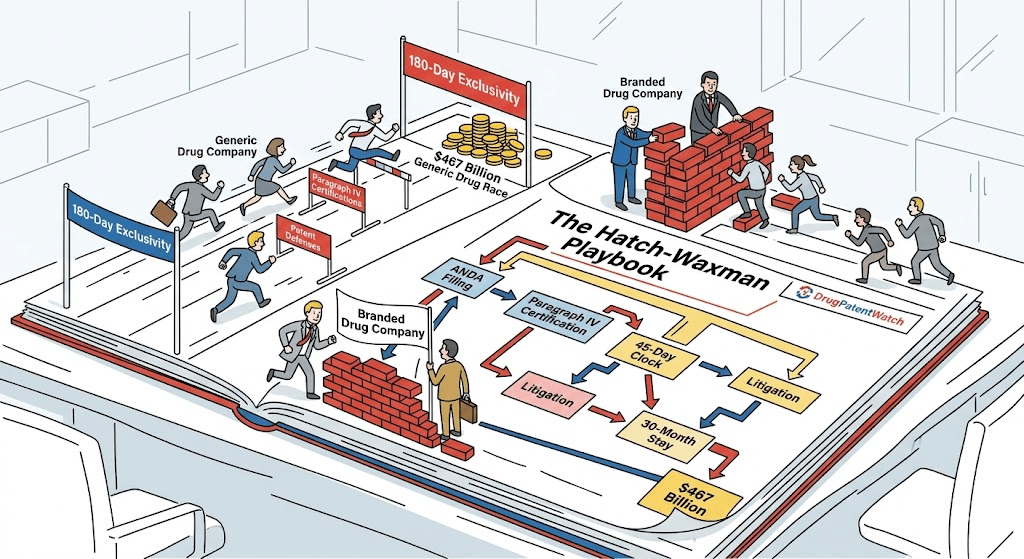
Every major pharmaceutical patent war starts with a single form filed with the FDA. It is not a lawsuit. No judge is involved. No jury is impaneled. A generic drug company submits an Abbreviated New Drug Application — an ANDA — and checks a box labeled “Paragraph IV.” In doing so, it accuses a brand-name manufacturer’s listed patents of being invalid, unenforceable, or not infringed by its product. The brand has 45 days to sue. If it does, a 30-month clock starts. If it does not, the generic can launch.
That one checkbox has reshaped the global pharmaceutical industry since 1984. It is the legal architecture that allowed Teva to become the largest generic drug manufacturer on earth. It produced the Cephalon pay-for-delay scandal, the FTC v. Actavis Supreme Court ruling, and billions in savings for American patients. It is, without question, the most financially consequential single regulatory mechanism in the history of U.S. drug law. <blockquote> “Generic and biosimilar medicines generated $467 billion in system-wide savings in 2024 alone and $3.4 trillion over the preceding decade.” — Association for Accessible Medicines, *Generic and Biosimilar Medicines Savings Report*, 2025 [1] </blockquote>
This guide covers the Hatch-Waxman Act from the ground up: how the system was designed, how it actually operates, what 180-day exclusivity is worth in real financial terms, and why — four decades after Senator Orrin Hatch and Representative Henry Waxman shook hands on a compromise — the law’s core tensions are sharper than ever. If you are a pharmaceutical attorney, a generic drug strategist, an investor evaluating a brand company’s revenue risk, or a policy professional tracking the IRA’s impact on the patent cliff, this is the operational context you need.
Part One: The Architecture — Why Congress Built This System
The Problem the Act Was Solving
Before 1984, getting a generic drug to market in the United States required clinical trials. Full ones. A generic manufacturer had to prove its product was safe and effective through the same costly human studies that the brand manufacturer had conducted years earlier. This was not a scientific requirement — the active molecules were identical — it was a regulatory artifact that functionally extended brand monopolies indefinitely. Generic manufacturers could not afford the trials, so they did not try. When patents expired on major drugs, brand manufacturers often faced no generic competition for years, sometimes a decade.
The political coalition that passed Hatch-Waxman in September 1984 was unusual: brand manufacturers and generic manufacturers both wanted it. Brand companies got two things — patent term restoration (up to five years to recover time lost to FDA review) and new regulatory exclusivity periods, including five years for new chemical entities and three years for new clinical studies. Generic companies got the Abbreviated New Drug Application pathway, allowing them to prove bioequivalence rather than repeat clinical trials, and the research exemption “safe harbor” allowing them to begin development work on patented drugs without infringement liability. The bargain worked. Before 1984, generics accounted for approximately 19% of U.S. prescriptions dispensed [2]. By 2024, that figure exceeded 90% [3].
The Paragraph IV mechanism was the most clever piece of the original design. Congress understood that patent expiration dates would not by themselves produce generic competition — brands could simply list additional patents and extend their exclusivity period organically. The system needed a way to accelerate competition before nominal patent expiration. Paragraph IV was that mechanism: a structured legal proceeding, initiated by a box on a form, that forced both parties into federal court while the generic was still in development.
The Orange Book: The Central Registry
The Approved Drug Products with Therapeutic Equivalence Evaluations — the Orange Book — is the FDA’s public list of approved drugs and the patents associated with each one. The Orange Book matters because it is the gatekeeper for the entire Hatch-Waxman certification system. A generic manufacturer must address every patent listed in the Orange Book for its reference listed drug (RLD). If a brand manufacturer lists a patent, the generic cannot simply ignore it.
The FDA accepts patent listings from brand manufacturers largely on their word. Companies submit patent information along with their New Drug Applications (NDAs), and the FDA has historically reviewed those listings for procedural completeness, not substantive accuracy. That hands-off approach created an obvious incentive problem, one that the FTC finally moved against aggressively in 2023 and 2024.
There are four types of certification a generic manufacturer can make regarding an Orange Book patent:
A Paragraph I certification states that the patent information has not been filed with the FDA. A Paragraph II certification states that the patent has already expired. A Paragraph III certification acknowledges the patent is valid and commits the generic manufacturer to waiting until it expires before launching. A Paragraph IV certification — the one that matters for this article — declares that the listed patent is invalid, unenforceable, or will not be infringed by the generic product.
Only Paragraph IV triggers litigation. Only Paragraph IV triggers 180-day exclusivity. Only Paragraph IV changes competitive timelines. The other three certifications are essentially administrative — Paragraph IV is a strategic declaration.
How Congress Designed the Patent Term Restoration Bargain
The 1984 Act’s patent term restoration provision gave brand manufacturers up to five years of additional patent protection to compensate for time spent in FDA review, subject to a cap of 14 years of remaining post-approval exclusivity. In exchange, generic manufacturers got the ANDA pathway and the safe harbor. The political calculus was sound: both sides gave something and got something.
Patent term extensions under the Act have since become routine. A brand manufacturer whose compound patent expires during the FDA review process can apply for a patent term extension through the USPTO, and the Hatch-Waxman system accommodates those extensions automatically. When a brand lists an extended patent in the Orange Book, the generic must address it through the certification system.
The exclusivity periods are legally distinct from patent protection and operate independently. A five-year New Chemical Entity (NCE) exclusivity period, for example, blocks FDA from accepting any ANDA at all — not just approving one — for five years from the date of the brand’s NDA approval. A three-year new clinical study exclusivity applies only to the specific use that was supported by the new studies. These exclusivities interact with patent protection in complex ways, and understanding those interactions is one of the core competencies distinguishing sophisticated Paragraph IV litigants from less experienced ones.
Part Two: The Mechanics — How a Paragraph IV Filing Works
Filing the ANDA and the Certification
A generic manufacturer that wants to challenge a brand’s Orange Book patents must first file a substantially complete ANDA. The ANDA contains bioequivalence data, manufacturing information, labeling, and the patent certifications. For Paragraph IV challenges, the ANDA must contain a detailed statement — required under 21 U.S.C. § 505(j)(2)(B) — explaining why the listed patents are invalid, unenforceable, or not infringed. This is known as the Paragraph IV notice letter.
The notice letter is one of the most strategically important documents in pharmaceutical law. It must be served on both the NDA holder (the brand manufacturer) and each patent owner of record. The letter must describe the factual and legal basis for the generic manufacturer’s belief that each patent is invalid, unenforceable, or not infringed. Courts have required that these letters provide real specificity — a letter that simply asserts invalidity without a theory is legally insufficient.
For the generic manufacturer, the notice letter is the first public disclosure of its patent challenge strategy. For the brand manufacturer, it is the opening bell of a multi-year legal conflict. Every subsequent decision — whether to sue, what claims to assert, how aggressively to pursue a settlement — flows from that initial notice.
The brand manufacturer has exactly 45 days from receiving a complete notice letter to file a patent infringement suit in federal court. That 45-day window is not negotiable. If the brand manufacturer files within that window, the 30-month stay triggers automatically. If it does not, the FDA may approve the ANDA immediately upon completing its review.
The 30-Month Stay
The 30-month stay is the Hatch-Waxman system’s most powerful defensive tool for brand manufacturers. When a brand sues within the 45-day window, the FDA cannot grant final approval to the ANDA for 30 months, unless a court decides the patent issue before then [4]. The stay expires when: (a) 30 months have passed; (b) a court finds the challenged patent invalid, unenforceable, or not infringed; or (c) a court enters a consent judgment or other final determination in the generic’s favor.
The 30-month stay effectively transforms every Paragraph IV filing into a compressed litigation proceeding with a hard deadline. Brand manufacturers know that if they can sustain the litigation for 30 months without losing, the stay will expire and the FDA can approve the generic — but during those 30 months, the brand has an automatic market protection mechanism regardless of how strong its underlying patent actually is.
This asymmetry bothered critics of the system for years. Under the original 1984 Act, brands could stack multiple patents and trigger multiple 30-month stays for each one, potentially accumulating years of stay protection per drug product. Congress fixed this with the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA), which limited brand manufacturers to one 30-month stay per ANDA, regardless of how many patents they listed.
Tentative Approval and the Approval Queue
If the FDA completes its review of an ANDA and the application is ready for approval but blocked by a 30-month stay, a patent, or another exclusivity period, the FDA grants tentative approval. A tentative approval is not a license to market — it is the FDA’s confirmation that the application meets all scientific and regulatory requirements and will receive final approval once the blockage clears.
Tentative approvals matter strategically for several reasons. They establish that the generic manufacturer has met the FDA’s standards. They also have implications for the MMA’s forfeiture provisions, discussed below. A generic that fails to obtain tentative approval within 30 months of filing its ANDA may forfeit its 180-day exclusivity.
The FDA publishes a public list of all Paragraph IV certifications that have been filed. This list, accessible through the FDA’s website and tracked extensively by services like DrugPatentWatch, allows industry participants to monitor competitive activity in real time. When a new Paragraph IV certification appears for a major drug, it signals competitive intent months or years before any litigation becomes public.
Part Three: The Prize — 180-Day Exclusivity
What the Exclusivity Is and Where It Comes From
The 180-day exclusivity is the carrot that makes the entire Paragraph IV system function. Congress understood that challenging a well-resourced brand manufacturer’s patents is expensive, risky, and time-consuming. Without a meaningful reward for the first generic challenger to succeed, rational economic actors would wait for other companies to bear those costs, then enter the market afterward as free riders. The 180-day exclusivity is Congress’s solution to that free-rider problem.
The first generic manufacturer to submit a substantially complete ANDA containing a Paragraph IV certification for a given drug and patent is designated the “first filer.” If the first filer wins its patent challenge — either through a favorable court decision or because the brand does not sue within 45 days — it earns the right to 180 days of market exclusivity during which the FDA cannot grant final approval to any other ANDA for the same drug product [5].
The exclusivity period begins when the first filer begins commercial marketing of its product or when a court enters a decision that the challenged patent is invalid or not infringed — whichever comes first. The FDA cannot approve any other generic’s ANDA for 180 days from that date. During those 180 days, the first filer competes only against the brand manufacturer, not against other generics.
The Financial Mathematics of 180-Day Exclusivity
The financial value of 180-day exclusivity depends entirely on the revenue profile of the reference drug. For a blockbuster with $1 billion in annual U.S. sales, 180 days of duopoly competition is worth hundreds of millions of dollars to the first-filer generic. When a generic enters the market, it typically prices its product at 15-30% below the brand’s price initially, then drops sharply as more generics enter. During the 180-day exclusive window, the generic price stays relatively high because there is no inter-generic competition.
Historical data confirms this value. In 2023 alone, first-filer generics earned an estimated $4.7 billion collectively from their 180-day exclusivity periods [6]. That figure represents the net financial benefit of being first — a premium that disappears the moment the exclusivity expires and additional generics flood the market.
The calculation is straightforward: if a drug earns $2 billion annually in U.S. branded sales, and a generic enters at 85% of the brand price, the duopoly market for 180 days (approximately half a year) might generate $500-700 million in revenue for the first-filer generic. Compare that to entry as the fourth or fifth generic, where inter-generic competition drives prices to 20-30% of the brand price, and the 180-day exclusivity is worth fighting for with hundreds of millions of dollars in legal fees.
First-Filer Status and the Same-Day Problem
The original Hatch-Waxman Act gave 180-day exclusivity to the single first filer — the one company that filed first, even if only by hours. This produced the spectacle of generic manufacturers literally camping outside the FDA’s offices in Rockville, Maryland, to ensure they submitted their applications before competitors. The FDA solved this problem in 2003 by announcing that all substantially complete ANDAs containing Paragraph IV certifications filed on the same day would share first-filer status and split the 180-day exclusivity period [7].
Shared exclusivity dilutes the prize but does not eliminate it. Three first-filers splitting 180 days of duopoly competition with the brand still earn significantly more than they would as later-entry generics. The same-day ruling also changed competitive strategy: companies now focus less on absolute timing and more on filing quality, since a deficient ANDA submitted on the same day will be found substantially incomplete and excluded from first-filer consideration.
The MMA Forfeiture Provisions
Congress added forfeiture provisions to the Hatch-Waxman system in 2003 precisely because first-filer exclusivity had become a weapon as well as a reward. A first-filer that refused to launch its generic — either because it settled with the brand for a delayed entry date or because it simply sat on its approval — could block all other generics from entering the market indefinitely. The 180-day clock does not start until the first filer actually begins commercial marketing, so a first-filer that never markets keeps the queue frozen.
Under the MMA’s forfeiture provisions, a first filer forfeits its 180-day exclusivity if it:
- Fails to market its product within 75 days of final approval (or tentative approval, if that was the FDA’s action)
- Has its ANDA withdrawn or abandoned
- Amends or withdraws its Paragraph IV certification
- Enters into an agreement with the NDA holder or another generic that is found to violate antitrust law
- Allows all challenged patents to expire before launching
The forfeiture provisions transformed the strategic calculus around settlement agreements. A first-filer that settles with the brand and accepts a delayed launch date must ensure that the delay does not trigger forfeiture. The MMA created a minimum standard of market activity that first-filers must meet or lose their exclusivity entirely.
Part Four: The Litigation — What Actually Happens in Court
Where These Cases Are Filed
Hatch-Waxman patent litigation is filed in federal district court, typically in the District of New Jersey, the District of Delaware, or the Southern District of New York — jurisdictions with deep dockets of pharmaceutical patent cases and experienced judges. The District of New Jersey and the District of Delaware together handle the substantial majority of ANDA-related patent litigation. Both districts have developed specialized procedures for managing these cases, including case management orders tailored to the 30-month stay deadline.
The litigation is formally a patent infringement case brought by the brand manufacturer against the generic. Under 35 U.S.C. § 271(e)(2)(A), filing an ANDA with a Paragraph IV certification constitutes an act of patent infringement — the “artificial act of infringement” that gives brand manufacturers standing to sue before the generic ever enters the market. This legal fiction is what the entire system is built on: Congress needed a mechanism to litigate patent validity before actual infringement occurred, and it accomplished this by treating the ANDA filing itself as the infringing act.
What Generic Manufacturers Actually Argue
Paragraph IV challenges usually advance two independent arguments: invalidity and non-infringement. These are legally distinct claims and require different evidence.
An invalidity argument attacks the patent itself. The most common invalidity theories are anticipation (the invention was already known or patented before the filing date), obviousness (the invention would have been obvious to a person of ordinary skill in the relevant field at the time), and lack of written description or enablement (the patent specification does not adequately describe or enable the claimed invention). Anticipation and obviousness are the dominant theories in pharmaceutical patent challenges.
A non-infringement argument accepts the patent’s validity but argues that the generic product does not practice the claimed invention. For formulation patents, non-infringement arguments focus on the specific composition of the generic’s formulation — arguing that the specific ingredients, ratios, or manufacturing processes fall outside the patent’s claims. For method-of-use patents, Section viii carve-outs (discussed below) are the dominant non-infringement strategy.
Generic manufacturers pursue both theories simultaneously because they are independently sufficient. A win on either invalidity or non-infringement clears the path to market. This creates an asymmetric burden on the brand manufacturer, which must defend every valid claim against every credible challenge.
Section viii Carve-Outs: Skinny Labels
A Section viii statement — commonly called a skinny label or carve-out — allows a generic manufacturer to receive ANDA approval for a subset of the brand’s approved indications, deliberately excluding those covered by valid method-of-use patents. If a brand drug is approved for indications A, B, and C, and only Indication C is protected by an unexpired Orange Book method-of-use patent, the generic can file an ANDA with a Section viii statement carving out Indication C from its label and seek approval for Indications A and B only.
Skinny labels are a significant competitive tool. They allow generics to enter the market earlier, capturing revenue from unpatented indications while waiting for method-of-use patents to expire. In 2023, approximately 37% of Paragraph IV filings employed some form of Section viii strategy [8]. The legal boundary of what constitutes “carving out” a patented use — versus infringing it through induced infringement of physicians who prescribe the generic for the patented indication — has been one of the most actively litigated questions in Hatch-Waxman law for the past decade.
The GSK v. Teva litigation involving labetalol (the brand drug Coreg) is the defining example of how skinny label strategies can go wrong. Teva carved out GSK’s patented indication for heart failure but was found liable for induced infringement because its label, despite the carve-out, still described the drug’s effectiveness in terms that a jury found encouraged physicians to prescribe it for the patented use. The Federal Circuit’s opinion in that case — issued in 2021 — chilled skinny label strategies significantly and remains one of the most consequential ANDA patent decisions of the past decade.
Win Rates and What They Mean
The statistics on Paragraph IV litigation outcomes require careful reading. A frequently cited figure holds that generic challengers prevail in approximately 76% of Paragraph IV cases overall [9]. That number is real but misleading if taken at face value. It includes settlements — which account for the overwhelming majority of resolutions — as well as the subset of cases where brands simply declined to sue (and the generic won by default). When you filter for cases that were fully litigated to a trial decision, the generic win rate falls to approximately 48% [9].
More recent data from 2024 further complicates the picture. Among cases that reached a court decision, innovator companies prevailed in about 20% of decided cases while generic companies prevailed in roughly 2%, with the remainder producing mixed or split outcomes across multiple patents [9]. The takeaway is not that generics have become less successful — most successful challenges never reach trial because the parties settle. The takeaway is that the cases that actually go to trial are the hardest ones, where the brand has its best patents and the generic has the most aggressive challenges.
For companies using tools like DrugPatentWatch to track live ANDA litigation activity, understanding this filtering effect is essential. A high volume of Paragraph IV filings against a particular drug does not mean those patents are weak — it may simply mean the drug is commercially important enough that multiple generics are willing to pay litigation costs for a chance at first-filer status.
The Role of IPRs in ANDA Litigation
The America Invents Act of 2012 introduced inter partes review (IPR) proceedings at the USPTO’s Patent Trial and Appeal Board (PTAB), and they have become a major tool in the pharmaceutical patent challenge arsenal. A generic manufacturer can file an IPR petition challenging the validity of an Orange Book patent on grounds of anticipation or obviousness, often in parallel with its Hatch-Waxman district court litigation.
IPRs are faster than district court proceedings — the PTAB must issue a final written decision within one year of institution — and they apply a lower claim construction standard than federal courts. The PTAB’s institution rates for pharmaceutical patents have been reasonably high, and the overall invalidity rate for instituted pharmaceutical IPRs is substantial. Generic manufacturers routinely use IPR petitions as a second front in their validity challenges: the PTAB proceeding may invalidate the patent before the district court litigation concludes, mooting the Hatch-Waxman case entirely.
The interaction between IPRs and Hatch-Waxman litigation is complex. A generic manufacturer that wins an IPR invalidating an Orange Book patent has not technically won its Paragraph IV certification challenge under the Hatch-Waxman framework — the 180-day exclusivity trigger requires a court decision in the district court litigation context, not a PTAB decision. The FDA’s interpretation of which decisions trigger the exclusivity clock remains one of the more technically intricate aspects of the entire system.
Part Five: The Pay-for-Delay Problem — Reverse Payments and FTC v. Actavis
How Reverse Payments Developed
The Hatch-Waxman system created a powerful economic logic for reverse payment settlements. A brand manufacturer facing a serious Paragraph IV challenge had a choice: litigate and risk having its patent invalidated (destroying years of remaining exclusivity), or pay the generic challenger to withdraw its challenge and delay market entry. The economics were compelling for both sides. The brand retained its monopoly profits for the full patent term. The generic received cash — essentially a share of the monopoly rents — without any manufacturing investment or market risk.
These arrangements were called “reverse payments” because money flowed in the opposite direction from a standard settlement: the defendant (generic) was being paid by the plaintiff (brand) to abandon its legal challenge. They were also called “pay-for-delay” settlements because the practical effect was to delay generic market entry beyond what the litigation outcome might have produced.
The FTC estimated that pay-for-delay agreements cost American consumers more than $3 billion per year during their peak prevalence in the 2010s [10]. The harm was structural: these settlements neutralized the first-filer generic — the one most likely to successfully challenge the patent — which under the MMA’s queue structure meant that no other generic could enter until after the first-filer’s delayed launch date.
FTC v. Actavis (2013) and Its Aftermath
The Supreme Court’s 2013 decision in FTC v. Actavis, Inc. resolved a circuit split on the legal status of reverse payment settlements. The case arose from Solvay Pharmaceuticals’ settlement with Actavis (then Watson Pharmaceuticals) and other generic manufacturers over the testosterone replacement drug AndroGel. Under the settlement, Solvay paid Actavis and its co-defendants to keep their generics off the market until 2015, deferring generic entry by approximately nine years [11].
The FTC argued that such settlements were presumptively anticompetitive. The Eleventh Circuit disagreed, holding that reverse payments were immune from antitrust scrutiny as long as their effects fell within the scope of the challenged patent. The Supreme Court reversed 5-3, holding that reverse payments were not per se immune from antitrust review and must be analyzed under the “rule of reason” standard [12]. The Court’s key insight was that a large, unjustified reverse payment is itself evidence that the brand manufacturer believed its patent was weak — a strong patent holder would litigate, not pay competitors to disappear.
The Actavis decision immediately affected settlement behavior. The number of potential pay-for-delay settlements filed with the FTC dropped from 40 in FY2012 (the last complete pre-Actavis year) to 29 in FY2013, 21 in FY2014, and 14 in FY2015 [13]. The overall number of pharmaceutical patent settlements continued to rise — from 140 in 2012 to 170 in 2015 — but the fraction involving explicit reverse payments declined sharply as brand manufacturers shifted to other settlement structures [14].
The Evolution of Settlement Structures After Actavis
Brand manufacturers did not stop settling Paragraph IV cases after Actavis — they restructured how they settled. Cash payments became rarer. Instead, settlements took non-cash forms: authorized generic agreements (the brand licenses its own generic version to the challenger for a defined period), business development agreements, supply agreements, co-promotion deals, and negotiated entry dates.
Courts and the FTC have recognized that non-cash “payments” can be just as anticompetitive as cash. The Third Circuit’s 2015 decision in King Drug Co. of Florence v. SmithKline Beecham held that authorized generic agreements can constitute reverse payments for Actavis purposes, extending the antitrust analysis beyond cash. The First Circuit’s 2016 decision in In re Loestrin Antitrust Litigation took the same approach [15]. The FTC has consistently argued that limiting Actavis to cash would give manufacturers “carte blanche” to structure anticompetitive deals through non-cash consideration.
The current enforcement environment reflects heightened scrutiny of settlement structures. Companies entering Paragraph IV settlements must report them to both the FTC and the Department of Justice under the MMA’s reporting requirements. The FTC’s Bureau of Competition reviews those reports and has brought enforcement actions based on non-cash settlement structures that it views as reverse payments in substance if not form.
Part Six: Orange Book Manipulation — Patent Thicketing and the FTC’s Counteroffensive
What Patent Thicketing Is
Patent thicketing is the practice of listing large numbers of patents in the Orange Book covering progressively peripheral aspects of a drug product — manufacturing methods, formulation details, delivery devices, packaging components — in order to multiply the number of potential Paragraph IV suits a generic challenger must navigate. Where a drug might once have had a single compound patent listed, brand manufacturers have increasingly listed dozens.
The financial logic is clear: each additional listed patent requires a generic’s ANDA to contain an additional certification. Each certification is a potential trigger for an additional 30-month stay (before the MMA) or, even now, additional rounds of litigation. More patents means more complexity, more cost, and more time for brand companies to earn monopoly returns.
The scale of thicketing has grown substantially over the past two decades. Data from DrugPatentWatch and other patent analytics platforms show that the average number of patents listed per branded drug in the Orange Book more than doubled between 2005 and 2024, with some blockbuster drugs carrying patent portfolios of 40 or more listed patents covering everything from the active molecule to the container closure system [16].
The FTC’s 2023-2024 Offensive
The FTC under Chair Lina Khan moved aggressively against what it characterized as improper Orange Book listings. In November 2023, the FTC challenged more than 100 patents as improperly or inaccurately listed, targeting asthma inhalers, epinephrine autoinjectors, and other drug-device combination products [17]. Warning letters went to ten companies including AbbVie, AstraZeneca, Boehringer Ingelheim, GSK, Mylan Specialty, and Teva.
The FTC’s legal theory was that patents covering delivery devices — inhalers, autoinjectors — should not be listed in the Orange Book at all if they do not claim the approved drug substance or product directly. A patent on the actuation mechanism of an inhaler, the FTC argued, is a device patent that belongs in device patent litigation, not in the Orange Book’s pharmaceutical patent listing system where it can trigger Hatch-Waxman stays.
In April 2024, the FTC expanded its challenge to more than 300 additional Orange Book patent listings across 20 brand-name drugs, adding injectable diabetes and obesity treatments and COPD inhalers [18]. The second wave targeted patents on injector pens and other delivery mechanisms — directly relevant to the GLP-1 market that had exploded with drugs like Ozempic and Wegovy.
The response from brand manufacturers was mixed. After the first wave of challenges, six of seventeen targeted products changed their Orange Book listings while eleven did not [19]. After the second wave, none of the twenty targeted products changed their listings, suggesting that manufacturers had concluded the FDA’s administrative dispute process carried insufficient legal force to compel delisting.
The FTC had more success through litigation. The Federal Circuit upheld a district court order requiring Teva to delist several asthma inhaler patents from the Orange Book, affirming the FTC’s position that device patents not claiming the approved drug substance are improperly listed [20]. That judicial win gave the FTC renewed authority. In May 2025, the agency issued a third round of warning letters renewing challenges to more than 200 listings that had remained in the Orange Book despite the earlier FTC challenges and the Federal Circuit ruling [21].
The GSK Advair Example
The GSK Advair case illustrates both the strategy and its commercial durability. Advair’s primary compound patent expired in 2010. Yet as of 2023, Advair was still earning GSK approximately 863 million pounds ($1.1 billion) in the first nine months of the year alone [22]. The product’s commercial persistence depended in part on device patents covering the Diskus inhaler delivery mechanism — patents the FTC argued were improperly listed. After the FTC’s challenge, GSK voluntarily withdrew its Advair patents from the Orange Book, and GSK, AstraZeneca, and Boehringer Ingelheim all subsequently announced caps on inhaler out-of-pocket costs at $35.
The commercial message is unambiguous: a single well-litigated device patent, improperly listed or not, can protect hundreds of millions of dollars in annual revenue for a decade after the compound patent expires. For generic challengers, identifying and successfully challenging such patents is the highest-value legal work in the pharmaceutical sector.
Part Seven: The Competitive Intelligence Layer — What Data Actually Shows
Using DrugPatentWatch to Track Paragraph IV Activity
For the pharmaceutical industry’s investment decision-makers, patent attorneys, and competitive intelligence professionals, tracking real-time Paragraph IV activity is indispensable. The FDA publishes its list of Paragraph IV certifications, but the raw FDA data requires aggregation, contextualization, and historical analysis to be actionable. This is where platforms like DrugPatentWatch — which aggregates FDA filings, Orange Book data, litigation records, and patent expiration timelines — provide practical value.
DrugPatentWatch allows users to identify which drugs are currently under Paragraph IV challenge, which companies have filed ANDAs, what the first-filer status landscape looks like, and how the litigation outcomes have evolved over time. For a brand company, this data indicates where generic challenge risk is highest — which of its drugs face imminent challenges and how many competitors are queuing up. For a generic company, it identifies the competitive landscape for a target drug: whether first-filer status is still available, how many other ANDAs have already been filed, and what the Orange Book patent structure looks like.
For investors, Paragraph IV certification data is a leading indicator of revenue risk. When DrugPatentWatch shows a cluster of new Paragraph IV filings against a brand’s flagship drug, that is actionable intelligence — it means the company faces potential generic entry years before nominal patent expiration, and the market may not have fully priced that risk.
The Competitive Value of First-Filer Intelligence
The 180-day exclusivity is awarded to the first filer, which means the most financially valuable intelligence is knowing, before your competitors do, whether first-filer status is still available for a given drug. The FDA publishes the date of the first substantially complete Paragraph IV ANDA filing for each drug on its website. Once that date is published, any subsequent filer knows it will not be a sole first-filer. But in the window before the FDA publishes that date — after the first ANDA has been filed but before the FDA formally confirms it — competitive intelligence from Paragraph IV notification letters and court filings can provide earlier signals.
Generic companies that discover they are the first to file on a valuable target have a strategic imperative to ensure their ANDA is substantially complete at the time of filing. A filing that the FDA finds deficient — missing bioequivalence data, inadequate patent certifications, incomplete labeling — may not qualify as “substantially complete” and could forfeit first-filer status. The FDA’s substantive completeness review is therefore the first critical hurdle after filing.
Patent Expiration Timelines and the Upcoming Cliff
The 2025-2030 period is the most significant patent cliff in the history of the pharmaceutical industry, with an estimated $200-$230 billion in annual branded drug revenue exposed to generic competition [23]. Drugs like Keytruda (pembrolizumab, Merck, $29 billion in 2024 sales), Eliquis (apixaban, BMS/Pfizer, over $13 billion in 2024 BMS-attributed revenue), and Opdivo (nivolumab, BMS) sit at the center of this cliff [24].
The Eliquis situation is a working illustration of how Paragraph IV litigation interacts with the patent cliff. BMS and Pfizer listed multiple Orange Book patents covering apixaban’s compound, specific dosing regimens, and manufacturing processes. Generic manufacturers including Sigmapharm, Aurobindo, MSN Pharmaceuticals, and others filed Paragraph IV ANDAs, triggering the 30-month litigation stay. Courts upheld some apixaban patents and invalidated others, producing a complex litigation landscape that BMS and Pfizer have actively managed through both litigation and negotiated settlement discussions. BMS reported over $13 billion in 2024 Eliquis revenue; a scenario in which generic entry occurs on schedule in 2026 produces an NPV impact in the range of $25-35 billion in lost future cash flows [25].
Part Eight: Recent Legal Developments and the Regulatory Horizon
The IRA’s Impact on Hatch-Waxman Dynamics
The Inflation Reduction Act of 2022 changed the economic calculus for pharmaceutical patents in ways that are still propagating through the industry. The IRA’s Medicare drug price negotiation provisions — which the CMS began exercising in 2023-2024, targeting ten high-cost Medicare Part D drugs — created a new competitive layer on top of the existing Hatch-Waxman framework.
For brand manufacturers, IRA negotiation reduces the financial ceiling of the monopoly period the Hatch-Waxman framework protects. A brand drug negotiated down to 40-60% of its list price through the IRA’s maximum fair price mechanism is worth substantially less to defend in Paragraph IV litigation. The calculus around litigation investment changes when the protected monopoly revenue is itself being capped by government negotiation.
For generic manufacturers, the IRA’s structure creates an unexpected complication. The Act provides longer negotiation-free periods for small-molecule drugs that faced earlier generic competition — specifically, small molecules get nine years before negotiation is available, while biologics get 13 years, but drugs that face generic competition early effectively start that clock sooner. This creates perverse incentives at the margin: a generic challenge that succeeds very early may, in some scenarios, make the brand drug eligible for IRA price negotiation sooner, lowering the floor price that subsequent generic competition races against.
The IRA’s full interaction with Hatch-Waxman incentive structures is still being analyzed by the industry. Several academic papers and policy analyses have identified potential unintended consequences for generic investment, and the FDA has acknowledged in its 2024 CDER guidance agenda that additional guidance on Hatch-Waxman matters — including the 30-month stay provisions in the supplement context — is forthcoming.
Competitive Generic Therapy Designations
The FDA Reauthorization Act of 2017 created the Competitive Generic Therapy (CGT) designation, which provides expedited ANDA review and a shorter exclusivity reward for generic manufacturers challenging drugs with no currently approved generic. CGT-designated ANDAs get a 180-day exclusivity period of their own, separate from the Paragraph IV first-filer exclusivity. This designation is available for drugs where there is inadequate generic competition — specifically, where there are three or fewer approved ANDAs.
CGT designation is most valuable for specialty drugs that brand manufacturers have deliberately kept generic-free through a combination of aggressive patent portfolios and REMS programs. The REMS exclusivity question — whether a brand manufacturer can use its Risk Evaluation and Mitigation Strategy program to restrict the samples required for bioequivalence testing, thereby blocking ANDA development — was partially resolved by the FDA Safety and Innovation Act of 2012, which created a formal mechanism for generic applicants to access samples over a brand manufacturer’s objection.
The Section 505(b)(2) Pathway and Its Paragraph IV Implications
The 505(b)(2) application pathway — which allows a manufacturer to rely on FDA’s prior finding of safety and efficacy for a listed drug plus its own new clinical studies — has become increasingly important for brand lifecycle management. A brand manufacturer can use a 505(b)(2) application to reformulate its product, change the delivery method, or seek approval for a new indication, then list new patents for the reformulated product in the Orange Book. Those new patents are subject to Paragraph IV challenges just like original NDA patents.
The 505(b)(2) pathway is central to the evergreening critique: the practice of brand manufacturers making incremental modifications to extend effective market exclusivity. An original capsule formulation becomes an extended-release tablet, which gets its own new three-year exclusivity period and new Orange Book patents. A quarterly injection is reformulated into a subcutaneous weekly autoinjector, with device patents that the FTC may challenge but which delay generic entry in the meantime.
The 30-month stay provisions for 505(b)(2) supplements have been one of the most litigated and least-resolved areas of Hatch-Waxman law. The FDA’s position has shifted over time on whether a 505(b)(2) supplement for a modified product triggers a separate 30-month stay period, creating uncertainty for both brands and generic challengers. The FDA’s 2024 CDER guidance agenda specifically flagged a forthcoming guidance on this issue.
FDA Orange Book Reform Under Consideration
The regulatory environment in 2025-2026 reflects increased governmental focus on the Orange Book as a competition policy instrument. Beyond the FTC’s patent listing challenges, Congress has considered multiple reform proposals targeting the patent listing system. The Drug Competition Enhancement Act, introduced in multiple sessions, would require drug manufacturers to justify each Orange Book patent listing under a heightened standard and would create a more accessible administrative process for challenging improper listings.
The FDA has also signaled that it plans to require brand manufacturers to justify each patent listing in the Orange Book more explicitly, moving away from the current honor system toward a document-supported listing requirement. Industry analysts tracking FDA rulemaking through DrugPatentWatch’s regulatory monitoring tools have noted that formal proposed rulemaking in this area is expected in 2026-2027.
Part Nine: Strategic Playbooks — What Brand and Generic Companies Actually Do
The Brand Company’s Defensive Toolkit
A brand manufacturer’s Hatch-Waxman defense involves four parallel activities: litigation, regulatory strategy, lifecycle management, and commercial preparation for the eventual generic entry.
The litigation strategy begins years before any ANDA is filed. Brand companies spend heavily on patent portfolio construction, filing continuation and continuation-in-part applications to extend their patent families, adding new Orange Book listings as new aspects of the product are patented, and timing divisional application filings to maximize the period during which pending applications can produce new listed patents. A well-managed pharmaceutical patent portfolio is constructed with the Paragraph IV litigation timeline in mind from the beginning.
Regulatory strategy includes the 505(b)(2) reformulation approach and the strategic filing of new NDA supplements with new Orange Book patents. Every new clinical study that produces approvable data — even a modest change in dosing flexibility or a new pediatric indication — can anchor a three-year new clinical study exclusivity period and new Orange Book patents. Brand companies routinely file ANDA citizen petitions raising safety and regulatory concerns about generic products, a practice the FDA has become increasingly skeptical of but which can delay ANDA approvals by months.
Lifecycle management means launching an authorized generic — a brand-produced generic version of the same product — before or alongside the first-filer generic. Authorized generic agreements with the first-filer challenger, or unilateral authorized generic launches timed to coincide with first-filer entry, can complicate the financial calculus of 180-day exclusivity. An authorized generic competes directly with the first-filer generic during the 180-day window, reducing the first-filer’s revenue premium significantly.
The Generic Company’s Offensive Toolkit
For a generic manufacturer, the Paragraph IV strategy begins with target identification. Not every drug with an Orange Book patent is worth challenging. The commercial opportunity must justify the litigation cost, and the patent landscape must present credible invalidity or non-infringement arguments. A drug with $500 million in annual sales and a single compound patent expiring in three years may not justify a Paragraph IV challenge. A drug with $3 billion in annual sales and a compound patent that cannot be extended, surrounded by a dense but potentially challengeable secondary patent portfolio, is a high-value target.
The patent landscape analysis — identifying which Orange Book patents are actually blocking entry, which have credible invalidity arguments, and which might be circumvented through non-infringement designs — is the core scientific and legal work of a Paragraph IV filing. Generic manufacturers with strong patent analysis capabilities gain significant advantages in target identification and litigation strategy. Platforms like DrugPatentWatch provide the baseline data — patent expiration dates, prosecution history flags, litigation history — that feed into these analyses.
After filing, generic manufacturers must manage their litigation timeline against the 30-month stay clock. The goal is to resolve the patent challenge — through litigation victory, settlement, or stay expiration — in a way that clears regulatory approval as quickly as possible. The 30-month stay is not necessarily the enemy: if the generic manufacturer can win a favorable claim construction ruling or summary judgment on non-infringement early in the litigation, it can potentially secure approval before the stay expires and launch immediately.
The Multi-Company Race
Most major Paragraph IV challenges involve multiple generic filers — sometimes dozens for blockbuster drugs. When multiple companies file Paragraph IV ANDAs on the same day or within a short window, several of them may share first-filer status. Those that file later — even by a single day — are not first filers and must wait for the 180-day exclusivity to expire before they can receive final ANDA approval.
This creates a bifurcated competitive strategy: the first-filer group manages its litigation to preserve first-filer status and maximize the financial value of the 180-day window, while the non-first-filer group focuses on obtaining tentative approvals and positioning for rapid entry on day 181 of the exclusivity period. The non-first-filer strategy is well understood: be ready to launch immediately when the exclusivity expires, with all manufacturing, distribution, and commercial infrastructure in place.
The scale of activity on blockbuster targets can be extraordinary. Drugs like Humira (adalimumab), Eliquis (apixaban), and Revlimid (lenalidomide) faced dozens of generic challengers, producing multi-year litigation involving coordinated but adversarial proceedings across multiple ANDA filers. The litigation management complexity for brands defending against 20 or 30 simultaneous ANDAs is substantial, and the resource allocation required to sustain 30-month stays across all of them is a significant business cost.
Part Ten: Case Studies — The Playbook in Action
Lipitor (Atorvastatin): The Textbook Case
The patent litigation over Pfizer’s Lipitor — atorvastatin calcium — is the canonical Hatch-Waxman case study and remains the largest single generic drug launch in U.S. history. Lipitor was generating over $10 billion annually in U.S. sales when the Paragraph IV challenges began in earnest. Ranbaxy Laboratories filed the first Paragraph IV ANDA, earning first-filer exclusivity status. Pfizer sued within 45 days and triggered the 30-month stay.
The litigation ran for years, involving multiple patent families and multiple rounds of litigation. Ranbaxy’s challenges to Pfizer’s atorvastatin patents were partially successful: some secondary patents were invalidated or not infringed, while others were sustained. The parties ultimately reached a settlement under which Ranbaxy received a negotiated entry date of November 2011, with 180-day exclusivity. In the first 180 days after generic entry, the market experienced a rapid price decline that saved the U.S. healthcare system billions. The Lipitor generic launch demonstrated that the system, when it works correctly, produces exactly the outcome Congress intended.
Cephalon’s Provigil and the $1.2 Billion Settlement
The Provigil (modafinil) pay-for-delay case illustrates the system’s failure mode. Cephalon paid four generic companies — Teva, Mylan, Barr, and Ranbaxy — a combined total approaching $300 million to keep their generic modafinil products off the market until 2012, four years before the relevant patent would have expired under litigation. The FTC challenged the arrangements.
After years of litigation and investigation, the FTC reached a $1.2 billion settlement with Teva (which had acquired Cephalon) in 2015 — one of the largest antitrust settlements in the agency’s history [26]. Teva disgorged the ill-gotten gains for distribution to consumers and businesses harmed by the delayed generic competition. The Cephalon/Provigil case became the central example in FTC v. Actavis briefing and has since been cited as the paradigm case of pay-for-delay harm.
Eliquis (Apixaban): The Current-Cycle Battle
BMS and Pfizer’s Eliquis is the highest-revenue small-molecule drug currently in active Paragraph IV litigation. The compound patent’s nominal expiration is in the 2026-2028 window, and multiple generic manufacturers filed Paragraph IV ANDAs years ago. Courts upheld some apixaban patents and invalidated others. The litigation produced complex outcomes, and the negotiated timelines for generic entry have been an active subject of both litigation and settlement discussions.
What makes Eliquis particularly instructive is the patent portfolio structure BMS and Pfizer built. Beyond the core compound patent, BMS listed patents covering specific dosing regimens — the 2.5 mg and 5 mg twice-daily dosing regimens used in the FDA-approved indications — as well as manufacturing process patents. Generic challengers have attacked these secondary patents as obvious variations. Courts have found some secondary apixaban patents invalid while upholding others, producing a situation where generic entry is imminent but the precise date depends on which patents survive appeal.
For investors tracking BMS through tools like DrugPatentWatch, the real-time status of the apixaban Paragraph IV litigation provides the most accurate available estimate of when Eliquis revenue will face full generic competition. The NPV difference between a 2026 generic entry and a 2028 generic entry on a $13 billion annual revenue drug is several billion dollars in present value terms [25].
Restasis (Cyclosporine 0.05%): The Citizens Petition Gambit
Allergan’s defense of Restasis, its cyclosporine ophthalmic emulsion for dry eye disease, combined citizen petitions, patent litigation, and an unusual attempted transfer of patents to the Saint Regis Mohawk Tribe to exploit tribal sovereign immunity from IPR proceedings. The tribal immunity gambit was ultimately rejected by the Federal Circuit in 2019, which held that the PTAB’s authority to conduct IPR proceedings cannot be blocked by tribal sovereign immunity claims. The citizen petition strategy also faced criticism, as the FDA ultimately rejected Allergan’s petitions as lacking sufficient grounds.
The Restasis litigation history is a case study in the full spectrum of defensive tactics available to a brand manufacturer and the limits of each. Legal creativity is not unlimited: courts eventually scrutinize novel defensive mechanisms, and what works procedurally in one context may fail in another.
Part Eleven: The International Dimension and What U.S. Practitioners Miss
Why Hatch-Waxman Is a U.S.-Specific Framework
The Hatch-Waxman framework has no direct international equivalent. The European Union’s approach to pharmaceutical patent protection — supplementary protection certificates (SPCs) extending patent terms, data exclusivity periods under Directive 2001/83/EC, and private patent enforcement after regulatory approval — operates entirely separately from the regulatory approval system. There is no mechanism in the EU analogous to the Orange Book that systematically links patent status to regulatory approval. EU generic manufacturers receive marketing authorization and then manage patent disputes through national courts as separate private litigation.
This divergence creates complications for pharmaceutical companies operating globally. A U.S. brand manufacturer’s Hatch-Waxman litigation strategy is irrelevant to its European IP management, and vice versa. Generic manufacturers with both U.S. ANDA and European generics operations must maintain separate patent monitoring and litigation infrastructures.
The recent Teva apixaban case in Spain illustrates how aggressive European patent defense can operate. Teva launched an apixaban generic in Spain after initial court rulings invalidated BMS’s supplementary protection certificate. In 2024, a Barcelona appellate court reversed that ruling, reinstating BMS’s SPC and forcing Teva’s product off the market — costing the Spanish public health system an estimated €138 million per year in foregone savings [27]. No equivalent reversal would be possible in the U.S. once a generic received final ANDA approval.
Canada, India, and the Compulsory License Dimension
Canada operates a system superficially similar to Hatch-Waxman under the Patented Medicines (Notice of Compliance) Regulations, but with significant structural differences, including a different patent listing mechanism and different procedural rules for automatic stays of generic approval. Indian pharmaceutical law operates under a distinct framework that has been the subject of significant controversy, particularly the Section 3(d) provision that limits patentability of new forms of known substances — a provision that has enabled India to deny patent protection for several drugs that hold valid U.S. patents.
For U.S. pharmaceutical companies, the international dimension primarily matters through the lens of manufacturing. Many of the largest Paragraph IV ANDA filers are Indian manufacturers — Teva (through its Indian operations), Sun Pharma, Dr. Reddy’s Laboratories, Cipla, Aurobindo — that manufacture the active pharmaceutical ingredients and finished dosage forms in Indian facilities subject to FDA inspection. Manufacturing quality events at these facilities have, on multiple occasions, delayed generic market entry and disrupted the 180-day exclusivity timeline.
Part Twelve: The Future of Hatch-Waxman
Patent Landscape Monitoring in the AI Era
The tools available for Paragraph IV intelligence have changed dramatically since 1984. Early Hatch-Waxman litigants worked from paper Orange Book listings and hand-searched patent databases. Today, platforms like DrugPatentWatch aggregate patent data, ANDA filing histories, litigation records, settlement databases, and regulatory approval timelines into integrated analytical environments. Machine learning tools scan patent claims for prior art and obviousness vulnerabilities, flagging candidate patents for challenge before the ANDA is filed.
The predictive dimension of these tools is their most valuable feature. A generic manufacturer that can predict, based on historical litigation patterns, which Orange Book patents are most likely to be held invalid at trial gains a material advantage in target selection. A brand manufacturer that can identify, based on ANDA filing patterns and paragraph IV notification letter timing, which of its products are most likely to face challenges in the next 12-24 months can allocate its patent litigation budget more effectively.
Congressional Reform Proposals
Multiple reform proposals have been introduced in Congress targeting different aspects of the Hatch-Waxman system. The Affordable Prescriptions for Patients Act, which has passed the Senate Judiciary Committee in various forms, would limit the number of patents a brand manufacturer can assert in Hatch-Waxman litigation — specifically targeting late-filed patents that appear designed to serially extend litigation rather than protect genuine innovation. The CREATES Act, enacted in 2019, addressed brand manufacturers’ use of REMS programs to withhold samples from generic developers, establishing a private right of action for generics denied sample access.
The Biosimilars Innovation Act proposals have sought to harmonize the 180-day exclusivity concept with the biosimilar interchangeability framework, recognizing that the line between complex small molecules and simple biologics has blurred. As peptide and oligonucleotide drugs occupy the regulatory space between traditional small molecules (governed by Hatch-Waxman) and biologics (governed by the Biologics Price Competition and Innovation Act), the regulatory framework has not fully kept pace with the chemistry.
The BPCIA and What Hatch-Waxman Can Learn From Its Biosimilar Analog
The Biologics Price Competition and Innovation Act of 2010 created a separate Paragraph IV analog for biosimilars — the “patent dance,” a structured exchange of patent information between reference product sponsors and biosimilar applicants. The BPCIA’s patent dance is more complex than Hatch-Waxman’s notification-and-sue structure and has generated its own extensive litigation over whether biosimilar applicants can bypass the exchange process.
The BPCIA does not include a 180-day exclusivity reward for the first biosimilar challenger. This is a significant structural difference from Hatch-Waxman, and it may partly explain why biosimilar market penetration for complex biologics has been slower and more contentious than small-molecule generic penetration was under Hatch-Waxman. The question of whether a 180-day-style exclusivity should be added to the BPCIA framework is an active policy debate.
Conclusion: A System Worth Understanding
The Hatch-Waxman Act, in its 42nd year of operation, is neither broken nor optimally functioning. It has delivered extraordinary value — $3.4 trillion in savings over the past decade alone — while creating incentive structures that sophisticated actors routinely exploit for anticompetitive advantage. Patent thicketing, pay-for-delay settlements, device patent abuse, and REMS weaponization are all real phenomena documented in FTC enforcement records and federal court opinions.
The system works when brand manufacturers hold genuinely innovative patents, generic manufacturers mount credible challenges to weak secondary patents, and the litigation produces a court ruling that clears the path to generic competition years before nominal patent expiration. It breaks when brand manufacturers use the listing mechanism to artificially extend exclusivity and when first-filer generics use their queue-blocking position to extract value from delay rather than competition.
The regulatory environment in 2025-2026 is actively addressing these failure modes: the FTC’s Orange Book patent challenges, the Federal Circuit’s Teva inhaler ruling, the IRA’s price negotiation mechanism, and forthcoming FDA guidance on 30-month stay provisions in the supplement context all push toward a system that better approximates the 1984 legislative design. Whether those adjustments are sufficient to address structural incentive problems that four decades of lobbying and litigation have embedded in the framework is a question that Congress will eventually need to confront directly.
For everyone operating in this system — brand manufacturer, generic challenger, investor, or policy analyst — the Paragraph IV certification is the document that matters most. It is where strategy meets law, where market access is won or lost, and where 42 years of legislative compromise are tested against the reality of a pharmaceutical market that neither Senator Hatch nor Representative Waxman could have anticipated.
Key Takeaways
- A Paragraph IV certification is a legal declaration — an artificial act of patent infringement — that triggers a 45-day window for brand manufacturers to sue and a potential 30-month stay of generic approval. Its strategic significance cannot be overstated.
- The 180-day exclusivity period is worth hundreds of millions of dollars for blockbuster drugs and is the primary financial incentive that drives generic patent challenges. First-filer status, once established, is closely guarded and subject to forfeiture.
- The FTC v. Actavis ruling in 2013 established that reverse payment settlements are subject to rule-of-reason antitrust scrutiny. Pay-for-delay settlements have declined since the ruling, but non-cash equivalent arrangements remain under active FTC scrutiny.
- Patent thicketing — the listing of device, formulation, and process patents beyond the core compound patent — has been the dominant brand lifecycle management tool for two decades. The FTC challenged more than 400 Orange Book patent listings in 2023-2024, and the Federal Circuit validated the agency’s core legal theory in Teva v. Amneal.
- The 2025-2030 patent cliff — representing $200-$230 billion in branded drug annual revenue — will produce the largest wave of Paragraph IV activity in the Hatch-Waxman system’s history. Eliquis, Keytruda, and Opdivo are the highest-value targets. DrugPatentWatch and similar patent analytics platforms are essential tools for tracking the evolving litigation landscape in real time.
- The Hatch-Waxman system was not designed to be gamed. It was designed around a simple bargain: brand companies get genuine innovation protection; generic companies get a fair path to market. When either side departs from that underlying bargain, the regulatory and judicial systems have shown they will eventually respond.
Frequently Asked Questions
Q1: Can a generic manufacturer file a Paragraph IV certification even if it is not yet ready to manufacture the drug?
Filing a substantially complete ANDA with a Paragraph IV certification does require meaningful scientific development work — particularly the bioequivalence studies demonstrating that the generic product has the same active ingredient, dosage form, route of administration, and strength as the reference listed drug, and that its bioavailability is not significantly different. The FDA will review the ANDA for completeness, and an application found not substantially complete will not establish first-filer status. In practice, most generic manufacturers have invested substantially in formulation development and bioequivalence testing before filing. The ANDA does not require full commercial-scale manufacturing validation at the time of filing, but the application must contain a complete description of the manufacturing process and facility information. Tentative approval — the FDA’s confirmation that the application is ready for final approval pending patent or exclusivity clearance — requires that manufacturing readiness meet FDA standards.
Q2: What happens if two generic manufacturers file their Paragraph IV ANDAs on the exact same day?
Under the FDA’s 2003 policy change, all substantially complete ANDAs containing Paragraph IV certifications submitted on the same day share first-filer status and split the 180-day exclusivity period. This means if five companies file on the same day, all five are designated first filers and none can receive final approval until the 180-day exclusivity clock — which runs from the date the first commercial marketing begins or a relevant court decision enters — expires. The FDA determines substantial completeness separately for each ANDA, so a deficient filing on a same-day basis will not be included in the shared first-filer group. Companies that file on the same day as an established first-filer group are also added to the group; the first-filer date remains the original date, not the later date of the new filer’s submission.
Q3: How does the 30-month stay interact with situations where the brand does not sue within 45 days?
If the brand manufacturer receives a proper Paragraph IV notification letter and does not file a patent infringement suit within 45 days, no 30-month stay is triggered. The FDA can approve the ANDA upon completing its review, with no waiting period based on the challenged patent. The brand manufacturer’s failure to sue within the window is sometimes strategic — it may reflect a judgment that the patent is not worth defending, or that the generic’s ANDA is deficient and will not receive approval regardless. In some cases, brands have declined to sue to avoid triggering the 180-day exclusivity for the first-filer generic, which would block other generics from the market and preserve the brand’s ability to compete against a fragmented generic market from the start.
Q4: Is the 180-day exclusivity period automatically triggered, or does the first-filer generic have to do something specific to start the clock?
The 180-day exclusivity clock starts on the date the first-filer begins commercial marketing of its approved generic product — or the date of a court decision (in the first-filer’s litigation) that the challenged patent is invalid or not infringed, whichever is earlier. The first-filer does not automatically start the clock simply by receiving final approval. If a first-filer receives final approval but does not immediately launch its product commercially, the clock does not run. This is the mechanism that enabled “bottlenecking” before the MMA — first-filers could sit on their approvals indefinitely. The MMA’s forfeiture provisions address this: a first-filer that fails to market within 75 days of final approval (or certain other triggering events) forfeits its exclusivity, clearing the queue for subsequent filers.
Q5: Can a brand company use an authorized generic to undermine the value of a first-filer generic’s 180-day exclusivity?
Yes. An authorized generic is a version of the brand drug that the brand manufacturer produces and markets itself (or licenses to a third party) at generic prices. Because the authorized generic is sold under the brand’s NDA, it is not an ANDA product and is not subject to the Hatch-Waxman exclusivity framework. An authorized generic can be launched at the same time as the first-filer generic, competing directly during the 180-day exclusivity window. From a first-filer’s perspective, authorized generic competition reduces the revenue premium the 180-day exclusivity was supposed to provide — instead of duopoly competition with only the brand at brand prices, the first-filer faces competition from an authorized generic pricing at near-generic levels. Brand manufacturers that settle with first-filer generics often offer authorized generic rights as part of the settlement consideration, which is one of the non-cash structures the FTC has scrutinized as a potential antitrust concern under Actavis.
Citations
[1] Association for Accessible Medicines. (2025). Generic and Biosimilar Medicines Savings Report 2024. Washington, D.C.: AAM.
[2] Drug Price Competition and Patent Term Restoration Act of 1984, Pub. L. No. 98-417, 98 Stat. 1585 (1984).
[3] Food and Drug Administration. (2024). Generic Drugs: Overview & Basics. U.S. Department of Health and Human Services.
[4] 21 U.S.C. § 355(j)(5)(B)(iii) (Hatch-Waxman Act 30-month stay provisions).
[5] 21 U.S.C. § 355(j)(5)(B)(iv) (180-day generic drug exclusivity provisions).
[6] Kidsforce Research. (2026, March 10). Paragraph IV Certifications: How Generics Challenge Patents Early. Retrieved from https://kidsforce.org/paragraph-iv-certifications-how-generics-challenge-patents-early
[7] Food and Drug Administration. (2003). Guidance for Industry: 180-Day Exclusivity When Multiple ANDAs Are Submitted on the Same Day. U.S. Department of Health and Human Services.
[8] Kidsforce Research. (2026). Paragraph IV certifications and Section viii carve-out usage statistics 2023. Op. cit.
[9] DrugPatentWatch. (2025). The Paragraph IV Playbook: Turning Patent Challenges into Market Dominance. Retrieved from https://www.drugpatentwatch.com/blog/the-paragraph-iv-playbook-turning-patent-challenges-into-market-dominance/
[10] Federal Trade Commission. (2013). Statement of FTC Chairwoman Edith Ramirez on the U.S. Supreme Court’s Decision in FTC v. Actavis, Inc. Washington, D.C.: FTC.
[11] Federal Trade Commission. (n.d.). Watson Pharmaceuticals, Inc., et al. (FTC v. Actavis). Retrieved from https://www.ftc.gov/legal-library/browse/cases-proceedings/071-0060-watson-pharmaceuticals-inc-et-al-ftc-v-actavis
[12] FTC v. Actavis, Inc., 570 U.S. 136 (2013).
[13] Federal Trade Commission. (2016, January 13). FTC Report on Drug Patent Settlements Shows Potential Pay-for-Delay Deals Decreased Substantially in the First Year Since Supreme Court’s Actavis Decision [Press release]. Washington, D.C.: FTC.
[14] Fialkoff, M. L. (2014). Pay-for-delay settlements in the wake of Actavis. Michigan Telecommunications and Technology Law Review, 20(2), 7. Retrieved from https://repository.law.umich.edu/mttlr/vol20/iss2/7/
[15] Carrier, M. A. (2018). FTC v. Actavis: Where we stand after 5 years. IPWatchdog.com. Retrieved from https://ipwatchdog.com/2018/06/18/ftc-v-actavis-stand-5-years/id=98536/
[16] Kidsforce Research. (2026). Patent thicketing and average Orange Book patent counts 2005-2024. Op. cit.
[17] Federal Trade Commission. (2023, November 7). FTC Challenges More Than 100 Patents as Improperly Listed in the FDA’s Orange Book [Press release]. Washington, D.C.: FTC. Retrieved from https://www.ftc.gov/news-events/news/press-releases/2023/11/ftc-challenges-more-100-patents-improperly-listed-fdas-orange-book
[18] Federal Trade Commission. (2024, April 30). FTC Expands Patent Listing Challenges, Targeting More Than 300 Junk Listings for Diabetes, Weight Loss, Asthma and COPD Drugs [Press release]. Washington, D.C.: FTC. Retrieved from https://www.ftc.gov/news-events/news/press-releases/2024/04/ftc-expands-patent-listing-challenges-targeting-more-300-junk-listings-diabetes-weight-loss-asthma
[19] Kagan, S., & Rosen, D. (2024, July 17). The FTC’s challenge to the listing of device patents in the Orange Book: What challenge? National Law Review. Retrieved from https://natlawreview.com/article/ftcs-challenge-listing-device-patents-orange-book-what-challenge
[20] Teva Branded Pharmaceutical Products R&D, Inc. v. Amneal Pharmaceuticals of New York, LLC, No. 2024-1936 (Fed. Cir. July 10, 2024).
[21] Federal Trade Commission. (2025, May). FTC Renews Challenge of More Than 200 Improper Patent Listings [Press release]. Washington, D.C.: FTC. Retrieved from https://www.ftc.gov/news-events/news/press-releases/2025/05/ftc-renews-challenge-more-200-improper-patent-listings
[22] Gardner, J. (2023, November 8). FTC challenges patents held by nine big drugmakers, citing unfair competition. BioPharma Dive. Retrieved from https://www.biopharmadive.com/news/ftc-orange-book-patent-advair-epipen-restasis/699132/
[23] DrugPatentWatch. (2026, March 22). The Patent Cliff Playbook: Pharmaceutical IP Valuation, Generic Entry Timing, and Biosimilar Strategy. Retrieved from https://www.drugpatentwatch.com/blog/patent-expirations-seizing-opportunities-in-the-generic-drug-market/
[24] DrugPatentWatch. (2026, March 22). Drug Patent Expiration: The Complete Strategic Guide to Loss of Exclusivity, Lifecycle Management, and the $400 Billion Cliff. Retrieved from https://www.drugpatentwatch.com/blog/the-impact-of-drug-patent-expiration-financial-implications-lifecycle-strategies-and-market-transformations/
[25] DrugPatentWatch. (2026). Eliquis apixaban NPV analysis and patent cliff implications, op. cit.
[26] Federal Trade Commission. (2015, May 28). FTC Settlement of Cephalon Pay for Delay Case Ensures $1.2 Billion in Ill-Gotten Gains Relinquished [Press release]. Washington, D.C.: FTC.
[27] IntuitionLabs. (2026, February 14). How Generics Challenge Patents: A Hatch-Waxman Act Guide. Retrieved from https://intuitionlabs.ai/articles/generic-drug-patent-challenge-guide