gross DCF produces an IP-risk-adjusted valuation that is more accurate than a model that ignores FTO entirely.
This methodology is not academic. It produces materially different valuations for specific asset types. A program in a crowded therapeutic area with an extensive originator secondary patent portfolio — think a biosimilar for a major biologic in the three to five years before primary patent expiration — has IP risk that can represent 20-40% of the unadjusted DCF in a worst-case scenario. An analyst who builds the FTO-adjusted model will make a different investment decision than one who does not.
Using Patent Cliff Data as a Market Entry Signal
Patent expiration timelines, properly modeled with PTE, SPC, data exclusivity, and litigation probability adjustments, are the most reliable forward-looking indicator of generic and biosimilar market entry available to pharmaceutical investors. The consensus generic entry date embedded in a branded pharmaceutical company’s analyst consensus model is typically the compound patent’s nominal PTE expiration. It is almost never the correct date.
The correct date is the later of the compound patent expiration and the resolution of all active Orange Book patent litigation — whether by final judgment, by settlement with authorized entry dates, or by expiration of remaining secondary patents. For biosimilars, the correct date is the later of the twelve-year BPCIA data exclusivity date and the resolution of all BPCIA patent dance litigation. Companies like DrugPatentWatch that track Orange Book and Purple Book patent listings, active Paragraph IV litigation dockets, and biosimilar patent dance filings in an integrated database allow analysts to construct materially more accurate generic and biosimilar entry timing models than those based on nominal patent expiration calendars alone.
Key Takeaways — Part XIV and XV
FTO-adjusted valuation models produce materially different asset values than models that treat patent risk as binary or ignore it entirely. Patent cliff modeling that accounts for secondary patent litigation timelines and settlement structures is substantially more accurate than modeling based on primary compound patent expiration dates. Maintaining an automated monitoring system for the patent landscape converts FTO from a one-time cost into an ongoing intelligence feed.
Part XVI: Frequently Asked Questions
How much does a biopharmaceutical FTO analysis cost?
Early-stage analyses focused on a single compound in a single jurisdiction run $50,000-$150,000. A comprehensive pre-Phase 3 analysis for a small-molecule drug covering the United States and major EU markets costs $150,000-$350,000. A full global FTO for a complex biologic or CGT, covering manufacturing, formulation, sequence, and device patents across eight to ten jurisdictions, can reach $500,000-$1,000,000. These figures represent a fraction of the Phase 3 investment they protect. The cost drivers are: technology complexity (CGT > biologic > small molecule), number of jurisdictions, density of the competitive patent landscape, and stage of development.
What is the most common FTO mistake biopharma startups make?
Starting the analysis too late. Most startups first engage with FTO when preparing for a Series B or clinical development funding round, by which point the product design, manufacturing process, and formulation are substantially fixed. Discovering a blocking patent at that stage forecloses the lowest-cost response, which is to redesign the product before development resources are committed. The second most common mistake is treating a patentability search conducted for the company’s own patent filing as equivalent to an FTO analysis. The two answer different questions and use different search methodologies.
Can AI replace patent attorneys in FTO analysis?
No. AI tools can reduce FTO project timelines by 30-40% by automating search and initial triage. They cannot interpret claim scope, apply prosecution history estoppel, assess infringement under the doctrine of equivalents, or produce the written opinion that constitutes a willful infringement defense. Using AI output as a final FTO determination without attorney review creates a paper trail demonstrating awareness of potentially blocking patents without the legal clearance that would protect against willful infringement findings.
How does biosimilar interchangeability affect FTO scope?
A program seeking FDA interchangeability designation requires clinical switching study data that must be generated using specific analytical methods. Some of those methods are patented. The FTO analysis for an interchangeability program must include clearance of the analytical methodologies required to generate the switching study dataset, not just clearance of the biosimilar drug product itself. This is a frequently overlooked category of exposure in biosimilar development programs.
We are acquiring a biotech. Should we rely on their existing FTO opinion?
No. Conduct independent FTO analysis for three reasons: the legal protection of a willful infringement opinion runs to its original recipient, not to an acquirer; the target’s analysis scope may not cover the acquirer’s full commercial plans; and the analysis may be outdated relative to the current patent landscape. At minimum, obtain an independent reliance letter from the original opinion’s authors. Commission a fresh analysis for any jurisdiction or indication not covered by the existing opinion.
What is the practical implication of the Unified Patent Court for EU FTO?
Since June 2023, Unitary Patents can be enforced across all participating EU member states through a single UPC proceeding. This means a missed blocking patent that is a Unitary Patent can result in an EU-wide injunction obtained in a single action rather than requiring parallel national court proceedings. The corresponding benefit is that a successful invalidity challenge at the UPC eliminates the patent across the entire EU market simultaneously. EU FTO analyses conducted before 2023 should be reviewed for patents that have since been registered as Unitary Patents and reassessed for the changed enforcement risk.
Consolidated Key Takeaways
FTO analysis costs between 0.002% and 0.02% of the R&D investment it protects. Without a written opinion, treble damages under 35 U.S.C. § 284 are live. Owning a patent does not authorize selling the patented drug. FTO, patentability, and validity searches answer three different questions and cannot substitute for each other.
For small molecules, the Orange Book, Paragraph IV history, and PTE/SPC calculations are as important as the compound patent. For biologics, FTO requires analysis of manufacturing process, host cell line, formulation, delivery device, and analytical method patents in addition to sequence analysis. For CGTs, FTO is platform-level from the outset, and platform selection decisions made in pre-IND research determine the FTO landscape for the entire program.
The CRISPR patent landscape, particularly the Broad Institute’s eukaryotic cell claims, is the most consequential IP factor in the CGT sector. Degenerate sequence patents, present in approximately 30% of biopharma patent claims, are systematically missed by standard BLAST/FASTA searches and require specialized parsing tools. India’s Section 3(d) creates a structurally different secondary patent landscape compared to the United States and EU.
AI tools reduce FTO timeline by 30-40% through search and triage automation. They cannot produce a willful infringement defense. The UPC’s 2023 launch materially raised the stakes of unresolved EU blocking patents. FTO-adjusted DCF models produce materially different biotech asset valuations than models that treat patent risk as binary.
This article is for informational purposes only and does not constitute legal advice. Consult qualified patent counsel for advice specific to your program.
Supported data sources include DrugPatentWatch, Tufts Center for the Study of Drug Development, USPTO PTAB statistics, PhRMA, I-MAK, and public FDA Orange Book and Purple Book databases.