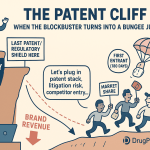
Every blockbuster drug is, at its core, a time-limited monopoly. The moment that monopoly expires, the revenue collapses. Pfizer watched Lipitor’s worldwide sales fall from $9.5 billion in 2011 to $3.9 billion in 2012, a 59% decline in a single year. AbbVie spent roughly 15 years and filed more than 100 patents to prevent the same fate from hitting Humira. Merck’s Keytruda generates $29.5 billion per year and faces a projected 19% first-year revenue decline when its core patents expire around 2028.
The companies that outperform don’t just discover drugs. They build IP architectures. They treat the patent portfolio as a financial instrument, stress-test it against litigation scenarios, prune it with the same discipline that a fund manager culls underperforming positions, and mine it for licensing revenue. This guide is the operational manual for doing exactly that across every stage of the drug lifecycle.
Part I: The Economic Foundation – Why Patent Portfolios Are Balance Sheet Items
The Innovation-Exclusivity-Reinvestment Cycle
The pharmaceutical R&D model only works because of one mechanism: market exclusivity. The Tufts Center for the Study of Drug Development estimated the fully capitalized cost of bringing a single new drug to market at $2.6 billion, accounting for the cost of capital over a 10-plus year development timeline and the loaded cost of every failed candidate that never reached approval. With only about 12% of drugs entering clinical trials eventually receiving regulatory clearance, the revenue from each approved product must subsidize eight or nine failures.
That math only pencils out if exclusivity exists long enough to generate meaningful returns. The cycle runs in one direction: a company takes high-risk capital into R&D, a patent converts that risk into a protected revenue stream, and that revenue funds the next generation of candidates. Cut the exclusivity period, and the investment calculus breaks. Slash prices during exclusivity, and the next cycle gets shorter. This is not an industry argument for high drug prices. It is the structural logic that every portfolio manager, IP team, and institutional investor must understand before making a single allocation or filing decision.
The practical implication for IP strategy is that a patent portfolio is not a legal archive. It is the mechanism through which R&D capital converts into return on investment, and managing it poorly is a direct financial failure.
IP Valuation as a Core Asset on the Balance Sheet
Pharma IP has three financial lives. The first is as a regulatory moat, protecting revenues during the exclusivity window. The second is as a transaction currency, since the strength of a portfolio determines licensing deal structure, M&A valuations, and royalty rates. The third is as a signal to capital markets.
For large-cap pharma, sell-side analysts routinely build patent cliff models that forecast when each major product loses exclusivity and what percentage of revenue is at risk within 12, 24, and 36 months. A company with a pipeline thin enough to leave a multi-year gap after its primary LOE event will see its EV/EBITDA multiple compress before any single patent expires. AbbVie’s aggressive filing strategy for Humira, which pushed effective biosimilar competition from 2016 to 2023 in the U.S., was not just a legal victory. It added approximately $65 billion in cumulative U.S. Humira revenue that would not have existed without the patent thicket holding.
For small and mid-cap biotechs, the calculus is even more acute. A company like Protagonist Therapeutics or Karuna Therapeutics before its acquisition by Bristol-Myers Squibb in 2024 carries relatively few products, meaning the composition of matter patent on the lead asset is the dominant driver of enterprise value. A successful IPR petition challenging that patent’s validity can destroy 40-60% of market capitalization in a single trading session.
Investment Strategy: When sizing a pharma or biotech position, obtain the Orange Book listing for all key products and map every patent expiration date, including formulation, method-of-use, and device patents. Calculate the ‘protected revenue window,’ which is the annual revenue attributable to the product multiplied by the number of years before the earliest credible generic entry. Discount that window at an appropriate rate to arrive at a patent-adjusted NPV. Then stress-test the position by modeling what happens if the composition of matter patent is successfully challenged via IPR two years ahead of its nominal expiration.
Part II: The Patent Cliff – Quantifying the Existential Threat
Revenue Erosion Mechanics for Small Molecules vs. Biologics
When a small-molecule blockbuster loses exclusivity, revenue erosion is rapid and deep. Generic manufacturers can enter simultaneously under Hatch-Waxman’s 180-day exclusivity for the first filer, and within 90 days of multi-generic entry, the brand typically retains only 10-20% of its original volume at a maintained price, or holds volume by slashing price to match generics. The math is unforgiving either way.
Biologics operate differently. The Biologics Price Competition and Innovation Act of 2009 established the abbreviated biosimilar pathway under section 351(k) of the PHS Act. But biosimilar market uptake is slower than generic entry for three structural reasons: manufacturers must demonstrate biosimilarity through extensive analytical, preclinical, and clinical data packages; FDA interchangeability designation (required before pharmacist-level substitution without prescriber sign-off) requires additional switching studies; and physician habit and patient persistence create stickier demand. This means the ‘cliff’ for a biologic behaves more like a long slope. Humira’s U.S. sales fell 30.8% in the first nine months of 2023 after biosimilar entry, significant but far less catastrophic than an equivalent small-molecule LOE.
That distinction matters for portfolio strategy. Small-molecule products need formulation or method-of-use patents with later expiration dates to buy time before the cliff arrives. Biologic products need manufacturing process patents, delivery device patents, and formulation patents that raise the technical bar for biosimilar developers, even when the composition of matter patent has expired.
The 2025-2030 Expiration Calendar: Products at Risk
The current wave of expirations concentrates more revenue at risk than any prior period. Keytruda alone, at roughly $33 billion in estimated peak sales, faces patent exposure around 2028. Eliquis, jointly owned by BMS and Pfizer and generating over $10 billion annually, faces patent challenges that began reaching courts in 2024-2025. Stelara (ustekinumab), Johnson & Johnson’s IL-12/23 inhibitor, began facing biosimilar entry in 2023 with the arrival of Hadlima and others. Ozempic and Wegovy (semaglutide) from Novo Nordisk will face composition of matter challenges, though Novo’s formulation and device portfolio provides meaningful secondary protection.
Each of these products has a unique IP architecture with a different litigation risk profile. Analyzing them in aggregate as a ‘patent cliff problem’ understates the strategic sophistication required. Each demands product-specific lifecycle management that starts years before the primary patent expires.
Key Takeaways for Part II: Revenue erosion from LOE is not uniform across drug classes. Small molecules collapse faster. Biologics erode more slowly but over a longer period. Portfolio managers who treat all LOE events equivalently will systematically mismodel revenue trajectories for biologic assets. The 2025-2030 window concentrates more brand revenue at risk than any prior period, making proactive lifecycle management a top-tier strategic priority now.
Part III: Architecting a Defensible Patent Portfolio
Strategic Alignment – Building IP Around Business Objectives
The most expensive IP mistake a pharma company makes is filing patents in a strategic vacuum. A patent that protects a feature no prescriber cares about, covers a manufacturing process the company no longer uses, or defends an indication with a shrinking patient population is not an asset. It is maintenance fee expenditure with no return.
Effective portfolio architecture starts from the commercial thesis and works backward to IP strategy. If the commercial strategy is best-in-class dosing convenience in a crowded oncology space, then R&D efforts aimed at extended-release or subcutaneous delivery mechanisms need immediate IP coverage, with filings timed to support the Phase II data package. If the strategy is rare disease market leadership, then orphan drug designation tracking and method-of-use patent applications for each discrete indication need to run in parallel with clinical development. These are not IP department decisions. They are cross-functional decisions made by teams that include R&D, regulatory, commercial, and IP counsel working from the same roadmap.
Patent mapping is the operational tool that keeps this alignment visible. A well-maintained patent map layers the portfolio against the product pipeline, showing which patents expire before which products reach peak revenue, where gaps exist that a competitor could exploit, and which patents are protecting assets no longer in the strategic core.
Building the Patent Thicket: Layer-by-Layer Analysis
A single composition of matter patent is a single point of failure. AbbVie’s Humira strategy is the canonical illustration of what a mature patent thicket looks like in practice. The company filed patents on adalimumab’s composition, its methods of manufacture for low-citrate formulations, its methods of use across more than a dozen indications, its concentration and dosage forms, and its delivery device. When Amgen launched Amjevita in January 2023 as the first U.S. Humira biosimilar, AbbVie had already negotiated a series of licensing agreements with biosimilar developers that granted delayed market entry dates in exchange for freedom to operate. The thicket did not block biosimilar entry permanently. It controlled the timing and terms of biosimilar entry, which allowed AbbVie to extract an additional seven years of largely unchallenged U.S. revenue.
The anatomy of a pharmaceutical patent thicket spans at least six distinct patent types, each with different strategic purposes and lifespans.
Composition of matter patents cover the active pharmaceutical ingredient itself. These are the broadest and most valuable, and they are the hardest to design around. They establish the initial 20-year term from the non-provisional filing date. For any new molecular entity, the composition of matter patent is the anchor of the entire IP structure.
Formulation patents protect the specific combination of API with excipients, coatings, or delivery mechanisms that constitute the final drug product. An extended-release formulation that enables once-daily dosing generates a new 20-year patent term that commonly outlasts the original composition of matter patent by 4-8 years. Shire’s transition of Adderall to Adderall XR, with the XR launch timed four months before original Adderall faced generic entry, is a textbook execution of this strategy.
Method-of-use patents protect specific therapeutic applications of an existing molecule. Pfizer’s sildenafil was originally investigated for pulmonary arterial hypertension and cardiovascular disease. Method-of-use patents for erectile dysfunction became the foundation of a Viagra franchise worth roughly $2 billion annually at peak. When the original compound patent expired, the indication-specific method-of-use coverage remained as a separate barrier.
Process patents cover novel manufacturing methods and are particularly strategic for biologics. The manufacturing process for a monoclonal antibody is deeply intertwined with the product’s biological activity. Amgen, Genentech, AbbVie, and other major biologics manufacturers hold large process patent estates that force biosimilar developers to demonstrate not only biological equivalence but also that their manufacturing process does not infringe on patented production methods. This is not a minor hurdle. Developing a non-infringing fermentation process, purification sequence, and formulation process can take 3-5 years of engineering effort and require constructing dedicated manufacturing infrastructure.
Polymorph patents protect specific crystalline forms of a drug molecule. Different polymorphs exhibit different solubility, stability, and bioavailability characteristics, and the patent covering the most pharmaceutically advantageous form can expire years after the original compound patent.
Delivery device patents cover the inhalers, auto-injectors, pre-filled syringes, and other administration systems that many modern drugs require. AstraZeneca’s dry powder inhaler portfolio, Novo Nordisk’s FlexPen and FlexTouch system, and Sanofi’s SoloStar injector represent IP estates that force biosimilar and generic developers to design, test, and seek regulatory clearance for their own non-infringing devices, adding substantial time and cost to market entry.
The IP Valuation Dimension of Each Patent Layer
Each layer of the thicket carries distinct IP valuation characteristics. Composition of matter patents in approved drugs command the highest licensing fees and the highest litigation stakes because they protect the broadest market access. In secondary market patent transactions, composition of matter patents for drugs with remaining commercial life trade at multiples of annual royalty income, commonly 10-20x depending on the remaining term, therapeutic area growth, and litigation exposure.
Formulation and device patents trade at lower multiples but with longer effective terms. A formulation patent filed during Phase II development and approved post-NDA may not expire until 10-15 years after the composition of matter patent, making it the primary defensive asset during the back half of a product’s commercial life.
Process patents are often the most difficult to value because they are rarely litigated openly, but they can be decisive in biosimilar entry timelines. A blocking process patent held by an innovator adds 2-4 years of operational effort for a biosimilar developer who must design around it, which translates directly into delayed generic competition and preserved brand revenue.
Investment Strategy: When evaluating an innovative pharma asset, do not stop at the primary patent expiration. Map the full thicket. Identify the last-expiring patent across all types and calculate the ‘effective exclusivity runway’ as distinct from the nominal composition of matter expiration. A 3-year difference in that runway can be worth $5-10 billion in NPV for a blockbuster drug.
Key Takeaways for Part III: A patent thicket functions as a portfolio of strategic options, not a single legal barrier. The commercial value of the thicket is determined by the last patent standing, not the first to expire. Companies that invest in only a composition of matter patent and neglect formulation, process, and device coverage leave the second half of their exclusivity window undefended.
Part IV: Claim Drafting and PTAB Resilience
The Strategic Architecture of Patent Claims
Patent claims are the only part of a patent that determines infringement. The specification, examples, and drawings exist to support the claims. In post-grant proceedings and district court litigation, the battle over a pharmaceutical patent is a battle over claim language: what it means, how broadly it reads, and whether it is supported by adequate written description and enablement.
Drafting pharmaceutical claims requires navigating a persistent tension. Claims drafted too broadly read on prior art and fail novelty or obviousness challenges. Claims drafted too narrowly invite design-arounds. The solution is a layered claiming strategy that functions as a series of nested fallback positions.
The broadest independent claim captures the largest possible commercial territory, typically protecting the compound class, the formulation type, or the method at the highest level of abstraction. Dependent claims progressively narrow the invention by adding specific limitations, such as a particular dosage range, a specific excipient class, a defined patient population, or a quantified clinical endpoint. Each dependent claim is a separate, enforceable right. An opponent who invalidates the broadest independent claim does not automatically invalidate the dependent claims. They must challenge each limitation independently.
The 2015 Supreme Court decision in Teva Pharmaceuticals v. Sandoz illustrates what happens when claim language fails. Teva’s patent on Copaxone, glatiramer acetate, referenced the drug’s molecular weight without specifying which of three different measurement methods applied. That ambiguity in a single technical term created enough uncertainty to generate a colorable indefiniteness argument, opening the door to generic competition. The lesson is that precision in claim drafting is a risk management activity, not a pedantic legal exercise.
PTAB Inter Partes Review: The New Litigation Reality
The America Invents Act of 2011 created the Patent Trial and Appeal Board and its Inter Partes Review proceeding, and the result has been a structural change in how patent challenges are litigated. IPR is faster, cheaper, and applies a lower evidentiary standard than district court. In district court, a patent enjoys a presumption of validity that requires challengers to prove invalidity by clear and convincing evidence. In IPR, no such presumption applies. The standard is preponderance of the evidence, roughly analogous to ‘more likely than not,’ and three technically trained administrative patent judges, not a lay jury, decide the outcome.
Bio/pharma patents account for roughly 6-7% of total IPR petitions filed, but they have one of the highest institution rates at approximately 73%. Once instituted, pharma patents have a somewhat better claim survival rate than patents in other technology areas, which suggests that innovators in pharma have generally invested more in patent quality than in other sectors. But even a ‘better’ survival rate in IPR means that a meaningful percentage of instituted petitions result in claim cancellation.
The strategic implication is that building a patent for PTAB resilience is now as important as building one for district court. A claim that a lay jury would find compelling based on a clinical story may be technically vulnerable to a prior art argument when scrutinized by expert judges applying a preponderance standard. Modern pharmaceutical patent prosecution must therefore be built with IPR attack scenarios modeled during drafting, not after grant.
Practical steps for PTAB-resilient claiming include conducting comprehensive prior art searches before drafting claims, not after; building specifications with extensive working examples and data that support a broad claim scope without overclaiming; using multiple independent claims across different claim types (composition, formulation, method, device) so that an IPR targeting one claim type does not invalidate the entire patent; and preparing claim amendment strategies in advance of any challenge.
Key Takeaways for Part IV: Claim quality is portfolio quality. A patent with a single broad claim and a thin specification is a liability in the IPR era. Every pharmaceutical patent should be stress-tested against a hypothetical IPR challenge before grant, and claim amendment contingencies should be documented as part of standard portfolio management practice.
Part V: Global Patent Filing Strategy
Where to File and Why
A U.S. patent stops at the U.S. border. The value of a pharmaceutical product protected by U.S. patent rights alone is significantly impaired if that product is manufactured abroad, sold in a dozen other markets, or if a competitor begins export to markets where no protection exists. Global filing strategy is the practice of acquiring IP rights in every jurisdiction where that gap would cause material financial harm.
The economics of global filing are not trivial. A single patent filed across 10 major jurisdictions accumulates over $250,000 in prosecution and maintenance costs across its lifetime. Translation fees alone can multiply prosecution costs by a factor of two or three in markets like Japan, South Korea, and the EU member states that require national language filings. A company with a 200-patent portfolio seeking global coverage cannot afford to file everywhere without a rigorous cost-benefit framework.
The PCT application is the cornerstone of efficient global coverage. Filing a single PCT application preserves the right to seek protection in over 150 member countries for up to 30 months from the priority date. That 30-month window is not wasted time. It is a period for gathering Phase II or Phase III clinical data that will improve the commercial case for national phase entry, refining which markets matter most as market access negotiations advance, and deferring significant prosecution costs while fundraising or partnership discussions are underway.
Strategic market prioritization follows a concentrated value approach. For most branded drugs, the top 6-8 markets capture 70-80% of global revenue: the United States, the five largest EU markets (Germany, France, Italy, Spain, and the UK), Japan, and China. Filing in all 150 PCT member states is not a strategy; it is a budget allocation error. Filing in the 8 markets that cover 75% of revenue at total prosecution cost is rational capital deployment.
Beyond market size, each jurisdiction requires analysis along three additional dimensions: the strength and predictability of patent enforcement, the probability of patent grant given the local examination standards and prior art landscape, and the competitive dynamics in that specific therapeutic area.
EU Strategy: SPCs, Bolar, and the Evolving Pharmaceutical Legislation
The EU patent system has historically offered robust protection through Supplementary Protection Certificates, which extend effective patent protection by up to five years to compensate for time consumed in regulatory review, with a further six-month extension available for drugs that complete pediatric studies. SPCs are granted at the national level, meaning a company must file in each EU member state individually, and the calculation of the SPC term depends on the date of first marketing authorization in the EU.
The European Commission’s proposed pharmaceutical legislation revisions, still under negotiation as of 2025-2026, have generated significant concern from innovator companies. EFPIA has argued that proposed reductions to data protection periods could effectively reduce the EU regulatory exclusivity baseline from 8 years to 6 years for many products, weakening incentives for companies to conduct EU-based clinical development or file for EU marketing authorization before the U.S. or other markets.
For portfolio strategists, the EU legislative uncertainty creates a 3-5 year window of ambiguity in European IP valuations. Products currently in Phase II or Phase III development should be modeled under both the current exclusivity framework and a downside scenario reflecting potential legislative changes, with the NPV gap informing both clinical development location decisions and EU filing timing.
China: High Win Rates, Hard Evidence Collection
China’s IP environment has shifted materially over the past decade. Foreign plaintiffs now achieve roughly a 77% success rate in patent infringement cases against Chinese defendants in specialized IP courts in Beijing, Shanghai, and Guangzhou. Average damage awards in Beijing rose from approximately $80,000 in 2018 to around $450,000 in 2022, with some high-profile pharmaceutical cases exceeding those averages. Approximately 70% of patent cases resolve within 12 months, faster than comparable U.S. district court timelines.
The structural challenge in China remains evidence collection. There is no U.S.-style discovery process. The burden of proving infringement falls on the patent holder, who must gather evidence without compulsory document production from the defendant. In pharmaceutical cases, this means proving that a Chinese manufacturer’s process or formulation infringes a specific claim without access to the manufacturer’s internal process documentation. Plaintiffs have increasingly used notarial seizure proceedings and administrative raids coordinated with market supervision bureaus to obtain physical evidence, but this adds cost and unpredictability.
China also excludes methods of treatment from patentability under Article 25 of the Patent Law on public policy grounds. This eliminates standard method-of-use claims. Companies protecting new indications in China must rely on ‘Swiss-type’ claims: ‘Use of compound X in the manufacture of a medicament for the treatment of disease Y.’ The drafting requirements and claim scope under Swiss-type format differ meaningfully from U.S. method-of-use claims, and the transition requires specialized prosecution strategy.
Japan: Opacity, Price Deflation, and Building Your Own Intelligence Layer
Japan operates a de facto two-stage patent linkage system without a formal equivalent to the U.S. Orange Book. During PMDA review of a generic application, the agency checks for potential infringement of originator patents covering the active substance and approved indications, relying on a list that the originator files with MHLW. That list is not publicly available, making it genuinely difficult for generic developers to assess their full infringement exposure before investing in clinical development.
After PMDA approval, the product faces a pre-NHI listing process during which brand and generic companies are expected to resolve remaining IP disputes, particularly around formulation and process patents. There is no mandatory stay equivalent to Hatch-Waxman’s 30-month freeze, and there is no statutory 180-day first-filer exclusivity reward equivalent to incentivize rapid generic challenges.
Pricing is the other defining feature of the Japanese market. The NHI conducts price revisions annually, almost always downward, with the magnitude depending on market volume and reference pricing against other indications and products. This creates a relentlessly deflationary commercial environment that compresses the NPV of any Japanese filing over time. For companies without a clear commercialization strategy in Japan and a credible plan to navigate the MHLW pricing process, the cost-benefit math on Japanese patent filing requires honest scrutiny.
Key Takeaways for Part V: PCT filing is not a substitute for a geographic prioritization framework. EU exclusivity is under legislative pressure. China’s courts are materially more favorable to patent holders than they were 10 years ago, but evidence collection remains the dominant procedural challenge. Japan requires building proprietary patent intelligence to compensate for a non-public patent linkage system.
Part VI: Active Portfolio Management – Audits, Pruning, and Intelligence
The Portfolio Audit Framework
A patent portfolio without a systematic audit cadence degrades. Individual patents expire or become commercially irrelevant as the pipeline evolves. Maintenance fees accumulate on assets protecting discontinued candidates or obsolete formulations. Patents covering technology that a competitor has already designed around provide theoretical rights but no practical commercial value.
A formal portfolio audit, conducted annually at minimum and triggered by any major strategic shift (pipeline addition, indication expansion, M&A activity, competitor regulatory filing), should answer five questions for every active patent and application:
Does this patent protect a commercial product or a near-term pipeline candidate? Does it align with the company’s current therapeutic area focus? Does it provide competitive advantage that a competitor cannot replicate without infringing, or has it been effectively designed around? Is the underlying technology still relevant given current science, or has it been rendered obsolete by a superior platform? What is the cost of maintaining this patent over its remaining life, and does that cost justify its projected defensive or commercial value?
The answers generate a prioritization matrix that separates core assets (high value, high strategic relevance), monitoring assets (moderate value, requiring periodic reassessment), and pruning candidates (low or zero value, consuming resources).
Patent Pruning: Reducing Costs and Unlocking Hidden Value
U.S. patent maintenance fees total over $14,000 across a patent’s full 20-year term, paid in escalating tranches at 3.5, 7.5, and 11.5 years post-grant. In Europe, annuity fees paid in each national jurisdiction can run to substantially more. A portfolio of 500 patents spread across 15 countries can carry annual maintenance costs exceeding $3 million, most of it invisible until someone performs a systematic cost-benefit review.
Pruning is not abandonment of IP. It is a resource reallocation decision. Once a patent is identified as a pruning candidate, three options exist: abandon it and allow it to expire by non-payment of maintenance fees; sell it outright to a third party for whom it has higher value, generating immediate cash; or license it on a non-exclusive basis to create a recurring royalty stream while retaining title.
The monetization angle is frequently underexplored. A formulation patent protecting a drug delivery technology that is no longer in your commercial portfolio may be extremely valuable to a CDMO or a specialty pharma company that has adopted that technology. A process patent on a synthetic chemistry method that you have replaced with a newer process may be worth licensing to a generic manufacturer who needs that specific route. The audit that identifies a pruning candidate should simultaneously trigger a market search for potential buyers or licensees. Pruning combined with monetization converts a cost center into a revenue generator.
Patent Landscape Analysis and Competitive Intelligence
Patent landscape analysis is a systematic scan of the patent filings in a defined technology space, designed to answer specific strategic questions: Who are the leading filers in this therapeutic area? What claim types dominate the landscape? Where are the ‘white spaces,’ areas with minimal patent coverage that represent potential innovation opportunities? Which companies are accelerating their filing pace, and what does that imply about their pipeline plans?
The intelligence value of landscape analysis extends beyond identifying competitive threats. A white space analysis can directly inform R&D prioritization. If a landscape analysis in antibody-drug conjugate technology reveals that linker chemistry for a specific tumor antigen target is sparsely patented while the antibody and payload spaces are heavily covered, that gap becomes an R&D opportunity with a clear path to exclusive protection.
Competitive intelligence at the company level is more targeted. The objective is a predictive model of a specific competitor’s next moves. Raw patent data is a lagging indicator; applications are published 18 months after filing, meaning you are reading about decisions made a year and a half ago. The intelligence value multiplies when patent data is fused with clinical trial registrations from ClinicalTrials.gov and its international equivalents, published scientific literature, conference presentations, regulatory meeting calendars, and executive commentary from investor calls and medical congresses.
A worked example: suppose a competitive intelligence review using a tool like DrugPatentWatch identifies that a key competitor has filed patent applications on sustained-release subcutaneous formulations of their oral GLP-1 agonist just as they have registered a Phase I study for a long-acting injectable version of the same compound. Cross-referencing those filings with FDA meeting calendars shows a Type B pre-NDA meeting request. The synthesis of these data points lets you predict with reasonable confidence that they intend to file an NDA for a monthly injectable within 24-30 months, positioned for the same patient convenience advantage your own XR oral formulation targets. That insight, extracted from publicly available data two years in advance, is worth more than any individual patent.
Key Takeaways for Part VI: A portfolio audit is not a legal exercise. It is a capital allocation exercise that has direct P&L implications through maintenance fee optimization and monetization of non-core assets. Competitive intelligence derived from patent data fused with clinical, regulatory, and commercial signals is more valuable than patent data alone, and the predictive horizon it creates should drive strategic decisions years before competitors make their moves publicly visible.
Part VII: Lifecycle Management and Evergreening – The Full Playbook
Regulatory Exclusivities as a Parallel Protection System
Patent rights and regulatory exclusivities are independent systems, and managing both in concert is essential. A regulatory exclusivity can block generic or biosimilar approval even if every relevant patent has expired, and a patent can block market entry even after a regulatory exclusivity has run. The two systems operate on different legal bases but interact to define the total protected commercial window.
In the U.S., the key regulatory exclusivities include New Chemical Entity exclusivity (5 years of data protection for drugs with a novel active moiety, blocking abbreviated NDA filings for the exclusivity period), New Clinical Investigation exclusivity (3 years for approved changes based on new clinical studies, such as new indications or formulations), Orphan Drug Exclusivity (7 years of market exclusivity for drugs treating conditions affecting fewer than 200,000 U.S. patients), Pediatric Exclusivity (6 months added to all existing patents and exclusivities as a reward for completing FDA-requested pediatric studies), and Biologics exclusivity (12 years of data exclusivity and 4 years of exclusivity against biosimilar applications under the BPCIA).
Orphan Drug Exclusivity deserves particular attention. The 7-year market exclusivity period is granted independently of patent status and blocks any competitor from receiving approval for the same drug in the same orphan indication, not merely from using your clinical data. For a drug with modest patent coverage, ODE can be a decisive commercial protection that outlasts the primary patents. It is also not mutually exclusive with other exclusivity types: a biologic drug with ODE, pediatric exclusivity, and BPCIA 12-year exclusivity can carry a protection stack that extends well beyond any individual exclusivity period.
Patent Term Extension under 35 U.S.C. 156 allows a company to apply for restoration of a portion of patent term consumed during regulatory review. The calculation is complex: the extension equals one-half the IND period plus the NDA review period, subject to a maximum of 5 years and a post-approval effective term cap of 14 years. Only one patent per approved drug can receive a PTE, so the selection of which patent to extend requires analysis of which patent would otherwise expire earliest and which one provides the broadest coverage of the approved indication.
Evergreening Strategies: Technology Roadmaps
‘Evergreening’ is the commercial practice; ‘lifecycle management’ is the internal framing. Both describe the same set of strategies for extending the commercially protected life of a drug beyond the primary composition of matter patent. The strategies are legally permissible, commercially rational, and politically contentious. A company executing them successfully should expect to manage reputational risk and antitrust scrutiny alongside the patent strategy.
The formulation pivot is the most commonly executed strategy. An immediate-release tablet is reformulated as an extended-release capsule with once-daily dosing, the new formulation is patented, and the brand transitions prescribing to the ER version before the IR version faces generic competition. Shire executed this with Adderall and Adderall XR. AstraZeneca executed a more complex version with omeprazole (Prilosec) and esomeprazole (Nexium). Esomeprazole is the S-enantiomer of omeprazole, offering marginally better bioavailability, and it was patented as a separate compound with a new 20-year term. AstraZeneca then marketed Nexium aggressively, transitioning the patient base before Prilosec faced generic entry in 2001-2002. The strategy generated approximately $5-6 billion in annual Nexium sales through the mid-2000s, buying roughly a decade of additional protected revenue.
The indication expansion strategy uses method-of-use patents to enter new patient populations. Pfizer’s tofacitinib (Xeljanz), originally approved for rheumatoid arthritis, received subsequent approvals and method-of-use patent coverage for psoriatic arthritis and ulcerative colitis. Each new indication carries its own market, its own pricing dynamics, and its own patent coverage. The same compound, with no changes to the molecule, generates a portfolio of method-of-use patents expiring years after the original compound patent.
The combination product strategy combines two previously separate drugs into a single dosage form, creating a new patent-protected product with formulation and method-of-use coverage. Bristol-Myers Squibb’s Atripla, which combined efavirenz, emtricitabine, and tenofovir into a single once-daily HIV tablet, is an example where the combination product’s patent coverage extended the effective exclusivity of the underlying components. Similarly, fixed-dose combinations in hypertension, diabetes, and oncology support markets are common execution vehicles for this strategy.
‘Product hopping,’ the most aggressive and controversial variant, involves making a minor modification to a drug and withdrawing the original formulation from the market before generic entry, forcing patients to transition to the new, patent-protected version. Because the original is no longer available, pharmacists cannot substitute a generic for the brand at the point of dispensing. New York and other states have challenged this practice. Abbott’s withdrawal of TriCor (fenofibrate) tablets in favor of capsules and then nanoparticle formulations, each iteration with new patent coverage and timed to precede generic entry, generated antitrust litigation that reached the Third Circuit. The line between legitimate product improvement and anticompetitive market manipulation is actively litigated, and any product hop strategy must be reviewed for antitrust exposure alongside the IP analysis.
Authorized Generics: The Revenue Defense Tool
An authorized generic (AG) is a version of a brand drug sold by the brand manufacturer itself, or its authorized partner, as a generic. The AG is marketed under the generic drug’s ANDA, uses the same manufacturing process and facilities as the brand, and is sold at a generic price. AGs are not separately regulated; they use the existing NDA approval.
The strategic purpose of an AG is to undercut the 180-day exclusivity period that the first Paragraph IV filer would otherwise enjoy alone. When a brand manufacturer launches an AG simultaneously with the first generic, the first filer’s market becomes a duopoly from day one. This reduces the first filer’s revenue during the 180-day period, making the economics of a Paragraph IV challenge less attractive. Over time, this can deter generic challenges by lowering the expected payoff, protecting the brand’s market position.
AbbVie launched an AG version of AndroGel (testosterone gel) during the generic exclusivity period. Pfizer launched an AG of Lipitor through Greenstone, its generic subsidiary, when atorvastatin faced generic entry. The AG strategy does not extend the primary patent exclusivity but reduces the financial attractiveness of the exclusivity window for the challenger, which has a deterrent effect on future Paragraph IV filings against the company’s other products.
Key Takeaways for Part VII: Lifecycle management is not a last-minute defensive move. The most successful executions, Nexium, Adderall XR, Humira’s biosimilar delay, were planned 7-10 years before the primary patent expiration. Any company with a major product 5 years from LOE that does not have an active lifecycle management program is late. The formulation pivot, indication expansion, and authorized generic strategies each require distinct regulatory and commercial lead times that must be mapped against the patent expiration calendar now.
Part VIII: Litigation Strategy – Hatch-Waxman, PTAB, and the Two-Front War
Paragraph IV Filings and the Hatch-Waxman Framework
The Drug Price Competition and Patent Term Restoration Act of 1984 created the U.S. generic drug industry as it currently exists. The Act established the Abbreviated New Drug Application pathway, allowing generic manufacturers to reference an innovator’s clinical data rather than conducting their own trials. In exchange for this shortcut, the Act requires generic applicants to certify one of four positions about each patent listed in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations (the Orange Book).
A Paragraph IV certification asserts that the listed patent is invalid, unenforceable, or would not be infringed by the proposed generic. Filing a Paragraph IV certification constitutes artificial infringement under the Act, giving the patent holder 45 days to sue the generic applicant. If the brand sues within that 45-day window, the FDA cannot approve the ANDA for 30 months unless the patent expires, the patent is adjudicated invalid or non-infringed, or the parties reach a settlement before the stay expires.
The first company to file a Paragraph IV certification against a patent is eligible for 180 days of generic marketing exclusivity from the date of first commercial marketing. This exclusivity period, which blocks subsequent generic entrants (but not the brand’s own authorized generic), provides the financial incentive that drives competitive Paragraph IV filings. For a blockbuster drug, 180 days of exclusivity in a duopoly with the brand at a 20-25% price discount can generate hundreds of millions in gross profit for the first filer, making the Paragraph IV challenge one of the highest-return investments in the generic pharmaceutical business.
The 30-month stay converts the Paragraph IV filing process into a litigation forcing function. Once the brand sues, the case must be resolved before the stay expires or the generic enters. For a drug with multiple listed patents, each with a separate Paragraph IV certification, a brand company can stack 30-month stays, provided the subsequent patents were not listed before the ANDA was filed. Strategic Orange Book listing, maintaining the listing current and accurate, is therefore a core activity for any brand company with a significant commercial product.
PTAB vs. District Court: Choosing the Right Battlefield
The table below captures the strategic distinctions that determine which venue a challenger should prefer and which a brand company should prepare to defend.
District court litigation operates under a presumption of patent validity. The challenger must prove invalidity by clear and convincing evidence, a high standard that benefits patent holders. Cases typically run 2-3 years to trial, cost $5-7 million for high-stakes matters, and can consider all invalidity grounds including enablement, written description, inequitable conduct, and obviousness. Juries, who are generally not technically expert, decide factual questions, and a compelling clinical narrative can be as influential as the technical merits.
IPR at the PTAB operates without any presumption of validity. Challengers need only a preponderance of evidence, roughly ‘more likely than not.’ Three technically expert administrative patent judges adjudicate the case. Proceedings are typically completed within 12-18 months of filing. Costs run in the hundreds of thousands of dollars rather than millions. The scope is limited to prior art challenges based on patents and printed publications; written description, enablement, and inequitable conduct arguments are not available in IPR.
The estoppel consequences after IPR are significant. A challenger who loses an IPR is barred from raising any invalidity ground in subsequent district court proceedings that was raised or reasonably could have been raised during the IPR. This ‘raised or reasonably could have raised’ standard is broader than the district court’s issue preclusion rules and means that a failed IPR can actually strengthen the patent’s district court position by limiting the grounds the challenger can argue in subsequent litigation.
For brand companies, the practical strategy now requires designing patents for both venues simultaneously. A claim that survives IPR based on strong prior art analysis and a robust specification is a far better commercial asset than one that might have been easier to enforce in district court but is vulnerable to an administrative challenge that can be filed for $30,000 and resolved in 18 months.
Paragraph IV Litigation: Select Case Studies and IP Valuation Implications
AbbVie’s defense of Humira’s adalimumab formulation patents against biosimilar developers proceeded through both district court and IPR. Samsung Bioepis, Mylan (now Viatris), Amgen, and others filed IPR petitions against multiple AbbVie patents covering low-citrate formulations and concentration ranges. AbbVie settled with most biosimilar developers through licensing agreements that provided delayed U.S. entry dates (2023 for most) in exchange for freedom to operate globally and royalty payments. The settlements effectively converted an uncertain litigation outcome into a structured revenue stream from biosimilar royalties while preserving U.S. market exclusivity for years beyond what the composition of matter patent alone would have provided.
Warner Chilcott’s litigation over Doryx (doxycycline hyclate) extended-release tablets illustrates the product hop antitrust risk. Warner Chilcott transitioned the market from tablets to capsules with a new tablet strength just before generic entry, generating antitrust challenges from generic manufacturers and the FTC. The Third Circuit found the conduct potentially anticompetitive. IP teams executing formulation transitions must retain antitrust counsel alongside patent counsel to assess the conduct under both bodies of law.
Merck’s defense of Keytruda (pembrolizumab) patents has already attracted scrutiny. Pembrolizumab’s composition of matter patent, U.S. Patent No. 8,952,136, expires in 2028. Merck holds additional patents on manufacturing processes, formulations, and methods of use across 18 approved indications. Biosimilar developers including Samsung Bioepis, Celltrion, and Incheon have filed BLAs with the FDA. The litigation landscape around Keytruda over the next 5 years will be one of the most closely watched IP battlegrounds in biopharma, with the outcome carrying implications for both Merck’s revenue trajectory and the commercial viability of the pembrolizumab biosimilar market.
Investment Strategy: For institutional investors, Paragraph IV certifications and IPR petition filings are public information that can be tracked in real time through the FDA ANDA database and the USPTO’s PTAB portal. A Paragraph IV filing against a brand’s lead product is a material event that typically prompts a litigation response within 45 days. Modeling the litigation outcome probability based on patent claim quality, prior art landscape, and the specific filer’s litigation track record can generate an early signal on LOE timing that is not yet reflected in sell-side consensus estimates.
Key Takeaways for Part VIII: Pharmaceutical patent litigation is a two-front war conducted simultaneously in district court and at the PTAB. A defense strategy optimized for one venue but not the other is half a strategy. The financial cost of full defense in a blockbuster drug case, $5-7 million per patent in district court plus additional costs for parallel IPR proceedings, is economically rational when set against the revenue being protected, but it requires pre-litigation preparation starting at patent prosecution.
Part IX: AI, Machine Learning, and the Future of Patent Portfolio Management
How AI Is Changing Patent Prosecution and Portfolio Analytics
Artificial intelligence is reshaping pharmaceutical patent work at every stage of the pipeline. In prosecution, AI-driven prior art search tools can scan tens of millions of patent documents and scientific publications in minutes, identifying relevant prior art that manual searches would miss and giving prosecution counsel the information needed to draft claims that avoid anticipation or obviousness rejections. Platforms like PatSnap and Anaqua integrate machine learning models that predict prosecution outcomes based on claim language, examiner assignment, and technology class, enabling firms to refine their arguments before submitting a response.
In portfolio analytics, AI enables classification and analysis at a scale that manual review cannot match. A 1,000-patent portfolio that would take a team of analysts months to classify and prioritize manually can be processed by an ML system in hours, generating a prioritized list of assets by estimated commercial value, litigation exposure, and maintenance cost-benefit ratio. This does not replace human judgment on the final decisions, but it dramatically reduces the time and cost of generating the information on which those decisions are based.
For competitive intelligence, natural language processing tools can scan competitor patent publications, clinical trial registrations, regulatory filings, and scientific literature simultaneously, generating automated alerts when a competitor files in a new technology area or registers a new clinical trial for an indication you also target. The latency between a competitive event occurring and an analyst becoming aware of it has collapsed from weeks to hours.
AI Inventorship and the Next Legal Frontier
AI-generated drug candidates raise a question that patent law has not yet resolved: if an AI system conceives of a novel compound with no meaningful human conceptual contribution, is the resulting innovation patentable? Current U.S. patent law requires a human inventor. The Federal Circuit’s 2022 decision in Thaler v. Vidal confirmed that only natural persons can be named as inventors on a U.S. patent application, explicitly rejecting an AI system (DABUS) as a named inventor.
The practical implication for pharmaceutical companies using AI-driven discovery platforms is that human scientists must be meaningfully involved in the conception of the invention, not merely in confirming results that an AI system generated. Companies like Insilico Medicine, Recursion Pharmaceuticals, and Schrödinger have developed internal documentation processes that record the specific human contributions to each AI-assisted discovery, preserving inventorship and patentability.
The deeper legal question, not yet resolved, is how AI-assisted discovery will affect the non-obviousness standard. If a compound can be identified by a computational screen that a skilled artisan would have run, courts may conclude that the compound was obvious to a person of ordinary skill in the art augmented by AI tools. This shifts the boundary of patentable invention upward, potentially requiring that a patentable drug candidate exhibit some property that was genuinely unexpected and could not be predicted by standard computational methods.
Key Takeaways for Part IX: AI tools provide competitive intelligence and portfolio analytics advantages that are now table stakes for large pharma IP operations. Companies without AI-augmented prosecution and landscape analysis capabilities are operating with an information disadvantage. On inventorship, the legal framework is clear for now (human inventors required) but the boundary of what is ‘obvious’ in an AI-augmented R&D environment will be litigated heavily over the next decade.
Part X: Investment Strategy Synthesis – Reading the Portfolio as a Financial Instrument
Patent-Adjusted Valuation Framework
For institutional investors and portfolio managers, a pharmaceutical company’s patent estate is a set of time-limited cash flow rights. Each patent, or cluster of patents protecting a single product, can be modeled as a bond with a maturity date (the last-expiring patent), a coupon (the annual revenue protected by exclusivity), and a credit risk (the probability of early call via successful generic challenge).
A patent-adjusted valuation model for a pharma company proceeds through four steps. First, identify every commercial product and map its full patent expiration profile across all patent types, using Orange Book data, PTAB filings, and proprietary patent intelligence. Second, estimate the annual revenue at risk for each product during each year of its remaining exclusivity window, accounting for competitive dynamics and indication mix. Third, model the LOE event as a revenue step-down with a timing probability distribution (the nominal patent expiration date as the base case, an early LOE scenario based on litigation risk, and a late LOE scenario based on successful lifecycle management execution). Fourth, discount each scenario at an appropriate cost of capital to generate a patent-adjusted NPV per product, then aggregate across the portfolio.
The resulting figure, compared against the company’s current enterprise value, tells you whether the market is pricing in the patent cliff accurately, optimistically, or pessimistically. Systematic deviations from patent-adjusted NPV are where the analytical edge lies.
Signal Detection from Public Patent Data
Every ANDA filing, Paragraph IV certification, and IPR petition is a public record. Orange Book data is updated monthly. The USPTO’s PTAB portal publishes petition filings in near-real time. For analysts willing to track these data sources systematically, they provide a proprietary signal on LOE timing that is often months or years ahead of sell-side consensus.
A Paragraph IV certification filing against a brand drug tells you three things: a generic developer has completed a bioequivalence study, they believe they have a colorable invalidity or non-infringement argument, and commercial launch is potentially 30 months away if the brand sues (or immediately if the brand does not sue within 45 days). Each of these facts has valuation implications that are often not reflected in consensus estimates for months after the filing is publicly available.
An IPR petition filing adds a fourth signal: a financially motivated third party with access to patent litigation counsel has reviewed the patent claims against the prior art landscape and concluded that at least one claim is more likely than not unpatentable. IPR petitions that reach institution proceed to final written decisions with a meaningful rate of claim cancellation or narrowing. A high institution rate across multiple IPR petitions against the same patent is a reliable warning signal on patent quality.
Investment Strategy: Build a tracking system that monitors Orange Book ANDA filings, Paragraph IV certifications, and PTAB petition filings for every company in your portfolio and watchlist. Set automated alerts for new certifications or petitions against products that represent more than 10% of a company’s revenue. Map the 30-month stay expiration dates for all active Paragraph IV litigations to generate a probabilistic LOE calendar that is more granular than consensus patent expiration dates.
Key Takeaways for Part X: A pharmaceutical company’s market value is, at a fundamental level, a claim on its remaining exclusivity windows. Patent-adjusted NPV modeling, driven by real-time monitoring of ANDA filings, Paragraph IV certifications, and IPR petitions, gives institutional investors an information edge on LOE timing that is quantifiable, systematic, and consistently underutilized by generalist investors without domain-specific patent intelligence infrastructure.
Conclusion: The Portfolio as a Living Asset
Drug patent portfolio management is not a legal compliance function. It is the discipline through which pharmaceutical companies convert R&D risk into durable competitive advantage, and through which investors translate scientific uncertainty into financial returns.
The companies that execute this well share a set of practices. They align every patent filing decision with commercial strategy. They build multi-layer patent thickets rather than relying on single composition of matter patents. They stress-test their claims against PTAB attack scenarios before grant, not after challenge. They audit and prune their portfolios on a defined cadence, converting non-core assets into licensing revenue rather than leaving them to accumulate maintenance fees. They treat patent landscape analysis and competitive intelligence as ongoing functions, not periodic projects. They run lifecycle management programs that start 8-10 years before LOE, not 2-3.
The companies that fail treat IP as a legal department deliverable, divorced from commercial and R&D strategy. They discover their portfolio weaknesses when a Paragraph IV certification arrives. They discover their competitive blind spots when a rival NDA appears in the FDA’s approval database.
In a business where the difference between a protected exclusivity window and an accelerated LOE can measure in billions of dollars per year, that distinction is not a management preference. It is the difference between companies that sustain R&D investment cycles and those that don’t.
Disclosure: This analysis is based on publicly available patent, regulatory, and litigation data. It does not constitute legal advice or investment advice. Patent-specific legal questions should be directed to qualified patent counsel in the relevant jurisdiction.