Executive Summary

Pfizer’s management of sildenafil citrate’s loss of exclusivity (LOE) in the United States is the closest thing small-molecule pharma has to a controlled experiment in patent cliff engineering. The company entered the generic transition with two layered U.S. patents, a pediatric exclusivity extension, a pre-negotiated litigation settlement with the first ANDA filer, and its own authorized generic subsidiary ready to launch on the exact same day as Teva’s independent product. The outcome: a “patent cliff” that looked, from a market-share perspective, more like a managed slope.
This report goes well beyond the standard case study treatment of Viagra’s LOE. It quantifies the IP asset value embedded in each patent layer, maps the Hatch-Waxman litigation mechanics that shaped every settlement, models the price erosion curve generic sildenafil followed relative to HHS benchmarks, and extracts a replicable evergreening roadmap applicable to any small-molecule franchise facing LOE within a five-to-seven-year window.
The core finding: Pfizer preserved roughly 45% of the total erectile dysfunction (ED) drugs market share for the combined Viagra franchise through 2024, years after the primary patent lapsed. That retention figure is not an accident of brand loyalty. It is the direct output of decisions made in patent prosecution, litigation strategy, and authorized generic deployment that began more than a decade before December 11, 2017, the date Teva’s sildenafil hit U.S. pharmacy shelves.
1. The Patent Cliff Myth: What Actually Happens at LOE
1.1 Generic Erosion Curves: The Industry Baseline
The standard narrative around patent cliffs is numerically accurate but strategically incomplete. HHS/ASPE data confirm that generic drug prices fall roughly 20% below brand levels when approximately three generic competitors are active in a market. With ten or more manufacturers present, erosion deepens to 70-80% within three years of first generic entry. Market share follows a parallel trajectory: brand-name drugs typically lose 80-90% of unit volume within twelve months of the first generic launch.
Those are the averages. Viagra’s trajectory did not follow them, and the divergence is entirely explained by deliberate IP and market management decisions, not by consumer behavior or physician prescribing inertia alone.
1.2 Viagra’s Revenue Profile: The Franchise at Risk
Sildenafil citrate launched as Viagra in 1998 and reached peak U.S. annual revenues of approximately $1.14 billion in 2012. Global sales exceeded $2 billion at peak. Between 2012 and 2018, worldwide Viagra revenues declined by close to 70%, driven primarily by generic entry in the European Union (2013) and sustained price competition in established markets. The U.S. market, still under patent protection through the settlement-derived December 2017 launch date, contributed disproportionately to revenue stability during that six-year window.
For Pfizer’s finance team, the LOE calculus was straightforward: every additional month of U.S. exclusivity at $65-per-pill retail pricing was worth hundreds of millions of dollars in gross profit. The pediatric exclusivity extension alone, which pushed the method-of-use patent from October 2019 to April 2020, represented approximately five to six months of protected U.S. revenue, with an estimated net present value in the range of $200-400 million depending on assumed discount rates and price erosion assumptions.
Key Takeaways: Section 1
Pfizer’s Viagra LOE illustrates that the 80-90% brand market share loss within year one is a median, not a ceiling. Strategic patent layering, authorized generic deployment, and litigation settlement can substantially compress that erosion. IP teams should model their own franchise LOE curves against the HHS/ASPE benchmark and identify the specific levers available to shift outcomes toward the Viagra end of the distribution rather than the typical unmanaged cliff.
2. Sildenafil’s IP Portfolio: Asset Valuation Layer by Layer
2.1 The Compound Patent (US5250534): Cardiovascular IP as a Dual-Purpose Asset
Pfizer filed US5250534 in 1992, covering sildenafil citrate as a compound and its use in cardiovascular disease. The FDA approved sildenafil under the brand name Revatio for pulmonary arterial hypertension (PAH) in 2005. This compound patent expired September 27, 2012.
The IP valuation of US5250534 requires separating its two economic functions. As a compound patent, it provided Pfizer a temporary barrier against generic sildenafil in any indication during its term. As the foundation for Revatio, it supported a distinct revenue stream in PAH with a different payer mix, prescriber base, and formulary position than Viagra. When the compound patent expired in 2012, generic sildenafil for PAH became legally accessible, putting downward pressure on Revatio pricing years before Viagra’s primary patent lapsed.
For IP valuation purposes, US5250534 should be treated as two separate cash-flow assets with distinct LOE dates, discount rates, and competitive exposure profiles. The compound layer had near-zero residual value by 2012. The Revatio indication layer, while tied to the same patent, had more complex dynamics because Revatio’s patient population is smaller, sicker, and more dependent on prescriber relationships, which partially insulated it from generic erosion despite patent expiry.
2.2 The Method-of-Use Patent (US6469012): The Viagra Core Asset
US6469012, filed in 1994 and published in 2002, specifically protects the use of sildenafil citrate for erectile dysfunction. This patent was the primary economic moat for the Viagra franchise in the U.S. market.
From a pure IP asset valuation standpoint, US6469012 was worth substantially more than a compound patent of equivalent remaining term because it controlled a specific, high-demand indication with a large addressable market (estimates put the global ED prevalence at 150 million men), strong brand recognition, direct-to-consumer advertising history, and prescription volumes that made it a material target for generic manufacturers. Method-of-use patents in high-demand consumer indications carry a valuation premium relative to compound or formulation patents because the demand signal is clear and the generic manufacturer’s return on ANDA investment is more predictable.
The initial expiry of October 22, 2019, already gave Pfizer a 27-year commercial window from first filing to LOE, well in excess of the nominal 20-year patent term because the filing predated U.S. commercialization by several years and Pfizer benefited from patent term restoration under 35 U.S.C. 156, which compensates for time lost during FDA review.
2.3 Pediatric Exclusivity as a Revenue Extension Tool
Under the Best Pharmaceuticals for Children Act (BPCA), FDA can request that a company perform pediatric studies on an approved drug. If the company completes those studies to FDA’s satisfaction, it receives six months of pediatric exclusivity that attaches to all existing patents and exclusivities on the product, effectively extending every remaining patent by six months.
Pfizer conducted pediatric testing for Revatio’s use in pulmonary arterial hypertension in children. The successful completion of those studies granted six months of pediatric exclusivity that pushed US6469012’s effective expiry from October 2019 to April 22, 2020. The mechanism applies to all approved applications sharing the active moiety, so the Viagra indication received the extension even though the studies were conducted in the context of Revatio, a separate NDA.
The financial logic behind pursuing pediatric exclusivity is straightforward. The cost of pediatric clinical trials for an already-approved drug, typically in the range of $20-80 million depending on indication complexity, is almost always dwarfed by the NPV of six additional months of exclusivity in a multi-hundred-million-dollar annual revenue market. Pfizer’s pediatric program generated a return-on-investment in extension costs that few R&D investments can match on a risk-adjusted basis.
2.4 The Evergreening Roadmap: How Pfizer Staggered Sildenafil’s IP Timeline
Sildenafil’s IP architecture follows a classic small-molecule evergreening pattern. The compound patent (filed 1992, expired 2012) anchors the timeline. The method-of-use patent for ED (filed 1994, effective expiry April 2020) extends it. Within that window, additional filings covering salt forms, formulation characteristics, dosing regimens, and combination therapies can add further layers of protection.
The broader evergreening toolkit available to any small-molecule franchise includes, but is not confined to, the following instrument classes: formulation patents (modified-release, dosage form innovations, drug delivery mechanisms), polymorph and salt form patents, combination product patents, pediatric exclusivity through BPCA, patent term extension under 35 U.S.C. 156, new chemical entity (NCE) exclusivity for structurally distinct follow-on compounds, and orphan drug designation for rare disease sub-indications.
Pfizer’s sildenafil strategy used a subset of these tools: the compound-to-use patent transition, pediatric exclusivity, and the dual-indication structure (Revatio/Viagra) that created two distinct FDA-regulated products from the same molecule, each with its own regulatory clock and competitive dynamic. What Pfizer did not pursue aggressively, at least not successfully enough to materially delay U.S. generic entry, was a reformulation strategy that might have created an extended-release or ODT (orally disintegrating tablet) variant with its own patent term.
2.5 IP Valuation: Quantifying the Patent Portfolio’s Dollar Value
Pharma IP teams and investors valuing a sildenafil-type patent portfolio should apply a risk-adjusted NPV (rNPV) framework that discounts future cash flows by both time and the probability of patent survival under litigation challenge.
For US6469012 as of 2010, when Teva filed its Paragraph IV certification, the relevant inputs were: remaining patent term (approximately nine years to October 2019, plus potential pediatric extension), annual U.S. net revenues attributable to patent exclusivity (approximately $800 million to $1 billion at the time), probability of patent survival in litigation (high, given the specificity of the method-of-use claim and the absence of clear prior art for sildenafil in ED), and the discount rate applied to pharmaceutical IP (typically 8-12% for established products with clear demand signals).
Under a base-case rNPV model with a 10% discount rate, a 75% probability of surviving litigation, and $900 million in annual U.S. net revenue declining at 5% per year over the nine-year remaining term, the asset value of US6469012 in 2010 was in the range of $4-6 billion. That number provides context for why Pfizer invested aggressively in litigation and settlement rather than accepting uncontrolled generic entry: the asset at stake was massive relative to the cost of defending it.
Key Takeaways: Section 2
Sildenafil’s IP architecture demonstrates that a single molecule can support multiple distinct patent assets with separate expiry dates, litigation risks, and revenue streams. IP teams should maintain a layered patent map for every franchise, with each patent categorized by type (compound, use, formulation, combination), expiry date, prosecution history risk factors, and estimated rNPV contribution. Pediatric exclusivity is consistently underutilized by mid-size pharma as an extension mechanism relative to its cost-to-benefit profile.
Investment Strategy: Section 2
Analysts evaluating a pharma company’s LOE exposure should request a complete Orange Book listing for each major franchise and cross-reference it with Paragraph IV certification history. A compound with only one Orange Book-listed patent and no pending pediatric exclusivity request is at maximum LOE risk. A compound with stacked method-of-use patents, active pediatric trial programs, and formulation patents covering commercially dominant dose forms is substantially more defensible. Sildenafil’s US6469012 extended the U.S. revenue window by approximately seven years beyond what a compound-only patent strategy would have delivered.
3. The Hatch-Waxman Framework: Rules of Engagement for Generic Entry
3.1 ANDA Filing Mechanics and the Paragraph IV Certification
The Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman) governs the procedural mechanics of generic drug market entry in the United States. A generic manufacturer seeking to market a product before the brand’s listed patents expire must file an Abbreviated New Drug Application (ANDA) with a Paragraph IV certification, which formally asserts that the relevant Orange Book-listed patents are either invalid, unenforceable, or will not be infringed by the proposed generic product.
The Paragraph IV filing triggers a specific litigation clock. The brand manufacturer has 45 days from receiving the Paragraph IV notice to file a patent infringement suit. If it does, an automatic 30-month stay goes into effect, during which the FDA cannot grant final approval to the generic ANDA. The 30-month stay is the brand company’s primary statutory delay mechanism and, in competitive markets, can represent $500 million to $2 billion in protected revenues for a single large-volume product.
Teva filed its Paragraph IV certification against US6469012 and initiated the litigation process that Pfizer responded to with its 2010 infringement complaint. The 30-month stay would have held FDA approval in limbo until approximately mid-2013 unless the litigation resolved earlier, which it did via settlement in December 2013.
3.2 The 30-Month Stay: Automatic Delay as a Revenue Shield
The 30-month stay is often described in terms of its burden on generic manufacturers, but brand companies engineer their patent listings with the stay mechanism explicitly in mind. A patent listed in the Orange Book at the time of an ANDA filing activates the 30-month clock. Patents listed after the ANDA is submitted do not trigger an automatic stay, a distinction that incentivizes brand manufacturers to file new patents and update Orange Book listings proactively, before competitive intelligence suggests a Paragraph IV filing is imminent.
For Pfizer, the 30-month stay on US6469012 provided a predictable litigation window. The 2011 trial win confirmed the patent’s validity and enforceability, at which point Pfizer held a court judgment keeping Teva off the market until October 2019. The subsequent settlement decision was therefore not driven by litigation weakness. It reflected a deliberate calculation that a structured entry in December 2017, on royalty-bearing terms, was more valuable than winning the full exclusivity period only to face an uncontrolled multi-generic flood in October 2019.
3.3 180-Day First-Filer Exclusivity: The Generic Applicant’s Prize
Hatch-Waxman grants the first ANDA applicant with a Paragraph IV certification 180 days of generic drug market exclusivity. During this window, the FDA will not approve other generic applications for the same product, creating a period in which the first generic competes only against the brand, not against subsequent generic entrants. In markets with strong demand and high brand pricing, 180-day exclusivity periods can generate $200 million to $1 billion in revenue for the first-filer.
Teva qualified as the first Paragraph IV filer for sildenafil citrate and was entitled to 180-day exclusivity from its launch date of December 11, 2017. In an unmanaged scenario, that 180-day window would have given Teva six months of effective duopoly against branded Viagra. Pfizer’s authorized generic strategy via Greenstone eliminated that duopoly. On day one, the market had Viagra at $65 per pill, Greenstone’s authorized generic at $30-35, and Teva’s independent generic at competitive discount. Teva’s 180-day exclusivity existed on paper but meant almost nothing commercially.
3.4 How Brand Manufacturers Counter Hatch-Waxman Incentives
Authorized generics launched during the 180-day exclusivity window are specifically permitted under Hatch-Waxman. Courts have confirmed that the first-filer exclusivity period applies only to other ANDA applicants, not to the NDA holder’s own generic product. Brand manufacturers exploit this gap systematically. A 2025 RAPS analysis found that the practice of delaying authorized generic launches, as opposed to concurrent launches, is declining, meaning more brand companies are choosing to compete directly against first-filers from day one rather than staying out of the market during the exclusivity window.
The Greenstone launch on December 11, 2017, is a textbook execution of this counter-strategy. Pfizer did not wait for Teva’s 180 days to expire. It priced its own generic below Teva’s street pricing in many pharmacy channels, used its manufacturing infrastructure and quality positioning to establish immediate credibility, and signed a supply agreement with Roman Health (then called Roman), one of the fastest-growing direct-to-consumer telehealth platforms for men’s health, to lock in a digital distribution channel before Teva could negotiate equivalent partnerships.
Key Takeaways: Section 3
Hatch-Waxman’s 180-day exclusivity provision is structurally weaker than it appears for first-filers facing brand manufacturers with active authorized generic strategies. IP teams advising generic clients should model scenarios in which the NDA holder launches its own generic concurrently, effectively collapsing the first-filer premium. Brand-side IP teams should identify the optimal authorized generic launch timing relative to any settled or anticipated Paragraph IV resolution.
4. Patent Litigation Strategy: From Trial Win to Strategic Settlement
4.1 Pfizer v. Teva: The 2010 Complaint and 2011 Trial Win
Pfizer filed its infringement complaint against Teva Pharmaceuticals USA in 2010, within the 45-day Hatch-Waxman window following Teva’s Paragraph IV notice. The case went to trial, and in August 2011 the federal district court ruled in Pfizer’s favor, finding US6469012 valid, enforceable, and infringed by Teva’s proposed generic product. The ruling entitled Pfizer to block Teva’s FDA approval until October 2019, the patent’s original expiry date.
From a pure litigation standpoint, this was an unambiguous win. Pfizer held a court judgment protecting eight more years of U.S. market exclusivity on a $1 billion-per-year revenue franchise. The question that immediately followed was whether holding that judgment to full term was the optimal economic outcome.
4.2 The 2013 Settlement: Why Pfizer Surrendered a Court Victory
In December 2013, Pfizer and Teva announced a confidential settlement allowing Teva to launch a generic version of Viagra in the U.S. on December 11, 2017, approximately twenty-eight months before the patent’s original expiry and roughly thirty-one months before the April 2020 pediatric exclusivity-extended expiry.
In exchange, Teva agreed to pay Pfizer a royalty on generic sildenafil sales. The royalty rate was not disclosed, but industry norms for reverse-payment settlements and royalty-bearing licenses for first-to-file generics entering under a valid patent typically range from 10-25% of generic net revenues, depending on patent strength and remaining term. At even a 10% royalty rate applied against Teva’s expected generic sildenafil revenues in 2017-2020, Pfizer would have captured tens to hundreds of millions of dollars in royalty income it would otherwise have received as brand sales, but with the cost structure stripped out.
The settlement accomplished several objectives simultaneously. It eliminated the risk of patent invalidation on appeal, which, while low given the trial win, was not zero. It gave Pfizer certainty over the LOE date, allowing Greenstone to prepare its authorized generic launch with precision. It delivered a royalty revenue stream that partially offset lost brand sales. And it preempted any attempt by Teva to seek an at-risk launch, which would have been unlikely given the trial outcome but not legally impossible if the Federal Circuit had reversed on appeal.
4.3 The Mylan Settlement: Establishing a Pattern
Pfizer settled separately with Mylan in April 2015, granting Mylan the right to launch its own generic sildenafil on December 11, 2017, also on a royalty-bearing license. By settling with both Teva and Mylan on the same launch date, Pfizer effectively choreographed the initial generic entry date. Rather than a staggered, unpredictable entry driven by separate litigation outcomes, the market saw a coordinated December 11, 2017, opening with Greenstone already in position.
Pfizer retained separate pending litigation against other generic manufacturers. The absence of scheduled trials in those cases suggests Pfizer was using litigation to maintain negotiating leverage rather than seeking additional trial wins. Multiple pending cases with no trial dates is a standard brand-side strategy: keep defendants under litigation cost pressure while negotiating settlements on your preferred terms.
4.4 Reverse Payment Settlements and FTC Scrutiny
The Pfizer-Teva and Pfizer-Mylan settlements were royalty-bearing licenses rather than cash reverse-payment arrangements (commonly called “pay-for-delay”). Under the Supreme Court’s 2013 ruling in FTC v. Actavis, cash payments from brand to generic in settlement agreements are subject to antitrust scrutiny under a rule-of-reason standard. However, the Actavis ruling specifically addressed cash payments, and its application to royalty-bearing licenses, where the generic pays the brand, is less direct.
Pfizer’s structure, where Teva and Mylan pay royalties to Pfizer in exchange for early entry, is facially pro-competitive relative to a scenario in which Pfizer pays generic manufacturers to stay out of the market. The FTC’s post-Actavis enforcement focus has centered primarily on cash reverse-payment arrangements. Royalty-bearing early-entry licenses occupy a more legally comfortable position, provided the royalty rate does not effectively eliminate generic price competition, which in sildenafil’s case it clearly did not, given the aggressive pricing at launch.
4.5 Litigation vs. Settlement NPV: A Decision Framework for IP Teams
IP teams modeling the litigation-versus-settlement decision for a patent analogous to US6469012 should build a decision tree with at least four nodes: full litigation win held to expiry, litigation win followed by at-risk launch attempt, settlement with controlled early entry, and litigation loss. Each node requires a probability estimate, a revenue model, and a cost structure.
For Pfizer in 2013, the most likely scenario without settlement was a Federal Circuit affirmance of the district court win, followed by a controlled October 2019 expiry with an immediate multi-generic flood. The settlement moved generic entry to December 2017 but on royalty-bearing terms, with Greenstone ready to capture market share from day one. The royalty income, combined with Greenstone’s revenue and the avoided multi-generic collapse in 2019, likely made the settlement NPV-positive relative to holding the judgment, particularly once the authorized generic strategy is incorporated into the model.
Key Takeaways: Section 4
Winning a Paragraph IV trial does not mean holding the judgment to expiry is the optimal strategy. IP teams should evaluate every major litigation win through a post-trial settlement lens, modeling the NPV of controlled early entry against the NPV of maximum exclusivity. The Pfizer-Teva precedent shows that a royalty-bearing settlement can deliver superior risk-adjusted returns compared to eight years of additional litigation risk management on a patent with a hard expiry date.
Investment Strategy: Section 4
Investors should treat a brand manufacturer’s Paragraph IV litigation win as the beginning of the settlement negotiation phase, not the end of the competitive threat. The Pfizer precedent also illustrates that settlement announcement dates, not trial outcomes, are the operative catalyst for modeling generic entry NPV. When a brand-side press release announces a royalty-bearing license to a first-filer, analysts should immediately revise their LOE date models and evaluate whether an authorized generic is positioned for concurrent launch.
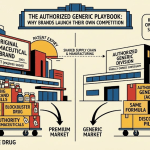
5. The Authorized Generic Weapon: Greenstone’s Defensive Cannibalization
5.1 What Authorized Generics Are and How They Work
An authorized generic (AG) is a product sold by the NDA holder (or its subsidiary) under a generic drug approval, without the brand name, at a lower price than the branded product. The AG is chemically identical to the brand, manufactured at the same facility, and approved under the original NDA rather than an ANDA. Because it is approved under the NDA, it does not require its own Paragraph IV certification and does not activate the 180-day exclusivity clock in favor of any other applicant.
The strategic value of an AG is twofold. First, it captures market share from price-sensitive patients and payers who would otherwise switch to an independent generic. Second, it limits the revenue upside for Paragraph IV filers during the 180-day exclusivity window, reducing their return on the investment in patent challenge litigation and, by extension, reducing the incentive for future challengers to invest in Paragraph IV filings against the same or similar patents.
5.2 Greenstone LLC: Pfizer’s Generic Subsidiary as a Strategic Vehicle
Greenstone LLC is Pfizer’s authorized generic subsidiary. It holds agreements with McKesson, AmerisourceBergen, and other major drug distributors under which it markets authorized generic versions of off-patent Pfizer products. Greenstone has operated this function for multiple Pfizer franchises, making it a standing infrastructure asset rather than a vehicle created specifically for sildenafil.
The existence of Greenstone as a pre-established entity meant Pfizer did not need to build authorized generic manufacturing, regulatory, and distribution capability from scratch for sildenafil. The infrastructure, distribution agreements, and sales force capable of reaching pharmacy networks were already in place. This operational readiness enabled the December 11, 2017, concurrent launch with Teva without the delays that would have accompanied a first-time AG build-out.
From an IP valuation and strategic planning perspective, Greenstone represents a residual asset extraction mechanism: a vehicle that converts expiring brand IP into continuing cash flow through the generic market rather than leaving that revenue entirely to independent manufacturers.
5.3 The December 11, 2017 Launch: Diluting Teva’s 180-Day Exclusivity
Teva’s FDA approval for generic sildenafil citrate came on March 9, 2016 (tentative approval, reflecting that final approval was blocked pending the settlement-derived December 11, 2017, launch date). Teva’s ANDA qualified it as the first Paragraph IV filer, entitling it to 180 days of generic exclusivity beginning on its first commercial marketing date.
Greenstone’s concurrent December 11, 2017, launch did not legally violate Teva’s 180-day exclusivity, because that exclusivity blocks other ANDAs, not the NDA holder. It did commercially gut the exclusivity’s value. Instead of competing only against $65-per-pill branded Viagra during its 180-day window, Teva competed against Greenstone’s $30-35 authorized generic from hour one. Independent pharmacies and pharmacy benefit managers negotiating contracts for the first-filer exclusivity window found that the AG immediately suppressed any premium Teva might have extracted during a true duopoly period.
The academic literature on AGs during 180-day exclusivity windows (see Alam and Conti, “Entry Delays and Fighting Brands”) confirms this effect systematically: AG concurrent launches during first-filer exclusivity windows reduce the first-filer’s revenues by 40-60% compared to scenarios without an AG. For Teva, the value of its first-filer status in sildenafil was reduced to a fraction of what it would have been without Greenstone’s presence.
5.4 The Roman Partnership: Telehealth as a Distribution Strategy
In January 2020, Greenstone entered an exclusive supply agreement with Roman Health (Roman), at the time one of the fastest-growing digital men’s health platforms in the United States. Under the agreement, Roman members received access to Greenstone’s sildenafil as the only FDA-approved authorized generic version of Viagra available through the platform.
The Roman partnership addressed a specific distribution gap. Greenstone’s traditional pharmacy distribution through AmerisourceBergen and McKesson positioned it well in brick-and-mortar retail pharmacy. The telehealth channel, where patients receive prescriptions via asynchronous consultation and drugs delivered by mail, was growing rapidly in the ED category and appealed to a demographic, notably men aged 25-45, who preferred online consultation over in-person physician visits for a stigmatized condition.
By partnering exclusively with Roman rather than licensing its AG to any telehealth platform, Greenstone secured a preferred distribution position in a high-growth channel while the partnership’s exclusivity gave Roman a product differentiation story (the “only FDA-approved authorized generic version of Viagra”) that its competitors, who dispensed independent generics, could not match on the same terms.
5.5 Authorized Generic IP Valuation: Residual Asset Extraction
IP teams and investors rarely model the authorized generic revenue stream as a distinct component of a patent portfolio’s terminal value. They should. In the sildenafil case, the AG strategy extended Pfizer’s effective revenue participation in the sildenafil market well beyond the April 2020 patent expiry date. Greenstone continued operating in the market post-April 2020, competing on a level field with all independent generics but retaining advantages in manufacturing quality positioning, distribution relationships, and brand-adjacent trust.
The residual cash flow from an authorized generic post-patent expiry is a declining but non-zero asset. In high-volume markets like sildenafil, where tens of millions of prescriptions are written annually and price erosion slows as the market approaches a competitive floor, AG revenues in years three through seven post-LOE can still be material, particularly if the AG maintains distribution relationships with large pharmacy chains that prefer to limit the number of generic suppliers they carry.
5.6 When Authorized Generics Backfire: Pricing Floor Risks
The Greenstone launch at $30-35 per pill set a pricing anchor for the market. Independent generics, for competitive and reimbursement positioning, needed to price below or near that level. Telehealth platforms priced even lower, with Ro, Hims, and Lemonaid Health offering sildenafil at $2-10 per dose by 2020. The AG accelerated price discovery on the downside, contributing to rapid erosion toward the competitive floor.
Brand companies considering AG strategies should model this dynamic explicitly: an early AG launch with aggressive pricing speeds total market price erosion, which eventually limits the AG’s own revenue potential. The optimal AG strategy balances maximum market share capture against the risk of triggering a pricing race to the bottom that compresses margins across all market participants, including the AG itself.
Key Takeaways: Section 5
The authorized generic strategy is the most potent defensive tool available to a brand manufacturer at LOE. Concurrent AG launch on the first-filer’s exclusivity start date is systematically superior to delayed AG launch because it limits the competitor’s first-mover revenue advantage from the outset. Greenstone’s sildenafil execution is the canonical example: concurrent launch, aggressive pricing, strategic telehealth partnership, and manufacturing quality positioning as a differentiator against independent generics.
Investment Strategy: Section 5
When evaluating a pharma company approaching a major LOE event, analysts should determine whether the company has an authorized generic subsidiary (or equivalent contractual arrangement with a generic manufacturer). The presence of a pre-established AG vehicle, comparable to Greenstone for Pfizer, suggests the company will defend market share more aggressively post-LOE than a company without one. Model the AG revenue as a separate line item in post-LOE revenue forecasts, with an initial market share assumption of 15-30% of total generic volume and a decay curve reflecting price erosion and competitive entry from additional ANDA holders.
6. Global IP Fragmentation: Jurisdiction-by-Jurisdiction LOE Analysis
6.1 European Union: 2013 Expiry and the Pace of Accelerated Erosion
The EU patent covering sildenafil’s use in erectile dysfunction expired in 2013, seven years before the U.S. expiry date. Generic manufacturers across EU member states launched sildenafil products within months of that expiry, triggering the standard European LOE pattern: rapid price erosion driven by reference pricing systems and mandatory generic substitution laws in markets like Germany, the Netherlands, and France.
The European experience gave Pfizer’s commercial teams an early-stage data set on sildenafil price erosion curves, payer behavior, and branded product resilience. In markets where direct-to-consumer advertising is more restricted than in the United States, brand retention rates post-LOE tend to be lower, as physician prescribing habits shift more quickly toward generic substitution. Pfizer’s ability to observe EU market dynamics from 2013 to 2017 presumably informed its U.S. authorized generic pricing and distribution strategy.
6.2 Canada: Patent Invalidation as a Worst-Case Scenario
The Canadian Viagra litigation offers the sharpest possible contrast with the U.S. outcome. In November 2012, the Supreme Court of Canada (Teva Canada Ltd v Pfizer Canada Inc) invalidated Pfizer’s Canadian Viagra patent, holding that Pfizer’s disclosure in the original application was insufficient to support the claims. The court found that Pfizer had identified sildenafil as the effective compound in its patent claims without adequately disclosing that finding in the patent specification, a disclosure failure under Canadian patent doctrine.
The decision had immediate commercial consequences. Teva Canada launched generic sildenafil (Novo-Sildenafil) on the same day the court issued its ruling. Pfizer, unable to rely on any remaining patent protection, cut the Canadian price of branded Viagra to remain marginally competitive. The Canadian scenario illustrates the worst-case LOE outcome: not a managed cliff with settlements and authorized generics, but an instantaneous court-ordered drop into full generic competition with no transitional revenue protection.
For pharma IP teams, the Canadian case is a disclosure quality warning. Patent prosecution teams in major markets must ensure that the disclosure supporting key claims, particularly method-of-use claims on high-value indications, is complete and unambiguous. The cost of inadequate disclosure is patent invalidation, and the financial damage from invalidation in a major market can exceed the cost of thorough prosecution by orders of magnitude.
6.3 Brazil: Early LOE and Compulsory License Dynamics
Pfizer’s sildenafil patent expired in Brazil in 2010, eight years before generic entry in the United States. Brazil’s patent framework includes compulsory license provisions that allow the government to authorize generic production of patented drugs deemed essential to public health. While no compulsory license was issued specifically for sildenafil, the broader compulsory license environment in Brazil shapes brand companies’ pricing and market access strategies even absent a formal compulsory license order.
The Brazilian LOE in 2010 established a large generic sildenafil market in a country with approximately 215 million people and significant unmet demand for ED treatment, particularly in income segments priced out of Viagra at brand pricing. Local generic manufacturers entered quickly, and by 2017 the Brazilian market had multiple generic producers with established distribution networks and competitive pricing structures.
6.4 China: Trademark Management in a Market with Contested IP Enforcement
China represents a structurally different IP challenge. Pfizer’s sildenafil patent claims faced inadequate enforcement in the Chinese market, and widespread manufacture and distribution of sildenafil-containing products outside the formal regulatory approval pathway remained a persistent feature of the market. Pfizer countered this primarily through trademark strategy rather than patent enforcement, positioning Viagra as a quality-assured product in a market where counterfeit products posed genuine safety risks to consumers.
The brand equity strategy worked to a degree: Viagra’s global Q3 2020 sales showed a 29% sequential increase, driven significantly by Chinese market recovery post-COVID-19 lockdowns. China was a revenue source that mitigated U.S. and EU sales declines. But the IP enforcement landscape in China forced Pfizer to compete on brand trust and quality signals rather than the patent exclusivity that defined its U.S. strategy.
6.5 Structuring a Global LOE Defense Strategy
A global LOE defense strategy for a product with sildenafil-type exposure requires different instruments in each jurisdiction. In the United States, the Hatch-Waxman framework, authorized generic capability, and settlement mechanics provide the primary tools. In the EU, reference pricing systems and mandatory substitution laws mean LOE management centers on managed market contracting, hospital tender strategy, and potentially an authorized generic launched through a wholly-owned European entity. In countries with compulsory license risk (Brazil, India, South Africa), the primary defense is proactive access pricing before government intervention forces a less favorable outcome. In markets with enforcement challenges (China), brand integrity and pharmacovigilance become competitive differentiators.
A single unified LOE strategy does not work across all jurisdictions. Companies that apply a U.S.-centric authorized generic and litigation settlement playbook to markets with different IP regimes consistently underperform in those markets.
Key Takeaways: Section 6
Sildenafil’s IP expiry was not a single global event. It was a sequence of jurisdiction-specific LOE episodes spanning from 2010 (Brazil) to 2020 (U.S. with pediatric exclusivity). Each market required distinct defensive instruments. The Canadian invalidation illustrates that poor patent prosecution disclosure creates risk that no amount of downstream litigation strategy can fully mitigate. IP teams should map every major patent in their portfolio against jurisdiction-specific disclosure standards, not just U.S. prosecution doctrine.
7. Post-LOE Market Dynamics: Pricing Erosion, Competitive Structure, and Telehealth Disruption
7.1 Price Erosion Curves: The HHS/ASPE Benchmark
The HHS/ASPE generic competition analysis provides the clearest benchmark for generic pricing dynamics by number of competitors. At one generic entrant, prices tend to be 20-30% below brand levels. At three entrants, prices reach 30-50% below brand. At ten or more competitors, prices settle 70-80% below brand within three years of first entry.
Generic sildenafil followed this curve but with an unusual starting point. Greenstone’s $30-35 authorized generic launch on December 11, 2017, anchored the market at roughly 50% of branded Viagra’s $65-per-pill retail price on day one, before any independent generic had a chance to establish pricing. This compressed the erosion curve’s early phase: instead of beginning at 20-30% below brand (as typical single-entrant scenarios), the market opened at 50% below brand because the NDA holder set the initial anchor.
By June 2018, seven or eight ANDA-holders had received FDA approval and were commercially active. By mid-2019, the number exceeded ten. At that point, telehealth platforms, dispensing sildenafil without pharmacy markups and with their own negotiating leverage, drove street-level pricing to $2-10 per dose in the direct-to-consumer channel. The HHS 70-80% erosion benchmark was reached in the telehealth channel within eighteen months of first generic entry.
7.2 Telehealth Disruption: Ro, Hims, and Lemonaid Health
The ED category was one of the first major therapeutic areas where direct-to-consumer telehealth platforms achieved meaningful scale. Ro, Hims, and Lemonaid Health all launched or expanded ED product lines within the 2017-2020 window, coinciding with sildenafil’s generic availability.
These platforms operate a vertically integrated model: online consultation, prescription issuance by a licensed prescriber, and generic drug dispensing through affiliated pharmacies, all delivered via mail. The model eliminates traditional pharmacy retail markups, reduces prescriber visit costs for patients, and creates a subscription-like revenue model with high retention among adherent patients.
Ro’s pricing for sildenafil started at approximately $4 per dose. Hims launched at comparable levels. Lemonaid Health offered sildenafil at $2 per pill, a level that reflects generic manufacturing costs (estimated at $0.05-0.20 per tablet for commodity small molecules like sildenafil) plus platform margin but stripped of every intermediate margin layer in the traditional supply chain. Greenstone’s partnership with Roman addressed this channel directly, giving Pfizer a presence in the DTC telehealth segment that independent generic manufacturers, without equivalent brand positioning, could not match on equal terms.
7.3 Viagra Franchise Market Share Retention: The 45.35% Story
The combined Viagra franchise, encompassing both branded Viagra and Greenstone’s authorized generic, held a 45.35% share of the total ED drugs market in 2024, according to Mordor Intelligence data. That figure, years after the primary patent expiry and amid a crowded generic market with ten-plus manufacturers and aggressive telehealth pricing, is substantially above what an unmanaged LOE would have produced.
For context: in markets where brand companies lack authorized generic capability and face uncontrolled multi-generic entry, brand-plus-AG market share typically falls to 10-20% within three years of first generic entry. The 45.35% figure reflects a combination of brand equity, authorized generic market capture, and the Mordor methodology (which may define market differently from pure prescription volume), but it still represents an outlier retention rate by any reasonable benchmark.
7.4 The PDE5 Inhibitor Competitive Landscape: Tadalafil and Vardenafil
Sildenafil does not compete in isolation. Tadalafil (Cialis, Eli Lilly) and vardenafil (Levitra, Bayer) are the other FDA-approved PDE5 inhibitors in the ED indication. Generic tadalafil entered the U.S. market in late 2017-2018, concurrent with sildenafil’s generic entry. Avanafil (Stendra, Vivus) holds a smaller market position.
Tadalafil’s competitive differentiation in the branded era rested on its 36-hour duration of action and its dual approval for ED and benign prostatic hyperplasia (BPH). The BPH indication, with its distinct prescriber base (urologists) and payer coverage pathway, provided tadalafil with a revenue stream partially insulated from ED generic competition. Generic tadalafil, launched by Lilly’s authorized generic subsidiary and multiple ANDA holders, followed a pricing trajectory similar to sildenafil.
The net effect for the PDE5 class as a whole was a dramatic reduction in per-episode treatment cost across all approved compounds. Telehealth platforms often offer bundled or subscription access to multiple PDE5 inhibitors, enabling patient switching based on individual response rather than cost. This dynamic compressed any remaining inter-product pricing differential and made the entire PDE5 class a commodity category at the generic level.
7.5 Sildenafil Market Projections: USD 4.4 Billion by 2037
Research Nester projects the global sildenafil citrate market at USD 4.4 billion by the end of 2037. The growth driver is not price: generic competition has permanently reduced average sildenafil prices by 60-80% from branded Viagra peak levels. Volume is the primary growth vector, driven by expanding awareness of ED as a treatable condition, particularly in emerging markets where stigma is decreasing and healthcare access is improving, aging demographics in developed markets, and continued adoption of telehealth delivery models that reduce barriers to diagnosis and treatment.
For investors, the sildenafil market at USD 4.4 billion is a volume story, not a margin story. The relevant investment exposures are manufacturing-scale generic producers with cost leadership in bulk sildenafil API synthesis (primarily Indian and Chinese API manufacturers), telehealth platforms with high patient lifetime value in men’s health categories, and distribution networks with formulary position for high-volume generic prescriptions.
Key Takeaways: Section 7
The ED market post-Viagra LOE restructured around two dynamics: commodity generic pricing driven by manufacturer competition, and direct-to-consumer telehealth disruption that compressed pharmacy markups and accelerated price erosion below HHS baseline projections. Brand companies entering LOE transitions in consumer-health-adjacent categories should proactively model telehealth channel dynamics and evaluate pre-LOE partnerships with DTC platforms as a mechanism to capture volume that would otherwise go to independent generics.
Investment Strategy: Section 7
Analysts covering telehealth platforms in therapeutically adjacent categories (sexual health, dermatology, mental health) should examine the degree to which generic drug pricing provides a structural tailwind to platform growth. At $2-4 per dose, sildenafil enabled Ro and Hims to price their total offering (consultation plus drug) at levels accessible to far broader income demographics than branded Viagra at $65 per pill. Generic LOE events in high-stigma, high-demand categories are effectively customer acquisition catalysts for DTC health platforms.
8. The Evergreening Technology Roadmap: A Replicable Model for Small-Molecule Franchises
8.1 Formulation Patents: Modified Release, Dosage Form, and Delivery System IP
Sildenafil is dosed as an immediate-release tablet. A modified-release formulation, whether extended-release or pulsatile-release, would in principle support a distinct patent layer with a later expiry date and potentially clinical differentiation (smoother plasma concentration curve, reduced adverse events, different dosing interval). Pfizer did not successfully commercialize a modified-release sildenafil product that achieved meaningful market penetration, which left one of the standard evergreening instrument classes unused in the sildenafil portfolio.
Generic-targeting IP teams should audit any incumbent brand’s formulation patent strategy as part of Paragraph IV challenge assessment. The absence of a commercially successful modified-release formulation in a high-volume oral solid dosage market is often a signal that the evergreening attempt either failed in clinical development, was not pursued, or was pursued but not broadly adopted. Any of those three scenarios weakens the brand’s post-LOE defensive posture.
8.2 Salt and Polymorph Patents
Sildenafil citrate, the salt form commercialized in Viagra, is itself a formulation decision: sildenafil as a free base has different solubility and stability properties. Additional patents on specific polymorph forms of sildenafil citrate, with particular X-ray diffraction patterns, melting points, and dissolution profiles, are possible but create defensive value only if the commercial product relies on a specific polymorph that generic manufacturers cannot readily avoid. Pfizer did not successfully use polymorph patents to extend sildenafil exclusivity in the U.S. market.
8.3 Method-of-Use Patents and New Indication Filing
The most productive evergreening instrument in Pfizer’s sildenafil portfolio was the method-of-use patent structure itself. By obtaining separate patents for the cardiovascular indication (US5250534) and the ED indication (US6469012), Pfizer created two distinct expiry dates for the same molecule. The Revatio-for-PAH indication created a smaller but distinct revenue stream with different competitive dynamics.
The replicable lesson is that new indication development, particularly in orphan or specialty segments where clinical trials are smaller and faster, can add method-of-use patents with later expiry dates to an existing molecule’s portfolio. The critical requirement is that the new indication must be clinically meaningful enough to attract prescribers and payers, not merely a paper indication designed to generate a new patent without commercial intent.
8.4 Pediatric Exclusivity Under BPCA and PREA
The Best Pharmaceuticals for Children Act (BPCA) and the Pediatric Research Equity Act (PREA) create two pathways for pediatric exclusivity. Under BPCA, the FDA issues a Written Request to a sponsor to conduct pediatric studies; completion earns six months of exclusivity attached to all existing patents. Under PREA, FDA can require pediatric studies as a condition of approval for new indications in adult populations; compliance earns no additional exclusivity but avoids a refusal to file.
Pfizer’s sildenafil/Revatio pediatric program was a BPCA-type engagement that generated the six-month exclusivity extension pushing US6469012 from October 2019 to April 2020. The financial return per dollar invested in pediatric trials for an established high-revenue franchise is almost always positive. Every company with a blockbuster product that has not yet pursued pediatric exclusivity should evaluate whether an FDA Written Request exists or could be requested for any currently approved indication.
8.5 Patent Term Extension Under 35 U.S.C. 156
35 U.S.C. 156 permits the holder of a product patent to apply for a patent term extension (PTE) to compensate for time lost during FDA regulatory review. The maximum PTE is five years, and the extended patent term cannot exceed 14 years from the date of FDA approval. PTE applies only to one patent per product, and the patent holder must apply within 60 days of FDA approval.
For sildenafil, the PTE mechanism contributed to the gap between the nominal 20-year patent term from filing date and the actual expiry date. US6469012, filed in 1994 and expiring in 2019, reflects both the nominal term and the regulatory review time Pfizer was compensated for. Companies that file PTAs (patent term adjustments, a related but distinct mechanism under 35 U.S.C. 154) for prosecution delays can add additional days to their patent term, sometimes cumulatively significant in a slow prosecution history.
8.6 Risk-Adjusted Evergreening Timeline: Building a Sildenafil-Style Model
An IP team building a comprehensive evergreening timeline for a current blockbuster should map each available instrument against three dimensions: estimated additional term generated, probability of successful execution (prosecution success, clinical success, regulatory success), and capital required. The output is a risk-adjusted LOE curve that replaces a single hard expiry date with a probability-weighted portfolio of extension scenarios.
For a hypothetical molecule with a primary compound patent expiring in Year 1, the risk-adjusted model might include: a method-of-use patent for a new indication with 60% probability of grant and an additional 4-year term; a modified-release formulation patent with 45% probability of successful clinical differentiation and 6-year additional term if successful; pediatric exclusivity with 85% probability of successful completion and 0.5-year term extension; and a PTE application with 90% probability and 2-year term extension. Each scenario requires its own probability adjustment and NPV calculation before the instruments can be ranked by expected value and incorporated into a strategic IP roadmap.
Key Takeaways: Section 8
Evergreening is not a single tactic; it is a portfolio management discipline that requires parallel investment in multiple extension instruments with varying probabilities and returns. Pfizer’s sildenafil execution used method-of-use separation (cardiovascular vs. ED), pediatric exclusivity, and patent term extension, while leaving formulation innovation, polymorph protection, and combination therapy patents either unused or insufficiently developed to contribute materially to LOE defense. Companies that systematically pursue all applicable extension instruments, rather than relying on a single strategy, produce higher expected values from their patent portfolios and smoother LOE transitions.
9. Lessons for IP Teams and R&D Strategy Directors
9.1 Build the Patent Stack Early
Pfizer’s sildenafil portfolio was structured over the 1992-1994 window, before the compound even had an indication, let alone a blockbuster revenue trajectory. The method-of-use patent for ED was filed in 1994, two years after the compound patent and four years before FDA approval. IP teams that file method-of-use patents reactively, after commercial launch, give up years of patent term during the highest-revenue phase of the product lifecycle. The correct approach is to file method-of-use claims as early as the clinical evidence supports them, even if that means filing before peak commercial projections are visible.
9.2 Treat Litigation as a Negotiation Tool, Not Just a Defense
The Pfizer-Teva sequence illustrates that patent litigation in the Hatch-Waxman context is primarily a negotiation mechanism with a court proceeding as its backdrop. Brand companies that pursue Paragraph IV litigation solely to win an injunction, rather than to establish a negotiating position for a favorable settlement, underutilize the strategic optionality that a strong litigation posture creates. A trial win, as Pfizer demonstrated, is most valuable as the leverage that produces a controlled settlement rather than as an endpoint in itself.
9.3 Pre-Position the Authorized Generic Infrastructure
Greenstone’s concurrent December 11, 2017, launch was possible because the authorized generic infrastructure, including manufacturing, regulatory filings, distribution agreements, and pricing strategy, was prepared well in advance of the settlement-derived launch date. Companies that wait until settlement announcement to begin building their authorized generic capability will miss the concurrent launch window and hand the first-filer a de facto exclusivity period that regulatory law does not require.
9.4 Model Telehealth as a Structural Force, Not a Channel Adjunct
The Ro and Hims market entry in sildenafil was not a peripheral channel development. It restructured the competitive floor for the entire ED category by eliminating pharmacy markup as a component of patient cost. Any brand or authorized generic strategy for a consumer-health-adjacent product with significant out-of-pocket payment must incorporate telehealth platform dynamics into both pricing models and distribution strategy. The Greenstone-Roman partnership was a recognition of this reality. Companies that ignore the telehealth channel in their LOE planning will consistently underestimate post-LOE price erosion.
9.5 Globalize the IP Strategy, Not Just the Commercial Footprint
Canada’s patent invalidation and Brazil’s early LOE produced LOE outcomes that were commercially painful and strategically preventable with better disclosure practice and proactive generic market preparation. A global pharmaceutical company’s IP strategy should be integrated across jurisdictions at the prosecution stage, not coordinated reactively when a specific market faces a legal challenge. Disclosure standards differ across major markets; patent prosecution teams must write applications that satisfy the most demanding standards in their target markets, not just the USPTO’s.
10. Investment Strategy: What Sildenafil’s LOE Tells Analysts About Future Patent Cliffs
10.1 Identifying the Next Managed Cliff
Sildenafil’s LOE management outperformed typical patent cliff outcomes on market share retention, revenue stability, and transition smoothness. The instruments Pfizer used, stacked patents, pediatric exclusivity, litigation-to-settlement strategy, concurrent authorized generic launch, and telehealth distribution partnership, are all available to any major pharmaceutical company facing LOE in the next five to seven years.
The key analytical question for investors is which companies approaching major LOE events have pre-positioned equivalent defensive infrastructure. A company with a major LOE in 2026-2028 and no authorized generic subsidiary, no pending pediatric exclusivity application, and no telehealth distribution partnerships is structurally exposed to a harder LOE curve than Pfizer experienced with Viagra.
10.2 Variables That Predict LOE Severity
Analysts should evaluate LOE-exposed compounds on at least five dimensions: patent stack depth (number of Orange Book-listed patents, remaining terms, and litigation history), authorized generic capability (existing subsidiary or contractual arrangement with a generic partner), Paragraph IV certification history (number of ANDA filers certified, litigation status, and settlement terms), telehealth channel exposure (whether the indication is served by DTC telehealth platforms that will accelerate generic adoption), and global LOE synchrony (whether U.S. expiry is the first or last major market LOE event).
A compound with a single Orange Book patent, no AG capability, multiple pending Paragraph IV certifications without settlement, strong telehealth channel penetration in the indication, and simultaneous global LOE events will experience sharper revenue decline than a compound with Viagra-type patent architecture. That spread in predicted LOE severity should be reflected in earnings model assumptions and target price derivations.
10.3 The Royalty Stream Valuation
When a brand manufacturer announces a settlement with a first-filer Paragraph IV generic, analysts should model the royalty stream as an above-the-line revenue contribution, not merely a below-the-line settlement cost recovery. Pfizer received royalties from both Teva and Mylan on sildenafil generic sales from December 2017 until the patent expiry. At even a 10% royalty rate on combined Teva and Mylan generic net revenues in the 2017-2020 period, that royalty contribution was likely in the range of $50-150 million annually, partially offsetting brand revenue declines.
Royalty streams from patent settlements are often not separately disclosed in company financials. Analysts should probe settlement announcement language for royalty structure details and build royalty contribution estimates into their LOE models rather than treating settlements purely as events that reduce patent exclusivity duration.
10.4 Authorized Generic Revenue as a Post-LOE Floor
Greenstone’s sildenafil AG revenue, while declining post-April 2020 as independent generic competition intensified, represents a post-LOE revenue floor that persists well beyond patent expiry. For modeling purposes, the AG revenue floor in a commodity generic market is approximately equal to the AG’s market share percentage multiplied by total market volume multiplied by the AG’s net selling price, which will typically be 15-25% above the lowest-cost independent generic due to quality positioning and distribution relationships.
Building this AG revenue floor into post-LOE models, rather than assuming total brand and AG revenue collapse to zero within two years of first generic entry, will consistently produce more accurate long-term earnings estimates for companies with active AG strategies.
Comprehensive Patent and Market Data Tables
Sildenafil U.S. Patent Expiry Timeline
| Patent | Indication | Original Expiry | Extension | Effective Expiry | Estimated rNPV (2010) |
|---|---|---|---|---|---|
| US5250534 | Cardiovascular (Revatio) | Sep 27, 2012 | None | Sep 27, 2012 | ~$300-500M |
| US6469012 | Erectile Dysfunction (Viagra) | Oct 22, 2019 | Pediatric exclusivity (+6 months) | Apr 22, 2020 | ~$4-6B |
Global LOE Timeline for Sildenafil
| Market | LOE Event | Date | Driver | Outcome |
|---|---|---|---|---|
| Brazil | Patent expiry | 2010 | Normal patent term | Generic market entry, local manufacturers |
| Canada | Patent invalidation | Nov 2012 / Apr 2013 | Supreme Court (Teva Canada v Pfizer Canada) | Same-day generic launch by Teva Canada; Pfizer price cuts |
| European Union | Patent expiry (use) | 2013 | Normal patent term | Multi-country generic entry; reference pricing erosion |
| United States | Settlement-derived entry | Dec 11, 2017 | Pfizer-Teva and Pfizer-Mylan settlements | Controlled entry; concurrent AG; royalty stream |
| United States | Primary patent expiry | Apr 22, 2020 | Pediatric exclusivity end | Full generic market; multi-manufacturer competition |
U.S. Generic Sildenafil Market Entry Timeline
| Manufacturer | Type | FDA Approval | Launch Date | Notes |
|---|---|---|---|---|
| Teva Pharmaceuticals | Independent ANDA | Mar 9, 2016 (tentative) | Dec 11, 2017 | First Paragraph IV filer; royalty-bearing license |
| Greenstone (Pfizer) | Authorized Generic | NDA-based | Dec 11, 2017 | Concurrent AG launch; $30-35/pill anchor pricing |
| Mylan | Independent ANDA | Mar 25, 2019 | Dec 11, 2017 (per settlement) | Royalty-bearing license; April 2015 settlement |
| Aurobindo Pharma | Independent ANDA | Jun 11, 2018 | Post-exclusivity | Standard ANDA approval |
| Hetero Labs | Independent ANDA | Jun 11, 2018 | Post-exclusivity | Standard ANDA approval |
| Amneal Pharms | Independent ANDA | Jun 27, 2018 | Post-exclusivity | Standard ANDA approval |
| Torrent Pharmaceuticals | Independent ANDA | Jun 11, 2018 | Post-exclusivity | Standard ANDA approval |
| Rubicon Research | Independent ANDA | Jun 11, 2018 | Post-exclusivity | Standard ANDA approval |
| Chartwell Rx | Independent ANDA | Mar 22, 2019 | Post-exclusivity | Standard ANDA approval |
| APPCO Pharma | Independent ANDA | Mar 2, 2020 | Post-exclusivity | Standard ANDA approval |
| Reyoung Pharmaceutical | Independent ANDA | Jun 12, 2020 | Post-exclusivity | Standard ANDA approval |
| Novitium Pharma | Independent ANDA | Aug 29, 2023 | Post-exclusivity | Standard ANDA approval |
| Cadila Pharmaceuticals | Independent ANDA | Nov 28, 2023 | Post-exclusivity | Standard ANDA approval |
| Jubilant Generics | Independent ANDA | Dec 20, 2024 | Post-exclusivity | Standard ANDA approval |
Sildenafil Pricing Structure: Launch to 2024
| Product | Manufacturer | Price per Dose at Launch | Price Range by 2020-2024 | Channel |
|---|---|---|---|---|
| Viagra (brand) | Pfizer / Viatris | ~$65 (2017) | ~$65 (brand retained) | Traditional pharmacy |
| Greenstone sildenafil | Greenstone LLC | $30-35 (Dec 2017) | Declining | Traditional pharmacy; Roman (exclusive) |
| Teva sildenafil | Teva | Deep discount vs. brand | Continuing decline | Traditional pharmacy |
| Sildenafil (Ro) | Ro / Roman Health | ~$4-10 | $4-10 | DTC telehealth, mail order |
| Sildenafil (Hims) | Hims & Hers | Starting at ~$4 | $4-8 | DTC telehealth, mail order |
| Sildenafil (Lemonaid) | Lemonaid Health | ~$2 | ~$2 | DTC telehealth, mail order |
Conclusion
Pfizer’s sildenafil LOE is not a story about brand loyalty overcoming generic competition. It is a story about sequential, deliberate IP and market decisions made across a thirty-year period, from the 1992 compound patent filing through the 2020 pediatric exclusivity end date, that systematically reduced the severity of a patent cliff that should, by industry averages, have been catastrophic.
The instruments Pfizer used are well-documented, individually available to any major pharmaceutical company, and collectively underutilized by most LOE-exposed franchises. Stacked method-of-use patents, pediatric exclusivity, strategic litigation followed by royalty-bearing settlement, concurrent authorized generic launch through a pre-established subsidiary, and a telehealth distribution partnership that captured the fastest-growing consumer channel in the indication: each of these decisions added value that accumulated into a market share retention figure, 45.35% of the combined ED market through 2024, that peers without equivalent strategies could not have achieved.
For IP teams, the Viagra case demands a portfolio management discipline: map every extension instrument for every major franchise, quantify the rNPV contribution of each, and invest in the highest-return instruments early, particularly pediatric exclusivity and method-of-use filing for new indications, where the cost-to-benefit ratio is consistently favorable. For investors, it demands a more nuanced LOE model: one that incorporates patent stack depth, authorized generic positioning, royalty stream revenue, and telehealth channel dynamics into every patent cliff analysis, rather than defaulting to a generic 80% brand market share loss within year one.
The sildenafil market is projected to reach USD 4.4 billion by 2037. Pfizer, through Greenstone and the brand, will hold a portion of that market. That persistence, years after the last patent expired, is the measured output of decisions made in a patent attorney’s office in 1992 and 1994, refined in a courtroom in 2011, and executed on a pharmacy shelf on December 11, 2017.
This analysis was produced for pharmaceutical IP teams, R&D strategy directors, and institutional investors. All revenue and market share figures are drawn from publicly available sources including HHS/ASPE, Mordor Intelligence, Research Nester, FDA ANDA approval records, company press releases, and federal court records. Patent expiry dates are drawn from USPTO and Orange Book public records. Market projections are third-party estimates and not investment advice.