Why Your LOE Strategy Depends on Molecule Type
A brand-name drug losing its final patent is not one event. It is a cascade of legal, regulatory, and commercial decisions that started years before the expiry date and will play out for years after. The mistake most companies make is treating that moment as a single cliff to fall off of, rather than a structured transition they could have shaped.
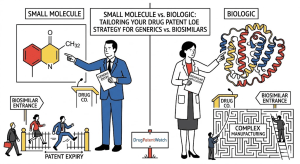
The shape of that transition depends almost entirely on one question: is the drug a small molecule or a biologic? The answer determines which regulatory pathway competitors use to enter the market, how many patents stand between them and launch, how much development capital they need, and how quickly the originator’s market share disappears once they arrive.
This article lays out the full mechanics of Loss of Exclusivity (LOE) for both categories, from patent architecture through regulatory strategy to real-world market erosion. It is written for executives, IP counsel, licensing professionals, and portfolio strategists who need to act on this information.
The differences between generics and biosimilars are not merely technical. They reshape the financial projections, the litigation calculus, and the competitive dynamics around every major product expiry. Getting them wrong is expensive. Getting them right creates durable competitive advantage.
The Core Distinction That Drives Everything
What Makes a Small Molecule
Small molecules are chemically synthesized compounds with defined, low molecular weight structures, typically under 1,000 daltons. Their chemical identity is precise and reproducible: given the same synthesis protocol, two manufacturers will produce structurally identical compounds. Aspirin made in Germany and aspirin made in India are the same molecule. That chemical equivalence is the foundation of the entire generic drug regulatory framework.
Examples span nearly every therapeutic area: atorvastatin (Lipitor), amlodipine (Norvasc), ibuprofen, sildenafil (Viagra), metformin, lenalidomide (Revlimid), and imatinib (Gleevec). The vast majority of the world’s most widely prescribed drugs are small molecules, which is why the generic industry generates over $300 billion in annual sales globally [1].
Small molecules can be produced in large batches using standard chemical synthesis equipment. The process is relatively predictable, scalable, and repeatable. The manufacturing cost difference between an originator and a generic manufacturer is primarily about volume and infrastructure, not fundamental capability. Any competent chemistry operation can learn to make a small molecule if it has the formula and regulatory approval to do so.
What Makes a Biologic Different
Biologics are large, complex molecules produced by living cells: bacteria, yeast, or mammalian cell lines. They include monoclonal antibodies, fusion proteins, peptides, vaccines, blood factors, and other protein-based therapeutics. Their molecular weight often exceeds 150,000 daltons, and their three-dimensional structure, which governs their clinical activity, emerges from an enormously complex manufacturing process.
Adalimumab (Humira), trastuzumab (Herceptin), bevacizumab (Avastin), etanercept (Enbrel), and insulin are all biologics. Monoclonal antibodies alone account for more than $200 billion in annual global pharmaceutical revenue [2]. The top ten best-selling drugs in the United States include a majority of biologics, and that ratio has been shifting further toward biologics for a decade.
The critical property of a biologic is this: the molecule and the manufacturing process are inseparable. Changing a cell line, fermentation condition, purification step, or even the supplier of a single raw material can alter the final molecule’s glycosylation pattern, aggregation state, or immunogenicity profile. This is not a manufacturing technicality. It has direct clinical consequences. The FDA acknowledges this complexity explicitly in its biosimilar guidance, which is why the approval standard for biosimilars is fundamentally different from the standard for generic drugs.
Why Manufacturing Complexity Changes the Competitive Calculus
A generic drug manufacturer needs to demonstrate bioequivalence: that its product delivers the same amount of drug to the bloodstream at the same rate as the reference listed drug. The analytical work is relatively straightforward, the clinical requirement is usually just a pharmacokinetic study in healthy volunteers, and the regulatory timeline is measured in months to a few years.
A biosimilar developer needs to demonstrate that its product is highly similar to the reference product with no clinically meaningful differences in safety, purity, or potency. That requires an extensive analytical characterization package, potentially multiple clinical pharmacology studies, and often at least one clinical efficacy study in a sensitive patient population. Development costs typically run between $100 million and $300 million per biosimilar program, compared to $1 million to $5 million for most generic drug applications [3].
This cost and complexity barrier dramatically changes who can participate in the market and how many competitors eventually enter. A small molecule patent expiry routinely attracts ten or more generic filers within the first year. A biologic patent expiry might attract three to five biosimilar developers over a much longer timeline, and several of those programs will not survive to approval.
Patent Architecture: How Protection Is Built Differently
Small Molecule Patent Anatomy
The patent estate around a small molecule drug is built in layers, with each layer serving a different commercial purpose. Understanding those layers is essential for anyone trying to forecast the real LOE date, which often has nothing to do with the date the primary composition-of-matter patent expires.
Composition of Matter Patents: The Crown Jewels
A composition-of-matter patent claims the chemical structure of the active ingredient itself. It is the strongest form of pharmaceutical patent protection because it bars any competitor from making, using, or selling the compound regardless of formulation, route of administration, or indication. These patents are filed early, typically around the time of lead compound identification, and they run for 20 years from the filing date, which in practice translates to 10 to 12 years of post-approval market exclusivity after accounting for development time.
The first generic to successfully challenge a composition-of-matter patent, or to wait out its expiry, gains access to the largest potential commercial prize. This is why composition-of-matter patents are the primary target of Paragraph IV certifications and inter partes review proceedings at the Patent Trial and Appeal Board (PTAB).
Formulation, Method, and Polymorph Patents
After the primary patent, brand manufacturers file secondary patents covering specific salt forms or polymorphs of the active ingredient, extended-release or modified formulations, methods of treating specific diseases, combination products with other active ingredients, and drug delivery devices.
These secondary patents often expire five to eight years after the primary patent. If they are listed in the Orange Book and the brand manufacturer asserts them against an ANDA filer, they can trigger a 30-month stay of generic approval and extend effective market exclusivity well beyond the primary patent’s life. Pfizer used this strategy extensively with atorvastatin. AstraZeneca deployed it with omeprazole (Prilosec) and later esomeprazole (Nexium), converting a primary-patent expiry into a much longer brand franchise.
The commercial rationality here is clear. Filing a secondary patent costs roughly $30,000 to $50,000 in direct prosecution costs. If it extends exclusivity by even a single month for a drug with $1 billion in annual sales, the return on investment is approximately 1,600:1. Critics call it evergreening. Patent holders call it rewarding innovation in delivery science and new indications. Both characterizations contain truth.
The Orange Book and the 30-Month Stay Mechanism
The FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, universally called the Orange Book, is the published list of patents that brand manufacturers have certified as covering an approved small molecule drug. When a generic manufacturer files an ANDA with a Paragraph IV certification alleging that a listed patent is invalid or not infringed, the brand manufacturer has 45 days to sue for infringement. If it does, the FDA cannot approve the ANDA for 30 months from the date of the notification, regardless of whether the patent has any merit.
This 30-month stay is arguably the most commercially powerful procedural mechanism in pharmaceutical law. It gives brand manufacturers a guaranteed nearly three-year period to litigate, settle, or develop lifecycle alternatives before a generic can reach the market. Abusive listing practices, where companies add weak patents to the Orange Book primarily to trigger stays, have been repeatedly challenged, but the core mechanism remains robust.
Equally important is what the Orange Book excludes. Process patents, metabolite patents, and patents on the manufacturing process are generally not eligible for Orange Book listing. This matters for LOE strategy because it means generic manufacturers often do not need to challenge or design around a brand’s manufacturing innovation to enter the market.
Biologic Patent Architecture
Biologics have their own patent ecosystem, governed by different legal rules and built around different commercial objectives. The key structural difference from small molecules is the emphasis on process patents and the formal role of the BPCIA’s patent exchange procedure.
The Patent Dance Under the BPCIA
The Biologics Price Competition and Innovation Act (BPCIA) of 2009 created the biosimilar approval pathway but also established a highly unusual patent resolution procedure, known in the industry as the ‘patent dance.’ A biosimilar applicant that receives FDA acceptance of its 351(k) application must provide the reference product sponsor with a detailed description of its manufacturing process within 20 days. The sponsor then has 60 days to identify the patents it believes are infringed. The parties then exchange claim charts and, in theory, negotiate in good faith toward a patent resolution framework before litigation begins.
In practice, the patent dance is rarely danced as Congress intended. Reference product sponsors, recognizing that the exchange of manufacturing information creates discovery advantages, have routinely chosen to omit the dance and proceed directly to litigation. The Supreme Court addressed parts of this in Sandoz Inc. v. Amgen Inc. [4], holding that participation in the dance is optional for biosimilar applicants and that a biosimilar applicant may give notice of commercial marketing before FDA approval. The decision clarified the rules but did not simplify the underlying litigation dynamics.
How Biologics Stack and Layer Patents
A mature biologic franchise like adalimumab or etanercept can have 70 to 100 patents in its portfolio, covering the amino acid sequence, glycosylation patterns, formulation (concentration, pH, excipients), delivery device (prefilled syringes, autoinjectors), manufacturing cell lines, purification processes, and methods of treating specific diseases. AbbVie’s adalimumab estate reportedly included over 100 patents at one point, with expiry dates stretching into the mid-2030s despite the primary sequence patent expiring in 2016 [5].
This layering achieves something structurally different from small molecule secondary patents. With small molecules, secondary patents typically do not prevent bioequivalent entry; a generic can use a different synthesis route or formulation and still achieve bioequivalence. With biologics, the manufacturing process is clinically relevant, so a biosimilar developer who alters the process to avoid a manufacturing patent risks changing the product in ways that require additional clinical data to justify. The process patent becomes, in effect, a clinical risk factor.
Manufacturing Process Patents as Competitive Moats
For biologics, process patents cover fermentation conditions, cell culture media, purification sequences, viral inactivation steps, fill-finish procedures, and storage conditions. These are not peripheral intellectual property. They represent decades of process development and optimization, and the resulting manufacturing quality is directly tied to product safety.
A biosimilar developer who reverse-engineers a competitor’s process faces two distinct risks. First, it may infringe the process patent and face litigation. Second, even if it designs around the patent by using a different process, it may produce a product with different characteristics that require more extensive clinical testing to establish biosimilarity. This dual risk substantially increases development cost and uncertainty, giving the process patent a commercial value well beyond its legal scope.
AstraZeneca demonstrated sophisticated use of this dynamic with its biologics programs, and Genentech’s extensive process patent portfolio around bevacizumab and trastuzumab created meaningful barriers for early biosimilar entrants in the European market, where biosimilar regulation has been active since 2006.
Loss of Exclusivity: The Revenue Cliff in Numbers
Abstract discussions of patent protection matter less than concrete financial outcomes. The numbers around generic and biosimilar market penetration are the most direct evidence of why LOE strategy deserves serious investment.
How Generics Erode Brand Sales
The generic erosion curve for small molecules is among the most reliable phenomena in commercial pharmaceuticals. Within the first six months after generic entry, branded products typically lose 70% to 90% of their prescription volume in the United States [6]. By the end of the first year, the brand’s market share by volume routinely falls below 10% in competitive categories. Price erosion follows unit erosion: as more generic manufacturers enter, average selling prices decline to 15% to 20% of the original brand price within 18 to 24 months of initial generic entry.
“Generic drugs account for 90% of prescriptions dispensed in the United States but only 18% of total drug spending, a ratio that reflects the price compression that follows small molecule LOE.” — Association for Accessible Medicines, Generic Drug & Biosimilars Access & Savings in the U.S. Report, 2023 [7]
The speed and severity of this erosion depend on several factors: the number of generic entrants (more competitors accelerate price erosion), whether the brand has an authorized generic in the market, formulary decisions by payers and pharmacy benefit managers, and the presence of any brand lifecycle management product such as an extended-release formulation.
Lipitor’s experience illustrates the extreme end of the erosion spectrum. When atorvastatin lost exclusivity in November 2011, Pfizer had negotiated a market arrangement with Watson Pharmaceuticals under which Watson received a license to sell an authorized generic. Multiple additional generics entered quickly. Atorvastatin brand revenue fell from approximately $5 billion annually in the U.S. to under $500 million within two years. The product did not disappear; Pfizer continued selling branded Lipitor at a substantial premium to a small, loyal patient segment. But the commercial franchise, as it had existed for a decade, was gone.
How Biosimilar Uptake Differs
Biosimilar market penetration follows a fundamentally slower and less predictable curve than generic penetration, and the U.S. experience has been notably weaker than European performance. Several structural factors explain this gap.
First, biologics are largely administered in clinical or specialty pharmacy settings, where prescriber habits and institutional formularies drive utilization rather than automatic substitution at the pharmacy level. The pharmacist automatic substitution rules that accelerate generic penetration in retail pharmacy do not apply to biosimilars unless the FDA has granted an interchangeability designation, a bar that no biosimilar had cleared until Semglee (insulin glargine-yfgn) in July 2021 [8].
Second, brand manufacturers of biologics have used patient assistance programs, co-pay cards, and direct contracts with hospital systems and specialty pharmacy networks to effectively insulate their patient base from biosimilar competition. AbbVie’s defense of Humira in the U.S. market after biosimilar entry in 2023 included rebate contracts with PBMs that positioned branded Humira more favorably on formulary than several biosimilars, despite the biosimilars carrying a substantial list price discount.
Third, physician comfort with biosimilars remains uneven. Surveys consistently show that rheumatologists and oncologists have higher acceptance rates than other specialties, but system-wide adoption still requires active management, including education, formulary incentives, and patient consent conversations that add friction to the transition.
The result is that biosimilar market share, by volume, typically reaches 30% to 40% in the U.S. within two years of initial entry in major therapeutic categories, compared to the 80% to 90% generic share that small molecules achieve in the same period [9]. European biosimilar markets, aided by more aggressive tendering systems and national formulary mandates, show penetration rates 15 to 25 percentage points higher than the U.S. in comparable therapeutic categories [10].
Side-by-Side Market Erosion Rates
The practical consequence of this difference is that a biologic LOE generates a much longer revenue tail for the originator than a small molecule LOE does. Brand revenues for a biologic can still represent 50% to 60% of the pre-LOE baseline two years after biosimilar entry. For a small molecule, that figure is typically 10% to 20% of the pre-LOE baseline at the same interval.
This changes the financial calculus for both sides. Generic manufacturers win by moving fast and taking volume at modest margins. Biosimilar developers need sustained commercial infrastructure, payer contracting capabilities, and patience. The investment thesis is different, the competitive dynamics are different, and the LOE strategy documents should reflect those differences from the start.
The Generic Drug Pathway: ANDA and Hatch-Waxman
The modern generic drug industry exists because of the Drug Price Competition and Patent Term Restoration Act of 1984, known as Hatch-Waxman after its sponsors. Before Hatch-Waxman, generic manufacturers had to repeat full clinical trials to demonstrate safety and efficacy, a cost and time barrier that effectively blocked generic competition for most products. The Act created the Abbreviated New Drug Application (ANDA) pathway, which substitutes bioequivalence data for clinical trials when the active ingredient, route of administration, dosage form, and strength are the same as an approved reference drug.
Hatch-Waxman Mechanics
An ANDA applicant submits data demonstrating that its product is bioequivalent to the reference listed drug and that its manufacturing facilities meet FDA current Good Manufacturing Practice (cGMP) standards. The clinical bar is relatively low: two single-dose pharmacokinetic studies (fed and fasted) in healthy volunteers, demonstrating that the 90% confidence intervals for Cmax and AUC fall within 80% to 125% of the reference product’s parameters.
The patent certification element is where the commercial strategy enters. For each patent listed in the Orange Book for the reference drug, the ANDA applicant must file one of four certifications: that no patent exists (Paragraph I), that the patent has expired (Paragraph II), that the ANDA will not launch until the patent expires (Paragraph III), or that the listed patent is invalid, unenforceable, or not infringed by the generic product (Paragraph IV).
Paragraph III certifications, called ‘wait-and-see’ filings, are the conservative choice. The generic manufacturer accepts the patent term and waits for expiry. Paragraph IV certifications are the aggressive choice: the manufacturer challenges the patent’s validity or argues non-infringement, and the brand can trigger the 30-month stay by suing within 45 days.
Paragraph IV Challenges and the First-to-File Race
The strategic importance of being the first company to file a Paragraph IV certification for a given drug cannot be overstated. Under Hatch-Waxman, the first applicant to file a substantially complete ANDA with a Paragraph IV certification for a particular drug product earns the right to 180 days of generic market exclusivity before any other ANDA filer can have its application approved.
This 180-day exclusivity window is extraordinarily valuable. When a single generic manufacturer has exclusive market access for six months, even at a price 20% to 30% below brand, the revenues from that window can exceed $100 million for a blockbuster product. Teva Pharmaceutical built much of its U.S. market leadership through aggressive first-to-file strategies, accumulating hundreds of 180-day exclusivity periods across its history.
The race to file first drives intense competitive intelligence activity. Generic manufacturers monitor brand patent filings at the USPTO, Orange Book additions, and FDA approval activity for reference drugs whose primary patents are approaching expiry. DrugPatentWatch is one of the most widely used professional tools for this monitoring: it aggregates patent expiry data, Orange Book listings, ANDA filing histories, and paragraph certification activity into a structured database that allows generic and brand manufacturers alike to track the competitive patent landscape across thousands of drug products simultaneously. For portfolio managers making filing decisions across dozens of molecules, having that level of organized patent intelligence is operationally critical.
The monitoring challenge has grown more complex as brand manufacturers have become more sophisticated about their Orange Book strategies. Some have added patents to the Orange Book shortly before generic filing activity was expected, specifically to trigger additional 30-month stays. The FDA’s final rule on patent listings, effective 2024, attempted to tighten eligibility criteria for Orange Book patents, but the strategic dynamic continues.
180-Day Exclusivity: The First-Filer Reward
The 180-day exclusivity period attaches to the first filer, not to the first manufacturer to actually market the product. This creates a layered strategic dynamic. The first filer’s 180-day clock begins on the date it first commercially markets the generic product, which means that if litigation delays are resolved in the brand’s favor, the clock may not start for years. Conversely, if the first filer wins the Paragraph IV litigation or settles on favorable terms, it can choose the precise moment to start the commercial clock based on its own launch readiness.
Forfeiture of the 180-day exclusivity is possible under specific circumstances codified by the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA). A first filer loses its exclusivity if it fails to market within a specified period after certain triggering events, if it enters into an agreement with the brand manufacturer that delays market entry, or if another applicant wins a court ruling that the challenged patent is invalid or not infringed. The forfeiture provisions were designed to prevent ‘parking’ of exclusivity rights, where a first filer holds the exclusivity without launching in order to accommodate a settlement with the brand.
Pay-for-delay settlements, also called reverse payment settlements, have been among the most controversial elements of Hatch-Waxman strategy. In a typical arrangement, the brand manufacturer pays the first generic filer a substantial sum in exchange for the generic delaying its entry until a specified date. The Supreme Court’s ruling in FTC v. Actavis, Inc. [11] in 2013 held that these settlements can violate antitrust law and must be evaluated under a rule-of-reason analysis. The legal risk has not eliminated pay-for-delay settlements, but it has made their structure considerably more complex.
Authorized Generics and Brand Defense
An authorized generic is a version of the brand-name drug that the brand manufacturer itself markets, either through a subsidiary or through a contract with a generic manufacturer, using the brand’s approved NDA. Because it is not a separate ANDA, it does not require a Paragraph IV certification and it is not subject to the 180-day exclusivity barrier.
The strategic logic is this: when a first-filer generic gains its 180-day exclusivity, an authorized generic competes directly against it during that exclusivity window. The brand manufacturer effectively destroys the first filer’s pricing power during the most valuable period of generic exclusivity, reducing the financial reward for having filed the Paragraph IV certification in the first place. The long-term effect is to reduce the incentive for future Paragraph IV challenges, which is exactly what brand manufacturers want.
Pfizer deployed this strategy aggressively with atorvastatin (Lipitor), partnering with Watson Pharmaceuticals as the authorized generic manufacturer. The result was that Watson, despite having 180-day exclusivity, faced immediate competition from Pfizer’s own authorized generic and never achieved the pricing premium that exclusivity periods normally deliver. Generic industry trade associations have repeatedly lobbied against authorized generics, but they remain entirely legal and are used routinely by brand manufacturers with valuable expiring products.
Patent Linkage and Orange Book Strategy
Orange Book strategy is one of the most consequential decisions a brand manufacturer makes in its IP lifecycle plan. What gets listed, when it gets listed, and how it is listed determines whether competitors face 30-month stays that must be litigated or expire uneventfully.
The FDA lists only those patents that claim the approved drug product or method of using it. Process patents are not eligible. But the brand manufacturer certifies the eligibility of its listed patents, and FDA historically has not reviewed that certification for accuracy. This self-certification model created opportunities for strategic over-listing, which the FTC challenged repeatedly in the early 2000s through its reports on brand/generic settlement practices.
From the generic manufacturer’s perspective, monitoring Orange Book additions in real time is essential intelligence work. A new patent listing six months before planned ANDA filing can trigger an entirely different litigation scenario and timeline than the one built into the original project plan. Companies that use tools like DrugPatentWatch to monitor Orange Book changes as they occur, rather than waiting for annual reviews, are better positioned to adjust their filing strategies before they become locked into a litigation path they did not price into their investment thesis.
The Biosimilar Pathway: 351(k) and the BPCIA
The BPCIA created Section 351(k) of the Public Health Service Act as the abbreviated pathway for biosimilar and interchangeable biological product applications. Where Hatch-Waxman’s ANDA pathway created a relatively standardized submission for small molecules, 351(k) requires a totality-of-the-evidence approach that gives FDA significant discretion in determining what data package is sufficient for any given biosimilar program.
What 351(k) Actually Requires
A 351(k) application must demonstrate biosimilarity to the reference product on the basis of: analytical studies showing the biological product is highly similar to the reference product notwithstanding minor differences in clinically inactive components; animal studies including the assessment of toxicity; and a clinical study or studies sufficient to demonstrate safety, purity, and potency in one or more appropriate conditions of use.
The FDA has progressively evolved its position on clinical study requirements, signaling in several guidance documents that sponsors with sufficiently robust analytical and pharmacology packages may be able to reduce or eliminate the need for comparative clinical efficacy trials. This is a meaningful development for the economics of biosimilar development. A pivotal clinical trial can cost $30 million to $80 million and take three to four years. Eliminating it, for programs where the analytical evidence is sufficiently compelling, can transform the financial profile of a biosimilar development program.
The totality-of-the-evidence standard means that no single element is determinative. A biosimilar program with exceptional analytical characterization may need less clinical data than one where analytical tools cannot fully characterize critical quality attributes. This flexible framework is scientifically appropriate but commercially challenging, because it makes it difficult to design a development program with high confidence in the regulatory outcome until you are deep into the analytical phase.
Interchangeability: What It Means for Market Access
Beyond biosimilarity, a biosimilar developer can seek an interchangeability designation, which requires demonstrating that the product produces the same clinical result as the reference product in any given patient and, for products administered more than once, that the risk in terms of safety or diminished efficacy of alternating or switching between the biosimilar and the reference product is not greater than the risk of using the reference product without switching.
Interchangeability is commercially significant for a specific reason: most state pharmacy laws allow pharmacists to automatically substitute an interchangeable biological product for the reference product without prescriber intervention, mirroring the automatic substitution rules that apply to generic drugs. Without interchangeability, a pharmacist who wants to dispense a biosimilar instead of the reference biologic must either get a new prescription or, depending on state law, get prescriber authorization.
As of early 2026, the FDA had granted interchangeability designations to products including insulin glargine products, aflibercept biosimilars, and a handful of others. The competitive advantage conferred by interchangeability is most significant in the retail and specialty pharmacy channels, where substitution at point of dispensing drives volume. For hospital-based biologics administered as infusions, interchangeability matters less because formulary decisions are made at the institutional level.
One nuance that biosimilar developers often underestimate is that the first interchangeable biosimilar for a given reference product earns a period of exclusivity before any other biosimilar can be granted interchangeability designation for the same reference product. This exclusivity period ranges from one to three years depending on circumstances, giving a meaningful first-mover advantage to programs that achieve interchangeability early.
Extrapolation of Indications
One of the most commercially valuable elements of the biosimilar framework is the ability to extrapolate approved indications. If a biosimilar demonstrates biosimilarity in one indication, the FDA may approve it for other indications held by the reference product without requiring separate clinical trials in each indication, provided the mechanism of action is the same and the totality of evidence supports extrapolation.
For reference products approved across multiple indications, this is important. Adalimumab (Humira) has over a dozen approved indications covering rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, plaque psoriasis, Crohn’s disease, ulcerative colitis, and others. A biosimilar approved in rheumatoid arthritis with extrapolation to other indications effectively competes across the full brand franchise without having run clinical trials in each individual disease. For a drug with $21 billion in global revenues, even a 20% market share across all indications is a $4 billion commercial opportunity.
The extrapolation framework is not automatic. FDA evaluates each extrapolation request on a product-specific and indication-specific basis. Biosimilar developers must address mechanistic rationale, safety considerations specific to each patient population, and any differences in clinical pharmacology that might affect performance. But when extrapolation is granted, it provides commercial leverage that fundamentally changes the economics of the development program.
Nonproprietary Naming Conventions and Their Consequences
The FDA requires that biosimilars have a nonproprietary name that includes a distinguishing suffix consisting of four lowercase letters attached with a hyphen to the core name of the reference product. For example, adalimumab-adaz is a biosimilar of adalimumab. This naming convention was intended to improve pharmacovigilance by enabling clear attribution of adverse events to specific products.
The commercial consequence is that biosimilars cannot rely on the reference product’s brand recognition in the way that generic drugs benefit from being labeled with the same active ingredient name and thereby being interchangeable under state pharmacy laws for non-interchangeable biosimilars. A physician who wants to prescribe the cheaper version of adalimumab must affirmatively prescribe the specific biosimilar by its suffix-modified name, rather than simply writing ‘adalimumab’ and having the pharmacist substitute. This is a structural barrier to volume growth that has no analogue in the generic drug market.
The Purple Book vs. The Orange Book
The Purple Book is the FDA’s registry of licensed biological products and their biosimilars and interchangeable products, analogous to the Orange Book for small molecules. Unlike the Orange Book, the Purple Book does not list patents. It records the exclusivity periods for the reference product and the biosimilar products, including the 12-year period of reference product exclusivity established under BPCIA during which no biosimilar may be approved, the four-year period after which a 351(k) application may be filed, and any interchangeability exclusivity periods.
The absence of patent information from the Purple Book reflects the fundamental difference in how biologic patent disputes are resolved: through the BPCIA patent dance or litigation outside of it, rather than through the automated linkage system that the Orange Book creates for small molecules. For LOE analysts, this means that biologic patent surveillance requires actively monitoring USPTO filings, PTAB proceedings, and litigation records rather than relying on a single government database.
LOE Timing: When to Enter and When to Wait
The decision of when to enter a market after LOE is not simply about being ready as soon as legally possible. Both generic and biosimilar manufacturers make strategic choices about entry timing that materially affect their financial outcomes.
First-Mover Advantage for Generic Entrants
For generic manufacturers, first-mover advantage is explicit and legislated through the 180-day exclusivity period. The first company to file a substantially complete ANDA with a Paragraph IV certification, and to successfully navigate any resulting litigation, earns the exclusive right to sell the only generic version of the drug for six months. During that period, the first filer can price its product at 20% to 30% below the brand and still capture enormous commercial value because it faces no generic price competition.
The financial modeling for Para IV challenges typically starts with this six-month exclusivity window. Analysts calculate the revenue at risk by taking the reference product’s annual U.S. sales, applying an estimated market share capture rate for a single generic entrant (often 60% to 70% of total prescriptions within the first three months), adjusting for the expected pricing discount, and then applying the probability of winning the litigation. The result is the risk-adjusted NPV of the first-to-file position.
For major drugs, this NPV can exceed $500 million. That is the financial justification for the legal fees, expert witness costs, and organizational investment that aggressive Para IV programs require. Companies like Teva Pharmaceutical, Mylan (now Viatris), and Dr. Reddy’s Laboratories built substantial portions of their U.S. revenue base on repeated successful execution of this strategy over the past 30 years.
The Biosimilar Paradox: Why Early Isn’t Always Best
Biosimilar market entry timing is more nuanced. Being first to market with a biosimilar does not guarantee market leadership, for reasons that have no parallel in generic drugs.
First, biosimilar launch requires substantial commercial infrastructure: medical affairs teams capable of educating physicians and payer medical directors, market access teams capable of negotiating formulary positions with PBMs and integrated delivery networks, and patient support programs capable of competing with the brand’s co-pay assistance programs. A biosimilar manufacturer that launches first but lacks this infrastructure can spend significantly on launch without achieving meaningful market penetration.
Second, payers typically prefer to have multiple biosimilar options available before conducting the formulary competitions that generate the deepest discounts. If only one biosimilar is available, a PBM has limited negotiating leverage and the biosimilar developer has limited competitive pressure to offer large rebates. When two or three biosimilars are available and competing for formulary exclusivity or preferred tier placement, the competitive dynamics intensify rapidly.
Third, clinical experience data accumulates over time. A biosimilar that launches first will have limited real-world evidence. Biosimilars that launch 12 to 18 months after the first entrant can cite the first entrant’s real-world safety and efficacy data as additional evidence for the overall biosimilar class, benefiting from the pioneer’s investment in post-market evidence generation.
Staggered Entry as Strategy
Savvy biosimilar developers explicitly plan for entry timing. Rather than treating FDA approval as the start of commercial launch planning, leading developers build 12 to 18 months of payer contracting, health system engagement, and physician education into their pre-launch timelines. They also build contingency plans for scenarios where the brand manufacturer’s defensive contracting has locked out a significant portion of the accessible market at the time of their approval.
The Humira biosimilar market in the U.S., which opened formally in January 2023, illustrates the consequences of inadequate entry timing preparation. Several biosimilar developers had approval but faced a market in which AbbVie had signed long-term contracts with major PBMs covering hundreds of millions of patients. Initial biosimilar penetration was slow despite significant list price discounts. Manufacturers who had invested heavily in payer relationships before launch fared substantially better than those who treated approval as the beginning of their commercial planning.
Competitive Intelligence: Watching the Pipeline
LOE strategy is not a document you draft once and update annually. It requires continuous competitive surveillance, because the patent and regulatory landscape shifts constantly. New patents are filed and granted. Orange Book listings are added and challenged. ANDA and 351(k) applications are filed without public notification. Litigation results change the timeline for specific competitors. Each of these events can materially alter the commercial outlook for a product.
Using DrugPatentWatch to Monitor Patent Activity
DrugPatentWatch is the pharmaceutical industry’s most comprehensive commercial database for tracking patent expirations, Orange Book listings, ANDA filing activity, and FDA approval history across approved drugs. For LOE strategy teams, it is an operational intelligence resource, not a research database.
The platform aggregates data from the USPTO, the FDA’s Orange Book, the Purple Book, the FDA’s drug approval database, and patent litigation records. For any approved drug product, a user can see which patents are listed in the Orange Book, when they expire, whether Paragraph IV certifications have been filed against them, the outcome of any resulting litigation, and the status of pending ANDA applications. For biologics, the database tracks Purple Book listings and biosimilar application activity.
For brand manufacturers, DrugPatentWatch provides visibility into competitive entry timelines, allowing LOE planning teams to accurately forecast when they should expect the first generic or biosimilar to enter their market. For generic and biosimilar developers, it provides comparable competitive intelligence about which products are already in development by competitors, reducing the risk of building a pipeline crowded with entries that will ultimately fight over a smaller market share than the NPV models assumed.
The practical use cases extend to M&A and licensing. When a company is evaluating the acquisition of a late-stage generic or biosimilar program, the patent intelligence available through databases like DrugPatentWatch is a critical input to due diligence. Understanding how many Paragraph IV certifications have been filed, whether any litigation has been resolved, and what the realistic exclusivity window looks like has a direct impact on the valuation of the program.
Reading FDA Databases for Market Signal
Beyond patent databases, the FDA’s own public systems provide rich intelligence for LOE planning. The Paragraph IV Certifications List, published on the FDA website, identifies drugs for which ANDA applicants have filed Paragraph IV certifications, sorted by date of first receipt. For brand manufacturers, this list is an early warning system. For generic manufacturers, it identifies competitive activity by other filers.
The FDA’s Drug Approval Database contains the Approval History for each approved NDA, including records of all supplements, pediatric exclusivity grants, and orphan drug designations that may extend effective exclusivity. A brand manufacturer that obtains pediatric exclusivity, six months of additional exclusivity granted for completing FDA-requested pediatric studies under the Best Pharmaceuticals for Children Act, effectively pushes out all competitive entry dates by six months. For a blockbuster drug, that six-month extension can be worth $500 million to $1 billion in preserved revenues, making the cost of conducting pediatric studies a trivially small investment.
Tracking Litigation and Settlement Activity
Paragraph IV litigation generates a public record of briefs, court filings, and judgments that constitutes one of the richest sources of patent intelligence in the pharmaceutical sector. The district court cases are publicly available through PACER, the federal courts’ electronic records system. Outcomes in major cases are widely covered in the specialized pharmaceutical law press.
Tracking settlement activity is equally important. When a brand manufacturer and a first-filer generic settle a Paragraph IV lawsuit, they must notify both the FTC and the DOJ under the MMA. The settlements filed under this notification requirement provide intelligence about entry date agreements, authorized generic provisions, supply agreements, and other commercial terms that reveal the parties’ assessments of the underlying patent validity and infringement questions.
For biosimilar developers, monitoring the publicly available records of BPCIA patent exchanges, where they have occurred, and tracking inter partes review petitions filed against biologic patents at the PTAB provides a comparable landscape view. PTAB trial statistics show institution rates for biologic patents that can inform biosimilar developers’ decisions about whether to pursue IPR challenges as part of their patent clearance strategy.
Pricing and Reimbursement After LOE
Patent strategy and regulatory strategy are upstream of the commercial decisions that ultimately determine how much value a company captures from either side of an LOE event. Pricing and reimbursement dynamics after LOE differ profoundly between small molecule generics and biosimilars.
Generic Price Erosion Curves
Generic price erosion follows a predictable pattern that has been extensively documented in FDA economic analyses, academic literature, and industry reports. During the 180-day exclusivity period, the first generic typically prices at 10% to 30% below the brand’s list price. Once additional generics enter, prices drop rapidly. After five or more generic entrants, average selling prices often fall to 15% to 25% of the original brand price [12].
The speed of the price decline depends on the number of ANDA approvals, the size of the market, and whether the brand has an authorized generic in the market. Authorized generics accelerate price erosion during the exclusivity period by creating two-supplier competition during what should be a single-supplier window.
After five years of generic competition, the remaining market structure typically consists of three to five commodity manufacturers competing on price, with the brand holding a small residual market share among patients resistant to switching and prescribers with strong brand preferences. The brand continues to sell at a price two to four times the generic price, serving this residual segment at very high gross margins on relatively low volumes.
Biosimilar Discounting Realities
Biosimilar pricing strategy is more complex than generic pricing and varies significantly between the list price and the net price after rebates. Initial biosimilar launches in the U.S. have typically offered list price discounts of 15% to 35% below the reference biologic’s list price. But the reference biologic’s list price, after rebates to PBMs and payers, may already be 40% to 50% below list. A biosimilar offering a 20% list price discount is thus not necessarily cheaper than the reference product on a net-price basis from the payer’s perspective.
This creates the perverse dynamic visible in the early U.S. Humira biosimilar market: biosimilars were listed at $1,000 to $2,000 below Humira’s list price per package, yet Humira remained on formulary at preferred status for many patients because AbbVie’s rebate structure made it more financially attractive to the PBM than the biosimilar alternatives.
Some biosimilar manufacturers responded by launching at even steeper discounts, 50% or more below list price, betting that the net-price advantage becomes compelling enough to override AbbVie’s contract terms. Amgen, Samsung Bioepis, and other manufacturers launched adalimumab biosimilars at these aggressive price points in 2023, creating a market in which the list price spread between branded Humira and the cheapest biosimilar exceeded $60,000 per patient per year.
PBM Formulary Levers and Rebate Dynamics
Pharmacy benefit managers control formulary placement for a majority of commercially insured lives in the United States. Their decisions about which drugs receive preferred tier status, which require prior authorization, and which qualify for automatic substitution drive prescribing patterns at a scale that no individual prescriber relationship can match.
For small molecule generics, PBMs typically move quickly: once a generic is available at 80% to 90% price reduction versus brand, generic products go to the lowest cost-sharing tier and brand products are relegated to non-preferred or specialty tiers. The financial incentive is clear and immediate.
For biosimilars, the calculus is murkier because the reference product manufacturers are negotiating rebate structures specifically designed to preserve their formulary position. AbbVie reportedly offered PBMs rebates of up to 80% off Humira’s list price in some contracts, effectively making net-priced branded Humira competitive with biosimilars that lacked equivalent rebate infrastructure. The FTC published a study in 2022 documenting PBM practices that limit biosimilar uptake [13], and CMS has proposed rule changes that could improve the biosimilar’s competitive position in Medicare Part D.
Case Studies: What History Teaches
Abstract strategy becomes concrete through real examples. The following case studies represent the full range of LOE outcomes, from catastrophic revenue cliffs to surprisingly durable brand franchises.
Lipitor (Atorvastatin): The Template for Generic Cliffs
Atorvastatin’s November 2011 U.S. patent expiry is the canonical example of small molecule LOE. At the time, Lipitor was the world’s best-selling drug, with approximately $10 billion in annual global sales and roughly $5.3 billion in U.S. revenues. Pfizer knew the expiry was coming for years and prepared accordingly, but the structural reality of Hatch-Waxman meant that no amount of preparation could prevent the fundamental revenue decline.
Pfizer’s defensive strategy had several components. It sued multiple Para IV filers, winning some patent claims and narrowing the field of potential first-filers. It negotiated the Watson authorized generic arrangement, which put a branded authorized generic into the market simultaneously with Watson’s 180-day exclusivity launch. It extended the Lipitor franchise through Caduet (atorvastatin/amlodipine) and pursued international lifecycle management. It ran an aggressive direct-to-consumer campaign reminding patients that brand Lipitor was ‘the only atorvastatin clinically proven in multiple large heart studies,’ a messaging strategy intended to retain the residual loyal segment.
None of it changed the fundamental outcome. Within 18 months of generic entry, atorvastatin was a generic commodity drug in the U.S. market, with over 15 manufacturers selling at prices below $0.10 per tablet. Pfizer’s U.S. Lipitor revenues fell by approximately 75% within two years of generic entry. The episode demonstrated that for a large-market oral solid dosage form small molecule, no amount of commercial preparation fully offsets the structural impact of commodity generic entry.
The lesson for brand manufacturers is not that preparation is useless. Pfizer’s authorized generic strategy did recover hundreds of millions of dollars in value during the exclusivity window. Its litigation strategy delayed entry for some filers. Its lifecycle management in other markets extended the overall franchise value. Preparation manages the shape of the cliff; it does not eliminate it.
Humira (Adalimumab): Patent Thickets and Delayed Entry
AbbVie’s adalimumab strategy is one of the most studied examples of biologic patent thicketing in pharmaceutical history. The primary sequence patents for adalimumab began expiring in 2016. Yet no U.S. biosimilar launched until January 2023, seven years after the first patent expiry.
The delay resulted from AbbVie’s construction of a formidable secondary patent estate, reportedly containing over 100 patents at its peak, covering formulation (specifically the high-concentration, low-volume subcutaneous formulation that enabled patients to self-inject at home), delivery devices (pre-filled syringes and autoinjector pens), manufacturing processes, and treatment methods for specific disease subsets. Biosimilar developers who sought to enter had to either design around this estate, challenge the patents through IPR proceedings, or negotiate settlement agreements that included entry date provisions.
AbbVie reached settlements with multiple biosimilar developers beginning in 2017, granting licenses with entry dates that clustered around January 2023 for the U.S. market, while allowing earlier entry in European markets where the patent estate was weaker. The settlements preserved AbbVie’s U.S. revenue stream for seven additional years after the primary patent expiry, during which time Humira generated approximately $140 billion in global revenues.
The EU Humira biosimilar market, where entries began in October 2018, provides the counterfactual. Multiple biosimilars entered the European market over the following 18 months, driving substantial price competition in markets that use national tendering. AbbVie maintained revenue in Europe through premium positioning in some markets and competitive rebating in others, but the European franchise experienced substantially greater biosimilar penetration than the U.S. market during the same period.
Remicade (Infliximab): The Contract-Based Defense
Infliximab (Remicade), marketed by Janssen in the U.S., faced biosimilar entry from Pfizer’s Inflectra and Merck’s Renflexis beginning in 2016 and 2017 respectively. Unlike Humira, Remicade’s key patents had expired without a comparably extensive secondary patent estate to defend the brand. Janssen’s strategy therefore pivoted from patent defense to contract-based market defense.
Janssen negotiated exclusive contract arrangements with hospital systems and specialty infusion centers that were the primary points of infliximab administration. These contracts, which offered substantial volume discounts and rebates tied to formulary exclusivity for branded Remicade, effectively created contractual barriers to biosimilar entry that operated in parallel with any remaining patent protection.
The FTC and multiple states challenged these contracting practices as anticompetitive. Janssen eventually reached settlements and modified its contracting practices, but not before sustaining Remicade’s market position through the critical early period of biosimilar competition. As of 2019, branded Remicade still held approximately 85% of the U.S. infliximab market by volume, despite being two to three years into the biosimilar competition period [14]. The contrast with small molecule LOE, where branded products typically fall below 20% market share within 12 months, is stark.
The Remicade case established a template that brand biologic manufacturers have used repeatedly: when patents are insufficient to block biosimilar entry, commercial contracting can substantially delay market penetration. It also established that these practices attract regulatory and antitrust scrutiny, creating legal risk that must be priced into the strategy.
Revlimid (Lenalidomide): Volume-Limited Authorized Generics
Bristol Myers Squibb’s Revlimid (lenalidomide) provides one of the most creative, and controversial, examples of LOE management for a small molecule drug. Lenalidomide’s key composition-of-matter patent began expiring in 2022, but BMS negotiated settlement agreements with generic manufacturers that limited the volume of generic lenalidomide that could be sold during a multi-year entry phase.
The settlement structure was unusual: generics were permitted to enter the market beginning in March 2022, but under the initial terms, each generic manufacturer was limited to a specific volume cap that increased over a scheduled ramp period through 2026. The practical effect was that generic lenalidomide existed in the market but could not fully displace branded Revlimid because the volume caps prevented generics from meeting the full demand of the approximately $5 billion U.S. market.
BMS retained billions in Revlimid revenue during a period when, under a standard Hatch-Waxman framework, branded Revlimid would have been fully commoditized. The FTC reviewed the settlements and ultimately did not block them, despite criticizing the structure. The episode demonstrated that LOE strategy innovation continues to find new forms, driven by the magnitude of the financial stakes.
The Next Wave: Immuno-Oncology and GLP-1s
The pharmaceutical sector is approaching a second major wave of patent cliffs, concentrated in two therapeutic areas: immuno-oncology (checkpoint inhibitors like pembrolizumab/Keytruda and nivolumab/Opdivo) and cardiometabolic disease (GLP-1 receptor agonists like semaglutide/Ozempic/Wegovy and tirzepatide/Mounjaro).
Pembrolizumab, Merck’s anti-PD-1 antibody, generates over $25 billion annually and faces biosimilar development programs that began in earnest in the early 2020s. The primary sequence patents expire in the late 2020s. Given the complexity of PD-1 antibody manufacturing and the extensive clinical evidence required for biosimilarity across multiple tumor types, meaningful biosimilar market penetration in immuno-oncology is unlikely before 2030 to 2032 even if technical development proceeds smoothly.
GLP-1 LOE strategy is structurally different because semaglutide exists in both small molecule (oral semaglutide, Rybelsus) and peptide/biologic forms. The patent strategy appropriate for the oral product differs from the strategy for the injectable formulation. This molecule-type complexity within a single commercial franchise is increasingly common as pharmaceutical manufacturers develop drug candidates across multiple modalities.
Building Your LOE Playbook
LOE planning is not an event-driven exercise. Companies that treat patent expiry as a project to manage once the date becomes imminent routinely leave value on the table. Effective LOE playbooks are built years in advance and updated quarterly as the competitive landscape shifts.
Brand Manufacturers: Playing Defense
Brand manufacturer defense strategy is about extending the period during which revenues remain near their pre-LOE peak, while managing the transition in a way that preserves long-term patient and prescriber relationships. The tools available differ between small molecules and biologics.
Lifecycle Management Tactics
For small molecules, lifecycle management typically involves new formulations (extended-release, fixed-dose combinations, new delivery forms), new indications with additional patent protection, line extensions to related compounds in the same class, and patient assistance programs designed to retain cost-sensitive patients on the branded product through the exclusivity period.
The financial logic of lifecycle management investments is straightforward. If a new extended-release formulation earns three additional years of market exclusivity for a $2 billion annual revenue product, the present value of those three years at even a 50% discount for expected market share loss is approximately $2.5 billion. Development costs for a new formulation rarely exceed $50 million to $150 million. The return on investment is compelling.
AstraZeneca’s esomeprazole (Nexium) strategy is the most cited example. When omeprazole (Prilosec) lost exclusivity in the early 2000s, AstraZeneca had already launched esomeprazole, the S-enantiomer of omeprazole, with new composition-of-matter protection. AstraZeneca successfully migrated a significant portion of the Prilosec patient base to Nexium before Prilosec went generic, creating a new branded revenue stream with fresh patent protection.
For biologics, new indications are the primary lifecycle management tool, because they generate both clinical data supporting expanded use and method-of-treatment patent protection for the new indication. Process improvements that generate manufacturing patents provide another layer. The transition to higher-concentration formulations or improved delivery devices can create patient preference for the newer product format that biosimilars may not initially match.
Authorized Generics and Partnerships
For brand manufacturers who cannot prevent generic or biosimilar entry, authorized generic and biosimilar partnerships offer a way to participate in the post-LOE market rather than ceding all revenues to third parties. An authorized generic arrangement allows the brand manufacturer to license the innovator’s NDA to a generic manufacturer, which then produces and sells the generic product under the brand’s approved specifications. The brand receives royalties or profit sharing without manufacturing the generic itself.
For biologics, several reference product sponsors have explored partnerships with biosimilar developers for their own products, in effect licensing their manufacturing expertise and reference product approval to biosimilar entrants. These arrangements are structurally complex but allow the originator to participate in a market it cannot prevent from opening.
Generic Manufacturers: The Offensive Posture
Generic manufacturers build their LOE strategies around maximizing the number of 180-day exclusivity periods they can capture, the timing and certainty of those periods, and the commercial execution quality that determines how much of the exclusivity window’s theoretical value they actually convert into revenue.
Para IV Filing Cadence and Portfolio Management
The largest generic manufacturers maintain systematic programs for monitoring upcoming small molecule patent expirations and prioritizing Paragraph IV filing opportunities. The selection criteria combine market size (larger markets justify more litigation investment), patent vulnerability (composition-of-matter patents are harder to challenge than secondary patents), competitive activity (how many other companies are also filing), and manufacturing readiness (can the company scale production in time for a potential early entry).
Maintaining the infrastructure for aggressive Para IV programs is expensive. A major Paragraph IV litigation case can cost $5 million to $20 million in legal fees over a multi-year lifecycle. Companies with large Para IV programs effectively run permanent litigation operations with specialist IP counsel, retained scientific experts, and dedicated litigation support staff. The ROI justifies this investment only if the programs are well-selected and well-managed.
Portfolio diversification matters as much in Para IV programs as in investment portfolios. A company that concentrates all its Para IV activity in one or two high-value products faces enormous binary risk from adverse litigation outcomes. The largest generic manufacturers spread their Para IV activity across dozens of simultaneous filings, accepting that some will succeed, some will settle on favorable terms, and some will fail, and that the portfolio-level return is positive even if individual case outcomes are unpredictable.
Settlement Risk and Reward Analysis
Para IV settlements are a standard part of the competitive landscape, and generic manufacturers need a clear analytical framework for evaluating settlement offers versus litigation continuation. The key variable is the probability of winning the litigation, which determines the expected value of the contested first-filer exclusivity period. A settlement that offers an entry date two years before patent expiry in exchange for no cash payment and no exclusivity challenge is worth calculating against the probability-weighted value of fighting for a potentially much earlier entry date.
The FTC’s post-Actavis scrutiny of pay-for-delay settlements means that any settlement in which a brand manufacturer transfers value to a generic manufacturer in exchange for delayed entry will be reviewed for antitrust compliance. Generic manufacturers who accept such arrangements take on legal risk that extends beyond the immediate business deal.
Biosimilar Developers: The Long Game
Biosimilar development requires a fundamentally different strategic orientation from generic drug development. The investment horizon is longer, the capital requirement is higher, the regulatory path is less predictable, and the commercial outcome depends more on sustained market-building than on the first-mover advantages that drive generic strategy.
Reference product selection is the most consequential early decision. The ideal biosimilar target has a large and growing market with multiple approved indications, a reference product patent estate that is manageable, a manufacturing process that is amenable to analytical characterization, and a payer and prescriber community that is receptive to biosimilar use. Products meeting all four criteria are rare, which is why the most attractive biosimilar targets attract multiple development programs.
Programs targeting adalimumab, bevacizumab, trastuzumab, and rituximab attracted five or more development programs each in the global biosimilar pipeline, creating competitive pressure on development timelines and launch economics. New entrants looking for biosimilar opportunities should systematically evaluate the second tier: products with annual revenues of $1 billion to $5 billion, fewer competing development programs, and patent estates that did not attract sustained litigation from multiple reference product sponsors.
Clinical development cost management is the area where biosimilar developers have the greatest control over program economics. The FDA’s evolving guidance on extrapolation and on reduced clinical study requirements for biosimilars with strong analytical packages creates an opportunity to design development programs that spend on analytical characterization (which is relatively cheap) in order to avoid or reduce clinical trial costs (which are very expensive). Programs that invest $5 million to $10 million in sophisticated analytical work at the start of development can potentially save $50 million to $100 million in avoided clinical trials, improving both economics and development speed.
Financial Modeling for LOE Events
LOE financial models are built around two related questions: how much revenue is at risk, and when is the most likely date that risk materializes. For brand manufacturers, these models inform investment decisions about lifecycle management, authorized generics, and LOE timing offsets. For generic and biosimilar developers, they inform investment decisions about which programs to fund and at what scale.
Revenue at Risk Calculations
A revenue-at-risk model for a brand manufacturer typically starts with a baseline forecast of revenues absent generic or biosimilar entry, then applies scenario-weighted adjustments for different competitive entry timelines and market penetration curves. The key inputs are: the probability and timing of first generic/biosimilar entry, the expected number of competitive entrants within 12 months of first entry, the expected market share erosion curve, and the pricing response the brand will employ post-LOE.
For small molecules, the erosion curve is relatively predictable based on historical precedent: 60% to 70% volume loss within six months of the first generic entry, 80% to 90% within 18 months. The primary uncertainty is the entry date itself, which depends on patent litigation outcomes. Sensitivity analysis around patent litigation probability is therefore the most important variable in the model.
For biologics, the erosion curve has much wider confidence intervals. Historical biosimilar penetration has ranged from essentially zero in markets with effective brand contracting to 60% to 70% in markets with formulary mandates. The right model for a specific product needs to reflect the payer landscape, the interchangeability status of the competing biosimilars, the brand’s contracting strategy, and the clinical setting (infusion versus self-injection).
NPV of a Paragraph IV Challenge
From the generic manufacturer’s perspective, the financial model for a Para IV challenge works backward from the 180-day exclusivity window. The model estimates: total U.S. prescriptions for the reference product in the year of anticipated generic entry, the market share expected for a single generic entrant (typically 60% to 70% during exclusivity), the price at which the generic will sell relative to the brand (typically 20% to 30% discount during exclusivity), and the cost of goods sold for generic manufacturing.
For a reference product with $2 billion in annual U.S. sales, a generic capturing 65% share at 25% below brand price during a six-month exclusivity window generates approximately $488 million in gross revenues during that window. At a 70% gross margin on generic manufacturing, that is $342 million in gross profit. Discounting for a 60% litigation success probability and $15 million in litigation costs yields a risk-adjusted NPV in the range of $190 million to $210 million. These numbers explain why Para IV programs for blockbuster drugs attract intense competition among generic manufacturers.
Global LOE Strategy
Patent expiry dates are not globally synchronized. A composition-of-matter patent filed under the Patent Cooperation Treaty (PCT) receives national phase patents in each designated country, but supplementary protection certificates (SPCs) in the EU, patent term extensions in the U.S. and Japan, and varying patent term lengths in other markets create different effective exclusivity end dates by geography. Managing global LOE strategy requires mapping these differences systematically.
Patent Expiry Timing Differences
In the European Union, supplementary protection certificates extend the effective patent term for pharmaceutical products by up to five years beyond the base patent’s 20-year term, subject to a maximum effective exclusivity of 15 years from first marketing authorization. EU generic manufacturers can prepare their products during the SPC term and begin commercial distribution immediately upon expiry, giving them an advantage in early market readiness.
U.S. patent term extensions under 35 U.S.C. 156 compensate for time spent in regulatory review by extending the patent term by up to five years, with a maximum of 14 years of effective exclusivity post-approval. Unlike EU SPCs, U.S. extensions apply to the specific product approved by the FDA under the subject patent. Understanding the precise scope of a U.S. patent term extension is essential for generic manufacturers designing around it.
Japan operates a patent term extension system similar to the U.S., with extensions of up to five years available. Korea, China, and India have adopted modified versions of these frameworks, with China introducing patent linkage regulations in 2021 that create an Orange Book-like system and a 30-month stay mechanism analogous to Hatch-Waxman.
EU Reference Pricing and Biosimilar Policy
European biosimilar markets are structurally more competitive than the U.S. market for several reasons. First, the European Medicines Agency has approved biosimilars since 2006 and has a longer track record of biosimilar regulation, giving prescribers and payers more comfort with the concept. Second, national tendering systems in markets like Germany, Norway, Denmark, and the Netherlands create direct price competition between biosimilars and reference products, with the lowest bidder gaining formulary exclusivity for 12-month contract periods.
The German national biosimilar tendering model, implemented through the Substitute Biosimilar Drug regulation, has produced dramatic results. Infliximab biosimilar market share in Germany exceeded 90% within three years of initial biosimilar availability, while branded Remicade retained less than 5% of the market. The same product in the U.S. market, during the same period, showed branded Remicade with over 80% market share under Janssen’s contracting strategy.
European LOE strategy for brand manufacturers therefore emphasizes different tools than U.S. strategy. Patient preference programs, clinical evidence differentiation, and disease management partnerships matter more than payer contracting, because European payers have less tolerance for the rebate-based contracting that American PBMs use to preserve branded market share.
Emerging Markets: India, China, and Beyond
India and China represent the largest emerging market pharmaceutical opportunities and the most complex LOE environments. India’s pharmaceutical sector is built around a robust generic manufacturing industry, with the world’s highest number of FDA-regulated manufacturing facilities outside the U.S. Indian patent law was modified by the 2005 Patents Amendment Act to comply with TRIPS requirements, but Section 3(d) of the Indian Patents Act bars patent protection for new forms of known substances that do not demonstrate enhanced efficacy, a provision that has been used to deny patents for many small molecule pharmaceutical compounds that would be patentable in the U.S. or EU.
The practical consequence for LOE strategy in India is that many drugs that still have 10 to 15 years of U.S. patent protection are already off-patent in India. Indian generic manufacturers can freely manufacture these compounds for the Indian domestic market and for export to countries where the patents have also expired. For global LOE strategy, this means that Indian generics supply chains are available for export to open markets before U.S. or EU patent expiry, potentially creating supply chain readiness advantages for companies that engage Indian manufacturing partners early.
China’s pharmaceutical patent system has been substantially modernized since 2021, with the introduction of a patent linkage regime, patent term extensions for innovative drugs, and data exclusivity protections aligned with international standards. The Chinese regulatory agency, the National Medical Products Administration (NMPA), has also streamlined its generic drug review process through its quality consistency evaluation program. China is now both an important biosimilar manufacturing hub and an increasingly important market for both branded and generic pharmaceuticals.
Key Takeaways
- Small molecules and biologics follow structurally different LOE pathways. Applying the generic drug playbook to a biologic LOE event, or vice versa, will produce systematically wrong financial projections and misallocated resources.
- Small molecule LOE produces rapid, predictable, and severe revenue erosion: 70% to 90% volume loss within 6 to 12 months of first generic entry, driven by automatic substitution and Hatch-Waxman’s Orange Book linkage mechanism.
- Biologic LOE produces slower, more variable, and commercially manageable transitions: 30% to 50% biosimilar market share within two years in the U.S., versus 60% to 90% in European tendering markets, with the difference driven by contracting, formulary policy, and interchangeability status.
- Patent architecture differs fundamentally. Small molecule Orange Book patents trigger 30-month stays and formal litigation linkage. Biologic patents are defended through the BPCIA dance or outside it, with process patents creating clinical as well as legal barriers for biosimilar developers.
- The 180-day generic exclusivity period is the primary financial engine for Para IV investment. Its NPV for blockbuster drugs regularly exceeds $150 million to $400 million on a risk-adjusted basis, justifying significant litigation and development investment.
- Biosimilar development costs of $100 million to $300 million per program require larger commercial ambitions and more sustained market-building than generic drug programs. Reference product selection, clinical development efficiency, and payer contracting capability are the three most important operational variables.
- Competitive intelligence tools, including DrugPatentWatch for patent and ANDA tracking, FDA databases for approval and exclusivity monitoring, and PTAB records for IPR activity, are not optional for LOE strategy teams. Competitive activity requires continuous surveillance, not annual reviews.
- Brand manufacturer defense strategies must distinguish between what patent protection can preserve and what commercial contracting can preserve. AbbVie and Janssen demonstrated that contract-based market defense can add years of revenue preservation beyond patent expiry for biologics, with commensurate antitrust risk.
- Global LOE timing varies by market due to SPC and patent term extension differences, data exclusivity periods, and local patent law variations. India’s Section 3(d) and China’s new patent linkage regime require specific treatment in any global LOE strategy.
- Second-tier biosimilar targets, products with $1 billion to $5 billion in revenues, fewer competing development programs, and manageable patent estates, offer better risk-adjusted returns than the most contested reference products for new entrants to the biosimilar market.
Frequently Asked Questions
1. What is the single most important difference between generic and biosimilar LOE strategy?
The most important difference is the speed and predictability of market erosion. Generic drugs lose 70% to 90% of volume within months of first generic entry because automatic substitution rules, Orange Book linkage, and commodity pricing dynamics move quickly and uniformly. Biosimilars typically capture 30% to 50% of the market within two years in the U.S., and the penetration rate is highly variable depending on payer contracting, interchangeability status, and institutional formulary decisions. A brand manufacturer defending a small molecule LOE needs to accept rapid and large-scale revenue loss as structurally inevitable. A brand manufacturer defending a biologic LOE has genuine tools to meaningfully delay and moderate that loss.
2. Can a biosimilar developer avoid the patent dance entirely, and should it?
Yes. The Supreme Court’s Sandoz v. Amgen decision confirmed that participation in the BPCIA patent dance is optional for a biosimilar applicant. The developer may decline to provide the manufacturing information disclosure and instead face litigation after giving 180-day notice of commercial marketing. Whether to participate is a product-specific decision. Participating means disclosing manufacturing process information that may be proprietary and that could be used by the reference product sponsor in subsequent litigation. Declining means foregoing the structured framework for identifying all disputed patents before litigation begins, potentially leading to serial litigation on patents that could have been consolidated. Many biosimilar developers have chosen not to participate, preferring to manage patent risk through IPR challenges at the PTAB and freedom-to-operate analyses rather than through the dance’s information exchange mechanism.
3. How does an interchangeability designation actually change biosimilar market dynamics?
Interchangeability allows pharmacists to substitute a biosimilar for the reference product without a new prescription in most U.S. states, mirroring the automatic substitution that drives rapid generic drug adoption. Without interchangeability, the prescriber must affirmatively prescribe the specific biosimilar. For oral self-administered biologics and injectable biologics dispensed through specialty pharmacy, interchangeability removes the primary friction point in biosimilar adoption for the retail and specialty pharmacy channels. For infused biologics administered in hospital or clinical settings, formulary decisions are made at the institutional level and interchangeability matters less. The first biosimilar to achieve interchangeability for a given reference product also earns a period of exclusivity before any other biosimilar can receive the same designation, creating meaningful first-mover value for programs that invest in the additional clinical requirements for interchangeability.
4. When does it make financial sense for a brand manufacturer to launch an authorized generic of its own product?
An authorized generic makes financial sense when the following conditions are present: the first Paragraph IV filer has a strong litigation position (making settlement or adverse judgment likely), the 180-day exclusivity period represents significant commercial value, and the brand manufacturer can negotiate a royalty or profit-sharing structure with an authorized generic partner that captures more value than would otherwise flow entirely to the first filer. The primary effect of an authorized generic is to destroy the first filer’s exclusivity-period pricing power by introducing a second generic competitor during the window that should be exclusivity-protected. The brand recovers some value from the exclusivity period while reducing the economic reward for having filed the Paragraph IV certification. For drugs with annual U.S. sales above $500 million, this calculation almost always favors an authorized generic strategy.
5. What is the most common analytical mistake in biosimilar LOE financial models?
The most common mistake is importing small molecule LOE erosion curves into biosimilar models without adjusting for the structural differences in market dynamics. Analysts who have built generic drug models are often tempted to use the 80% to 90% first-year volume erosion figure as a baseline for biosimilar entry as well. The U.S. market data clearly does not support this: the average biosimilar’s market penetration in the first two years post-launch has been significantly lower than generic drug penetration at comparable time intervals, and the variance is enormous depending on therapeutic category, interchangeability status, and brand contracting. A more accurate approach models two distinct scenarios: a competitive scenario resembling European tendering outcomes and a contract-protected scenario resembling the early Humira biosimilar market, weights them by probability based on product-specific factors, and uses that probability-weighted curve as the base case for financial projections. Single-point estimates for biosimilar market penetration are almost certain to be wrong; range-based modeling is essential.
References
[1] Association for Accessible Medicines. (2023). Generic Drug & Biosimilars Access & Savings in the U.S. Report. Washington, DC: AAM.
[2] IQVIA Institute for Human Data Science. (2023). The Global Use of Medicines 2023: Outlook to 2027. IQVIA.
[3] Blackstone, E. A., & Fuhr, J. P. (2013). The economics of biosimilars. American Health & Drug Benefits, 6(8), 469-478.
[4] Sandoz Inc. v. Amgen Inc., 582 U.S. 1 (2017).
[5] Feldman, R., & Frondorf, E. (2017). Drug wars: How big pharma raises prices and keeps generics off the market. Harvard Business Review Press.
[6] U.S. Food and Drug Administration. (2019). Generic Competition and Drug Prices. FDA Office of Generic Drugs. https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/generic-competition-and-drug-prices
[7] Association for Accessible Medicines. (2023). Generic Drug & Biosimilars Access & Savings in the U.S. Report. Washington, DC: AAM.
[8] U.S. Food and Drug Administration. (2021). FDA approves first interchangeable biosimilar insulin product for treatment of diabetes [Press release]. https://www.fda.gov/news-events/press-announcements
[9] IQVIA Institute for Human Data Science. (2022). Biosimilars in the United States 2017-2021: Competition, Savings, and Sustainability. IQVIA.
[10] European Medicines Agency. (2022). Biosimilars in the EU: Information guide for healthcare professionals. EMA.
[11] FTC v. Actavis, Inc., 570 U.S. 136 (2013).
[12] Congressional Budget Office. (2021). Research and development in the pharmaceutical industry. CBO.
[13] Federal Trade Commission. (2022). Pharmacy benefit managers: The powerful middlemen inflating drug costs and squeezing main street pharmacies. FTC Report.
[14] Lotvin, A., Luthra, R., Demas, M., & Audet, A. M. (2019). US biosimilar market performance during a period of increasing regulatory approvals. Health Affairs, 38(10), 1718-1725.