The global pharmaceutical industry currently faces a historic loss-of-exclusivity super-cycle. Between 2025 and 2030, an estimated $200 billion to $400 billion in annual brand-name drug revenue enters the risk zone as primary patents expire.1 For generic manufacturers, this era represents a high-stakes race to secure the 180-day exclusivity period, often described as the “brass ring” of the sector.1 This six-month window provides a temporary duopoly with the brand-name sponsor, allowing the first generic entrant to capture a disproportionate share of the drug’s lifetime profits.1 However, this incentive carries a significant burden: the ‘Use It or Lose It’ framework established by the Medicare Prescription Drug, Improvement, and Modernization Act (MMA) of 2003.7 This report decodes the technical mechanisms of 180-day exclusivity forfeiture, providing the strategic intelligence necessary for professionals to navigate the regulatory minefield and transform complex patent data into a competitive advantage.
The Architecture of Generic Competition
The foundation of the modern generic industry rests on the Drug Price Competition and Patent Term Restoration Act of 1984, better known as the Hatch-Waxman Act.9 Before this legislation, generic firms faced a cost-prohibitive landscape. They had to perform independent clinical trials to prove safety and efficacy, even though the innovator had already done so.11 Hatch-Waxman removed these barriers by creating the Abbreviated New Drug Application (ANDA) pathway.9
Under this regime, generic applicants rely on the safety and efficacy data of the Reference Listed Drug (RLD).13 To gain approval, they must demonstrate bioequivalence through pharmacokinetic and pharmacodynamic testing.3 This ensures the generic delivers the same amount of active ingredient to the bloodstream over the same period as the pioneer drug.11 In exchange for this expedited entry, the Act gave innovators patent term extensions and a 30-month stay of generic approval during patent litigation.4
The Paragraph IV Certification
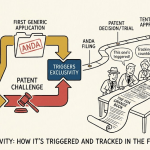
The legal engine of generic disruption is the Paragraph IV certification.11 When filing an ANDA, an applicant must certify against every patent listed in the FDA’s Orange Book for the RLD.11
| Certification Type | Legal Basis | Strategic Implication |
| Paragraph I | Patent information not filed in the Orange Book. | Rare for modern blockbusters; suggests an “open” market.4 |
| Paragraph II | The listed patent has already expired. | Routine entry; usually results in a crowded market.4 |
| Paragraph III | The applicant seeks approval after the patent expires. | Predictable entry; avoids litigation but yields no exclusivity.3 |
| Paragraph IV | The patent is invalid, unenforceable, or not infringed. | Triggers litigation and grants 180-day exclusivity to first filers.11 |
A Paragraph IV (PIV) filing is an “artificial act of infringement” under 35 U.S.C. § 271(e)(2).1 This mechanism grants the brand manufacturer subject matter jurisdiction to sue the generic applicant before any commercial harm occurs.1 If the brand files suit within 45 days of receiving a notice letter, the FDA stays the generic approval for up to 30 months.4 This stay acts as a critical defensive shield for innovators, preserving their monopoly while the court adjudicates the patent dispute.4
The 180-Day Bounty
To encourage challenges against weak or non-innovative patents, Congress created a powerful carrot: 180 days of marketing exclusivity for “first applicants”.7 This exclusivity blocks the FDA from approving any subsequent generic application for the same drug during that window.1 This period typically accounts for 60% to 80% of a generic product’s total lifetime profits.5
The value of being first is disproportionate. Statistics from DrugPatentWatch and the FDA indicate that a single generic entrant maintains high margins by pricing at a significant discount—often 20% to 40%—below the brand price.1 However, as the number of entrants increases, price erosion accelerates.
| Number of Generic Entrants | Price Reduction vs. Brand | Market Dynamic |
| 1 (First-to-File) | 20% – 40% | High-margin duopoly; “brass ring” phase.1 |
| 2 Competitors | ~50% – 55% | Prices crack; margins compress.1 |
| 3 – 5 Competitors | 60% – 70% | The “commoditization cliff” begins.1 |
| 6+ Competitors | > 95% | Pure commodity; profits depend on volume.20 |
The Forfeiture Regime: MMA 2003
Before the MMA 2003, the generic industry exploited a massive loophole: “exclusivity parking”.5 A first filer could settle with a brand manufacturer and agree to delay its launch indefinitely. Because the 180-day clock only started upon commercial launch or a final court decision, this “parked” exclusivity acted as a bottleneck, preventing any other generic from ever reaching the market.1
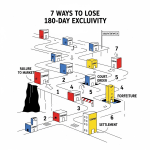
The 2003 amendments sought to clear this bottleneck by introducing mandatory forfeiture triggers.7 These triggers force first applicants to launch or lose their lucrative exclusivity.
Trigger 1: Failure to Market
The most complex forfeiture event is the “failure to market” under 21 U.S.C. § 355(j)(5)(D)(i)(I). Exclusivity is forfeited if the first applicant fails to market the drug by the “later of” two specific dates: Date (aa) and Date (bb).23
The formula for the forfeiture date is:
$$\text{Forfeiture Date} = \max(D_{aa}, D_{bb})$$
Date (aa) is the earlier of:
- 75 days after the ANDA receives final approval.23
- 30 months after the date of submission of the application.23
Date (bb) is 75 days after the date on which at least one of the following occurs regarding the patents that qualified the applicant for exclusivity:
- A final court decision of invalidity or non-infringement from which no appeal can be taken.23
- A court signs a settlement order or consent decree with a finding of invalidity or non-infringement.23
- The patent information is withdrawn (delisted) from the Orange Book.7
This trigger creates the “Catch-22” for generic strategists.25 If a generic firm wins its patent challenge early but other patents still block its path, it may be forced to launch “at-risk” or forfeit exclusivity before it is ready to compete.4
Trigger 2: Failure to Obtain Tentative Approval
Under § 355(j)(5)(D)(i)(IV), a first applicant forfeits exclusivity if it fails to obtain “tentative approval” within 30 months of filing the application.23 Tentative approval means the ANDA meets all scientific and manufacturing standards but cannot receive final approval due to existing patents or exclusivities.27
The FDA applies a strict “bright-line rule” here. If the clock hits 30 months and 1 day, exclusivity is generally gone.28 However, the statute provides a critical safety valve: forfeiture is avoided if the failure is “caused by a change in or a review of the requirements for approval” imposed after the filing date.26
Case Study: Mutual Pharmaceutical and Carvedilol Phosphate
Mutual Pharmaceutical qualified as a first applicant for generic Coreg CR (Carvedilol Phosphate).27 It missed its 30-month deadline, as its application was still deficient in bioequivalence and product quality.27 However, just months before the deadline, the FDA issued new draft guidance requiring dissolution testing with ethanol to prevent “alcohol dose dumping”.27 Because Mutual was actively addressing this new requirement at the 30-month mark, the FDA ruled the delay was “caused by” the requirement change.27 Mutual preserved its exclusivity.27
Case Study: Zydus and Mesalamine (Lialda)
A similar situation occurred with Zydus and generic Lialda.32 Zydus failed to obtain approval within 30 months, but the FDA had revised bioequivalence standards for mesalamine while the application was pending.32 The Agency “punted” on the initial decision but eventually confirmed that Zydus’s efforts to comply with the new methodology saved its exclusivity.32
Trigger 3: Withdrawal or Amendment of Application
Exclusivity is forfeited if the first applicant voluntarily withdraws its ANDA or the FDA deems it withdrawn due to non-compliance.4 Furthermore, amending or withdrawing the PIV certification—switching to a Paragraph III or a Section viii carve-out—will trigger forfeiture for all patents qualifying the applicant for exclusivity.23
Trigger 4: Anticompetitive Agreements
The MMA requires all patent settlement agreements between brand and generic firms to be filed with the FTC and the Department of Justice.11 If a court or the FTC finds that the first applicant entered into an agreement with the brand or another applicant that violates antitrust laws (often called “reverse payment” or “pay-for-delay” deals), exclusivity is forfeited.23
Trigger 5: Expiration of All Patents
This is a logical forfeiture event. If every patent for which the applicant filed a PIV certification expires, the exclusivity window disappears.13 There is no “vested right” to exclusivity once the legal barriers to other generics have naturally lapsed.34
The Lipitor Crisis and the AIP Barrier
The launch of generic Lipitor (atorvastatin) provides the ultimate investigative study in regulatory risk.1 Ranbaxy Pharmaceuticals was the sole first-to-file applicant, positioning it for a windfall on a drug generating $10 billion annually.1 However, the FDA’s discovery of fraud and manufacturing violations at Ranbaxy’s Paonta Sahib and Dewas plants triggered the Application Integrity Policy (AIP).1
The AIP effectively froze Ranbaxy’s applications.1 Because Ranbaxy could not receive approval, and no other generic could launch until Ranbaxy’s 180-day clock ran out, the entire generic market for atorvastatin was paralyzed.1 This created a multi-billion dollar bottleneck.
The solution came through a strategic, albeit controversial, collaboration.1 Teva entered into an agreement with Ranbaxy in 2010 to assist in manufacturing and supply chain compliance.1 This allowed a launch in November 2011, with Teva and Ranbaxy sharing the profits.1 This case proves that regulatory assets like exclusivity are tradable commodities; even a company with crippled manufacturing can monetize its first-to-file status through partnership.1
The “Authorized Generic” Tax
While the 180-day period blocks other ANDA applicants, it does not block the brand manufacturer from selling its own “authorized generic” (AG).1 Brands use AGs as a standard defensive moat to capture a portion of the generic market and preserve revenue.1
| Impact Category | Statistic / Value | Economic Ripple Effect |
| First-Filer Revenue Loss | 40% – 52% | Significantly reduces the “prize” for patent challenges.36 |
| Retail Price Reduction | 4% – 8% lower | Increases consumer savings during exclusivity.36 |
| Wholesale Price Reduction | 7% – 14% lower | Aggressive competition from day one.36 |
| Post-Exclusivity Impact | 53% – 62% lower revenue | Long-term market share remains suppressed for first filer.36 |
Brands recognize that launching an AG at the moment of generic entry increases their own profits while simultaneously slashing the profits of their rivals.36 For skeptical generic strategists, modeling a 50% revenue reduction during the 180-day window due to AG entry is now standard operating procedure.1
The 2026 “Super-Cliff” Watchlist
As we move into 2026, the convergence of the biologic “super-cliff” and traditional small-molecule expirations creates a hyper-dynamic transition.2 Analysts using DrugPatentWatch mark these dates in red, as multi-billion dollar revenue streams face imminent erosion.1
| Drug Name | Primary Manufacturer | Estimated Peak Revenue at Risk | Strategic Status (2026) |
| Eliquis (Apixaban) | BMS / Pfizer | $12 Billion | Patent extension ends Nov 21, 2026; “Clean Cliff” erosion expected.6 |
| Januvia (Sitagliptin) | Merck | $2.2 Billion | Multi-source generic entry expected Spring 2026 via settlement.2 |
| Ozempic (Semaglutide) | Novo Nordisk | $5 – $10 Billion (U.S.) | Key patent expires March 20, 2026; device patents extend to 2031.37 |
| Stelara (Ustekinumab) | J&J | $10 Billion | Complex conflict between biosimilar entry and IRA negotiation.6 |
| Imbruvica (Ibrutinib) | AbbVie / J&J | $2.8 Billion | Supplemental patents expire Sep 20, 2026.37 |
For small molecules like Januvia and Eliquis, price erosion will reach 90% within months of launch.2 In contrast, biologics like Humira and Stelara experience a slower “slope” erosion because biosimilar entry costs $100 million to $250 million, far higher than the $1 million to $5 million required for small-molecule generics.6
Strategic Intelligence: Transforming Data into Alpha
In this environment, static data is a liability. High-level professionals use competitive intelligence (CI) platforms to identify “soft targets” in the patent landscape.1 Platforms like DrugPatentWatch have become essential for monitoring three distinct signals.1
1. Paragraph IV Monitoring
Analysts track the “starting gun” of litigation. By identifying when a competitor has filed an ANDA, firms can prepare to litigate in parallel or identify markets with “inadequate competition” for Competitive Generic Therapy (CGT) status.1
2. Supply Chain Forensics
By identifying choke points in the API supply chain from India and China, analysts can predict a generic launch years before an ANDA is actually filed.14 A manufacturer’s ability to guarantee supply in the face of shortages is a key differentiator that justifies premium contracts.40
3. AI-Driven Opportunity Scoring
Modern CI teams utilize agentic AI to ingest 70,000 International Patent Classification (IPC) fields.14 They evaluate targets based on “strategic fit,” “technical credibility,” and “market pull”.14 This allows firms to identify internal programs with high litigation risk early, generating ROI through avoided legal costs.14
Second-Order Insights: The Ripple Effects of Forfeiture
The “Use It or Lose It” rule does more than just regulate timing; it reshapes corporate behavior and R&D philosophy across the entire pharmaceutical ecosystem.
The Shift from Law to Science
Because the forfeiture triggers increasingly focus on regulatory speed (the 30-month tentative approval rule), generic firms are shifting resources from legal departments to regulatory and manufacturing compliance.1 Manufacturing quality is no longer just a cost center; it is a strategic asset.1 A single warning letter from the FDA can now lead to the loss of a $500 million exclusivity window.
Negotiation as the New Exclusivity
With the Inflation Reduction Act (IRA), high-revenue drugs are selected for price negotiation in their 9th year.6 Generic challengers must now calculate ROI based on the negotiated brand price, not the launch price.6 This narrows the profit margin for generics and may discourage challenges in smaller markets where the “brass ring” is already partially tarnished by government-mandated discounts.
The “Volume-Limited” Masterstroke
Innovators like Celgene (BMS) have learned to avoid the patent cliff through volume-limited settlements.38 For Revlimid (lenalidomide), they negotiated agreements that cap generic market share until 2026.38 This structure preserves brand revenue while offering generics a guaranteed, if limited, revenue stream.38 It avoids the “flash crash” of brand revenue, allowing for a “controlled descent”.14
Conclusion: The New Playbook for 180-Day Exclusivity
The ‘Use It or Lose It’ rule has effectively turned 180-day exclusivity from an entitlement into a high-stakes performance contract. To win and keep the “brass ring,” generic manufacturers must master a multi-disciplinary playbook:
- Speed to File: The first-to-file status remains the primary value driver, requiring rapid pre-ANDA preparation.1
- Regulatory Precision: Missing the 30-month tentative approval window is the most common path to forfeiture.27 Firms must aggressively use the “caused by requirement changes” exception to preserve their status.27
- Supply Chain Resilience: In an era of disruptive shortages, the ability to guarantee inventory for “Day 1” readiness is a competitive weapon.1
- Data-Driven Intelligence: Using platforms like DrugPatentWatch to monitor litigation outcomes and settlement “date certain” entries provides the alpha needed to outpace rivals.1
For the skeptical business professional, the lesson is clear: exclusivity is king, but it is fragile.1 Success in the contemporary market requires more than manufacturing efficiency; it demands a sophisticated integration of legal acumen, regulatory savvy, and real-time competitive intelligence. As the 2026 super-cliff approaches, those who decode the forfeiture maze will capture the lion’s share of the staggering $400 billion market rotation.1
Works cited
- A Strategic Analysis of Generic Drug Launches, Patent Litigation, and Market Exclusivity, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/a-strategic-analysis-of-generic-drug-launches-patent-litigation-and-market-exclusivity/
- 7 Strategies to Analyze Patent Cliffs for Generic Opportunities in 2026 – Patsnap, accessed February 6, 2026, https://www.patsnap.com/fr/resources/blog/articles/analyze-patent-cliffs-generic-opportunities-2026/
- The First-Mover’s Gambit: A Strategic Guide to Maximizing the 180-Day Generic Exclusivity Advantage – DrugPatentWatch – Transform Data into Market Domination, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/the-first-movers-gambit-a-strategic-guide-to-maximizing-the-180-day-generic-exclusivity-advantage/
- A Strategic Playbook for Timing ANDA Submissions Using Drug Patent Data, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/a-strategic-playbook-for-timing-anda-submissions-using-drug-patent-data/
- Earning Exclusivity: Generic Drug Incentives and the Hatch-‐Waxman Act1 C. Scott – Stanford Law School, accessed February 6, 2026, https://law.stanford.edu/wp-content/uploads/2015/06/ssrn-id1736822.pdf
- Mastering LOE: Expert Strategies to Predict Drug Patent Expiry and Seize Generic Market Share – DrugPatentWatch, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/mastering-loe-expert-strategies-to-predict-drug-patent-expiry-and-seize-generic-market-share/
- Earning Exclusivity: Generic Drug Incentives and the Hatch …, accessed February 6, 2026, https://law.stanford.edu/index.php?webauth-document=publication/259458/doc/slspublic/ssrn-id1736822.pdf
- Issues in the Interpretation of 180-Day Exclusivity – University of Missouri School of Law, accessed February 6, 2026, https://scholarship.law.missouri.edu/facpubs/554/
- The Balance Between Innovation and Competition: The Hatch-Waxman Act, the 2003 Amendments, and Beyond – Harvard DASH, accessed February 6, 2026, https://dash.harvard.edu/bitstreams/7312037c-94cf-6bd4-e053-0100007fdf3b/download
- Continuing Abuse of the Hatch-Waxman Act by Pharmaceutical Patent Holders and the Failure of the 2003 Amendments – UC Law SF Scholarship Repository, accessed February 6, 2026, https://repository.uclawsf.edu/cgi/viewcontent.cgi?article=3715&context=hastings_law_journal
- Landmark Paragraph IV Patent Challenge Decisions: A Strategic Playbook for Generic Manufacturers – DrugPatentWatch – Transform Data into Market Domination, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/landmark-paragraph-iv-patent-challenge-decisions-a-strategic-playbook-for-generic-manufacturers/
- The Hatch-Waxman Act and Market Exclusivity for Generic Manufacturers: An Entitlement or an Incentive? – Scholarly Commons @ IIT Chicago-Kent College of Law, accessed February 6, 2026, https://scholarship.kentlaw.iit.edu/cgi/viewcontent.cgi?article=3541&context=cklawreview
- September 28, 2015 VIA REGULATIONS.GOV Maryll W. Toufanian Acting Deputy Director Office of Generic Drug Policy U.S. Food and Dr, accessed February 6, 2026, https://downloads.regulations.gov/FDA-2015-N-2713-0031/attachment_1.pdf
- Navigating Paragraph IV Challenges, the Biologic Super-Cliff, and AI-Driven IP Valorization, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/what-every-pharma-executive-needs-to-know-about-paragraph-iv-challenges/
- Brief for the United States as Amicus Curiae – Department of Justice, accessed February 6, 2026, https://www.justice.gov/atr/case-document/brief-united-states-amicus-curiae-5
- Guidance for Industry 180-Day Exclusivity: Questions and Answers – FDA, accessed February 6, 2026, https://www.fda.gov/media/102650/download
- 1 OFFICE OF THE ATTORNEY GENERAL OF THE STATE OF NEW …, accessed February 6, 2026, https://www.naag.org/wp-content/uploads/2020/10/666.civil_.NY-AOD_Teva_Ranbaxy_Signed-3.pdf
- The Paragraph IV Playbook: Turning Patent Challenges into Market Dominance, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/the-paragraph-iv-playbook-turning-patent-challenges-into-market-dominance/
- 21 U.S.C. § 355 – U.S. Code Title 21. Food and Drugs § 355 – Codes – FindLaw, accessed February 6, 2026, https://codes.findlaw.com/us/title-21-food-and-drugs/21-usc-sect-355/
- The Hatch-Waxman 180-Day Exclusivity Incentive Accelerates Patient Access to First Generics, accessed February 6, 2026, https://accessiblemeds.org/resources/fact-sheets/the-hatch-waxman-180-day-exclusivity-incentive-accelerates-patient-access-to-first-generics/
- Are You Falling Behind? The New Rules of Generic Drug Portfolio Management, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/are-you-falling-behind-the-new-rules-of-generic-drug-portfolio-management/
- An Administrative Meter Maid: Using Inter Partes Review and Post-Grant Review to Curb Exclusivity Parking via the “Failure, accessed February 6, 2026, https://repository.law.umich.edu/cgi/viewcontent.cgi?article=1226&context=mlr
- 180-Day Generic Drug Exclusivity – Forfeiture – UC Berkeley Law, accessed February 6, 2026, https://www.law.berkeley.edu/wp-content/uploads/2024/05/180-Day-Generic-Drug-Exclusivity-%E2%80%93-Forfeiture.pdf
- ORI GI NAL – Regulations.gov, accessed February 6, 2026, https://downloads.regulations.gov/FDA-2007-N-0269-0008/attachment_1.pdf
- District Court Decision Acts to Preserve 180-Day Exclusivity …, accessed February 6, 2026, https://www.thefdalawblog.com/2009/12/district-court-decision-acts-to-preserve-180day-exclusivity-eligibility-an-interesting-solution-arou/
- Norwich Pharms., Inc. v. Kennedy Jr. | Robins Kaplan LLP Law Firm, accessed February 6, 2026, https://www.robinskaplan.com/newsroom/insights/generically-speaking-hatch-waxman-bulletin-2025-q2-norwich-pharms-inc-v-kennedy-jr
- 180-day Exclusivity for Carvedilol Phosphate Extended-Release …, accessed February 6, 2026, https://www.axinn.com/a/web/v2r5VKx7mxMi53nKW2XnHi/carvedilol-er-capsules-fda-forfeiture-ltr-decision.pdf
- A SANDOZ – Regulations.gov, accessed February 6, 2026, https://downloads.regulations.gov/FDA-2012-P-0661-0001/attachment_1.pdf
- Definition: forfeiture event from 21 USC § 355(j)(5) | LII / Legal Information Institute, accessed February 6, 2026, https://www.law.cornell.edu/definitions/uscode.php?width=840&height=800&iframe=true&def_id=21-USC-1613252815-174134459&term_occur=999&term_src=
- Definition: 180-day exclusivity period from 21 USC § 355(j)(5) – Cornell Law School, accessed February 6, 2026, https://www.law.cornell.edu/definitions/uscode.php?width=840&height=800&iframe=true&def_id=21-USC-1727272510-174134459&term_occur=999&term_src=title:21:chapter:9:subchapter:V:part:A:section:355
- FDA’s Draft Guidance for Industry on 180-Day … – Duane Morris LLP, accessed February 6, 2026, https://www.duanemorris.com/alerts/fda_draft_guidance_for_industry_on_180_day_exclusivity_0317.html
- FDA Says No 180-Day Exclusivity Forfeiture for Generic LIALDA Based on Changed Bioequivalence Recommendations, accessed February 6, 2026, https://www.thefdalawblog.com/2018/03/fda-says-no-180-day-exclusivity-forfeiture-for-generic-lialda-based-on-changed-bioequivalence-recommendations/
- The Hatch-Waxman Act: A Primer – Congress.gov, accessed February 6, 2026, https://www.congress.gov/crs_external_products/R/PDF/R44643/R44643.3.pdf
- APOTEX, INC. – FDA Law Blog, accessed February 6, 2026, https://www.thefdalawblog.com/wp-content/uploads/archives/docs/losartan—apotex-emergency-stay-of-judgment-pending-appeal.pdf
- Patent Expiration and 180-Day Generic Exclusivity Under the Hatch-Waxman Act: Teva v. Sebelius – Bloomberg Law News, accessed February 6, 2026, https://news.bloomberglaw.com/pharma-and-life-sciences/patent-expiration-and-180-day-generic-exclusivity-under-the-hatch-waxman-act-teva-v-sebelius
- Authorized Generic Drugs: Short-Term Effects and Long-Term …, accessed February 6, 2026, https://www.ftc.gov/sites/default/files/documents/reports/authorized-generic-drugs-short-term-effects-and-long-term-impact-report-federal-trade-commission/authorized-generic-drugs-short-term-effects-and-long-term-impact-report-federal-trade-commission.pdf
- Drug Patents Expiring in 2026: A Comprehensive Guide – IntuitionLabs, accessed February 6, 2026, https://intuitionlabs.ai/articles/drug-patent-expirations-2026
- Evergreening by Lawsuit: Strategic Patent Actions and Generic Entry Stagnation, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/evergreening-by-lawsuit-strategic-patent-actions-and-generic-entry-stagnation/
- Competitive Generic Therapy Exclusivity: Maximizing the 180-Day Advantage, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/competitive-generic-therapy-exclusivity-maximizing-the-180-day-advantage/
- Crafting a Winning Marketing Strategy for Generic Drugs – DrugPatentWatch, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/crafting-a-winning-marketing-strategy-for-generic-drugs/
- Congressional Investigation of RevAssist-Linked and General Pricing Strategies for Lenalidomide | JCO Oncology Practice – ASCO Publications, accessed February 6, 2026, https://ascopubs.org/doi/10.1200/OP.23.00579
- Lower Cost Revlimid: Generic Lenalidomide is Here – International Myeloma Foundation, accessed February 6, 2026, https://www.myeloma.org/blog/lower-cost-revlimid-generic-lenalidomide-here
- How to Win Post-ANDA: Drug Development & Patent Hacks – DrugPatentWatch, accessed February 6, 2026, https://www.drugpatentwatch.com/blog/how-to-win-post-anda-drug-development-patent-hacks/