A comparative analysis of the statutory stay periods that define when generic and biosimilar manufacturers can enter the world’s two largest pharmaceutical markets — and what the difference means for your competitive strategy.
The Clock That Controls Billions
There is a moment in pharmaceutical competition that every generic drug manufacturer knows intimately: the day a patent linkage challenge begins its statutory count. From that day, a timer runs. When it expires, the path to market either opens or remains blocked by ongoing litigation. The length of that timer is not an accident of drafting. It reflects a deliberate policy judgment about who bears the burden of uncertainty while courts decide whether an originator’s patent is valid and infringed.
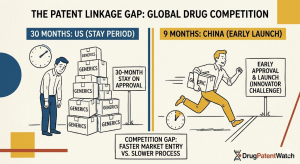
In the United States, that timer runs for 30 months.
In China, it runs for 9 months.
The difference — 21 months — sounds modest compared to the decade-long development timelines of branded pharmaceuticals. In commercial terms, it is enormous. A single month of generic exclusion on a blockbuster drug can be worth hundreds of millions of dollars in sustained originator revenue. Twenty-one additional months of protection, or the denial of that protection, redraws the competitive map for every multinational pharmaceutical company that operates in both markets.
This article examines how the US 30-month stay under the Hatch-Waxman Act and China’s 9-month stay under its 2021 patent linkage framework were designed, how they function procedurally, and what the gap between them means for originators, generic manufacturers, investors, and regulators navigating global drug launches.
The analysis draws on public regulatory filings, court records, the NMPA’s (National Medical Products Administration) implementing rules, academic literature, and patent data tracked through platforms including DrugPatentWatch, which catalogs the US Orange Book listings, paragraph IV certifications, and associated litigation timelines that form the empirical backbone of any serious Hatch-Waxman analysis.
What Patent Linkage Actually Does
Patent linkage is a regulatory mechanism that connects a drug’s marketing approval process to the status of patents covering that drug. Without linkage, a generic manufacturer could theoretically obtain regulatory approval for a drug, begin selling it, and then face a patent infringement lawsuit — all while the approved product sits on the shelf waiting for the dispute to resolve. The regulatory system and the IP system operate on separate tracks, potentially pushing in conflicting directions.
Patent linkage solves this by making approval conditional, or at least delayed, when a patent dispute exists. The generic applicant, in filing for approval, must take a position on every listed patent: either it certifies that the patent has expired, that the drug will not be marketed until the patent expires, or that the patent is invalid or will not be infringed by the generic product. That last position — a “Paragraph IV” certification in US terminology, or a “Category IV declaration” in China’s framework — triggers the linkage mechanism and potentially activates a stay on regulatory approval.
The policy rationale has two sides. Originator companies argue that linkage protects patent rights during the regulatory approval window, preventing generic manufacturers from gaining approval while simultaneously challenging patents — effectively creating a race to market entry that undermines IP protection. Generic manufacturers, and many health economists, counter that linkage allows originators to weaponize the regulatory process, converting it into an additional delay mechanism even when patents are weak or clearly inapplicable to the specific generic formulation at issue.
Both arguments have merit. The policy compromise is expressed in the length and conditions of the stay.
The length of a statutory stay period is, in effect, a thumb on the scale. A long stay favors originators: even if they ultimately lose the patent dispute, they collect additional revenue during the litigation period. A short stay favors generic manufacturers: if litigation cannot be resolved quickly, the stay expires and the generic enters anyway, leaving the originator to pursue damages rather than injunctive relief. Every pharmaceutical market that uses patent linkage must pick a point on this spectrum. The US chose 30 months in 1984. China chose 9 months in 2021. Those choices deserve careful examination.
The US System: Hatch-Waxman and the 30-Month Stay
The Legislative Context of 1984
The Drug Price Competition and Patent Term Restoration Act of 1984, universally known by the names of its Senate and House sponsors — Orrin Hatch and Henry Waxman — remains one of the most consequential pieces of pharmaceutical legislation ever enacted. Before Hatch-Waxman, generic drug manufacturers had to conduct full clinical trials to prove safety and efficacy even for drugs that were chemically identical to already-approved products. The regulatory cost was prohibitive. Hatch-Waxman created the Abbreviated New Drug Application (ANDA) process, which allows generics to rely on the innovator’s clinical data and demonstrate only bioequivalence.
In exchange, Hatch-Waxman created the Orange Book — officially the FDA’s “Approved Drug Products with Therapeutic Equivalence Evaluations” — where originator companies list patents covering their approved drugs. When a generic manufacturer files an ANDA and certifies that a listed patent is invalid or not infringed (the Paragraph IV certification), it must notify the patent holder. If the patent holder sues within 45 days, the 30-month stay triggers automatically.
The 30-month figure was not derived from any empirical analysis of how long patent litigation actually takes. It was a negotiated compromise. Congressional testimony from 1984 indicates that the pharmaceutical industry argued for a longer automatic stay, and consumer advocates pushed for shorter periods or no automatic stay at all. Thirty months was the number that passed.
The Orange Book: The Foundation of Linkage
The Orange Book is the central registry that makes Hatch-Waxman work. Originators are required to list patents that claim the approved drug (the active ingredient, the formulation, or the method of use) and for which a claim of patent infringement could reasonably be asserted. The listing obligation is self-executing — there is no pre-listing review by the FDA for validity or scope. The FDA takes the listing at face value and records it.
This design choice has profound consequences. Because listing is automatic and unchecked, originators have strong incentives to list every arguably applicable patent, including patents of dubious validity or uncertain applicability to generic formulations. Each listed patent is a potential litigation trigger, and each litigation trigger is a potential stay. A drug protected by four Orange Book patents, challenged by a single Paragraph IV ANDA, generates the possibility of four separate infringement suits, though courts typically consolidate them.
Data from DrugPatentWatch and the FDA’s public ANDA database shows that the average number of Orange Book patents per new drug application has increased substantially since 1984. Drugs approved in the 1980s typically had one or two listed patents. Many drugs approved in the 2010s and 2020s have five, ten, or even more. The practical result is that originator companies have used Orange Book listing as a portfolio strategy — layering patents on formulation, dosage form, metabolites, and methods of treatment to create multiple potential litigation triggers for any generic challenger.
The Mechanics of the 30-Month Stay
When the stay triggers, the FDA may not approve the ANDA until the earlier of two events: the court enters judgment that the patent is invalid or not infringed, or the 30-month period expires. The FDA also cannot approve the ANDA while the 30 months run unless the court enters an early resolution.
The stay begins from the date the NDA holder receives notice of the Paragraph IV certification, not from the date the ANDA was filed or from the date the lawsuit was filed. This timing detail matters: an originator who receives notice and delays filing suit until near the end of the 45-day window extends the effective protection period.
There are two modifications to the basic 30-month rule worth noting. First, the Medicare Modernization Act of 2003 amended Hatch-Waxman to provide that for ANDA applicants that file after the first ANDA filer (second-wave filers), a 30-month stay only attaches if the patent in question was listed in the Orange Book before the ANDA was filed. This prevents originators from listing new patents after ANDAs are filed and using those late-listed patents to generate fresh 30-month stays against already-pending applications. Second, courts have authority to shorten or extend the 30-month period for “reasonable cause.” In practice, extensions are rare. Shortenings — specifically where the generic manufacturer has failed to reasonably cooperate in expediting the litigation — are also uncommon.
The 180-Day Exclusivity Incentive
Hatch-Waxman’s patent challenge mechanism is reinforced by a commercial incentive: the first generic manufacturer to file a Paragraph IV ANDA receives 180 days of market exclusivity against other generic entrants once it receives approval. This means that the first ANDA filer — even if it ultimately loses its patent challenge — holds a gate-keeping position: no subsequent generic can enter the market until either the first filer’s exclusivity is exhausted or forfeited.
The 180-day exclusivity has produced a distinct category of patent litigation strategy known as the “authorized generic.” Originator companies responded to the 180-day exclusivity by entering into agreements with the first ANDA filer, sometimes paying that filer to delay market entry (so-called “reverse payment” or “pay-for-delay” settlements) or by launching their own authorized generic through a subsidiary or licensee to compete with the first filer during the exclusivity period. Both strategies have generated substantial antitrust litigation, culminating in the Supreme Court’s 2013 decision in FTC v. Actavis, Inc., which held that reverse payment settlements can violate antitrust law.
How the 30-Month Stay Works in Practice
Filing Dynamics
Between 1984 and 2022, over 3,000 Paragraph IV certifications were filed in the United States across hundreds of branded drug products [1]. The practical reality of this litigation load is that patent disputes under Hatch-Waxman are not ordinary commercial cases. They proceed under compressed timelines mandated by statute, typically with scheduling orders that push trial within 30 months of filing — not always successfully, but with more urgency than typical commercial IP cases.
The Hatch-Waxman case is filed in federal district court as a normal patent infringement action. The generic manufacturer, as the ANDA filer, is technically the defendant, though it often takes an active role in driving the litigation forward to resolve uncertainty about its market entry date. The 30-month stay means that even a generic manufacturer that is highly confident of prevailing must wait out the clock (or win in court) before receiving FDA approval to sell.
What Happens in Court
Hatch-Waxman cases concentrate in a small number of federal districts. The District of Delaware and the District of New Jersey handle the substantial majority, largely because many pharmaceutical companies are incorporated in Delaware or headquartered in New Jersey. Judges in these districts have developed significant familiarity with pharmaceutical patent issues, which has produced a body of precedent specifically tailored to the Hatch-Waxman context.
The legal issues in these cases typically fall into a predictable pattern. Invalidity arguments dominate: lack of written description, obviousness, and anticipation by prior art. Non-infringement arguments focus on whether the generic’s formulation actually uses the patented technology or falls within the patent’s claims. Because the ANDA specifies the exact generic formulation, the infringement analysis is often more tractable than in other patent contexts — the product is fully described before any sales occur.
Settlement rates in Hatch-Waxman litigation are high, historically running above 70% of filed cases [2]. Many settlements include agreements on entry dates and licensing arrangements. The FTC reviews settlements involving reverse payments under the antitrust framework established by Actavis, but settlements that simply grant an entry date without payment from the originator to the generic have generally not faced antitrust scrutiny.
The Stay as a Revenue Protection Tool
From an originator company’s perspective, the 30-month stay is most valuable not when the patent is strong but when the patent is weak. A clearly valid and clearly infringed patent will withstand challenge in any case; the originator does not need a stay to protect its revenue because it will win in court. The stay is most valuable when the originator is uncertain about the outcome — when the patent might be invalid, or when infringement is genuinely debatable.
In that scenario, the 30-month stay guarantees revenue collection during litigation regardless of the ultimate legal outcome. If the originator wins, it gets both the stay protection and a post-judgment injunction. If it loses, it still collected 30 months of branded revenue during litigation. The expected value of weak-patent litigation is therefore positive for originators under the US system, which critics argue creates systematic incentives to list questionable patents and file marginal infringement suits.
A study analyzing Hatch-Waxman litigation outcomes between 1992 and 2002 found that generic manufacturers prevailed in about 73% of cases that went to final judgment [3]. If the stay mechanism were operating as intended — protecting valid patents during litigation — one would expect a higher originator win rate among litigated cases. The high generic win rate is consistent with the hypothesis that some stays are used to delay competition on weak patents rather than to protect legitimate IP rights.
Litigation Strategy Under Hatch-Waxman
The 45-Day Decision Window
When an originator receives a Paragraph IV notice, it has 45 days to decide whether to file suit. This window is not just administrative — it is strategic. The originator must assess the strength of each listed patent against the specific generic formulation, the likely legal venue, the quality of its litigation team, and the commercial stakes.
For drugs with less than 18 months of stay protection remaining before patent expiry (where 30 months would run past the patent expiry date anyway), the stay serves limited purpose. For drugs with substantial remaining exclusivity and high revenue, the 45-day decision is almost always yes: file suit, trigger the stay, and litigate.
For drugs with weaker patent positions, the calculus is more complicated. The originator must weigh the cost of litigation (typically $5-20 million per case through trial) against the commercial value of 30 months of additional exclusivity. On a drug generating $1 billion per year, 30 months represents approximately $2.5 billion in retained revenue even before accounting for post-generic price competition. The math strongly favors filing suit in most commercially significant cases regardless of patent strength.
Multi-Patent Strategies
Modern Hatch-Waxman defendants — the generic manufacturers — face not single-patent challenges but multi-patent portfolios. A single ANDA filing against a major branded drug may require challenging ten or more Orange Book-listed patents, each of which independently can trigger a 30-month stay (subject to the 2003 modifications limiting late-listed patent stays).
This creates what practitioners call the “patent thicket” problem: even if a generic manufacturer is confident it can invalidate or design around most of a patent portfolio, the uncertainty introduced by any single patent with plausible infringement arguments may delay market entry by years. The cumulative effect of multiple overlapping patents — each requiring individual analysis and potential litigation — extends the period of market exclusivity well beyond the nominal patent expiry of the primary compound patent.
The pharmaceutical industry’s use of secondary patents — covering formulations, dosage forms, manufacturing processes, and metabolites rather than the active compound itself — has expanded significantly since 1984. These secondary patents typically have later expiry dates than the original compound patent, allowing originators to maintain Orange Book listings that trigger fresh stays even after the core compound patent expires or is invalidated.
Section 8 Carve-Outs
One partial remedy to multi-patent litigation available to generic manufacturers is the “Section viii” statement. Rather than challenging a method-of-use patent, a generic manufacturer can submit a statement with its ANDA certifying that it will not seek approval for the specific method of use claimed in the listed patent. This carve-out removes the patent from the litigation universe for that ANDA but limits the label for the generic drug — it cannot legally promote the carved-out indication.
The Section viii carve-out is widely used for drugs that have method-of-use patents on secondary indications. It allows generic entry on the primary indication while the originator retains patent protection on the secondary indication. The FDA and courts have grappled with how skinny labels interact with off-label prescribing, because physicians may prescribe the generic for all indications regardless of the label.
The Cost of the 30-Month Stay for Generic Manufacturers
Capital and Opportunity Cost
Receiving a 30-month stay does not mean the generic manufacturer stops working. During the stay period, the generic company bears all costs of litigation while earning no revenue from the challenged product. Litigation counsel fees, expert witness costs, and the opportunity cost of regulatory resources devoted to the ANDA all continue to accumulate.
For large generic manufacturers with diversified pipelines, the cost of a single Hatch-Waxman litigation is manageable. For smaller companies that have concentrated their ANDA pipeline on a single blockbuster drug — a rational strategy given the 180-day exclusivity incentive — a 30-month stay combined with an adverse judgment can be financially catastrophic. Several generic manufacturers have faced bankruptcy or acquisition after losing key Hatch-Waxman cases.
The financial asymmetry is structural. Originator companies fighting to protect blockbuster revenue can commit virtually unlimited resources to Hatch-Waxman litigation. Generic manufacturers, fighting for future revenue that has not yet materialized, must litigate on projected cash flows discounted for litigation risk.
The Broader Consumer Cost
The downstream effect of the 30-month stay on drug prices is the subject of substantial economic literature. A Federal Trade Commission study estimated that rapid generic entry — defined as entry within 12 months of patent expiry — results in branded prices falling by 80% or more within two years as generic market share grows [4]. Every month of delayed generic entry is therefore a month during which consumers pay branded prices for drugs with no meaningful clinical differentiation from the eventual generic equivalent.
The Congressional Budget Office has estimated the annual consumer savings from generic drugs at approximately $338 billion [5]. Conversely, studies of Hatch-Waxman litigation delays suggest that even modest reductions in generic entry delays could produce savings of tens of billions of dollars annually.
China’s 2021 Patent Linkage System: The Architecture
The Legislative History
China’s patent linkage system is new. It came into force on July 4, 2021, implemented through coordinated amendments to the Drug Administration Law and the Patent Law, supplemented by the “Measures for the Implementation of Drug-Related Patent Linkage” issued jointly by the NMPA and the China National Intellectual Property Administration (CNIPA) [6].
Before 2021, China had no formal patent linkage mechanism for pharmaceuticals. Generic manufacturers could obtain marketing authorization from the NMPA without regard to patent status. Patent holders had to pursue infringement actions in the courts separately, without any regulatory backstop. This was a significant point of friction in US-China trade negotiations, particularly under the phase one trade agreement signed in January 2020, which included specific commitments on pharmaceutical IP protection.
China’s patent linkage system was, in no small part, a direct response to US trade pressure. The United States, through the USTR, had specifically identified China’s lack of patent linkage as a market access barrier for US pharmaceutical companies. The phase one agreement’s IP chapter contained commitments to establish patent linkage that closely tracked the structure of the US system, though with significant differences in detail.
The Patent Registration Platform
Where the US uses the FDA’s Orange Book, China uses the “Patent Registration Platform for Marketed Drugs” (药品专利信息登记平台), a database maintained by the NMPA. The registration platform serves the same conceptual function as the Orange Book: it is the official registry of patents that generic challengers must address when filing marketing authorization applications (MAAs).
The registration categories differ somewhat from the US model. China’s platform uses a classification system that distinguishes between drug substance patents (Category I), preparation patents (Category II), and method of use patents (Category III), plus a Category IV for additional patent types. The classification affects the rights and procedures available during the linkage process.
The Four-Category Declaration System
When a generic manufacturer files a MAA with the NMPA for a drug covered by patents registered on the platform, it must make one of four declarations:
- Category I: No relevant patents are registered for the reference drug.
- Category II: The relevant patents have expired or been invalidated, or the applicant has obtained a license.
- Category III: The applicant will not market the drug until the relevant patents expire.
- Category IV: The registered patents are invalid or will not be infringed by the generic drug.
Only a Category IV declaration triggers the patent linkage procedures and the potential 9-month stay. Categories I, II, and III are self-executing: the NMPA processes the MAA without involving the patent holder.
The Category IV declaration initiates a notification process. The generic applicant must notify the patent holder of its declaration within the time limits specified in the implementing rules. The patent holder then has 45 days to initiate proceedings — either administrative proceedings before the CNIPA or civil proceedings before the Beijing Intellectual Property Court or the courts of competent jurisdiction in China [7].
The 9-Month Stay: Design Choices and Their Consequences
Why 9 Months?
China’s choice of a 9-month stay reflects several deliberate policy judgments that diverge from the US approach.
First, China’s pharmaceutical policy is oriented toward accelerating generic drug access as part of its volume-based procurement (VBP) system. Since 2018, China has run a series of centralized drug procurement rounds under the “4+7” procurement program and its successors, which have driven generic prices down dramatically — in some cases by 90% or more from branded levels. A 30-month patent linkage stay would directly conflict with this policy by preventing generic manufacturers from obtaining the marketing approvals needed to compete in procurement rounds.
Second, Chinese policymakers appear to have concluded that a shorter stay is less likely to be weaponized for thin patents. A 9-month stay is long enough to allow a genuine patent dispute to be initiated and some preliminary process to occur, but short enough that it does not allow weak-patent litigation to produce commercially significant delays by itself. The implicit model is that if an originator has a strong patent, it will obtain an injunction from a court before the 9-month period expires. If it cannot obtain preliminary relief, that suggests the patent is not strong enough to warrant a longer automatic delay.
Third, China was conscious of its position as a major generic manufacturing country. China is the world’s largest producer of active pharmaceutical ingredients (APIs) and a major generic drug manufacturer for both domestic and export markets. A patent linkage system with a long automatic stay would impose significant costs on the domestic pharmaceutical industry.
The Procedural Structure of the 9-Month Stay
When a patent holder initiates proceedings in response to a Category IV declaration within the 45-day window, the NMPA must stay the marketing approval of the generic drug. The stay runs for 9 months from the date the NMPA receives notice that proceedings have been initiated.
Unlike the US system, where the stay is purely passive — the FDA simply withholds approval — China’s 9-month period has a more active design. The proceedings initiated by the patent holder are expected to make meaningful progress within 9 months. If the patent holder obtains a favorable court ruling or a preliminary administrative determination within 9 months, further protection is available. If not, the NMPA may proceed with generic approval regardless of ongoing proceedings.
The CNIPA’s patent status determination process is one mechanism available during the 9-month period. Unlike the civil court system, CNIPA proceedings are administrative and are designed to move faster. The CNIPA has committed to completing patent status determinations within 9 months for linkage-related cases, aligning the administrative process with the stay period.
Limitations on the Stay
China’s stay has more explicit limitations than the US version. The 9-month stay only applies to drug substance patents (Category I), preparation patents (Category II), and new use patents (Category III-type patents). Not every patent registered on the platform automatically generates a stay-eligible linkage claim. If a registered patent falls outside these categories, or if the registration is found to be improper, the NMPA may decline to honor the stay trigger.
The implementing rules also provide that the NMPA will not delay approval beyond the 9-month period even if litigation or CNIPA proceedings remain pending, unless a court has issued an injunction or other order directing the NMPA to halt processing. This places the burden on the patent holder to obtain independent judicial relief rather than relying on the regulatory stay as a self-executing protection mechanism.
The Parallel Track: CNIPA vs. Court
China’s system offers two parallel tracks for patent challenges during the 9-month period. The CNIPA handles patent validity (invalidity declarations). The People’s Courts — primarily the Beijing Intellectual Property Court and the Supreme People’s Court’s IP tribunal for high-profile matters — handle infringement determinations.
The availability of fast-track CNIPA proceedings for generic challengers is a feature with no direct US analog. In the US, a generic manufacturer that wants to challenge patent validity through an administrative process must use the USPTO’s inter partes review (IPR) process, which is not formally linked to the Hatch-Waxman stay. In China, the CNIPA validity determination process is integrated into the patent linkage framework, and a successful invalidity declaration during the 9-month period can result in the NMPA proceeding with generic approval even before the stay expires.
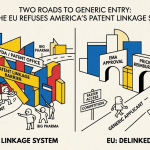
Head-to-Head: Where the Two Systems Diverge
Duration
The most visible difference is the one announced in the title: 30 months versus 9 months. But the practical difference is larger. In the US, the 30-month stay is absolute — it does not matter how weak the patent is, how quickly litigation is progressing, or whether the originator is genuinely pursuing the case. The stay runs unless and until the court rules or the patent holder voluntarily dismisses the case.
In China, the 9-month stay is closer to an initial protective window. A patent holder that cannot secure preliminary court relief within 9 months effectively loses the administrative protection, though litigation may continue in courts with the possibility of injunctive relief. A US patent holder in a weak case can sit back and collect 30 months of protected revenue; a Chinese patent holder in a weak case must win some form of interim relief within 9 months or watch the generic enter the market.
Scope of Eligible Patents
The US Orange Book system is broadly self-reporting. Originators can list patents that “claim” the drug in several ways: the active moiety, the formulation, a method of using the drug. The FDA’s review of listing eligibility is minimal — the agency generally accepts listings unless the patent on its face is clearly non-applicable. This has allowed significant creative listing, including patents claiming polymorph forms, salts, prodrugs, and other variations on approved drugs.
China’s registration platform has built-in eligibility restrictions. The implementing rules specify patent categories, and patents outside those categories do not generate linkage rights. Although the categories are fairly broad, the explicit classification system creates more potential for challenge to the registration itself. A generic manufacturer can dispute whether a registered patent belongs in a category that generates stay rights, adding a threshold dispute layer absent in the US system.
The Role of Courts
In the US, Hatch-Waxman patent disputes are adjudicated exclusively in federal district courts, with appeal to the Federal Circuit. The courts play a central role throughout the 30-month period. The typical timeline sees claim construction briefing, then summary judgment motions, then trial, all within the 30-month window (though many cases settle before reaching trial).
In China, the system allocates cases between courts and the CNIPA, creating complexity. A generic manufacturer may simultaneously pursue invalidity at CNIPA and contest infringement in court. A patent holder may obtain preliminary relief from a court while the CNIPA proceeding is pending. These parallel tracks can produce conflicting outcomes that the courts and CNIPA must then reconcile. The Supreme People’s Court has issued guidance on coordination between the tracks, but early experience suggests coordination challenges persist [8].
Transparency and Data
The US Orange Book is a mature, well-documented database with decades of public data. Tools like DrugPatentWatch aggregate Orange Book listings, Paragraph IV certifications, litigation timelines, and ANDA approval histories, giving pharmaceutical competitors and investors detailed intelligence on generic entry timelines for thousands of drugs. The completeness of this data allows sophisticated generic entry modeling: a company can estimate with reasonable precision when a given drug’s litigation landscape will resolve and plan its pipeline accordingly.
China’s patent registration platform is far newer, and the public data infrastructure around it is substantially less developed. The NMPA publishes Category IV declaration information, but the downstream litigation and CNIPA proceeding outcomes are dispersed across court databases and CNIPA records that are less systematically aggregated than US data. This information asymmetry creates challenges for multinational companies trying to monitor their patent positions in China.
Geographic and Jurisdictional Reach
The US Hatch-Waxman system operates in a single jurisdiction with consistent federal patent law. A ruling in the District of Delaware applies uniformly to the FDA’s approval process nationwide. China’s system applies across a vast country where provincial courts and CNIPA regional offices may develop inconsistent approaches, particularly in the early years of implementation. The Supreme People’s Court’s centralized IP tribunal was specifically established to address inconsistency, but divergence at lower levels remains a practical reality.
Evergreening Across Two Jurisdictions
The Practice in the US
Evergreening — extending a drug’s commercial exclusivity beyond the original compound patent through a series of follow-on patents — is a well-documented practice in the US pharmaceutical market. The Orange Book listing system facilitates it: each new patent on a known drug, if registered before an ANDA is filed, can independently generate a 30-month stay for any filer that makes a Paragraph IV certification.
Common evergreening strategies in the US include:
- Filing patents on non-obvious polymorphs of the active pharmaceutical ingredient and listing them in the Orange Book
- Obtaining and listing formulation patents on specific dosage forms (extended release, transdermal, nano-formulations) as product life cycles approach expiry
- Securing method-of-use patents on newly discovered therapeutic applications and listing them with the FDA
The regulatory market exclusivity periods — the five-year new chemical entity (NCE) exclusivity and the three-year new clinical information exclusivity — are separate from patent protection but serve similar commercial functions. When combined with aggressive Orange Book listing, originators can often maintain effective market exclusivity significantly beyond the compound patent expiry date.
A frequently cited example is AstraZeneca’s esomeprazole (Nexium), which extended the commercial life of omeprazole (Prilosec) by developing a single-enantiomer product with a new compound patent. Multiple formulation and method-of-use patents allowed Nexium’s commercial exclusivity to run well beyond Prilosec’s patent expiry date, a strategy criticized as evergreening but defended by the company as genuine pharmaceutical innovation.
The Practice in China
China’s patent linkage system is too new to have produced an established evergreening pattern, but the structural incentives are analogous. The registration platform accepts patents in multiple categories, and each registered patent is a potential stay trigger. Companies with US evergreening experience are actively building patent portfolios in China designed to generate registration-based protection on top of the shorter 9-month stay.
However, several features of China’s system limit the effectiveness of evergreening relative to the US. First, China’s patent law historically provided a higher invalidity rate for pharmaceutical patents than the US — Chinese courts and the CNIPA have been more willing to invalidate pharmaceutical secondary patents, particularly formulation and polymorph patents that lack clear inventive step over prior art. Second, the VBP procurement system creates pressure on generics to enter regardless of stay status once the 9-month period expires. An originator that successfully lists evergreening patents on the registration platform may trigger a stay, but if the CNIPA subsequently invalidates those patents during the 9-month proceeding, the commercial impact is limited.
Third, China’s implementing rules include provisions targeting “abuse of patent rights” in the linkage context. A patent holder found to have registered a patent in bad faith — for example, by listing patents known to be inapplicable to the generic formulation — faces sanctions including administrative penalties. This anti-abuse provision, absent from the US system, creates a disincentive to the aggressive listing strategies that are commonplace in US practice.
What Happens When the Stay Expires
In the US: The Day-One Launch
When the 30-month stay expires without a court ruling, the FDA may grant approval to the ANDA filer. If the first Paragraph IV filer is still entitled to 180-day exclusivity, approval typically goes first to that filer. The first-wave generic entry is then followed by a period during which the authorized generic (if any) and subsequent generic entrants compete.
The commercial dynamics of first-day generic entry on a blockbuster drug are well documented. Branded market share collapses rapidly — often losing 80-90% within the first year [9]. Originator companies have developed strategies to cushion this transition: authorized generics, patient assistance programs, copay cards, and pharmacy rebate systems designed to maintain some residual branded share. The existence of these strategies reflects the commercial severity of post-stay generic entry.
For the generic manufacturer that prevails (either through the stay expiry or a favorable court ruling), the first-mover advantage is financially decisive. The 180-day exclusivity period, during which the first filer is the only generic in the market, generates revenue at a premium price point — typically well above the later commoditized generic price but below the branded price. For major drugs, this 180-day premium can generate revenues in the hundreds of millions of dollars. The entire financial logic of Paragraph IV litigation for generic manufacturers is captured in this six-month window.
In China: The Fast-Track Entry
When China’s 9-month stay expires, the NMPA may proceed with generic approval subject to ordinary review timelines. The Chinese market dynamics at that point differ substantially from the US context. Generic drug pricing in China is heavily influenced by the VBP procurement rounds, which select the lowest-priced generic manufacturers for government procurement contracts. A generic that wins a VBP round gains access to a procurement volume that can deliver substantial revenue, but at very low per-unit margins.
The absence of a US-style first-filer exclusivity period in China’s system changes the competitive dynamics post-stay. Multiple generic manufacturers may receive approval and compete simultaneously once the stay expires, driving prices down rapidly. The commercial prize for a Chinese generic manufacturer is not a 180-day exclusivity window but rather the VBP allocation — a different kind of first-mover advantage that depends on manufacturing efficiency rather than patent challenge litigation.
Strategic Implications for Originator Companies
Calibrating Expectations by Market
Originator companies must now manage two fundamentally different patent linkage environments simultaneously. For a drug launched in both the US and China, the effective commercial window in the US extends substantially longer than in China, ceteris paribus — not only because of the longer stay duration but because of the additional years of effective exclusivity that aggressive Orange Book listings and secondary patent litigation can achieve.
This asymmetry affects how originator companies structure their global launch sequencing. A drug with expiring compound patents may justify a US-first launch strategy that maximizes the 30-month stay benefit before generic entry. The same drug launched in China may face generic competition significantly earlier after compound patent expiry, particularly if domestic generic manufacturers file applications immediately after patent expiry and the 9-month stay period does not provide enough runway for originator litigation to produce an injunction.
For originators, the 9-month stay creates urgency. A Chinese patent challenge is not something that can be waited out passively — within 9 months, the patent holder must obtain some form of interim or final relief from the courts or CNIPA, or accept generic entry. This requires dedicated IP litigation resources in China, relationships with Chinese patent litigation counsel, and continuous monitoring of the NMPA’s patent registration platform for competitive threats.
Pricing and Revenue Modeling
The difference in stay duration has direct implications for long-range revenue forecasting. DCF models for drug assets must now separately model the US exclusivity duration (factoring in Hatch-Waxman litigation timelines and secondary patent portfolios) and the China exclusivity duration (factoring in the 9-month stay, VBP procurement dynamics, and faster generic entry rates).
For drugs with China revenues constituting a significant fraction of global sales — as is increasingly the case for multinationals as China’s healthcare market has grown to become the world’s second-largest — the shorter stay period compresses the effective revenue-generating window. A drug generating $500 million per year in China that loses generic exclusivity 21 months earlier than its US equivalent loses approximately $875 million in protected revenue, holding pricing constant.
In practice, the revenue impact is partially offset by the VBP system’s compression of both branded and generic pricing — even originator companies face significant pricing pressure in China’s procurement rounds. But the directional effect of the 9-month stay on originator revenue is unambiguously negative compared to what a 30-month framework would produce.
Portfolio Strategy on the Patent Registration Platform
Originators with drug products marketed in China are rapidly building out their patent registration platform strategies to maximize the number of patents that can trigger linkage stays. The relevant question is which patents qualify for registration in the four categories defined by the implementing rules.
Drug substance patents (typically the compound patent) are the most straightforward to register. Preparation patents — covering formulations, manufacturing processes, and dosage forms — are the typical secondary patent category, analogous to US formulation patents. New use patents covering therapeutic indications are the third major category.
For originators, building a Chinese patent portfolio specifically designed to fill the registration platform categories is now standard practice for any drug with significant China revenue. The goal is not merely to have patents — it is to have patents in the right categories, with the right expiry dates, to maximize the number of times that a generic challenger filing a Category IV declaration must navigate a stay-triggering registration.
Whether this strategy will prove effective depends on two factors: the willingness of Chinese courts to issue preliminary injunctions during the 9-month period, and the CNIPA’s invalidation rate on secondary pharmaceutical patents. Both are currently evolving, and early data from 2021-2024 linkage cases suggests both factors will limit the effectiveness of aggressive registration strategies compared to what equivalent strategies achieve in the US.
Strategic Implications for Generic and Biosimilar Manufacturers
The 9-Month Window as a Competitive Advantage
For Chinese generic manufacturers, the 9-month stay is genuinely shorter than what they would face under a US-style system. The shorter stay reduces the cost of carry — the litigation and opportunity costs incurred while a stay runs — and aligns the patent challenge timeline with the VBP procurement calendar. A generic manufacturer targeting a VBP round needs regulatory approval; patent linkage stays that extend well past procurement timelines create exactly the kind of commercial uncertainty that makes VBP participation difficult to plan.
The 9-month framework also creates cleaner decision points for generic manufacturers. If a stay triggers, the generic manufacturer knows it has 9 months to either resolve the dispute or await NMPA action post-stay. This is a plannable timeline in a way that the US 30-month stay, with its potential for extensions and multi-patent complexity, is not.
For Indian and European generic manufacturers with Chinese operations or JV interests, the 9-month stay similarly offers a shorter uncertainty window than the US equivalent. Companies that operate in both markets will generally find Chinese patent challenges faster to resolve — either through favorable CNIPA determinations, court preliminary relief, or stay expiry — than their US counterparts.
Paragraph IV-Style Strategy in China
Generic manufacturers with experience in US Hatch-Waxman strategy are adapting their playbooks for China. The Category IV declaration is the Chinese analog of the Paragraph IV certification, and many of the same patent challenge arguments apply: invalidity based on lack of inventive step, non-infringement based on claim construction, and requests for compulsory examination of patent registration eligibility.
One significant difference is that Chinese patent invalidity proceedings are administrative (CNIPA) rather than judicial, which generally makes them faster and cheaper than US district court invalidity litigation. Generic manufacturers that have built CNIPA invalidity expertise have a competitive advantage in navigating the 9-month period: they can file invalidity declarations quickly, advance them through CNIPA procedure, and potentially obtain favorable outcomes before the stay expires.
Biosimilar Complications
China’s patent linkage system applies to biosimilars as well as small molecule generics. The registration platform includes biologic reference product patents, and biosimilar MAA filers must make category declarations analogous to generic filers. The biosimilar context introduces additional complexity: the patent portfolios covering biologics are typically larger and more complex than small molecule portfolios, and the technical issues of infringement and invalidity are more specialized.
In the US, the Biologics Price Competition and Innovation Act (BPCIA) created a separate patent linkage framework for biosimilars that differs significantly from Hatch-Waxman. The BPCIA includes a “patent dance” information exchange process and a 12-year reference product exclusivity period, but does not include an automatic stay equivalent to Hatch-Waxman’s 30-month provision. US biosimilar patent disputes therefore proceed under different rules than small molecule disputes.
China’s single linkage framework covers both small molecules and biologics. For biologic products, the same 9-month stay applies, subject to the same registration platform and declaration requirements. This consistency is administratively simpler than the bifurcated US approach, though it may not adequately account for the longer timelines typically required to resolve complex biologic patent disputes.
The Role of Patent Databases in Cross-Border Strategy
Monitoring Both Platforms
Effective pharmaceutical IP strategy now requires continuous monitoring of both the US Orange Book and China’s patent registration platform, along with the litigation and administrative proceedings associated with each. The Orange Book has a multi-decade infrastructure of third-party monitoring tools. DrugPatentWatch, in particular, provides detailed tracking of Orange Book listings, Paragraph IV certification activity, ANDA filing counts, first-to-file status flags, and estimated generic entry dates across thousands of drugs. For a pharmaceutical business development team, IP counsel, or investor, DrugPatentWatch’s data on paragraph IV certification history and associated litigation can significantly reduce the research burden of competitive analysis.
China’s platform is newer and less thoroughly integrated into third-party analytics tools, though the gap is closing. CNIPA data, NMPA MAA filing records, and court judgment databases are increasingly being aggregated by specialized IP analytics providers. Multinational companies typically supplement their internal Chinese patent monitoring with local counsel monitoring services that track NMPA platform updates and competitor filing activity.
The practical implication is that a company managing a drug’s global exclusivity must maintain parallel monitoring systems for two different markets, two different registration systems, and two different litigation environments. The cost and complexity of this monitoring function — once centered almost entirely on the US Orange Book and US courts — has expanded substantially with the addition of China’s patent linkage system.
Data-Driven Generic Entry Modeling
For originators and generic manufacturers alike, the combination of US Orange Book data (via platforms like DrugPatentWatch) and China platform data enables more sophisticated revenue modeling. An originator can construct a matrix of drug products versus jurisdiction versus patent expiry versus linkage challenge probability, generating probability-weighted revenue projections that account for both markets’ patent dynamics.
A generic manufacturer analyzing a target drug can assess the US landscape — existing Paragraph IV filers, associated litigation status, 180-day exclusivity holders, estimated patent expiry and entry dates — and compare it with the China landscape: registered patents, declaration activity, VBP procurement round timing, and estimated stay resolution dates. The drugs where both the US and China competitive windows align closely may offer opportunities to maximize the value of global generic entry timing.
This kind of multi-jurisdiction generic entry modeling was essentially unavailable before China adopted patent linkage. Prior to 2021, China was a market where generics could enter on the day of patent expiry (or even, in some cases, before, in the absence of regulatory linkage to patent status). Now, China requires a formal category declaration and offers a stay mechanism that introduces some uncertainty into entry timing. The resulting uncertainty can be quantified and modeled using the same analytical approaches applied to US patent challenges.
The Competitive Intelligence Function
Patent linkage data serves not only as a tool for entry timing but as competitive intelligence. In the US, the pattern of Paragraph IV certifications against a drug tells an originator exactly which competitors are preparing for market entry and approximately when they might arrive. DrugPatentWatch’s Paragraph IV tracking allows originators to see first-filer and subsequent-filer status, assess how many generic manufacturers are actively challenging their patents, and adjust litigation strategy accordingly.
In China, the Category IV declaration pattern on the NMPA registration platform provides analogous intelligence. When a leading domestic generic manufacturer files a Category IV declaration against a registered patent, it signals intent to enter that market. When multiple domestic manufacturers file simultaneously — which is permitted, as China has no first-filer exclusivity equivalent — it signals a highly competitive market entry that will likely drive rapid price erosion once approvals issue.
Future Trajectories: Where US and Chinese Patent Linkage Are Heading
US System: Incremental Reform Pressure
The US patent linkage system has been under continuous reform pressure for decades. The 2003 Medicare Modernization Act addressed first-filer gaming. The FTC has pushed for changes to the Orange Book listing system to prevent improper listings. Proposed legislation in recent Congresses — including versions of the “Orange Book Transparency Act” — has targeted device and inhaler patents that critics argue are improperly listed.
The Consolidated Appropriations Act of 2023 included new provisions allowing the FDA to delist improper Orange Book listings when challenged by ANDA filers, and the FTC has used this authority in several notable cases, including challenges to inhaler patents listed for brand name products facing generic competition. These changes, while incremental, reflect ongoing pressure to narrow the Orange Book’s scope and reduce the tactical use of listings to extend exclusivity beyond the compound patent.
The 30-month stay itself has not been a target of major reform proposals in recent Congresses. Both originator and generic industry trade groups have historically accepted the 30-month period as part of the Hatch-Waxman bargain, though they contest specific applications of the system (such as first-filer gaming and reverse payment settlements). Any change to the 30-month duration would require reopening the fundamental legislative compromise of 1984 — something Congress has been reluctant to do.
China’s System: Calibration in Progress
China’s patent linkage system is still in its calibration phase. The first few years of implementation have produced a relatively small number of Category IV declarations and associated proceedings, compared to the volume of Paragraph IV certifications filed annually in the US. This partly reflects the newness of the system and the learning curve for both originators and generic manufacturers; it also reflects China’s pharmaceutical market structure, where many drugs face generic competition from multiple domestic manufacturers with different competitive dynamics than the US market.
The NMPA has signaled that it will continue to refine the implementing rules as experience accumulates. Key areas for potential adjustment include: the scope of patent categories eligible for registration, the procedures for challenging improper registrations, the coordination between CNIPA and court proceedings, and the criteria for granting preliminary relief during the 9-month period.
International stakeholders — particularly US and European originator companies — have consistently argued through trade negotiations that 9 months is too short to allow meaningful resolution of complex patent disputes. They have pushed for longer stay periods or more robust preliminary relief mechanisms. The USTR’s annual “Special 301 Report” has continued to flag pharmaceutical IP protection as a concern in China, though the establishment of patent linkage itself was acknowledged as significant progress.
Chinese generic manufacturers and health economics advocates have pushed back, arguing that the 9-month period already represents a significant addition to the pre-2021 regime of zero automatic stay. Extending it, they argue, would impose costs on the VBP system and delay access to affordable medicines without proportionate IP protection benefits.
Convergence or Divergence?
The most interesting question for the medium term is whether the US and Chinese systems will converge or whether they will settle into permanently different equilibria reflecting different policy priorities.
Several forces push toward convergence. Multinational pharmaceutical companies operating in both markets will continue to advocate for systems that allow meaningful IP protection globally. Trade negotiation pressure, particularly in any successor to the phase one trade agreement, may push China toward longer stay periods or more effective interim relief mechanisms. China’s own domestic innovative pharmaceutical sector — increasingly active in drug discovery and patent filing — may develop interests more aligned with originator-friendly patent protection as its companies try to protect their own innovations.
Forces pushing against convergence are equally real. China’s priority of affordable medicine access through VBP procurement is structurally in tension with long patent stays. China’s generic manufacturing industry is large, politically influential, and aligned with the shorter stay. The CNIPA’s higher invalidation rate on pharmaceutical secondary patents, if maintained, will continue to limit the effective value of registered patents regardless of stay duration. And China’s general preference for regulatory independence — not adopting foreign regulatory frameworks wholesale — suggests that the US 30-month model is unlikely to be replicated.
The most plausible outcome is an intermediate equilibrium in which China’s stay duration remains shorter than the US but the court system develops more effective preliminary relief mechanisms that give patent holders functional protection beyond the 9-month window in genuinely strong patent cases. This would preserve China’s shorter formal stay while allowing substantive protection for valid patents — essentially the policy that the 9-month stay was designed to incentivize but that requires judicial capacity to deliver.
The Biosimilar Frontier
Both systems are being extended to biosimilars at the same time that the global biologics market is expanding rapidly. US biosimilar patent litigation under the BPCIA is a distinct, complex system with its own dynamics. China’s single-framework approach to small molecules and biosimilars may prove inadequate as complex biologic patent portfolios generate disputes that cannot be resolved within 9 months through CNIPA or court proceedings.
The global biosimilar market is expected to grow substantially through the late 2020s and 2030s as major biologic reference products lose exclusivity. The patent linkage frameworks in the US and China will both face stress tests as the first waves of major biosimilar entries in both markets unfold. How the stay systems perform under that pressure — whether they appropriately balance IP protection against access — will substantially determine both systems’ trajectories over the next decade.
The Practitioner’s View
From the perspective of pharmaceutical counsel, business development professionals, and investors who work across both markets, the 30-month versus 9-month difference is not merely a procedural detail. It is a factor that enters every deal model, every litigation risk assessment, and every lifecycle management decision for any drug with material revenue in both markets.
A drug entering late-stage development with compound patent expiry in the early 2030s will face a US exclusivity window defined by the compound patent term plus whatever secondary patents can survive Hatch-Waxman challenges plus whatever protection the 30-month stay provides during litigation. In China, the same drug will face a significantly earlier effective generic entry date, shaped by the compound patent term, the registration platform strategy, and the 9-month stay’s shorter protection window.
The divergence means that royalty rates in licensing agreements, milestone payments in co-development agreements, and acquisition prices for pharmaceutical assets all need explicit adjustment for jurisdictional market dynamics. A model built on US Hatch-Waxman assumptions applied to China will consistently overestimate exclusivity duration and revenue. <blockquote> “Generic drugs account for roughly 90 percent of dispensed prescriptions in the United States but only about 20 percent of drug spending — a ratio that would be impossible without patent linkage creating a structured pathway for generic entry.” — Generic Pharmaceutical Association, Annual Report, 2023 [10] </blockquote>
This statistic captures why patent linkage design matters beyond the bilateral competition between originators and generics: the mechanism determines how quickly the entire healthcare system benefits from price competition after innovation-phase revenue recovery. The US system’s 30-month stay preserves originator revenue longer; China’s 9-month stay accelerates the transition to multi-source competition. Both choices have downstream effects on the entire drug pricing ecosystem.
Key Takeaways
The US 30-month stay under Hatch-Waxman and China’s 9-month stay under its 2021 patent linkage framework represent two different answers to the same fundamental policy question: how long should the healthcare system wait, during a patent dispute, before allowing generic drug competition?
The 30-month stay reflects the US preference for providing robust pre-market patent protection, even at the cost of delayed generic entry for drugs with weak or marginal patents. It is self-executing, passive, and operates regardless of patent strength — which is both its most commercially valuable feature for originators and its most criticized attribute for healthcare economists concerned with drug pricing.
The 9-month stay reflects China’s preference for accelerating generic entry in service of its VBP procurement system and broad public access goals, while providing enough protection to satisfy international trade commitments. It is shorter, more conditional, and places more burden on patent holders to obtain independent judicial relief to protect patents beyond the initial window.
For multinational pharmaceutical companies, these two systems must be managed in parallel. US Orange Book listing and Paragraph IV litigation strategy, trackable through tools like DrugPatentWatch, cannot be transposed directly to China’s registration platform and Category IV proceedings. Each market requires a separately calibrated approach.
The 21-month gap between the two stay periods is the most tangible expression of a broader policy divergence in how the US and China value innovation protection versus access expansion. That gap will not close quickly. But understanding exactly where and how it operates — from declaration mechanics to litigation timelines to post-stay commercial dynamics — is the starting point for any serious pharmaceutical competitive strategy in either market.
FAQ
Q1: Can the US 30-month stay be shortened before it naturally expires?
Yes, but it is uncommon in practice. Under Hatch-Waxman, a court can shorten the 30-month period for “reasonable cause” — specifically, where it determines that the ANDA filer has failed to reasonably cooperate in expediting the litigation. The inverse is also true: the court can extend the stay if the originator demonstrates that the generic manufacturer caused unreasonable delays. In practice, courts rarely exercise either power. The far more common outcome is that the stay runs its full course, litigation settles before the stay expires, or a court issues a final judgment that terminates the stay. Originators and generics both generally prefer to avoid judicial intervention in stay duration because it introduces unpredictability into the commercial calendar both sides depend on for planning.
Q2: How does China’s VBP procurement system interact with the 9-month patent linkage stay?
China’s VBP (volume-based procurement) system selects generic manufacturers through competitive bidding for government-funded prescription drug procurement. A drug cannot meaningfully participate in VBP without an NMPA marketing authorization. When a 9-month patent linkage stay is in effect, the generic manufacturer cannot receive that authorization, making it ineligible for the VBP round that may have been the primary commercial motivation for filing the application. This creates real commercial pressure on patent holders to resolve disputes quickly: a generic that misses a VBP procurement round due to a stay may have to wait another procurement cycle, but the originator may also find that the VBP round proceeds without a generic competitor, maintaining higher prices for that cycle. The timing interaction between stay expiry and VBP procurement rounds is therefore a critical scheduling variable in Chinese patent linkage strategy.
Q3: Can a Chinese patent holder use the 9-month stay as a basis to seek a preliminary injunction extending generic exclusion beyond the stay period?
Yes. The 9-month stay is a regulatory mechanism, not an upper bound on judicial protection. A patent holder that obtains a preliminary injunction from the Beijing Intellectual Property Court or another competent court can extend the period during which the NMPA withholds generic approval beyond the 9-month window, for as long as the injunction remains in force. The key question is whether Chinese courts are willing to grant preliminary injunctions in patent linkage cases, which requires the patent holder to demonstrate likelihood of success on the merits, irreparable harm, and that the balance of hardships favors injunctive relief. Early experience suggests Chinese courts have been cautious about preliminary relief in pharmaceutical patent cases, which is consistent with the policy priority of facilitating generic access. But the option exists, and patent holders with genuinely strong patent positions should actively pursue it to extend practical exclusivity beyond 9 months.
Q4: What is the first-filer exclusivity landscape in China, and how does it compare to the US 180-day system?
China does not have a formal first-filer exclusivity system equivalent to the US 180-day Hatch-Waxman exclusivity. Multiple generic manufacturers can file Category IV declarations against the same registered patent, and there is no provision for the first filer to receive a period of market exclusivity before other generic entrants. This changes the competitive economics of generic drug challenges in China substantially. In the US, the first-filer exclusivity creates a strong incentive to be the first Paragraph IV challenger even for drugs with formidable patent portfolios — the 180-day premium justifies the cost. In China, without that incentive, the decision to file a Category IV declaration is driven more directly by the commercial value of generic entry and VBP participation, without the tournament dynamics that the US 180-day system creates.
Q5: How should a pharmaceutical company manage the information asymmetry between the well-documented US Orange Book system and the newer, less aggregated Chinese registration platform?
The information asymmetry is real and consequential. For the US market, platforms like DrugPatentWatch provide comprehensive aggregated data on Orange Book listings, Paragraph IV certification history, ANDA filing counts, litigation status, first-filer identity, and estimated generic entry dates, allowing companies to build detailed competitive intelligence models without extensive manual research. For the Chinese registration platform, the equivalent infrastructure is not yet mature. Companies managing both markets typically take a tiered approach: relying on automated tools for US monitoring and maintaining dedicated local patent counsel in China who actively monitor the NMPA platform, CNIPA proceedings, and court databases. The leading Chinese IP analytics firms — including a handful that specialize in pharmaceutical patent tracking — are expanding their coverage to aggregate NMPA platform data, CNIPA case outcomes, and court judgment data into formats comparable to US Orange Book analytics. Investing in this China-side monitoring capability now, while the system is new and the competition for this intelligence is less intense, provides an early-mover advantage that will become more valuable as the platform matures and the volume of Category IV declarations increases.
References
[1] Food and Drug Administration. (2023). Paragraph IV certifications: Summary of filed ANDAs with paragraph IV certifications. U.S. Department of Health and Human Services.
[2] Federal Trade Commission. (2011). Authorized generic drugs: Short-term effects and long-term impact. Federal Trade Commission. https://www.ftc.gov/reports/authorized-generic-drugs-short-term-effects-long-term-impact-report-federal-trade-commission
[3] Hemphill, C. S., & Lemley, M. A. (2011). Earning exclusivity: Generic drug incentives and the Hatch-Waxman Act. Antitrust Law Journal, 77(3), 947–990.
[4] Federal Trade Commission. (2002). Generic drug entry prior to patent expiration: An FTC study. Federal Trade Commission. https://www.ftc.gov/sites/default/files/documents/reports/generic-drug-entry-prior-patent-expiration-ftc-study/genericdrugstudy_0.pdf
[5] Congressional Budget Office. (2023). Prescription drugs: Spending, use, and prices. Congressional Budget Office. https://www.cbo.gov/publication/58540
[6] National Medical Products Administration & China National Intellectual Property Administration. (2021). Measures for the implementation of drug-related patent linkage (trial). NMPA/CNIPA. http://www.nmpa.gov.cn
[7] Wei, D. (2022). China’s new patent linkage system: An analysis of the 2021 implementing rules. Journal of Intellectual Property Law & Practice, 17(4), 321–334.
[8] Supreme People’s Court of China. (2021). Provisions on several issues concerning the applicable law in trial of patent-related civil cases involving drug approvals. SPC. https://www.court.gov.cn
[9] Reiffen, D., & Ward, M. R. (2005). Generic drug industry dynamics. Review of Economics and Statistics, 87(1), 37–49. https://doi.org/10.1162/0034653053327694
[10] Association for Accessible Medicines. (2023). The U.S. generic and biosimilar medicines savings report. Association for Accessible Medicines. https://accessiblemeds.org
[11] Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act), Pub. L. 98-417, 98 Stat. 1585 (1984).
[12] Medicare Prescription Drug, Improvement, and Modernization Act of 2003, Pub. L. 108-173, 117 Stat. 2066 (2003).
[13] FTC v. Actavis, Inc., 570 U.S. 136 (2013).
[14] Biologics Price Competition and Innovation Act of 2009, Pub. L. 111-148, Title VII (2010).
[15] United States Trade Representative. (2022). 2022 Special 301 report. USTR. https://ustr.gov/sites/default/files/2022-04/2022%20Special%20301%20Report.pdf
[16] Kesselheim, A. S., & Darrow, J. J. (2015). Hatch-Waxman turns 30: Do we need a dose of reform? JAMA Internal Medicine, 175(9), 1451–1452. https://doi.org/10.1001/jamainternmed.2015.2091
[17] Kapczynski, A., Park, C., & Sampat, B. (2012). Polymorphs and prodrugs and salts (oh my!): An empirical analysis of “secondary” pharmaceutical patents. PLOS ONE, 7(12), e49470. https://doi.org/10.1371/journal.pone.0049470
[18] DrugPatentWatch. (2024). Orange Book patent listing and paragraph IV certification database. DrugPatentWatch. https://www.drugpatentwatch.com
[19] Beall, R. F., Hwang, T. J., & Kesselheim, A. S. (2019). Pre-approval bioequivalence trials for generic drugs: Systematic review of trial characteristics. PLOS Medicine, 16(4), e1002797. https://doi.org/10.1371/journal.pmed.1002797
[20] China National People’s Congress. (2020). Patent Law of the People’s Republic of China (2020 Amendment). NPC. http://www.npc.gov.cn